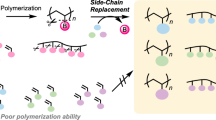
Abstract
Although vinyl ketones (VKs) exhibit excellent reactivity toward radicals and nucleophiles, their application in polymer chemistry has been limited compared with that of acrylates. One of the reasons is the difficulty of the synthesis, particularly that of multivalent VKs. Herein, we report the facile synthesis of divalent VKs via Friedel‒Crafts acylation of fluorene and subsequent elimination reactions. For example, via this approach, 2,7-diacryloyl-9,9-dimethylfluorene was obtained at high yields (78%). Because the monomer was obtained at high purity through recrystallization and washing, the procedure is suitable for industrial applications. The addition of dithiols and diamines via thia- and aza-Michael addition afforded the corresponding polythioesters and polyamines, respectively. In addition, the divalent VKs exhibited high reactivity in the Morita‒Baylis‒Hillman reaction with formaldehyde, affording a diol monomer. The polycondensation of the diol monomer and isophthaloyl dichloride yielded a poly(conjugated-ketone ester). Consequently, the divalent VKs described herein are attractive monomers and monomer precursors with ready accessibility and sufficient electrophilicity.
Similar content being viewed by others
Introduction
Since vinyl ketones (VKs) are more electrophilic than acrylates are [1], they behave as excellent substrates for nucleophilic conjugate addition (Michael addition) reactions [2]. Owing to this property, VKs have been employed not only in organic synthesis [3] but also in polymer chemistry. In particular, VKs have been recognized as highly reactive monomers in anionic polymerization reactions, which can be initiated by weak nucleophiles such as alkali alkoxides [4] and trialkylaluminum [5]. VKs can also be polymerized as typical vinyl monomers via the use of a radical initiator. Notably, the resulting polymers exhibit a glass transition temperature (Tg) higher than that of the corresponding polyacrylate; the Tg of poly(phenyl acrylate) is 57 °C [6], whereas that of poly(phenyl vinyl ketone) is 74 °C [6]. The high Tg is attributed to the high rotational barrier of the phenyl pendants directly linked to the backbone.
Despite their excellent monomer reactivity and polymer properties, the synthesis of multivalent VKs remains challenging. One of few examples involves a divalent VK prepared through the reaction of a Weinreb amide and a vinyl Grignard reagent [3], and the Stille coupling of an acyl chloride also affords divalent VKs [7]. Although Friedel–Crafts (FC) acylation and the subsequent elimination reaction constitute a general synthetic strategy for preparing VKs, this approach is not effective for the synthesis of divalent VKs if the FC reaction progresses with insufficient selectivity and incomplete conversion; for example, the FC reaction of thianthrene results in a mixture of mono- and diacylated products [8]. Therefore, divalent VKs have not been applied in industry, as far as we know, which is in sharp contrast to multivalent acrylates, which are easily prepared by the condensation reaction of acryloyl chloride with polyols and can be applied as a curing agent [9, 10]. Therefore, the purpose of this research is to provide an efficient synthetic procedure for divalent VKs.
As mentioned above, the key reaction for preparing divalent VKs is FC acylation, which requires quantitative conversion of the reactants to a disubstituted product with high regioselectivity. Fluorene (1a) meets this requirement and can undergo FC acylation twice at the 2- and 7-positions selectively. In addition, the solubility of the products can be tuned by substitution at the 9-position [11, 12]. As these features are suitable for the synthesis of divalent VKs, VK monomers were prepared by FC acylation followed by the E1cB reaction (Scheme 1). Herein, we report the synthesis of divalent VKs containing fluorene skeletons and their reactivity in polymerization reactions.
Results and discussion
Synthesis of divalent monomers
FC acylation of 1a was conducted with 2.2 equimolar 3-chloropropyonyl chloride using aluminum trichloride as a catalyst (yield: 18.3%). The solubility of the products in common organic solvents, such as CHCl3, toluene, and EtOAc, was poor, leading to a low yield after purification. The subsequent E1cB reaction with triethylamine (Et3N) yielded 3a in 45% yield. To improve the yield in the first step, 9,9-dimethylfluorene (1b) was examined because of its high solubility. As expected, the FC acylation of 1b afforded 2b in an almost quantitative yield (93.3%). The subsequent E1cB reaction resulted in 3b (83.1%). The structure of 3b was determined via 1H (Fig. 1A) and 13C (Fig. S7) spectra. Note that the purification in each step only involved washing, which enabled the scale-up of the synthesis; 3b (42.5 g) was obtained from 1b (35.9 g). Similarly, the FC reaction of 1b with 2-bromopropionyl bromide followed by elimination afforded 5b (total yield: 34.7%).
