Abstract
Background
Although it is commonly accepted that upper airway obstruction (UAO) increases gastroesophageal reflux (GER), the link is poorly understood and insufficiently documented. In addition, while hypoxia is often encountered in infants with UAO, its consequences on GER are virtually unknown. The two aims of the present study were to characterize the effects of (1) UAO and (2) hypoxia on GER.
Methods
Seventeen lambs underwent polysomnographic and esophageal impedance/pH-metry monitoring during UAO vs. a control condition (6 h, ten lambs) or 10% hypoxia vs. normoxic condition (3 h, seven other lambs).
Results
Moderate-to-severe UAO was maintained throughout monitoring (inspiratory tracheal pressure of −13 (−15, −12) cm H2O vs. −1 (−1, −1) cm H2O in control condition, p = 0.005). While the number of GERs increased with UAO (2 (1, 4) vs. 0 (0, 3) in the control condition, p = 0.03), the increase was less than anticipated and inconsistent among the lambs. Also, sustained 10% hypoxia did not alter the number of GERs (2 (1, 3) vs. 0 (0, 5) in the control condition, p = 0.9).
Conclusions
The presence of an UAO for 6 h mildly increased the number of GERs, whereas hypoxia for 3 h had no significant effect.
Impact
-
The effect of upper airway obstruction and hypoxia on gastroesophageal reflux is poorly documented in the neonatal period.
-
A moderate-to-severe upper airway obstruction for 6 h results in a mild, inconsistent increase in the number of gastroesophageal refluxes.
-
Overall, a hypoxia of 10% for 3 h had no significant impact on gastroesophageal reflux.
-
The prescription of an antireflux medication in infants with upper airway obstruction must not be systematic but rely on objective signs of a pathologic gastroesophageal reflux.
Similar content being viewed by others
Introduction
A number of chronic conditions in children and infants—such as maxillofacial malformations, adenotonsillar hypertrophy, laryngomalacia, hypotonia, and various syndromes—can cause significant upper airway obstruction (UAO). While gastroesophageal reflux (GER) has been linked to UAO in clinical practice,1,2,3 it is still unclear whether UAO induces GER or if GER worsens UAO.1,2,4 Importantly, persistent GER can lead to significant problems, such as esophagitis, chronic lung aspiration, and recurrent pneumonia. In addition, it can contribute to brief resolved unexpected events5 as well as aggravate UAO consequently to laryngopharyngeal reflux. Hence, antireflux medications are often given to infants and children presenting with UAO, even without evidence of GER, while ignoring the potential for deleterious adverse events of such medications.6
To our knowledge, only two animal studies have yielded some evidence that UAO can be responsible for GER. A tripling of the thoracoabdominal pressure gradient was observed following the induction of a partial airway obstruction in anesthetized adult rats.7 Acid GERs were triggered in adult dogs following 1 week of partial tracheal obstruction.8 No results are available on early life, although it is well known that esophageal immaturity is responsible for frequent GER in virtually all infants.9 We have recently shown that the newborn lamb during its first week of life is a relevant model to study neonatal GER.10 The first aim of the present study was therefore to assess the effects of UAO on GER in lambs.
Hypoxia is another condition frequently encountered in preterm infants with chronic lung disease, as well as in full-term infants and children, yet its effect on GER remains virtually unknown. To our knowledge, the only study documenting the relationship between hypoxia and GER revealed that hypoxia induces a significant increase in lower esophageal sphincter pressure in piglets, suggesting that hypoxia is protective against GER.11 Hence, the second aim of this study was to characterize the effect of a sustained short-term hypoxic exposure on GER in lambs.
Materials and methods
The study was carried out in accordance with the recommendations of the Canadian Council on Animal Care. The protocol was approved by the Ethics Committee for Animal Care and Experimentation of the Université de Sherbrooke (protocol #283-11). A total of 17 full-term, mixed-bred male lambs, aged 4–5 days were included in the study (n = 10 for the UAO group; n = 7 for the hypoxia group).
