Abstract
Heavy alcohol consumption is a major cause of morbidity and mortality. Globally, alcohol per-capita consumption rose from 5.5 litres in 2005 to 6.4 litres in 2016 and is projected to increase further to 7.6 litres in 2030. In 2019, an estimated 25% of global cirrhosis deaths were associated with alcohol. The global estimated age-standardized death rate (ASDR) of alcohol-associated cirrhosis was 4.5 per 100,000 population, with the highest and lowest ASDR in Africa and the Western Pacific, respectively. The annual incidence of hepatocellular carcinoma (HCC) among patients with alcohol-associated cirrhosis ranged from 0.9% to 5.6%. Alcohol was associated with approximately one-fifth of global HCC-related deaths in 2019. Between 2012 and 2017, the global estimated ASDR for alcohol-associated cirrhosis declined, but the ASDR for alcohol-associated liver cancer increased. Measures are required to curb heavy alcohol consumption to reduce the burden of alcohol-associated cirrhosis and HCC. Degree of alcohol intake, sex, older age, obesity, type 2 diabetes mellitus, gut microbial dysbiosis and genetic variants are key factors in the development of alcohol-associated cirrhosis and HCC. In this Review, we discuss the global epidemiology, projections and risk factors for alcohol-associated cirrhosis and HCC.
Key points
-
Global alcohol consumption per capita rose from 5.5 litres in 2005 to 6.4 litres in 2016 and is projected to increase further to 7.6 litres in 2030.
-
Currently, Europe has the highest levels of alcohol consumption; however, it is projected to be surpassed by countries/regions in the Western Pacific region by 2030.
-
Alcohol was estimated to be associated with one-quarter of global cirrhosis deaths and one-fifth of liver cancer deaths in 2019.
-
Alcohol was the second-fastest-growing cause of liver-cancer deaths from 2010 to 2019.
-
Patients with alcohol-associated hepatocellular carcinoma (HCC) tend to present with advanced tumours, which relates at least in part to late diagnosis and limited access to HCC screening in comparison to other aetiologies of liver disease.
-
The risk factors for the development of cirrhosis and HCC include the amount of alcohol consumed, age, obesity, diabetes, smoking and PNPLA3 variants.
Similar content being viewed by others
Introduction
Heavy alcohol consumption, which is defined in this Review as the consumption of >40 g of pure alcohol per day over a sustained period, is a major cause of mortality and morbidity1,2. The World Health Organization (WHO) Global Status Report on Alcohol and Health estimated that 2.3 billion people are current alcohol drinkers, who consume an average of 32.8 g of pure alcohol per day3. Global per-capita alcohol consumption increased from 1990 to 2017 and is projected to increase further by 20304.
Alcohol use was associated with 3 million deaths and 132.6 million disability-adjusted life years (DALY) in 2016 alone3. Of those aged 15–49 years, 12% of male deaths and 4% of female deaths were associated with alcohol-related diseases and injuries in 2016 worldwide5. Alcohol was the seventh-leading cause for both deaths and DALY in 2016, leading to 7% and 2% of male and female age-standardized deaths, respectively5. Among alcohol-attributable deaths, the highest percentage was secondary to digestive diseases (21%) and >90% of these were secondary to liver cirrhosis3.
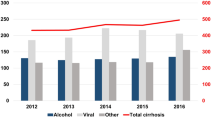
Alcohol is one of the most common causes of liver diseases worldwide and causes a wide spectrum of direct liver injury ranging from steatosis, alcoholic hepatitis, cirrhosis and hepatocellular carcinoma (HCC)2,6,7,8,9,10,11. One-quarter of the global deaths due to cirrhosis in 2019 were associated with alcohol12. Liver cancer is now the third-leading cause of cancer death worldwide13. In 2019, alcohol was associated with an estimated 19% of deaths from liver cancer globally, although this varied markedly by region12,14. The age-standardized death rate (ASDR) for alcohol-associated cirrhosis declined between 2012 and 2017, whereas the ASDR for alcohol-associated liver cancer increased15.
Given the projected increase in global alcohol exposure, the burden of alcohol-associated liver disease (ALD), cirrhosis and HCC might increase in parallel4. In this Review, we highlight the global trends for alcohol consumption by country/region. We discuss epidemiology, trends, risk factors and projections for alcohol-associated cirrhosis and HCC.
Overview of global alcohol consumption
The WHO Global Status Report on Alcohol and Health, Global Burden of Disease Study and the Global Information System on Alcohol and Health provide comprehensive data on alcohol exposure and have facilitated modelling studies and projections up until 20303,4,5,16.
Trends in alcohol per capita consumption
Alcohol per capita consumption (APC) is defined as alcohol consumption within a calendar year in litres of pure alcohol in people aged 15 years or older. The global APC rose from 5.5 litres in 2005 to 6.4 litres in 20163. Between 2005 and 2016, APC rose steadily in the Western Pacific region (4.6 litres in 2005 to 7.3 litres in 2016) and the Southeast Asia region (2.1 litres in 2005 to 4.5 litres in 2016)3, largely owing to increased APC in China and India, respectively. Europe had the highest total APC in the world in 2016, although its APC had declined over time (12.3 litres in 2005 to 9.8 litres in 2016)3. The rise in APC in the Western Pacific and Southeast Asia might in part be related to rising economic wealth and per capita income, whereas measures such as the enforcement of a minimum price for alcohol and increased alcohol taxation might contribute to the decline in APC in Europe17,18.
Projections in alcohol per capita consumption
The global APC is projected to increase to 7.6 litres in 2030, fuelled by increased consumption in low to middle-income countries/regions3,4. The greatest increases are anticipated in the Southeast Asia region (6.9 litres in 2030) and the Western Pacific region (9.9 litres in 2030), driven by increasing economic wealth in highly populated countries/regions such as India and China3,4,19,20,21. By contrast, the APC in Europe is expected to fall to 9.3 litres in 2030. For each country/region, the models accounted for economic wealth and the proportion of the population that identified as Muslim, but not alcohol-related policies. In addition, the uncertainty of forecasts increases with the length of the forecasting interval. The projected APCs require cautious interpretation as the models used in the projections relied on several assumptions, utilized surveys for estimates of unrecorded alcohol consumption, and did not account for potential alcohol-related policy changes. Box 1 provides a discussion of the limitations of the models used to generate these estimates.
Trends in patterns of alcohol consumption
The percentage of current drinkers (defined by any alcohol consumption within the previous 12 months) increased from 45% in 1990 to 47% in 2017 globally4. Heavy episodic drinking is defined as the consumption of 60 g or more of pure alcohol on at least one occasion at least once per month22, whereas binge drinking has been defined in the USA as the consumption of 70 g of pure alcohol for men or 56 g of pure alcohol for women within a 2-hour period, although definitions vary by region23. In this Review, we report the degree of alcohol consumption by grams of pure alcohol, given that the ‘standard unit’ of alcohol differs by country/region, ranging from 8 g in the UK, to 14 g in the US and 20 g in Japan24. Binge drinking or heavy drinking is associated with an increased risk of developing alcohol-induced liver damage25. Across all WHO regions, heavy episoFignifdic drinking was most prevalent among people aged 20–24 years in 2016 (ref.3), and the percentage of heavy episodic drinking in men and women in the general population in 2017 was 29% and 11%, respectively4.
Epidemiology of alcohol-associated cirrhosis
The Global Burden of Disease Study provides an overview of the estimated frequency of incident cases and deaths due to alcohol-associated cirrhosis and other chronic liver diseases (collectively referred to as cirrhosis in the Global Burden of Disease Study) and liver cancer12,26. Although the Global Burden of Disease Study provides the only peer-reviewed estimates of cause-specific mortality for each country/region, the quality of data is dependent on the quality of each country/region’s vital registration system15. In countries/regions with missing data, the estimates rely on modelling studies and past trends. In addition, cirrhosis and liver cancer might have been underdiagnosed in countries/regions with inadequate access to care and diagnostic tools. Therefore, the estimates of cirrhosis and liver cancer mortality might be underestimated in countries/territories with a lower standard of healthcare and require cautious interpretation. Globally, 25% of cirrhosis deaths in 2019 were estimated to be associated with alcohol12. The highest percentage of cirrhosis deaths for each region that were associated with alcohol was in Europe (42%), and the lowest percentage was in the Eastern Mediterranean region (8%)12 (Table 1).