1H NMR spectra of monomer 3b (A) and polymer P1 (B) (400 MHz, 25 °C, CDCl3). The full-scale spectra are shown in Fig. S13
Polyaddition of divalent VKs with dithiols and diamines
The nucleophilic thiol-ene reaction involves typical click chemistry that proceeds quantitatively under ambient conditions without any side reactions [13, 14]. Therefore, the polyaddition reaction of 3a and 1,10-decandithiol (DT1) was carried out in the presence of a catalytic quantity of tributylphosphine (Bu3P) in tetrahydrofuran (THF) (Scheme 2, Table 1, Entry 1). DT1 was chosen as an aliphatic dithiol with a sufficiently long distance between the two mercapto groups to avoid neighboring effects. The 1H NMR signals of the vinyl protons, observed at 7.24, 6.49, and 5.96 ppm in the spectrum of 3a (Fig. S5), disappeared after polymerization, whereas the signals of the methylene protons formed by the thiol-ene reaction clearly appeared at 3.26 and 2.51 ppm in the 1H NMR spectrum of P0 (Fig. S13). Similarly, the polyaddition reaction of 3b and DT1 resulted in P1 (Entry 2), confirmed by the disappearance of the 1H NMR signals of the vinyl protons at 7.24, 6.48, and 5.98 ppm and the appearance of methylene signals at 3.33 and 2.95 ppm (Fig. 1B). The polyaddition reactions of 4,4′-thiobisbenzenethiol (DT2), an aromatic dithiol, and ethylene glycol bis(3-mercaptpropyonate) (DT3), an ester-containing dithiol, also yielded the corresponding polymers (Entries 3 and 4). Similarly, 5b afforded the desired polymers via polyaddition reaction with dithiols (Entries 5–7). These results indicated that 3b and 5b functioned as divalent monomers. The chemical structures of the obtained polymers were determined via 1H NMR spectroscopy (Figs. S13–18).
An advantage of vinyl ketones over acrylates is their efficient reaction with amines. Thus, polymerization with divalent secondary amines via aza-Michael addition was investigated (Entries 8–10). The polyaddition reaction of 3b and 1,4-piperadine (DA1) afforded a product insoluble in toluene, THF, CHCl3, and acetone (Entry 8). To examine whether the insolubility was caused by cross-linking via side reactions such as enamine formation or poor solubility, a model reaction using DA1 and 2-acryloyl-9,9-dimethylfluorene (9), ‘half’ of 3b, was employed under similar conditions for polymerization (Scheme S1). The resulting product was an aza-Michael adduct 10, and side reactions, such as enamine formation, were not observed in the 1H and 13C NMR spectra (Figs. S25 and 26). On the other hand, the solubility of the product was poor, and precipitation was observed during the reaction in THF. These results suggest that the insolubility of the product in Entry 8 was caused not by crosslinking due to side reactions but by the poor solubility of the resulting polymers. The polyadditions of 3a (Entry 9) and 3b (Entry 10) with N,Nʹ-dimetyl-1,6-hexanediamine (DA2) resulted in polymers that were soluble in THF. The SEC curves of the obtained polymers exhibited a low molar mass (Mn,SEC), although the 1H NMR spectra (Figs. S19 and S20) indicated the quantitative consumption of vinyl groups and sharp NMR signals assignable to each expected polymer structure. It has been reported that polyamines interact with the stationary phase of SEC columns, making the exact evaluation of their molar mass difficult [15]. For such polymers, 1H DOSY NMR is known to be effective in evaluating the weight-average molar mass (Mw,DOSY) [16]. The Mw,DOSY values correlated with polystyrene standards in CHCl3 were 39800 and 8300 g·mol−1 for P8 and P9, respectively, which were higher than those obtained by SEC analysis (Table S1 and S2, and Fig. S27). Therefore, polymerization via the aza-Michael reaction was as effective as that via thia-Michael addition.