Chronic instrumentation and recording equipment
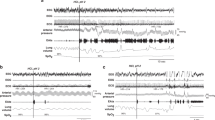
Chronic surgical instrumentation was performed under general anesthesia (2% isoflurane) to insert custom-built catheters (i) into the left carotid artery for blood-gas measurement and (ii) transcutaneously into the trachea to monitor pressure variations.12 Following a postoperative recovery period of 48 h, noninvasive instrumentation was added immediately before recordings started and included (i) subcutaneous needle electrodes (S43-638 Technomed subdermal needle electrode, CA) for electrocardiogram (ECG) recordings; (ii) elastic bands around the chest and abdomen to monitor lung-volume variations semiquantitatively via respiratory inductance plethysmography; (iii) a pulse oximetry probe placed at the base of the tail for continuous monitoring of hemoglobin oxygen saturation (SpO2; Masimo, Montreal, Canada) and (v) a naso-esophageal multichannel intraluminal impedance/pH-metry (MII-pH) catheter (six French antimony impedance-pH catheters, Laborie, Mississauga, Canada) to record GERs (Greenfield, Medical Measurement Systems, Dover, NH) connected to an ambulatory recorder using Bluetooth wireless transmission (Ohmega, Medical Measurement Systems, Dover, NH).10
The lambs were comfortably positioned in a sling with loose restraints. Physiological signals were transmitted wirelessly via our custom-made radiotelemetry system13 and continuously recorded on a PC using AcqKnowledge software (version 4.1, Biopac Systems, Montreal, Canada). The entire recording was video recorded and an experimenter was present throughout the recordings to note all relevant events.
Design of the study
Upper airway obstruction experiment
Two polysomnographic recordings were performed in nonsedated lambs, 1 h after bottle-feeding (15 mL/kg of ewe milk). Two randomly ordered conditions, namely UAO and control, were studied for 6 h on 2 consecutive days in each lamb. A custom-made, leak-proof nasal mask with unidirectional inspiratory and expiratory valves was installed on the lamb’s muzzle in both conditions. For the UAO condition, a 10-cm-long piece of soft plastic tube (diameter = 0.5 cm) was attached to the inspiratory valve and compressed with an adjustable clamp to obtain the maximally tolerated inspiratory resistance in each lamb (see Fig. 1). Such continuously applied resistance corresponded to a peak inspiratory tracheal pressure around −13 cmH2O and was tolerated by all lambs for the vast majority of the 6-h UAO recording. If a lamb became agitated, the clamp was immediately loosened to reduce the inspiratory resistance for 1 or 2 min, then rapidly reset to the initially determined level when the animal had calmed down. Of note, simultaneous measurement of tracheal and esophageal pressure in the first two lambs of the study showed that a peak inspiratory tracheal pressure around −13 cmH2O corresponded to esophageal pressure swings around −18 cmH2O. A soft plastic hood was loosely tied around the lamb’s head and continuously flushed with a flow (10 L/min) of air or an oxygen mixture adjusted to maintain PaO2 > 65 mmHg throughout the UAO recordings. Arterial blood gases were measured at 30-min intervals during the recordings, in addition to the continuous SpO2 recording.
Our custom-made nasal mask, specifically designed for newborn lambs, has a double nasal cannula (diameter = 4.5 mm) placed into the nares of the animal. Unidirectional inspiratory and expiratory valves were added for the purpose of this study. On the inspiratory branch, a 10-cm-long piece of soft plastic tube (diameter = 0.5 cm) was attached and compressed with an adjustable clamp to obtain the maximally tolerated inspiratory resistance in each lamb.
Hypoxia experiment
Two 3-h recordings were performed on the same day in seven additional nonsedated lambs. A soft plastic hood was loosely tied around the lambs’ head and continuously flushed with 10 L/min of air or a hypoxic mixture. Each lamb underwent an initial 3-h recording in normoxia, followed by a 1-h pause. Then, each lamb underwent a second 3-h recording with 10% hypoxia. The oxygen concentration in the hood was continuously monitored throughout the recordings, and the nitrogen flow adjusted if needed. Lambs were fed 1-h before the beginning of each condition (15 mL/kg of ewe milk). Arterial blood gases were measured every hour in the control condition; they were measured every 15 min during the first hour of hypoxia, then at 30-min intervals until the end of the recording.
Data analysis
Gastroesophageal refluxes
Multichannel intraluminal impedance/pH-metry recordings were automatically analyzed with MMS software (Medical Measurement Systems, Enschede, The Netherlands) and visually verified as previously described.10,14 The total number of GERs was calculated, and GER characterized as liquid, purely gaseous, or mixed according to published criteria.15 Furthermore, the bolus exposure index (BEI; % total time spent in liquid reflux), the bolus clearance time (time required for the entire bolus to be cleared from the esophagus by the secondary peristaltism), and the number of proximal GERs (GER reaching Z1, the most rostral channel of the MII/pH catheter) were computed. In addition, the mean electrical impedance baseline (EIB, an index of impaired mucosal integrity16,17) was computed in the hypoxic experiment.