Age-standardized death rates for alcohol-associated cirrhosis
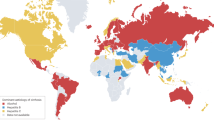
The global estimated ASDR for alcohol-associated cirrhosis in 2019 was 4.5 deaths per 100,000 population (95% confidence interval (CI) 3.8–5.3)12. The estimated ASDRs for alcohol-associated cirrhosis in 2019 by country/territory are summarized in Fig. 1. The estimated ASDRs in 2019 by country/territory ranged from 0.5 deaths (95% CI 0.4–0.7) per 100,000 population in Singapore, to 29.3 deaths (95% CI 21.6–38.9) per 100,000 population in Mongolia12. Between 2012 and 2017, there was a reduction in the global estimated ASDR for alcohol-associated cirrhosis (annual percentage change −0.44% (95% CI −0.78 to −0.10)15. In our opinion, the ASDRs might be an underestimate owing to underreporting of deaths due to alcohol-associated cirrhosis.
Data for the age-standardized death rate in 2019 were estimated by the Global Burden of Disease study 2019 for each country/territory12. These data were obtained from the GBD Results Tool, which is maintained by the Institute for Health Metrics and Evaluation119. Where data for certain countries or regions were unavailable, the Global Burden of Disease Study 2019 results depended on modelling and past trends, potentially resulting in discrepancies in the accuracy of the data.
Projections for mortality and morbidity due to ALD
In the USA, alcohol has resulted in rising rates of mortality from cirrhosis, especially among individuals aged 25–34 (ref.27). A modelling study utilized data from the National Epidemiologic Survey on Alcohol and Related Conditions, the National Institute on Alcohol Abuse and Alcoholism, National Death Index, National Vital Statistics System and other published data to forecast outcomes from alcohol-associated liver disease in the USA up to 2040 (ref.28). The ASDRs related to alcohol-associated liver disease are projected to increase from 8.2 deaths per 100,000 patient-years in 2019 to 15.2 per 100,000 patient-years by 2040, if current trends in alcohol consumption are left unchecked. Similarly, the age-standardized incidence of decompensated alcohol-associated cirrhosis in the USA is projected to increase by 77% from 9.9 cases per 100,000 patient-years in 2019 to 17.5 cases per 100,000 patient-years in 204028. However, the researchers used data from multiple sources, and made assumptions regarding the past rates of heavy alcohol consumption in the birth cohorts from 1995 to 2017 owing to a lack of data; therefore, the results of this modelling study should be interpreted cautiously. The projected burden of ALD in the USA might be underestimated in this study, as the models relied partially on self-reported survey data of alcohol consumption and did not account for underreporting of alcohol-related deaths. The limitations of the models used in this study are further discussed in Box 1. There are limited data for projected mortality and morbidity due to ALD outside the USA.
Effect of COVID-19 on ALD
COVID-19 has caused a severe burden to healthcare systems worldwide, adversely affecting the care of patients with liver disease29,30,31,32. National sales of alcohol increased in the USA, resulting in an increase of 14% in the frequency of alcohol consumption33 (Box 2). A cross-sectional telephone survey of individuals from the UK who were current drinkers prior to lockdown revealed that 21% had been drinking more frequently since lockdown, and 15% had been drinking more per session34. In the USA, there was an increase of 107% in the rate of adding patients with acute alcohol-associated hepatitis to the liver transplant waiting list, and a 210% increase in the rate of receipt of liver transplantation between March 2020 and February 2021 compared with March 2018 through February 2020 (ref.35). An international registry study of 745 patients with chronic liver disease and SARS-CoV-2 infection reported that among various causes of liver disease, ALD was the only statistically significant independent predictor of death (adjusted OR 1.8; 95% CI 1.0–3.1, P = 0.04)36. The global burden of ALD could increase over the next few years owing to increased alcohol consumption during the pandemic37, especially if the COVID-19 pandemic persists.
Epidemiology of alcohol-associated HCC
The Global Burden of Disease Study provides a comprehensive overview of the various aetiologies of liver cancer, stratified by country and region12,15,38. Although this study encompassed various histologies of liver cancer including HCC, cholangiocarcinoma and mixed tumours, HCC is by far the most common form of liver cancer39. In 2019, 91,000 deaths from liver cancer were estimated to be associated with alcohol12,14. However, the frequencies of liver cancer deaths are likely to be an underestimate owing to under-reporting of liver cancer during death certification, lack of disease awareness, or lack of diagnostic capacity in regions of low sociodemographic index40,41.
Incidence of alcohol-associated HCC
The annual incidence of alcohol-associated HCC ranges from 0.86% to 5.6% across studies42,43 (Table 2). In our opinion, this wide range in the reported incidence rate of alcohol-associated HCC might be due to inter-study differences in the prevalence of diabetes, obesity, and severity of hepatic decompensation. Hence, the variance in the reported incidence rate of HCC should be interpreted with caution and might not be generalizable to all patients with alcohol-associated cirrhosis. A systematic review and meta-analysis of reconstructed individual patient data of 18 studies (148,333 patients) determined that the cumulative incidence of HCC in patients with alcohol-associated cirrhosis at 1-, 5- and 10-year follow-ups was 1%, 3% and 9%, respectively44.
A retrospective Swedish nationwide population-based study of 2,042 patients with biopsy-proved alcohol-associated cirrhosis evaluated patients between 1969 and 2016 and reported an average annual HCC incidence of only 0.86% (8.6 per 1,000 patient-years)42. HCC awareness and diagnosis rates in the earlier years of this study were likely to be suboptimal and might have contributed to a lower than expected HCC incidence. Another nationwide, population-based study from Denmark evaluated 8,482 patients with alcohol-associated cirrhosis45. This retrospective cohort study was conducted among individuals with a hospital diagnosis of alcohol-associated cirrhosis between 1993 and 2005 and also reported a low HCC incidence of 4.0 per 1,000 patient-years. Neither the studies from Sweden or Denmark reported data on the proportion of patients with hepatic decompensation. It is difficult to interpret the estimates of HCC incidence in the Swedish and Danish studies owing to the long study period, because treatment of cirrhosis has substantially improved over time, resulting in a reduction of cirrhosis mortality rates but increased HCC incidence rates.
In a retrospective study of 16,175 patients with alcohol-associated cirrhosis from the Veteran Affairs healthcare system in the USA, the annual incidence of HCC was 1.4%46. In this study, 36.5% of patients had decompensation (ascites, encephalopathy or variceal bleeding). A study from Europe evaluated 652 patients with biopsy-proven alcohol-associated cirrhosis from 22 centres in France and Belgium and found a higher annual HCC incidence, of 2.9 per 100 patient-years47. This study had a higher percentage of patients with a history of hepatic decompensation (65.3%), which contributes to higher incidence of HCC. Similarly, a study from Taiwan evaluated 202 patients with alcohol-associated cirrhosis (46% were Child-Pugh class B or C) from two tertiary hospitals and found an annual HCC incidence of 2.1%48. Patients with decompensated alcohol-associated cirrhosis should be monitored carefully for the development of HCC where clinically appropriate.
Projected incidence of alcohol-associated HCC
The above-mentioned modelling study from the USA provided projections for the incidence of alcohol-associated HCC. The incidence is estimated to increase from 1.1 per 100,000 person-years in 2019 to 2.0 per 100,000 person-years in 2040, if current trends continue28. Data for projected HCC incidence outside the USA are scarce. In our opinion, the estimates used in the models for alcohol-associated HCC might have underestimated the projected incidence of alcohol-associated HCC owing to underreporting of deaths secondary to alcohol-associated HCC49,50.
Percentage of liver cancer deaths attributed to alcohol
Globally, an estimated 19% of all liver cancer deaths in 2019 were associated with alcohol, which was lower than for liver cancer deaths associated with hepatitis B virus infection (40%) and hepatitis C virus infection (29%)14. There was marked regional variation in the percentage of liver cancer deaths related to alcohol, with the highest percentage found in Europe (35%) and the lowest percentage in the eastern Mediterranean region (10%)14 (Table 1). As adherence to surveillance among patients with alcohol-associated cirrhosis is lower compared with patients with cirrhosis from other causes of liver disease, and patients with alcohol-associated cirrhosis often have competing medical co-morbidities that might result in death before HCC diagnosis, the percentage of liver cancer deaths associated with alcohol are likely to be an underestimate11.