Except for Entry 8, the obtained polymers were readily soluble in THF and CHCl3. Moreover, the polymer obtained from 5b showed excellent solubility in other solvents, such as toluene and acetone (Entries 5–7), indicating its high solubility. The 5% weight loss temperatures (Td5) of the polymers, except those in Entries 3, 9 and 10, were above 250 °C (Figs. S28–S36). The Td5 of P2 (Entry 3) was 105 °C, which was much lower than those of the other polymers. TG-DTA analysis was repeated several times with to obtain similar results. Therefore, the low Td5 was not a technical error. The TG curves (Fig. S30) revealed degradation in three steps, and a 14% weight loss corresponding to benzene liberation was observed in the first step below 150 °C (molecular weight; benzene: 78.11, P2: 552.77. Proportion of the molecular weight of benzene in the polymer: 14%). On the other hand, P3, synthesized via polyaddition with DT3 in the same way as P2, did not decompose at that temperature. Therefore, the 1,4-dithiaphenylene skeletons of the DT2 moieties might specifically lead to thermal degradation in this step, although further investigations using TG-GC/MS, for example, are needed to draw concrete conclusions. P3, P4, P6, and P8 presented Tg values of 43, 17, 30, and 30 °C, respectively (Figs. S39–44).
Modification with methylol groups for polycondensation
Polyesters containing α,β-unsaturated carbonyl skeletons, such as acrylates and VKs, have attracted attention as polymers carrying reactive sites on the backbone [17,18,19,20,21,22,23]. To prepare such polyesters, the Morita‒Baylis‒Hillman (MBH) reaction is often applied both in monomer synthesis and polymerization reactions [18, 24,25,26]. MBH reactions are useful for incorporating a methylol group at the α-position of the acrylic compound [27, 28]. In general, however, the MBH reaction of acrylates is slow and competitive with some side reactions, such as dimerization of the product [24]. For this reason, a large excess of a catalyst or reagent (an aldehyde) is required in the MBH reaction of diacrylate to promote the reaction and to avoid side reactions as much as possible. In contrast, the high electrophilicity of VKs appears to be suitable for the MBH reaction. Therefore, the MBH reaction of 3b with formaldehyde was investigated (Scheme 3). Surprisingly, the reaction was completed within 35 min, confirming the high reactivity of the VKs. After purification by washing with CHCl3, 6b was obtained with 50.9% yield, confirmed by 1H NMR spectrum (Fig. 2A).
6b, which has two methylol groups on the VK skeleton, was polymerized with isophthaloyl dichloride (7) (Table 2). A solution of 7 was added dropwise to a solution of 6b and Et3N. The reaction was performed in dichloromethane (CH2Cl2), THF, and DMF (Entries 1–3) and resulted in the expected polyesters, confirmed by 1H NMR spectrum (Fig. 2B). Notably, Et3N reportedly acts as a nucleophile to cause main-chain scission in similar polyesters; Lu et al. explained that the main-chain scission occurred by acyl substitution [17], whereas conjugate substitution was also a possible mechanism [22]. Moreover, Et3N may cause main-chain scission and may not be a suitable base for polycondensation reactions. Therefore, a similar polycondensation reaction was employed using EtNiPr2, a base with low nucleophilicity (Entry 4). The resulting molar mass was greater than that in Entry 1, although the difference was not significant. In addition, polycondensation reactions were carried out using an excess (4.0 equimolar to 7) of Et3N (Entry 5). The molar mass of the resulting product decreased slightly compared with that in Entry 2. Because the differences in the resulting molar masses in these entries were small, the influence of main-chain scission by Et3N seems insignificant in this polymerization system.
The thermal properties of the polymers obtained were also investigated. Td5 was determined to be 91 °C by TG-DTA, and no exothermic peaks assignable to the curing point were observed (Fig. S37). The low thermal stability of the molecule despite the rigid aromatic skeleton was somewhat surprising; notably, monomer 6b also exhibited low thermal stability (Fig. S38). Thus, the low stability was attributed to the VK skeletons.