Respiratory changes during upper airway obstruction
The following variables were calculated and averaged over 1 min at each 30 min during the 6-h recording to assess the severity of inspiratory obstruction in our model: (i) the respiratory rate (RR, breaths/min); (ii) the duty cycle, i.e., the ratio of inspiratory duration to total duration of the breathing cycle (Ti/Ttot); (iii) the amplitude of the peak inspiratory negative tracheal pressure (Ptrach, cmH2O); (iv) the minute tracheal pressure-time product (PTP • RR, cmH2O.s/min) obtained by computing the area over the Ptrach curve during inspiration and multiplying it by RR,18 as an index of the inspiratory work of breathing; and (v) arterial blood gases.
Respiratory variables during hypoxia
Respiratory changes were characterized by the following variables: (i) RR; (ii) the percentage of time spent in apnea (defined as at least two missed breaths with an amplitude < 30% compared to baseline breathing) during the recordings; and (iii) arterial blood gases.
Statistical analysis
Statistical analyses were performed on raw data for all dependent variables. All data were expressed as median (quartile 1 (Q1); quartile 3 (Q3)). The Wilcoxon signed-rank test was used on all dependent variables to assess the effects of increased inspiratory resistance or hypoxia on GER, respiratory variables, and arterial blood gases. Statistical analyses were performed with SPSS Statistics 17.0 (SPSS Inc, Chicago, IL, USA). Differences were considered significant if p < 0.05.
Results
Animals
The UAO experiment was conducted on ten full-term lambs weighing 4.3 (3.9; 4.7) kg, whereas the hypoxia experiment was conducted on seven full-term lambs weighing 3.8 (3.5; 4.0) kg.
Upper airway obstruction experiment
Respiratory effects
Table 1 provides detailed results. The peak inspiratory tracheal pressure was much more negative with UAO (p = 0.005) in all lambs, indicating they were all subjected to moderate-to-severe UAO. Simultaneously, respiratory rate was significantly increased (p = 0.005). Consequently, the minute tracheal pressure-time product was markedly increased (p = 0.02) in the UAO condition. No significant difference was noted for Ti/Ttot (p = 0.1).
Although statistically significant (p = 0.005), the decrease in PaO2 with UAO was very limited, with PaO2 > 75 mmHg in all lambs. Similarly, the statistically significant alterations in PaCO2 and arterial pH (p = 0.05 and p = 0.02, respectively) with UAO were very limited.
Gastroesophageal reflux
Upper airway obstruction significantly increased the number of GERs (2 (1; 4) vs. 0 (0; 3) in control condition, p = 0.03) and the percentage of mixed GERs (Table 2). Seven lambs out of ten presented a greater number of GERs in the presence of UAO (Table 2). No statistical differences were, however, observed for the number of proximal GERs, the bolus exposure index, or the median bolus clearance time (Table 3).
Hypoxic experiment
Respiratory effects
Hypoxic breathing was responsible for a severe hypoxemia (PaO2 ~ 30 mmHg) with respiratory alkalosis (Table 1). Respiratory rate was significantly increased (58 (57; 66) vs. 52 (48; 57) min−1; p = 0.03) and the percent of time spent in apnea was halved (0.5% (0.1, 0.6) vs. 1% (0.8; 1.2); p = 0.02).
Gastroesophageal reflux
Overall, the alterations in the number of GERs (2 (1; 3) vs. 0 (0; 5) in the control condition, p = 0.9) brought about by 3-h hypoxic exposure were not significant. The number of GERs was, however, lower in five lambs out of seven during hypoxia (Table 2). Notably, the median bolus clearance time was greatly decreased during hypoxia (p = 0.03), while no other indices of GER were significantly altered (Table 3). Finally, the mean electrical impedance baseline was significantly increased in the proximal (Z1 channel: 5069 (4773, 6319) ohms in hypoxia vs. 4568 (4305, 5228) ohms in normoxia, p < 0.03) but not the distal (Z6 channel: 6252 (3623, 6389) ohms in hypoxia vs. 5411 (3797, 5538) ohms in normoxia, p = 0.2) esophagus.