Age-standardized death rates for alcohol-associated liver cancer
The global estimated ASDR for alcohol-associated liver cancer in 2019 was 1.1 deaths (95% CI 0.9–1.3) per 100,000 population12,14. The global estimated ASDR for alcohol-associated liver cancer increased between 2010 and 2019 (annual percentage change 0.23%, 95% CI 0.09–0.37), and alcohol was the second-fastest-growing cause of liver cancer deaths, after nonalcoholic steatohepatitis14. Estimated ASDRs for alcohol-associated liver cancer in 2019 by country/territory are summarized in Fig. 2. The ASDR by country/region ranged from 0.1 deaths per 100,000 population (95% CI 0.1–0.2) in Niger to 34.2 deaths per 100,000 population (95% CI 23.1–47.8) in Mongolia. In our opinion, these ASDRs might be underestimated in countries/regions with a lower socioeconomic status owing in part to a lack of access to care and the diagnostic services required for diagnosis of liver cancer.
Data for the age-standardized death rate in 2019 were estimated by the Global Burden of Disease Study 2019 for each country/territory12. These data were obtained from the GBD Results Tool maintained by the Institute for Health Metrics Evaluation119. Where data for certain countries or regions were unavailable, the Global Burden of Disease Study 2019 results depended on modelling, potentially resulting in discrepancies in the accuracy of the data. These ASDRs may be an underestimate owing to underreporting, especially in countries/regions with a lack of access to the diagnostic services required for the diagnosis of liver cancer.
Gaps in the care of patients with alcohol-associated HCC
Several studies from Europe have shown that patients with alcohol-associated liver disease present with advanced tumours, which relates at least in part to late referrals and limited access to HCC screening in comparison to other aetiologies51,52,53,54. The rate of access to screening is noticeably lower than for patients with other aetiologies because a high proportion of patients with alcohol-associated liver disease has not been diagnosed and consequently these patients are not in a screening programme51,55,56. In addition, patients with alcohol-associated HCC are more likely to be diagnosed at a more advanced stage than patients with other aetiologies53. There are multiple potential reasons for a lower HCC surveillance rate among patients with alcohol-associated cirrhosis, including lack of disease awareness, clinic time constraints due to other active medical issues, and provider beliefs regarding the likelihood of adherence56. These factors lead to lower access to curative therapies and reduction in overall survival of alcohol-associated HCC compared with viral-related HCC57.
A study of the USA Scientific Registry of Transplant Recipients from 2002 to 2016 estimated the trends in the prevalence of HCC among liver transplant candidates58. Of the 24,431 patients with a listing diagnosis that provided information on the aetiology of their HCC, 10.3% were secondary to alcohol and 7.9% were secondary to a combination of alcohol and hepatitis C virus infection. Between 2002 and 2016, the prevalence of alcohol-associated HCC among liver transplant candidates increased by 3.1-fold, and the combination of hepatitis C virus infection and alcohol as a cause of HCC increased by 6.4-fold. In addition, these patients have poor disease awareness and social circumstances that further limit their access to care, which contribute to the large discrepancy between the number of deaths and the number of transplantations related to alcohol-associated HCC12,59.
The combination of the anti-programmed cell death ligand 1 (PDL1) antibody atezolizumab and the vascular endothelial growth factor (VEGF)-neutralizing antibody bevacizumab has been established as the current standard of care for advanced HCC60,61. A meta-analysis of three randomized controlled phase III trials of immunotherapy in patients with advanced HCC determined that immunotherapy improved survival in the overall analysis and in subgroup analysis for patients with viral-associated HCC, but not in subgroup analysis for patients with non-viral HCC (including HCC related to nonalcoholic fatty liver disease (NAFLD) and ALD)62. However, it is unclear whether patients with alcohol-associated HCC have poorer response to immunotherapy, as the researchers of this study grouped NAFLD-related HCC and alcohol-associated HCC together in their analysis. By contrast, a multicentre European study (comprising of patients from Austria, Germany, Italy and Switzerland) of 292 patients with HCC treated with PDL1-based immunotherapy found that aetiology of liver disease (viral versus non-viral) was not a predictor of survival63. Further data are required to determine whether patients with alcohol-associated HCC derive the same degree of benefit from immunotherapy as those with viral-associated HCC.
Risk factors
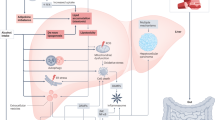
Alcohol-associated liver disease encompasses a spectrum of diseases ranging from alcohol-associated fatty liver, steatohepatitis and, finally, cirrhosis. Alcohol-associated fatty liver develops in more than 90% of individuals who drink more than 60 g of pure alcohol per day, even in the absence of obesity64. Up to 35% of hospitalized patients with ALD have alcohol-associated steatohepatitis64. In a study of 330 consecutive patients from Denmark who consumed more than 50 g of pure alcohol per day for more than one year and underwent a liver biopsy, 18% of patients either had histology consistent with cirrhosis, or suspicious for cirrhosis65. Although most people who regularly consume heavy amounts of alcohol develop alcohol-associated fatty liver disease, only a small proportion go on to develop cirrhosis or cancer, which suggests that genetics and environmental factors have an important role in determining susceptibility to advanced liver disease2. The pathogenesis of alcohol-associated cirrhosis and HCC has been reviewed elsewhere and is summarized in66 Fig. 3. The risk factors for alcohol-associated cirrhosis and HCC include age, sex, obesity, amount, pattern and duration of alcohol consumption, genetics and gut microbial dysbiosis43,47,67 (Fig. 4).
Alcohol is metabolized to acetaldehyde by alcohol dehydrogenase (ADH) and cytochrome P450 2E1 (CYP2E1) within hepatocytes124, resulting in the formation of reactive oxygen species (ROS), which activates hepatic stellate cells and leads to fibrosis in conjunction with acetate. Acetaldehyde triggers inflammation, forms DNA adducts and results in DNA hypomethylation, which leads to hepatocarcinogenesis125. Formation of ROS also leads to the formation of DNA adducts via lipid peroxidation and subsequent carcinogenesis126. Alcohol induces microbial dysbiosis and alters gut permeability, enabling translocation of lipopolysaccharide (LPS) and gut bacteria to the liver114. This translocation results in the activation of Kupffer cells and production of pro-inflammatory cytokines and chemokines, which promotes fibrosis and activation of the oncogenic interleukin-6 (IL-6)–signal transducer and activator of transcription 3 (STAT) and tumour necrosis factor (TNF)–nuclear factor-κB (NF-κB) pathways127.
Sex
Women are more susceptible than men to alcohol-induced liver injury, although the exact mechanisms are still unclear68,69,70,71. In a prospective study (n = 13,285) conducted in Denmark, an increased risk of developing alcohol-associated liver disease and cirrhosis was observed when alcohol consumption exceeded 168 g of alcohol per week for men and 84 g per week for women72. In the prospective Million Women Study conducted in the UK among 401,806 women, consuming approximately 100 g of alcohol per week or more was associated with an increased risk for cirrhosis in women, compared with consuming only 12 g of alcohol a week73. Despite the greater susceptibility of women to alcohol-induced liver injury, the global prevalence of alcohol-associated cirrhosis is lower in women, compared with men, owing to their substantially lower levels of alcohol consumption3,4.
Among patients with alcohol-associated cirrhosis, the incidence of HCC is higher among men. In the previously mentioned study of patients with alcohol-associated cirrhosis in Denmark, the HCC incidence (per 1,000 patient-years) was 5.8 and 0.7 in men and women, respectively45. Similarly, a study from France analysed 652 patients with alcohol-associated cirrhosis and found that male sex was an independent predictor for HCC development (adjusted HR 2.66; 95% CI 1.12–6.32)47. A prospective study of 55,917 individuals from the Danish Diet, Cancer and Health study demonstrated that compared with non-drinkers, the hazard ratio for cirrhosis increased disproportionately in women versus men as the volume of alcohol consumed increased74. These studies collectively emphasize that women are more susceptible to alcohol-induced liver injury than men. However, as alcohol consumption is substantially higher in men, ASDRs of alcohol-associated liver cancer are higher in men compared with women, globally75.
Obesity
A prospective study of 9,559 men from Scotland found that obesity was an independent predictor of liver disease mortality in men who consumed at least 120 g of alcohol per week (adjusted relative risk 18.9; 95% CI 6.84 to 52.4)76. In this study, alcohol and obesity interacted in a synergistic manner (synergy index 2.89). Similarly, a population-based study (n = 2,364 participants) in California found that consuming more than 30 g of pure alcohol per day, and the presence of obesity, were both independently associated with elevation of serum alanine aminotransferase levels77. In this study from California, the combination of both factors (consuming more than 30 g of pure alcohol per day and the presence of obesity) appeared to have a multiplicative effect on increasing alanine aminotransferase levels. In addition, a population-based study of 13,580 individuals in the USA determined that the consumption of 14 g of alcohol per day was associated with a substantial risk of elevation in serum alanine transaminase levels in individuals with obesity78. In a French cohort of 268 patients with heavy alcohol consumption, BMI was an independent risk factor for liver fibrosis (r = 0.11 ± 0.04; P < 0.002)79. These studies provide clear evidence that obesity potentiates the toxic effects of alcohol and hastens progression to cirrhosis.