Conclusion
Divalent VKs were synthesized via FC acylation and elimination reactions. 1b was effective for quantitative and regioselective FC acylation, providing products with good solubility. In particular, the synthesis of 3b did not require complicated purification processes, such as column chromatography; therefore, the preparation process is suitable for industrial production. As expected, the divalent VKs exhibited high reactivity not only in thia-/aza-Michael addition polymerization to polysulfides/polyamines but also in the MBH reaction to yield diol monomers with VK skeletons. Despite the high reactivity of VKs, the polymer chemistry of divalent VKs has not been well established because of the difficulty in monomer synthesis. As this technical issue is now resolved, we believe that research on divalent VKs will advance to the next stage.
References
Allgäuer DS, Jangra H, Asahara H, Li Z, Chen Q, Zipse H, et al. Quantification and theoretical analysis of the electrophilicities of Michael acceptors. J Am Chem Soc. 2017;139:13318–29. https://doi.org/10.1021/jacs.7b05106
Mather B, Viswanathan K, Miller K, Long T. Michael addition reactions in macromolecular design for emerging technologies. Prog Polym Sci. 2006;31:487–531. https://doi.org/10.1016/j.progpolymsci.2006.03.001
Goldup S, Leigh D, Lusby P, McBurney R, Slawin A. Active template synthesis of rotaxanes and molecular shuttles with switchable dynamics by four-component Pd-II-promoted Michael additions. Angew Chem Int Ed. 2008;47:3381–4. https://doi.org/10.1002/anie.200705859
Thomas PR, Tyler GJ, Edwards TE, Radcliffe AT, Cubbon RCP. The anionic polymerization of some alkyl vinyl ketones. Polymer. 1964;5:525–31. https://doi.org/10.1016/0032-3861(64)90201-0
Tsuruta T, Fujio R, Furukawa J. Anionic polymerization of vinyl ketones. Die Makromol. Chem. 1964;80:172–84. https://doi.org/10.1002/macp.1964.020800115
Pizzirani G, Magagnini P, Giusti P. Glass transition temperatures of some linear polymers containing phenyl side groups. J. Polym. Sci. Part A-2: Polym. Phys. 1971;9:1133–45. https://doi.org/10.1002/pol.1971.160090609
Lee CW, Choi TL, Grubbs RH. Ring expansion via olefin metathesis. J. Am. Chem. Soc. 2002;124:3224–5. https://doi.org/10.1021/ja017790c
Hatakeyama-Sato K, Masui T, Serikawa T, Sasaki Y, Choi W, Doo S-G, et al. Nonconjugated redox-active polymer mediators for rapid electrocatalytic charging of lithium metal oxides. ACS Appl Energy Mater. 2019;2:6375–82. https://doi.org/10.1021/acsaem.9b01007
Zhang Y, Wang R, Hua Y, Baumgartner R, Cheng J. Trigger-responsive poly(β-amino ester) hydrogels. ACS Macro Lett. 2014;3:693–7. https://doi.org/10.1021/mz500277j
Zhu J, Zhang Q, Yang T, Liu Y, Liu R. 3D printing of multi-scalable structures via high penetration near-infrared photopolymerization. Nat Commun. 2020;11:3462 https://doi.org/10.1038/s41467-020-17251-z
Neher D. Polyfluorene homopolymers: conjugated liquid-crystalline polymers for bright blue emission and polarized electroluminescence. Macromol Rapid Commun. 2001;22:1365–85. https://doi.org/10.1002/1521-3927(20011101)22:17<1365::AID-MARC1365>3.0.CO;2-B.