Discussion
The present study provides novel findings on GER in nonsedated newborn ovine models of marked UAO or severe hypoxemia. While the overall number of GERs was statistically elevated in the presence of an UAO, this increase was less than anticipated and absent in some lambs. Moreover, short-term severe hypoxemia did not alter GER overall, but increased the proximal electrical impedance baseline and markedly decreased the bolus clearance time.
The newborn lamb model for studying gastroesophageal refluxes
Over the past 7 years, we have gained unique, extensive expertise in studying GER in nonsedated newborn lambs using MII/pH.10,14,19,20 During the first week of life, the preruminant, monogastric newborn lamb presents spontaneous GERs, which bear several features similar to those of the human infant.10 In addition, we have used manometric measurements to demonstrate the presence of a high-pressure zone at the level of the distal esophagus,20 corresponding to the gastroesophageal junction. Unpublished histological studies on newborn lambs conducted by our team have also shown that, similarly to humans, the gastroesophageal junction is composed of the right diaphragmatic crus around the lower esophageal sphincter (smooth muscle) (see Supplemental Fig. S1). The newborn lamb in its first week of life hence offers a unique means to test various research hypotheses on GER in controlled conditions and in the same subject.
Effects of upper airway obstruction on gastroesophageal reflux in animal models
As already alluded to, while it is widely accepted in clinical settings that UAO increases GER, the cause and effect relationship between GER and UAO is still poorly understood. An extensive review of the literature on the effects of airway obstruction on GER revealed only two studies in adult animal models of partial inspiratory + expiratory airway obstruction, mimicking a fixed central-airway obstruction. In a first study, an airway obstruction was induced in anesthetized adult rats by successively intubating the trachea with three progressively narrower cannulas. Results showed a progressive increase in the thoracoabdominal end-inspiratory pressure gradient, up to tripling of the baseline value, leading the authors to suggest that the antireflux barrier function of the gastroesophageal junction could be overcome during a severe airway obstruction. Gastroesophageal reflux was not, however, assessed in this study.7 In another study, adult dogs breathed for 1 week through a fenestrated capped tracheostomy tube. Results from pH-metry recordings performed without sedation showed that while no dog had acid GER at baseline, acid GERs were observed in three of the dogs following tracheal obstruction for 1 week.8
Of note, although the above results point to a positive association between GER and airway obstruction, the studies were not designed to specifically mimic an UAO, which is mainly characterized by increased inspiratory resistance.21 In addition, to our knowledge, the potential links between UAO and GER have not been previously studied in early life, despite the unique and well-known propensity for GER in young infants.9 Our nonsedated newborn lamb model was designed to closely reproduce a severe UAO. Relevance of the model was reflected by the markedly augmented inspiratory efforts (increased inspiratory Ptrach) and work of breathing (increased minute tracheal pressure-time product). While an increase in Ti/Tot has been reported in children with UAO,22 the increase in RR (without Ti/tot increase) we observed in newborn lambs in the UAO condition is consistent with past observations in infants with UAO.23,24,25,26 Overall, the results obtained in newborn lambs with regard to increased GER in the UAO condition are consistent with adult dog studies.8 The increase in GER number was, however, less than anticipated, being even absent in some lambs and dogs. At first glance, this may seem surprising, given the marked increase in inspiratory negative intrathoracic pressure being responsible for an increased thoracoabdominal pressure gradient, in turn, favoring GER.7 The operating mechanisms underlying GER occurrence during UAO are, however, more complex. There is convincing evidence in animal and human studies that the normal inspiratory contraction of the crural-diaphragm significantly increases the pressure of the gastroesophageal junction during inspiration, hence preventing GER.27,28,29,30 Similarly, the addition of inspiratory resistance was shown to increase the gastroesophageal-junction pressure during inspiration due to an augmented inspiratory crural-diaphragm contraction,31 hence opposing GER occurrence, even in the presence of a greater thoracoabdominal pressure gradient. On the contrary, the increased abdominal-muscle expiratory contraction observed with UAO32 in the absence of the forceful inspiratory crural-diaphragm contraction, might increase the chance of GER occurrence via the increased intra-abdominal pressure during expiration. Overall, this leads us to hypothesize that the inconsistent increase in GER with UAO in dogs and lambs might be related to the different balance between the increase in gastroesophageal-junction pressure in inspiration and the increase in abdominal pressure in expiration between the animals. Of note, the situation might even be more complex if the UAO is responsible for hypoxemia and/or hypercapnia, because the consequent increase in abdominal-muscle activity33,34,35 would likely favor GER occurrence.