Besides being a strong risk factor for cirrhosis, obesity is also an independent risk factor for HCC development. A prospective, population-based study of 23,712 Taiwanese residents determined that the presence of alcohol drinking (defined in this study as the consumption of alcohol for at least 4 days per week for at least 1 year) and obesity resulted in an increased risk of incident HCC (adjusted HR 3.82; 95% CI 1.94–7.52), with a multiplicative interaction between alcohol and obesity80. In a study of 2,260 Taiwanese men with chronic hepatitis B virus infection, alcohol use (defined in this study as the consumption of alcohol for at least 4 days per week for at least 1 year) and obesity had synergistic effects to increase the risk of incident HCC, with an increased risk for each unit increase in BMI81. A study of 622 patients with HCC from the USA that compared past weight and height (self-reported) of HCC cases against healthy control individuals determined that early-adult obesity was associated with an increased risk of developing HCC at a younger age, with each unit increase in BMI at early adulthood associated with a 3.9-month decrease in age at HCC diagnosis82. An analysis of the United Network for Organ Sharing database showed that obesity was an independent risk factor for HCC in patients with alcohol-associated cirrhosis (adjusted HR 3.2; 95% CI 1.5–6.6)83. A study from France followed up 478 patients with biopsy-proved alcohol-associated cirrhosis and found 108 incident HCCs; the researchers also concluded that high BMI was an independent predictor for HCC occurrence43. Likewise, a USA Veteran Affairs healthcare system study analysed data from 16,175 patients with alcohol-associated cirrhosis and found that BMI > 25.2 kg/m2 was an independent predictor for HCC development46. These studies collectively demonstrate that the presence of obesity is a strong and independent predictor of HCC development in alcohol-associated cirrhosis.
Diabetes mellitus
Type 2 diabetes mellitus (T2DM) is a risk factor for the development of cirrhosis owing to the pro-fibrogenic effect of glucose on hepatic stellate cells and subsequent induction of chronic inflammation84,85. A study of 268 patients with alcohol use disorder who underwent liver biopsy determined that fasting glucose independently correlated with liver fibrosis score (r = 0.115 ± 0.045, P < 0.011)79. In the previously mentioned French study of patients with alcohol-associated cirrhosis, the presence of T2DM (adjusted HR 1.6; 95% CI 1.1–2.3; P = 0.029) was an independent predictor for HCC43. Similarly, the USA study conducted within the Veteran Affairs healthcare system found that T2DM was an independent predictor for HCC development (adjusted HR 1.46; P < 0.001)46. Taken together, these findings indicate that the presence of T2DM increases the risk of HCC by approximately 50% in patients with alcohol-associated cirrhosis. Efforts should be undertaken to screen individuals with heavy alcohol consumption for diabetes to identify patients at higher risk of cirrhosis and HCC.
Pattern and degree of alcohol consumption
A meta-analysis of 5,505 individuals (from the USA, Italy, Denmark, the UK and China) with alcohol-associated cirrhosis demonstrated that the risk of cirrhosis increased exponentially in women with any degree of alcohol consumption, whereas for men the risk increased only beyond consumption of 12 g of alcohol per day86. A study of 55,917 individuals from Denmark found that the risk of alcohol-associated cirrhosis in men was higher for daily drinkers compared with those who drink 2–4 days a week (HR 3.65; 95% CI 2.39–5.55)74. The Million Women Study from the UK found that daily consumption of alcohol was associated with a higher risk for cirrhosis compared with non-daily consumption (RR 1.61, 95% CI 1.40–1.85; P < 0.0001)73. In addition, alcohol consumption with meals was associated with a lower risk for cirrhosis versus alcohol consumption outside meals (RR 0.69, 95% CI 0.62–0.77; P < 0.0001), after adjusting for the volume of alcohol consumed73. The researchers speculated that consuming alcohol with meals might result in delayed gastric emptying and less rapid absorption in the intestines, leading to lower blood alcohol concentrations. Additional studies are needed to validate this hypothesis87. Individuals who consume heavy amounts of alcohol daily are at the highest risk of cirrhosis and subsequent HCC88. Countries/regions with a rising burden of alcohol-associated cirrhosis and HCC should consider policies to reduce heavy alcohol consumption, such as the enforcement of a minimum price for alcohol, increased alcohol taxation and implementation of alcohol advertising bans18,89,90. In addition, resources should be provided to primary healthcare providers to enable screening for alcohol-use disorders to aid early detection and treatment of ALD before the development of cirrhosis and HCC91,92.
Smoking
Smoking increases the risk of advanced liver fibrosis and cirrhosis, potentially by activation of hepatic stellate cells through nicotinic acetylcholine receptors93. In addition, smoking results in the development of 4-aminobiphenyl-DNA adducts, which increases the risk of HCC94. A population-based study from Denmark involving 18,479 individuals determined that smoking was associated with an increased risk of alcohol-associated cirrhosis (adjusted HR 3.9; 95% CI 1.6–9.4; P = 0.001)95. Similarly, a study of 128,934 individuals from California, USA, found that cigarette smoking was independently associated with three times the risk (P < 0.05) of alcohol-associated cirrhosis in the general population96.
Smoking is an established risk factor for the development of HCC in general. A study of 1,423 patients with HCC (not specific to alcohol-associated HCC) from a consortium of 14 prospective USA cohort studies found that current smokers had an increased risk for HCC (HR 1.86; 95% CI 1.57–2.20), compared with those who have never smoked97. In addition, the researchers reported that previous smokers who had ceased smoking for more than 30 years had a similar risk of HCC to those who have never smoked. A systematic review involving 81 studies reported that the pooled odds ratio for HCC development in current smokers was 1.55 (95% CI 1.46 to 1.65; P < 0.00001) compared with non-smokers98. Studies investigating the interaction between alcohol and smoking have produced conflicting results; for example, a study of 693 individuals in Greece reported a multiplicative effect of drinking and smoking in the development of HCC99, whereas a Korean study of 1,283,112 individuals did not find any interaction between smoking and drinking on the risk of HCC development100. Collectively, these studies indicate that smoking increases the risk of developing alcohol-associated cirrhosis and HCC in general; therefore, care providers should advise smoking cessation for patients with heavy alcohol consumption.
Genetics
A growing body of evidence demonstrates that the susceptibility to developing ALD and alcohol-associated HCC is influenced by genetic factors. A genome-wide association study (GWAS) was performed in individuals of European descent and included 712 patients with alcohol-associated cirrhosis and 1,426 individuals who drink heavily without liver disease101. The researchers showed that rs738409 in the gene encoding patatin-like phospholipase domain-containing 3 (PNPLA3) was a major determinant of susceptibility towards developing cirrhosis (OR 2.19, 95% CI 1.97–2.43; P = 1.54 × 10−48) after heavy alcohol consumption101. A meta-analysis of five studies studying patients with alcohol-associated cirrhosis showed a higher prevalence of the PNPLA3 rs738409 GG genotype compared with the CC genotype, compared with healthy controls (OR 3.37 95% CI 2.49–4.58)102. This meta-analysis also demonstrated that the rs738409 GG genotype had a higher prevalence among patients with alcohol-associated cirrhosis with HCC (OR 2.81, 95% CI 1.57–5.01) compared with the CC genotype. PNPLA3 has an important role in lipid metabolism103. Further studies are required to clarify how it influences the pathogenesis of alcohol-associated cirrhosis and HCC. In addition, the GWAS study mentioned earlier101 identified variants in the membrane-bound O-acyltransferase domain-containing protein 7 (MBOAT7) (OR 1.35, 95% CI 1.23–1.49) and transmembrane 6 superfamily member 2 (TM6SF2) (OR 1.72, 95% CI 1.44–2.04) gene loci as additional factors influencing the risk of alcohol-associated cirrhosis.
A GWAS and meta-analysis involving 1,128 individuals of European ancestry from Europe, the USA and Australia reported a novel locus at rs374702773 in FAS-associated factor 2 (FAF2) (OR 0.61, P = 2.56 × 10−8) that was protective against the development of alcohol-associated cirrhosis104. FAF2 influences a lipid pathway and helps to regulate lipid droplet organization80,81. Additional studies are required to define its role in the development of cirrhosis.