Li L, He F, Wang X, Ma N, Li L. Reversible ph-responsive fluorescence of water-soluble polyfluorenes and their application in metal ion detection. ACS Appl Mater Inter. 2012;4:4927–33. https://doi.org/10.1021/am301286q
Lowe AB. Thiol-ene “click” reactions and recent applications in polymer and materials synthesis. Polym Chem. 2010;1:17–36. https://doi.org/10.1039/B9PY00216B
Lowe AB. Thiol–ene “click” reactions and recent applications in polymer and materials synthesis: a first update. Polym Chem. 2014;5:4820–70. https://doi.org/10.1039/C4PY00339J
Rieger E, Manhart A, Wurm FR. Multihydroxy polyamines by living anionic polymerization of aziridines. ACS Macro Lett. 2016;5:195–8. https://doi.org/10.1021/acsmacrolett.5b00901
Li W, Chung H, Daeffler C, Johnson JA, Grubbs RH. Application of 1H DOSY for facile measurement of polymer molecular weights. Macromolecules. 2012;45:9595–603. https://doi.org/10.1021/ma301666x
Xu YC, Ren W-M, Zhou H, Gu G-G, Lu X-B. Functionalized polyesters with tunable degradability prepared by controlled ring-opening (co)polymerization of lactones. Macromolecules. 2017;50:3131–42. https://doi.org/10.1021/acs.macromol.7b00239
Ji S, Bruchmann B, Klok H-A. Synthesis of side-chain functional polyesters via baylis–hillman polymerization. Macromolecules. 2011;44:5218–26. https://doi.org/10.1021/ma2006238
Kohsaka Y. Conjugate substitution reaction of α-(substituted methyl)acrylates in polymer chemistry. Polym J. 2020;52:1175–83. https://doi.org/10.1038/s41428-020-0376-z
Kohsaka Y, Yamashita M, Matsuhashi Y, Yamashita S. Synthesis of poly(conjugated ester)s by ring-opening polymerization of cyclic hemiacetal ester bearing acryl skeleton. Eur Polym J. 2019;120:109185 https://doi.org/10.1016/j.eurpolymj.2019.08.012
Kohsaka Y, Nagai K. Controls and effects of monomer junctions and sequences in curable and degradable polyarylate containing acrylate moieties. Macromol Rapid Commun. 2021;42:2000570 https://doi.org/10.1002/marc.202000570
Robert T, Friebel S. Itaconic acid – a versatile building block for renewable polyesters with enhanced functionality. Green Chem. 2016;18:2922–34. https://doi.org/10.1039/C6GC00605A
Noda T, Kitagawa T, Kohsaka Y. Degradation of poly(conjugated ester)s using a conjugate substitution reaction with various amines and amino acids in aqueous media. Polym J. 2024;26:343–351. https://doi.org/10.1038/s41428-023-00859-5
Mathias LJ, Kusefoglu SH. New difunctional methacrylate ethers and acetals: readily available derivatives of .alpha.-hydroxymethyl acrylates. Macromolecules. 1987;20:2039–41. https://doi.org/10.1021/ma00174a065
Ji S, Bruchmann B, Klok HA. Exploring the scope of the Baylis–Hillman reaction for the synthesis of side-chain functional polyesters. Macromol. Chem. Phys. 2011;212:2612–8. https://doi.org/10.1002/macp.201100482
Günay KA, Theato P, Klok HA. Standing on the shoulders of Hermann Staudinger: Post-polymerization modification from past to present. J Polym Sci Part A: Polym Chem. 2013;51:1–28. https://doi.org/10.1002/pola.26333
Basavaiah D, Rao AJ, Satyanarayana T. Recent advances in the Baylis−Hillman reaction and applications. Chem. Rev. 2003;103:811–92. https://doi.org/10.1021/cr010043d
Wang T, Zhang B-S, Liu J-J, Liu X-J, Wang X-C, Quan Z-J. Visible light promoted polyhalomethylation of alkenes: alkylation and cyclization. Org. Che. Front. 2022;9:1004–9. https://doi.org/10.1039/D1QO01662H
Acknowledgements
This research was supported by JST, the establishment of university fellowships towards the creation of science and technology innovation (Grant Number JPMJF2118). The isophthaloyl dichloride was a gift from Iharanikkei Chemical Industry Co. Ltd.
Funding
Open Access funding partially provided by Shinshu University.
Author information
Authors and Affiliations
Contributions
MO performed all the experiments and drafted the manuscript. RY examined the reproducibility of the monomer synthesis. SM, HK, and YK supervised this study. YK planned and directed the project, designed all the molecules, and refined the draft prepared by MO. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Rie Yasuda and Shinsuke Miyauchi reported a relationship with Osaka Gas Chemicals Co., Ltd. that included employment. Masatoshi Ohyama, Rie Yasuda, Shinsuke Miyauchi, and Yasuhiro Kohsaka have patents #JP, 2021-127317,A issued to Shinshu University and Osaka Gas Chemicals Co., Ltd.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ohyama, M., Yasuda, R., Miyauchi, S. et al. Divalent vinyl ketones derived from fluorene: a facile synthesis of bifunctional acrylic monomers with high reactivity in thia-/aza-Michael addition and Morita-Baylis-Hillman reactions. Polym J (2024). https://doi.org/10.1038/s41428-024-00939-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41428-024-00939-0