Effects of 10% hypoxia on GER
To our knowledge, our study is the first to specifically assess the effect of hypoxia on GER occurrence. One previous study found that a 3-min hypoxia (inspiratory fraction of oxygen of 0.08) significantly increased the gastroesophageal-junction pressure in newborn piglets, presumably due to an increase in inspiratory crural-diaphragm activity. This result led the authors to suggest that a short hypoxia protects against GER.11 Similar to UAO, however, the situation is likely not so simple, especially during sustained hypoxia. Indeed, the increase in inspiratory crural-diaphragm activity is paralleled by an increase in expiratory abdominal electrical activity.36 The latter, that we have demonstrated in lambs breathing a fraction of inspiratory oxygen of 0.08 for 15 min, would rather favor GER occurrence by increasing abdominal pressure. On the contrary, given that the gastroesophageal-junction pressure is decreased during central apnea,11 the decrease in the time spent in apnea we found in hypoxia in this study would tend to decrease GER occurrence. The preeminence of one or the other mechanism might explain the variable effect of hypoxia, with a decrease in GER occurrence in four out of seven lambs, but an increase in two out of seven lambs.
The consistent decrease in bolus clearance time we found in hypoxia is noteworthy. Volume clearance of the esophagus following GER is achieved by primary and secondary peristalsis.36,37 In this regard, we have previously shown in lambs that a sustained hypoxia similar to the one in this study decreased nonnutritive swallowing.38 In addition, hypoxia has been shown to decrease esophageal muscle activity.39 These two past results appear contradictory with a decrease in bolus clearance time in hypoxia, hence the latter remains unexplained.
Finally, the increase in basal electrical impedance, especially in the proximal esophagus, suggests that mucosal integrity is impaired by sustained hypoxia.16,17
Limitations of the study
Although our experiments provide unique information to better understand the effects of UAO and hypoxia on GER in infants, several limitations must be mentioned. First, despite our careful attempt to be clinically relevant, there are significant differences between the type of UAO we applied to lambs in the study and the UAO clinically encountered in infants. In lambs, UAO was permanent and identical for all breaths. On the contrary, infants not only present with incomplete UAO, which most often is intermittent and highly variable with time, but also with epochs of complete obstructed breaths (obstructive apneas). Our study was not designed, in particular, to assess the effect of complete UAO on the GER occurrence. Hence, our results constitute only one step toward understanding the effects of UAO on GERs in infants. Secondly, our results do not contribute any direct information on the effects of increased inspiratory resistance or hypoxia on the various components of the esophagogastric junction. This requires further research, which could involve recording of activity of the lower esophageal sphincter and crural-diaphragm muscle via electromyographic electrodes and/or sonomicrometry.40 Thirdly, both experiments were carried out for only a few hours, whereas UAO and hypoxia might present as long-term conditions. Hence, our results might better apply to short-term conditions such as viral croup or rhinitis in young infants. Finally, these results do not shed any light on conditions that associate both UAO and hypoxia. We, however, consider these experiments as a necessary first step in assessing the effects of UAO and/or hypoxia on GER.
Conclusion
Overall, a short-term, marked increase in inspiratory resistance increased the number of GERs in newborn lambs. This increase, however, is inconsistent, variable, and somewhat less than we anticipated. Although these results might call into question the systematic prescription of potentially dangerous antireflux medications (such as proton-pump inhibitors) in infants with UAO, further studies are obviously needed before making such a recommendation. Such studies should especially assess the effects of intermittent and variable UAO, including obstructive apneas. Moreover, a sustained short-term hypoxic exposure of 10% also had a variable effect on the number of GERs.
From a physiological standpoint, results from the present study should be complemented by more direct physiological studies on the effects of increased inspiratory resistance or hypoxia on the gastroesophageal junction.
References
Bibi, H. et al. The prevalence of gastroesophageal reflux in children with tracheomalacia and laryngomalacia. Chest 119, 409–413 (2001).
Matthews, B. L., Little, J. P., McGuirt, W. F. Jr & Koufman, J. A. Reflux in infants with laryngomalacia: results of 24-hour double-probe pH monitoring. Otolaryngol.-Head. Neck Surg. 120, 860–864 (1999).