A 2021 case-control GWAS of European patients compared 775 patients with alcohol-associated HCC with 1,332 control individuals with ALD without HCC105. The researchers identified a risk locus on chromosome 1 involving the WNT3A and WNT9A genes (both of which are involved in the WNT–β-catenin pathway) and determined that the WNT3A–WNT9A rs708113[T] minor allele was associated with a lower risk of developing alcohol-associated HCC. Alcohol-associated HCC is associated with somatic mutations in ARID1A, CTNNB1, SMARCA2 and TERT in clinical studies, however, genetic data have yet to be integrated into routine clinical risk assessment models for patients with alcohol-associated cirrhosis106,107. Collectively, these data demonstrate that genetic risk scores might help to risk stratify patients with ALD and might help identify patients who are at a higher risk of developing cirrhosis and HCC. Those identified to be at a higher risk could benefit from HCC screening and surveillance. Further research is needed to better define the role of genetic testing in HCC screening and surveillance.
The gut microbiota
Alcohol disrupts the intestinal barrier, predisposes individuals to gut dysbiosis, and causes an influx of endotoxins and inflammatory cytokines to the liver108,109,110. Among individuals with alcohol-use disorder, there is a decrease in abundance of the Ruminococcaceae family and an increase in abundance of the Lachnospiraceae family, along with increased intestinal permeability110. Transplanting human intestinal microbiota from a patient with severe alcoholic hepatitis into conventional mice resulted in substantially greater liver inflammation compared with mice who had been transplanted with microbiota from a patient with alcohol-use disorderwithout hepatitis111. In addition, chronic alcohol feeding in mice resulted in increased levels of unconjugated bile acids in the small intestine112. One study compared actively drinking patients (with AUDIT score > 8; ref.113) with alcohol-associated cirrhosis (n = 37) against patients with cirrhosis who were abstinent (n = 68), as well as healthy control individuals (n = 34), and reported a toxic intestinal milieu with substantially higher markers of intestinal and systemic inflammation in patients with cirrhosis who were actively drinking114. The researchers proposed that dysbiosis in patients with cirrhosis leads to farnesoid X receptor activation, which, by increasing circulating bile acids, further damages the intestinal barrier and results in a vicious cycle of increased endotoxin and inflammatory cytokine delivery to the liver108. Further studies are needed to characterize the intestinal microbiota in patients with alcohol-associated cirrhosis and to determine subgroups of patients that might benefit from therapies targeting the gut microbiota115.
In a study, the gut microbiota was characterized in 150 patients with HCC, 40 patients with cirrhosis and 131 healthy control individuals116. The investigators found that microbial diversity was lower in patients with cirrhosis than in healthy control individuals, but higher in patients with early-stage HCC compared with cirrhosis. Among patients with early-stage HCC, levels of genera belonging to butyrate-producing bacterial families decreased, whereas Klebsiella and Haemophilus species, which produce lipopolysaccharide (LPS), increased. A reduction in bacteria protecting the intestinal barrier (butyrate-producing bacterial families) and increased LPS delivery to the liver activates the NF-κB pathway, increases levels of pro-inflammatory cytokines (tumour necrosis factor (TNF), IL-6 and IL-1) and triggers hepatocarcinogenesis117,118. More data are required specifically in alcohol-associated HCC to better define the link between the gut microbiome and HCC to discover biomarkers and therapeutic targets. Box 3 summarizes future directions for research in alcohol-associated cirrhosis and HCC.
Conclusion
Globally, the consumption of alcohol has increased in the past two decades and is projected to increase further by 2030. Europe currently has the highest levels of alcohol consumption; however, this is projected to be surpassed by countries/regions in the Western Pacific by 2030. Alcohol is one of the leading causes of cirrhosis worldwide, accounting for an estimated one-quarter of cirrhosis deaths and one-fifth of liver cancer deaths. The burden of ALD is projected to increase in the USA, but projections for ALD burden outside the USA are limited. The risk factors for the development of cirrhosis and HCC include degree of alcohol consumption, sex, age, obesity, diabetes, PNPLA3 variants and gut microbial dysbiosis. Measures are required to slow the rising trend of alcohol consumption to reduce the burden of alcohol-associated cirrhosis and HCC.
References
Rehm, J., Samokhvalov, A. V. & Shield, K. D. Global burden of alcoholic liver diseases. J. Hepatol. 59, 160–168 (2013).
Seitz, H. K. et al. Alcoholic liver disease. Nat. Rev. Dis. Prim. 4, 16 (2018).
World Health Organization. Global Status Report On Alcohol And Health 2018. WHO https://www.who.int/publications/i/item/9789241565639 (2018). Comprehensive work from the World Health Organization that provides global and country/region-level data on alcohol consumption and burden.
Manthey, J. et al. Global alcohol exposure between 1990 and 2017 and forecasts until 2030: a modelling study. Lancet 393, 2493–2502 (2019). An important study that forecasts alcohol exposure in 2030.
GBD 2016 Alcohol Collaborators. Alcohol use and burden for 195 countries and territories, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 392, 1015–1035 (2018).
Crabb, D. W., Im, G. Y., Szabo, G., Mellinger, J. L. & Lucey, M. R. Diagnosis and treatment of alcohol-associated liver diseases: 2019 practice guidance from the American Association for the Study of Liver Diseases. Hepatology 71, 306–333 (2020).
Mathurin, P. & Bataller, R. Trends in the management and burden of alcoholic liver disease. J. Hepatol. 62, S38–S46 (2015).
European Association for the Study of the Liver. EASL Clinical Practice Guidelines: management of alcohol-related liver disease. J. Hepatol. 69, 154–181 (2018).
Rehm, J. et al. Alcohol as a risk factor for liver cirrhosis: a systematic review and meta-analysis. Drug Alcohol Rev. 29, 437–445 (2010).
Mathurin, P. et al. Fibrosis progression occurs in a subgroup of heavy drinkers with typical histological features. Aliment. Pharmacol. Ther. 25, 1047–1054 (2007).
Singal, A. K. & Mathurin, P. Diagnosis and treatment of alcohol-associated liver disease: a review. JAMA 326, 165–176 (2021).
Collaborators, G. D. A. I. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1204–1222 (2020). An important source of data providing peer-reviewed estimates for the burden of liver disease associated with alcohol.
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Huang, D. Q. et al. Changing global epidemiology of liver cancer from 2010 to 2019: NASH is the fastest growing cause of liver cancer. Cell Metab. 34, 969–977.e962 (2022). This study utilized data from the Global Burden of Disease Study and determined that alcohol was the second fastest growing cause of liver cancer deaths.
Paik, J. M., Golabi, P., Younossi, Y., Mishra, A. & Younossi, Z. M. Changes in the global burden of chronic liver diseases from 2012 to 2017: the growing impact of NAFLD. Hepatology 72, 1605–1616 (2020).
WHO. Global Information System on Alcohol and Health (GISAH). Global Health Observatory https://www.who.int/data/gho/data/themes/global-information-system-on-alcohol-and-health (2016).
Blas, E. and Sivasankara Kurup, A. (eds) Equity, social determinants and public health programmes. Social Determinants of Health (WHO) https://www.who.int/publications/i/item/9789241563970 (2015).
WHO. European action plan to reduce the harmful use of alcohol 2012–2020. WHO https://www.euro.who.int/__data/assets/pdf_file/0008/178163/E96726.pdf (2012).
Schmidt, L. A. & Room, R. Alcohol and inequity in the process of development: contributions from ethnographic research. Int. J. Alcohol Drug Res. 1, 41–55 (2013).
Wang, H., Ma, L., Yin, Q., Zhang, X. & Zhang, C. Prevalence of alcoholic liver disease and its association with socioeconomic status in north-eastern China. Alcohol. Clin. Exp. Res. 38, 1035–1041 (2014).
Charatcharoenwitthaya, P., Liangpunsakul, S. & Piratvisuth, T. Alcohol-associated liver disease: East versus West. Clin. Liver Dis. 16, 231–235 (2020).
Rehm, J. et al. The relationship between different dimensions of alcohol use and the burden of disease-an update. Addiction 112, 968–1001 (2017).
US Department of Health and Human Services and US Department of Agriculture. 2015–2020 Dietary Guidelines for Americans. health.gov https://health.gov/our-work/nutrition-physical-activity/dietary-guidelines/previous-dietary-guidelines/2015 (2015).
Ritchie, H. Alcohol Consumption (Our World in Data, 2019).
Bellentani, S. et al. Drinking habits as cofactors of risk for alcohol induced liver damage. The Dionysos Study Group. Gut 41, 845–850 (1997).
The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 5, 245-266, (2020).
Tapper, E. B. & Parikh, N. D. Mortality due to cirrhosis and liver cancer in the United States, 1999-2016: observational study. Br. Med. J. 362, k2817 (2018).