Noronha, A. C. et al. Gastroesophageal reflux and obstructive sleep apnea in childhood. Int. J. Pediatr. Otorhinolaryngol. 73, 383–389 (2009).
Katz, E. S., Mitchell, R. B. & D’Ambrosio, C. M. Obstructive sleep apnea in infants. Am. J. Respir. Crit. Care Med. 185, 805–816 (2012).
Tieder, J. S. et al. Clinical practice guideline: Brief resolved unexplained events (formerly apparent life-threatening events) and evaluation of lower-risk infants. Pediatrics 137, e20160590 (2016).
El-Mahdy, M. A., Mansoor, F. A. & Jadcherla, S. R. Pharmacological management of gastroesophageal reflux disease in infants: current opinions. Curr. Opin. Pharmacol. 37, 112–117 (2017).
Wang, W., Tovar, J. A., Eizaguirre, I. & Aldazabal, P. Airway obstruction and gastroesophageal reflux: an experimental study on the pathogenesis of this association. J. Pediatr. Surg. 28, 995–998 (1993).
Boesch, R. P. et al. Relationship between upper airway obstruction and gastroesophageal reflux in a dog model. J. Investig. Surg. 18, 241–245 (2005).
Vandenplas, Y., Hauser, B. & Salvatore, S. Functional gastrointestinal disorders in infancy: impact on the health of the infant and family. Pediatr. Gastroenterol., Hepatol. Nutr. 22, 207–216 (2019).
Djeddi, D., Samson, N., Boudaa, N. & Praud, J. P. Newborn lamb as a new model for studying gastroesophageal reflux. J. Pediatr. Gastroenterol. Nutr. 55, 745–746 (2012).
Kiatchoosakun, P., Dreshaj, I. A., Abu-Shaweesh, J. M., Haxhiu, M. A. & Martin, R. J. Effects of hypoxia on respiratory neural output and lower esophageal sphincter pressure in piglets. Pediatr. Res. 52, 50–55 (2002).
Fortier, P. H., Reix, P., Arsenault, J., Dorion, D. & Praud, J. P. Active upper airway closure during induced central apneas in lambs is complete at the laryngeal level only. J. Appl. Physiol. 95, 97–103 (2003).
Samson, N., Dumont, S., Specq, M. L. & Praud, J. P. Radio telemetry devices to monitor breathing in non-sedated animals. Respir. Physiol. Neurobiol. 179, 111–118 (2011).
Cantin, D., Djeddi, D., Samson, N., Nadeau, C. & Praud, J. P. Nasal high-frequency oscillatory ventilation inhibits gastroesophageal reflux in the neonatal period. Respir. Physiol. Neurobiol. 251, 28–33 (2018).
Wenzl, T. G. et al. Indications, methodology, and interpretation of combined esophageal impedance-pH monitoring in children: ESPGHAN EURO-PIG standard protocol. J. Pediatr. Gastroenterol. Nutr. 55, 230–234 (2012).
Borrelli, O. et al. Relationship between baseline impedance levels and esophageal mucosal integrity in children with erosive and non-erosive reflux disease. Neurogastroenterol. Motil. 24, 828–e394 (2012).
Martinucci, I. et al. Esophageal baseline impedance levels in patients with pathophysiological characteristics of functional heartburn. Neurogastroenterol. Motil. 26, 546–555 (2014).
Dodd, D. S., Collett, P. W. & Engel, L. A. Influence of inspiratory flow rate and frequency on O2 cost of resistive breathing in humans. J. Appl. Physiol. 65, 760–766 (1988).
Cantin, D. et al. Inhibitory effect of nasal intermittent positive pressure ventilation on gastroesophageal reflux. PLoS ONE 11, e0146742 (2016).
Djeddi, D., Cantin, D., Samson, N. & Praud, J. P. Nasal continuous positive airway pressure inhibits gastroesophageal reflux in newborn lambs. PLoS ONE 9, e107736 (2014).
Khemani, R. G. et al. Respiratory inductance plethysmography calibration for pediatric upper airway obstruction: an animal model. Pediatr. Res. 77, 75–83 (2015).
Praud, J. P., Gaultier, C., Buvry, A., Boule, M. & Girard, F. Lung mechanics and breathing pattern during wakefulness and sleep in children with enlarged tonsils. Sleep 7, 304–312 (1984).