Julien, J., Ayer, T., Bethea, E. D., Tapper, E. B. & Chhatwal, J. Projected prevalence and mortality associated with alcohol-related liver disease in the USA, 2019-40: a modelling study. Lancet Public Health 5, e316–e323 (2020).
Toyoda, H., Huang, D. Q., Le, M. H. & Nguyen, M. H. Liver care and surveillance: the global impact of the COVID-19 pandemic. Hepatol. Commun. 4, 1751–1757 (2020).
Tan, E. X.-X. et al. Impact of COVID-19 on liver transplantation in Hong Kong and Singapore: a modelling study. Lancet Regional Health West. Pacif. https://doi.org/10.1016/j.lanwpc.2021.100262 (2021).
Boettler, T. et al. Impact of COVID-19 on the care of patients with liver disease: EASL-ESCMID position paper after 6 months of the pandemic. JHEP Rep. 2, 100169 (2020).
Li, J. et al. Epidemiology of COVID-19: a systematic review and meta-analysis of clinical characteristics, risk factors, and outcomes. J. Med. Virol. 93, 1449–1458 (2021).
Pollard, M. S., Tucker, J. S. & Green, H. D. Jr Changes in adult alcohol use and consequences during the COVID-19 pandemic in the US. JAMA Netw. Open 3, e2022942 (2020).
The Lancet Gastroenterology, H. Drinking alone: COVID-19, lockdown, and alcohol-related harm. Lancet Gastroenterol. Hepatol. 5, 625 (2020).
Bittermann, T., Mahmud, N. & Abt, P. Trends in liver transplantation for acute alcohol-associated hepatitis during the COVID-19 pandemic in the US. JAMA Netw. Open 4, e2118713–e2118713 (2021).
Marjot, T. et al. Outcomes following SARS-CoV-2 infection in patients with chronic liver disease: an international registry study. J. Hepatol. 74, 567–577 (2021).
Julien, J. et al. Effect of increased alcohol consumption during COVID-19 pandemic on alcohol-associated liver disease: a modeling study. Hepatology 75, 1480–1490 (2022).
Akinyemiju, T. et al. The burden of primary liver cancer and underlying etiologies from 1990 to 2015 at the global, regional, and national level: results from the Global Burden of Disease Study 2015. JAMA Oncol. 3, 1683–1691 (2017).
Altekruse, S. F., Devesa, S. S., Dickie, L. A., McGlynn, K. A. & Kleiner, D. E. Histological classification of liver and intrahepatic bile duct cancers in SEER registries. J. Regist. Manag. 38, 201–205 (2011).
Percy, C., Ries, L. G. & Van Holten, V. D. The accuracy of liver cancer as the underlying cause of death on death certificates. Public Health Rep. 105, 361–367 (1990).
Polednak, A. P. Using cancer registries to assess the accuracy of primary liver or intrahepatic bile duct cancer as the underlying cause of death, 1999–2010. J. Regist. Manag. 40, 168–175 (2013).
Hagström, H. et al. Risk of cancer in biopsy-proven alcohol-related liver disease: a population-based cohort study of 3,410 persons. Clin. Gastroenterol. Hepatol. https://doi.org/10.1016/j.cgh.2021.01.005 (2021).
N’Kontchou, G. et al. Risk factors for hepatocellular carcinoma in patients with alcoholic or viral C cirrhosis. Clin. Gastroenterol. Hepatol. 4, 1062–1068 (2006).
Huang, D. Q. et al. Hepatocellular carcinoma incidence in alcohol-associated cirrhosis: systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. https://doi.org/10.1016/j.cgh.2022.06.032 (2022).
Jepsen, P., Ott, P., Andersen, P. K., Sørensen, H. T. & Vilstrup, H. Risk for hepatocellular carcinoma in patients with alcoholic cirrhosis: a Danish nationwide cohort study. Ann. Intern. Med. 156, 841–847 (2012).
Ioannou, G. N., Green, P., Kerr, K. F. & Berry, K. Models estimating risk of hepatocellular carcinoma in patients with alcohol or NAFLD-related cirrhosis for risk stratification. J. Hepatol. 71, 523–533 (2019).
Ganne-Carrié, N. et al. Estimate of hepatocellular carcinoma incidence in patients with alcoholic cirrhosis. J. Hepatol. 69, 1274–1283 (2018).
Lin, C. W. et al. Heavy alcohol consumption increases the incidence of hepatocellular carcinoma in hepatitis B virus-related cirrhosis. J. Hepatol. 58, 730–735 (2013).
Aguilera, V. et al. Cirrhosis of mixed etiology (hepatitis C virus and alcohol): posttransplantation outcome — comparison with hepatitis C virus-related cirrhosis and alcoholic-related cirrhosis. Liver Transpl. 15, 79–87 (2009).
Asrani, S. K., Larson, J. J., Yawn, B., Therneau, T. M. & Kim, W. R. Underestimation of liver-related mortality in the United States. Gastroenterology 145, 375–382 (2013).
Bucci, L. et al. Comparison between alcohol- and hepatitis C virus-related hepatocellular carcinoma: clinical presentation, treatment and outcome. Aliment. Pharmacol. Ther. 43, 385–399 (2016).
Schutte, K. et al. Delayed diagnosis of HCC with chronic alcoholic liver disease. Liver Cancer 1, 257–266 (2012).
Costentin, C. E. et al. Hepatocellular carcinoma is diagnosed at a later stage in alcoholic patients: results of a prospective, nationwide study. Cancer 124, 1964–1972 (2018).
Costentin, C. E. et al. Geographical disparities of outcomes of hepatocellular carcinoma in France: the heavier burden of alcohol compared to hepatitis C. Dig. Dis. Sci. 65, 301–311 (2020).
Eskesen, A. N., Bjøro, K., Aandahl, E. M., Line, P. D. & Melum, E. Low use of surveillance and early diagnosis of hepatocellular carcinoma in Norway — a population-based cohort study. Cancer Epidemiol. 38, 741–747 (2014).
Singal, A. G. et al. Failure rates in the hepatocellular carcinoma surveillance process. Cancer Prev. Res. 5, 1124–1130 (2012).
Goutté, N. et al. Geographical variations in incidence, management and survival of hepatocellular carcinoma in a Western country. J. Hepatol. 66, 537–544 (2017).
Younossi, Z. et al. Nonalcoholic steatohepatitis is the fastest growing cause of hepatocellular carcinoma in liver transplant candidates. Clin. Gastroenterol. Hepatol. 17, 748–755.e743 (2019).
Mathurin, P. & Lucey, M. R. Liver transplantation in patients with alcohol-related liver disease: current status and future directions. Lancet Gastroenterol. Hepatol. 5, 507–514 (2020).
Sangro, B., Sarobe, P., Hervás-Stubbs, S. & Melero, I. Advances in immunotherapy for hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 18, 525–543 (2021).
Finn, R. S. et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N. Engl. J. Med. 382, 1894–1905 (2020).
Pfister, D. et al. NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature 592, 450–456 (2021).
Scheiner, B. et al. Prognosis of patients with hepatocellular carcinoma treated with immunotherapy — development and validation of the CRAFITY score. J. Hepatol. https://doi.org/10.1016/j.jhep.2021.09.035 (2021).
O’Shea, R. S., Dasarathy, S. & McCullough, A. J., Practice Guideline Committee of the American Association for the Study of Liver Diseases & Practice Parameters Committee of the American College of Gastroenterology. Alcoholic liver disease. Hepatology 51, 307–328 (2010).
Christoffersen, P. & Nielsen, K. Histological changes in human liver biopsies from chronic alcoholics. Acta Pathol. Microbiol. Scand. A 80, 557–565 (1972).
Ganne-Carrié, N. & Nahon, P. Hepatocellular carcinoma in the setting of alcohol-related liver disease. J. Hepatol. 70, 284–293 (2019).
Mancebo, A. et al. Annual incidence of hepatocellular carcinoma among patients with alcoholic cirrhosis and identification of risk groups. Clin. Gastroenterol. Hepatol. 11, 95–101 (2013).
Janele, D. et al. Effects of testosterone, 17β-estradiol, and downstream estrogens on cytokine secretion from human leukocytes in the presence and absence of cortisol. Ann. N. Y. Acad. Sci. 1069, 168–182 (2006).
Yin, M. et al. Estrogen is involved in early alcohol-induced liver injury in a rat enteral feeding model. Hepatology 31, 117–123 (2000).
Baraona, E. et al. Gender differences in pharmacokinetics of alcohol. Alcohol. Clin. Exp. Res. 25, 502–507 (2001).
Ikejima, K. et al. Estrogen increases sensitivity of hepatic Kupffer cells to endotoxin. Am. J. Physiol. 274, G669–G676 (1998).
Becker, U. et al. Prediction of risk of liver disease by alcohol intake, sex, and age: a prospective population study. Hepatology 23, 1025–1029 (1996).
Simpson, R. F. et al. Alcohol drinking patterns and liver cirrhosis risk: analysis of the prospective UK Million Women Study. Lancet Public Health 4, e41–e48 (2019).
Askgaard, G., Grønbæk, M., Kjær, M. S., Tjønneland, A. & Tolstrup, J. S. Alcohol drinking pattern and risk of alcoholic liver cirrhosis: a prospective cohort study. J. Hepatol. 62, 1061–1067 (2015).
Tan, D. J. H. et al. Global burden of liver cancer in males and females: changing etiological basis and the growing contribution of NASH. Hepatology https://doi.org/10.1002/hep.32758 (2022).
Hart, C. L., Morrison, D. S., Batty, G. D., Mitchell, R. J. & Davey Smith, G. Effect of body mass index and alcohol consumption on liver disease: analysis of data from two prospective cohort studies. BMJ 340, c1240 (2010).
Loomba, R., Bettencourt, R. & Barrett-Connor, E. Synergistic association between alcohol intake and body mass index with serum alanine and aspartate aminotransferase levels in older adults: the Rancho Bernardo Study. Aliment. Pharmacol. Ther. 30, 1137–1149 (2009).
Ruhl, C. E. & Everhart, J. E. Joint effects of body weight and alcohol on elevated serum alanine aminotransferase in the United States population. Clin. Gastroenterol. Hepatol. 3, 1260–1268 (2005).
Raynard, B. et al. Risk factors of fibrosis in alcohol-induced liver disease. Hepatology 35, 635–638 (2002).
Loomba, R. et al. Synergism between obesity and alcohol in increasing the risk of hepatocellular carcinoma: a prospective cohort study. Am. J. Epidemiol. 177, 333–342 (2013).
Loomba, R. et al. Obesity and alcohol synergize to increase the risk of incident hepatocellular carcinoma in men. Clin. Gastroenterol. Hepatol. 8, 891–898 (2010).
Hassan, M. M. et al. Obesity early in adulthood increases risk but does not affect outcomes of hepatocellular carcinoma. Gastroenterology 149, 119–129 (2015).
Nair, S., Mason, A., Eason, J., Loss, G. & Perrillo, R. P. Is obesity an independent risk factor for hepatocellular carcinoma in cirrhosis? Hepatology 36, 150–155 (2002).
Elkrief, L. et al. Diabetes mellitus in patients with cirrhosis: clinical implications and management. Liver Int. 36, 936–948 (2016).
Fehrenbach, H., Weiskirchen, R., Kasper, M. & Gressner, A. M. Up-regulated expression of the receptor for advanced glycation end products in cultured rat hepatic stellate cells during transdifferentiation to myofibroblasts. Hepatology 34, 943–952 (2001).
Roerecke, M. et al. Alcohol consumption and risk of liver cirrhosis: a systematic review and meta-analysis. Am. J. Gastroenterol. 114, 1574–1586 (2019).
Gentry, R. T. Effect of food on the pharmacokinetics of alcohol absorption. Alcohol. Clin. Exp. Res. 24, 403–404 (2000).
Munaka, M. et al. Genetic polymorphisms of tobacco- and alcohol-related metabolizing enzymes and the risk of hepatocellular carcinoma. J. Cancer Res. Clin. Oncol. 129, 355–360 (2003).
Pimpin, L. et al. Burden of liver disease in Europe: epidemiology and analysis of risk factors to identify prevention policies. J. Hepatol. 69, 718–735 (2018).
Sheron, N. Alcohol and liver disease in Europe — simple measures have the potential to prevent tens of thousands of premature deaths. J. Hepatol. 64, 957–967 (2016).
Anderson, P. et al. Improving the delivery of brief interventions for heavy drinking in primary health care: outcome results of the optimizing delivery of health care intervention (ODHIN) five-country cluster randomized factorial trial. Addiction 111, 1935–1945 (2016).
Manns, M. P., Burra, P., Sargent, J., Horton, R. & Karlsen, T. H. The Lancet-EASL commission on liver diseases in Europe: overcoming unmet needs, stigma, and inequities. Lancet 392, 621–622 (2018).
El-Zayadi, A. R., Selim, O., Hamdy, H., El-Tawil, A. & Moustafa, H. Heavy cigarette smoking induces hypoxic polycythemia (erythrocytosis) and hyperuricemia in chronic hepatitis C patients with reversal of clinical symptoms and laboratory parameters with therapeutic phlebotomy. Am. J. Gastroenterol. 97, 1264–1265 (2002).
Wang, L. Y. et al. 4-Aminobiphenyl DNA damage in liver tissue of hepatocellular carcinoma patients and controls. Am. J. Epidemiol. 147, 315–323 (1998).
Dam, M. K., Flensborg-Madsen, T., Eliasen, M., Becker, U. & Tolstrup, J. S. Smoking and risk of liver cirrhosis: a population-based cohort study. Scand. J. Gastroenterol. 48, 585–591 (2013).
Klatsky, A. L. & Armstrong, M. A. Alcohol, smoking, coffee, and cirrhosis. Am. J. Epidemiol. 136, 1248–1257 (1992).
Petrick, J. L. et al. Tobacco, alcohol use and risk of hepatocellular carcinoma and intrahepatic cholangiocarcinoma: the Liver Cancer Pooling Project. Br. J. Cancer 118, 1005–1012 (2018).
Abdel-Rahman, O. et al. Cigarette smoking as a risk factor for the development of and mortality from hepatocellular carcinoma: an updated systematic review of 81 epidemiological studies. J. Evidence-Based Med. 10, 245–254 (2017).
Kuper, H. et al. Tobacco smoking, alcohol consumption and their interaction in the causation of hepatocellular carcinoma. Int. J. Cancer 85, 498–502 (2000).
Jee, S. H., Ohrr, H., Sull, J. W. & Samet, J. M. Cigarette smoking, alcohol drinking, hepatitis B, and risk for hepatocellular carcinoma in Korea. J. Natl Cancer Inst. 96, 1851–1856 (2004).
Buch, S. et al. A genome-wide association study confirms PNPLA3 and identifies TM6SF2 and MBOAT7 as risk loci for alcohol-related cirrhosis. Nat. Genet. 47, 1443–1448 (2015).
Salameh, H. et al. PNPLA3 gene polymorphism is associated with predisposition to and severity of alcoholic liver disease. Am. J. Gastroenterol. 110, 846–856 (2015).
Romeo, S. et al. Genetic variation in PNPLA3 confers susceptibility to nonalcoholic fatty liver disease. Nat. Genet. 40, 1461–1465 (2008).
Schwantes-An, T. H. et al. Genome-wide association study and meta-analysis on alcohol-related liver cirrhosis identifies novel genetic risk factors. Hepatology https://doi.org/10.1002/hep.31535 (2020).
Trépo, E. et al. Common genetic variation in alcohol-related hepatocellular carcinoma: a case-control genome-wide association study. Lancet Oncol. https://doi.org/10.1016/s1470-2045(21)00603-3 (2021).
Schulze, K. et al. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat. Genet. 47, 505–511 (2015).
Nahon, P. & Nault, J. C. Constitutional and functional genetics of human alcohol-related hepatocellular carcinoma. Liver Int. 37, 1591–1601 (2017).
Bajaj, J. S. Alcohol, liver disease and the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 16, 235–246 (2019).
Dubinkina, V. B. et al. Links of gut microbiota composition with alcohol dependence syndrome and alcoholic liver disease. Microbiome 5, 141 (2017).
Leclercq, S. et al. Intestinal permeability, gut-bacterial dysbiosis, and behavioral markers of alcohol-dependence severity. Proc. Natl Acad. Sci. USA 111, E4485–E4493 (2014).
Llopis, M. et al. Intestinal microbiota contributes to individual susceptibility to alcoholic liver disease. Gut 65, 830–839 (2016).
Hartmann, P. et al. Modulation of the intestinal bile acid/farnesoid X receptor/fibroblast growth factor 15 axis improves alcoholic liver disease in mice. Hepatology 67, 2150–2166 (2018).
Saunders, J. B., Aasland, O. G., Babor, T. F., de la Fuente, J. R. & Grant, M. Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption-II. Addiction 88, 791–804 (1993).