Guilleminault, C. & Khramtsov, A. Upper airway resistance syndrome in children: a clinical review. Semin. Pediatr. Neurol. 8, 207–215 (2001).
Katz, E. S. & D’Ambrosio, C. M. Pathophysiology of pediatric obstructive sleep apnea. Proc. Am. Thorac. Soc. 5, 253–262 (2008).
Leboulanger, N. et al. Physiologic and clinical benefits of noninvasive ventilation in infants with Pierre Robin sequence. Pediatrics 126, e1056–e1063 (2010).
Balfour-Lynn, I. M. & Rigby, M. L. Tachypnoea in a well baby: what to do next? Arch. Dis. Child. 100, 722–727 (2015).
Martin, C. J. et al. Diaphragmatic contribution to gastroesophageal competence and reflux in dogs. Am. J. Physiol. 263, G551–G557 (1992).
Mittal, R. K. & Balaban, D. H. The esophagogastric junction. N. Engl. J. Med. 336, 924–932 (1997).
Lin, Z. et al. Novel 3D high-resolution manometry metrics for quantifying esophagogastric junction contractility. Neurogastroenterol. Motility 29, e13054 (2017).
Rengarajan, A. & Gyawali, C. P. High-resolution manometry can characterize esophagogastric junction morphology and predict esophageal reflux burden. J. Clin. Gastroenterol. 54, 22–27 (2020).
Nobre e Souza, M. A. et al. Inspiratory muscle training improves antireflux barrier in GERD patients. Am. J. Physiol. Gastrointest. Liver Physiol. 305, G862–G867 (2013).
Praud, J. P. et al. Sleep-related abdominal muscle behavior during partial or complete obstructed breathing in prepubertal children. Pediatr. Res. 26, 347–350 (1989).
Praud, J. P. et al. Abdominal muscle activity during CO2 rebreathing in sleeping neonates. J. Appl. Physiol. 70, 1344–1350 (1991).
Praud, J. P., Kianicka, I., Leroux, J. F. & Dalle, D. Laryngeal response to hypoxia in awake lambs during the first postnatal days. Pediatr. Res. 37, 482–488 (1995).
Helm, J. F. et al. Effect of esophageal emptying and saliva on clearance of acid from the esophagus. N. Engl. J. Med. 310, 284–288 (1984).
Praud, J. P., Canet, E., Kianicka, I., Gaultier, C. & Bureau, M. Vagal and chemoreceptor influences on abdominal muscle activity in awake lambs during hypoxia. J. Appl. Physiol. 74, 1689–1696 (1993).
Zerbib, F. et al. Normal values and day-to-day variability of 24-h ambulatory oesophageal impedance-pH monitoring in a Belgian-French cohort of healthy subjects. Aliment. Pharmacol. Therapeut. 22, 1011–1021 (2005).
Duvareille, C. et al. Effects of hypoxia and hypercapnia on nonnutritive swallowing in newborn lambs. J. Appl. Physiol. 103, 1180–1188 (2007).
Dombkowski, R. A. et al. Hydrogen sulfide (H(2)S) and hypoxia inhibit salmonid gastrointestinal motility: evidence for H(2)S as an oxygen sensor. J. Exp. Biol. 214, 4030–4040 (2011).
Adelson, D. W. & Million, M. Tracking the moveable feast: sonomicrometry and gastrointestinal motility. News Physiol. Sci. 19, 27–32 (2004).
Acknowledgements
This work was supported with an operating grant from Canadian Institutes of Health Research and Canada Research Chair in Neonatal Respiratory Physiology held by J.-P.P.
Author information
Authors and Affiliations
Contributions
A.M., D.D., J.-P.P., N.S., and W.L.J. conceived and designed the study. A.M., C.N., N.S., and W.L.J. performed the animal experiments. A.M., C.N., D.D., N.S., and W.L.J. analyzed the data. A.M., D.D., J.-P.P., N.S., and W.L.J. interpreted the results obtained. A.M. and N.S. drafted the manuscript. D.D., J.-P.P., and N.S. revised the manuscript. All authors approved the final version of the manuscript and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Michaud, A., Jia, W.L., Djeddi, D. et al. Effects of upper airway obstruction or hypoxia on gastroesophageal reflux in newborn lambs. Pediatr Res 89, 496–501 (2021). https://doi.org/10.1038/s41390-020-0920-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-020-0920-8