Bajaj, J. S. et al. Continued alcohol misuse in human cirrhosis is associated with an impaired gut-liver axis. Alcohol. Clin. Exp. Res. 41, 1857–1865 (2017).
Sarin, S. K., Pande, A. & Schnabl, B. Microbiome as a therapeutic target in alcohol-related liver disease. J. Hepatol. 70, 260–272 (2019).
Ren, Z. et al. Gut microbiome analysis as a tool towards targeted non-invasive biomarkers for early hepatocellular carcinoma. Gut 68, 1014–1023 (2019).
Dapito, D. H. et al. Promotion of hepatocellular carcinoma by the intestinal microbiota and TLR4. Cancer Cell 21, 504–516 (2012).
Schwabe, R. F. & Greten, T. F. Gut microbiome in HCC — mechanisms, diagnosis and therapy. J. Hepatol. 72, 230–238 (2020).
GHDx GBD Results Tool. Global Burden of Disease Study https://ghdx.healthdata.org/gbd-results-tool (2019).
Sharma, S. A. et al. Toronto HCC risk index: a validated scoring system to predict 10-year risk of HCC in patients with cirrhosis. J. Hepatol. https://doi.org/10.1016/j.jhep.2017.07.033 (2017).
Toshikuni, N. et al. Comparison of outcomes between patients with alcoholic cirrhosis and those with hepatitis C virus-related cirrhosis. J. Gastroenterol. Hepatol. 24, 1276–1283 (2009).
Kodama, K., Tokushige, K., Hashimoto, E., Taniai, M. & Shiratori, K. Hepatic and extrahepatic malignancies in cirrhosis caused by nonalcoholic steatohepatitis and alcoholic liver disease. Alcohol. Clin. Exp. Res. 37 (suppl. 1), E247–E252 (2013).
The SURF Report 2. WHO https://apps.who.int/iris/bitstream/handle/10665/43190/9241593024_eng.pdf (2005).
Lieber, C. S., Rubin, E. & DeCarli, L. M. Hepatic microsomal ethanol oxidizing system (MEOS): differentiation from alcohol dehydrogenase and NADPH oxidase. Biochem. Biophys. Res. Commun. 40, 858–865 (1970).
Seitz, H. K. & Stickel, F. Molecular mechanisms of alcohol-mediated carcinogenesis. Nat. Rev. Cancer 7, 599–612 (2007).
Linhart, K., Bartsch, H. & Seitz, H. K. The role of reactive oxygen species (ROS) and cytochrome P-450 2E1 in the generation of carcinogenic etheno-DNA adducts. Redox Biol. 3, 56–62 (2014).
Chiba, T., Marusawa, H. & Ushijima, T. Inflammation-associated cancer development in digestive organs: mechanisms and roles for genetic and epigenetic modulation. Gastroenterology 143, 550–563 (2012).
Callinan, S. & Livingston, M. Increases in alcohol consumption in middle-income countries will lead to increased harms. Lancet 393, 2471–2472 (2019).
COVID-19 and increased alcohol consumption: NANOS poll summary report. Canadian Centre on Substance Use and Addiction (CCSA) https://www.ccsa.ca/covid-19-and-increased-alcohol-consumption-nanos-poll-summary-report (2020).
Vanderbruggen, N. et al. Self-reported alcohol, tobacco, and cannabis use during COVID-19 lockdown measures: results from a web-based survey. Eur. Addict. Res. 26, 309–315 (2020).
Sidor, A. & Rzymski, P. Dietary choices and habits during COVID-19 lockdown: experience from Poland. Nutrients https://doi.org/10.3390/nu12061657 (2020).
Kim, J. U. et al. Effect of COVID-19 lockdown on alcohol consumption in patients with pre-existing alcohol use disorder. Lancet Gastroenterol. Hepatol. 5, 886–887 (2020).
Mahmud, N., Hubbard, R. A., Kaplan, D. E. & Serper, M. Declining cirrhosis hospitalizations in the wake of the COVID-19 pandemic: a national cohort study. Gastroenterology 159, 1134–1136.e1133 (2020).
White, A. M., Castle, I.-J. P., Powell, P. A., Hingson, R. W. & Koob, G. F. Alcohol-related deaths during the COVID-19 pandemic. JAMA https://doi.org/10.1001/jama.2022.4308 (2022).
Marjot, T. et al. COVID-19 and liver disease: mechanistic and clinical perspectives. Nat. Rev. Gastroenterol. Hepatol. 18, 348–364 (2021).
Acknowledgements
R.L. receives funding support from the NIAAA (U01AA029019), the NIEHS (5P42ES010337), the NCATS (5UL1TR001442), the NIDDK (U01DK130190, U01DK061734, R01DK106419, P30DK120515, R01DK121378 and R01DK124318), the NHLBI (P01HL147835) and the DOD PRCRP (W81XWH-18-2-0026). D.Q.H. receives funding support from Singapore’s Ministry of Health’s National Medical Research Council under its NMRC Research Training Fellowship (MOH-000595-01). P.M. receives funding support from the Programme Hospitalier de Recherche Clinique (French Minister for Health). H.C.-P. receives funding support from the FCT: Projectos De Investigação Científica E Desenvolvimento, Portugal.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to all aspects of the manuscript.
Corresponding author
Ethics declarations
Competing interests
R.L. serves as a consultant or advisory board member for Anylam/Regeneron, Arrowhead Pharmaceuticals, AstraZeneca, Bristol-Myers Squibb, CohBar, Eli Lilly, Galmed, Gilead Sciences, Glympse Bio, Inipharm, Intercept, Ionis, Janssen, Merck, Metacrine, NGM Biopharmaceuticals, Novartis, Novo Nordisk, Pfizer, Promethera, Sagimet, 89bio and Viking Therapeutics. In addition, his institution has received grant support from Allergan, Boehringer-Ingelheim, Bristol-Myers Squibb, Cirius, Eli Lilly, Galectin Therapeutics, Galmed Pharmaceuticals, Genfit, Gilead, Intercept, Inventiva, Janssen, Madrigal Pharmaceuticals, Merck, NGM Biopharmaceuticals, Pfizer, pH Pharma and Siemens. He is also co-founder of Liponexus. D.Q.H. serves as an advisory board member for Eisai. P.M. serves as a consultant or advisory member for Ipsen, Eisai, Abbvie, Sanofi, Gilead Sciences, Evive Biotech, Novo Nordisk, Bayer Healthcare, Intercept, Surrozen and Pfizer. H.C.-P. lectures and receives advisory board fees from Intercept, Genfit, Promethera Bioscience, Orphalan, Novo Nordisk and Roche Portugal.
Peer review
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks Ramon Bataller, Shiv Sarin and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Review criteria
PubMed was searched using the terms ‘alcoholic liver disease’, ‘alcohol-associated cirrhosis’, ‘alcoholic cirrhosis’, ‘alcohol-related liver disease’, ‘alcohol-associated hepatocellular carcinoma’ and ‘alcohol-associated liver cancer’ without language restrictions. Guidelines, original articles and reviews were evaluated. The literature search was performed in February 2021.
Glossary
- Heavy alcohol consumption
-
The consumption of >40 g of pure alcohol per day over a sustained period of time.
- Current alcohol drinkers
-
Individuals who have consumed alcoholic beverages in the previous 12-month period.
- Age-standardized death rate
-
(ASDR). A weighted average of the age-specific death rates, where the weights are the proportions of a standard population in the corresponding age groups.
- Alcohol-associated
-
A disease state that is attributed to heavy consumption of alcohol.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, D.Q., Mathurin, P., Cortez-Pinto, H. et al. Global epidemiology of alcohol-associated cirrhosis and HCC: trends, projections and risk factors. Nat Rev Gastroenterol Hepatol 20, 37–49 (2023). https://doi.org/10.1038/s41575-022-00688-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-022-00688-6
This article is cited by
-
Global burden of liver cirrhosis and other chronic liver diseases caused by specific etiologies from 1990 to 2019
BMC Public Health (2024)
-
The association between triglyceride-glucose index and its combination with obesity indicators and cardiovascular disease: NHANES 2003–2018
Cardiovascular Diabetology (2024)
-
Fluorescence/electrochemical dual-mode strategy for Golgi protein 73 detection based on molybdenum disulfide/ferrocene/palladium nanoparticles and nitrogen-doped graphene quantum dots
Microchimica Acta (2024)
-
Addiction and Liver Disease: Exploring the Complex Relationship and Implications for Clinical Management
Current Hepatology Reports (2024)
-
TRIM5 as a promising diagnostic biomarker of hepatocellular carcinoma: integrated analysis and experimental validation
Functional & Integrative Genomics (2024)