Featured
-
-
News & Views |
 Manganese catalyst enables exploration of the magic methyl effect
Manganese catalyst enables exploration of the magic methyl effect
The addition of a methyl group to a drug molecule can greatly alter the drug’s pharmacological properties. A catalyst has been developed that enables this ‘magic methyl effect’ to be rapidly explored for drug discovery.
- Emily B. Corcoran
- & Danielle M. Schultz
-
Article |
 A general carbonyl alkylative amination for tertiary amine synthesis
A general carbonyl alkylative amination for tertiary amine synthesis
The synthesis of tertiary amines is achieved through a carbonyl alkylative amination reaction facilitated by visible light, in which an aldehyde and an amine condense to form an iminium ion that subsequently reacts with alkyl radical.
- Roopender Kumar
- , Nils J. Flodén
- & Matthew J. Gaunt
-
Article |
 A map of the amine–carboxylic acid coupling system
A map of the amine–carboxylic acid coupling system
All possible chemical transformations between amine and carboxylic acid groups are mapped using an automated string-based combinatorics method, showing that the properties and functions of the products vary considerably over all plausible reactions.
- Babak Mahjour
- , Yuning Shen
- & Tim Cernak
-
News & Views |
 Strong chemical reducing agents produced by light
Strong chemical reducing agents produced by light
An electrically neutral radical has been found to be a potent chemical reducing agent when excited by light. Remarkably, it is produced from a positively charged precursor that has long been used as a strong excited-state oxidizing agent.
- Radek Cibulka
-
Article |
 Discovery and characterization of an acridine radical photoreductant
Discovery and characterization of an acridine radical photoreductant
Photoexcited acridine radical catalysts are found to have redox potentials more reducing than lithium, which is attributed to the population of higher-energy doublet excited states via a twisted intramolecular charge-transfer species.
- Ian A. MacKenzie
- , Leifeng Wang
- & David A. Nicewicz
-
News & Views |
 Radial flow system decouples reactions in automated synthesis of organic molecules
Radial flow system decouples reactions in automated synthesis of organic molecules
Organic compounds can be synthesized in a continuous flow of solutions, but the need to balance mass flow across multiple reactors complicates the development of such systems. A new platform for flow chemistry addresses this issue.
- Klavs F. Jensen
-
Article |
 Automated radial synthesis of organic molecules
Automated radial synthesis of organic molecules
An automated synthesis instrument comprising a series of continuous flow modules that are radially arranged around a central switching station can achieve both linear and convergent syntheses.
- Sourav Chatterjee
- , Mara Guidi
- & Kerry Gilmore
-
Research Highlight |
 Turning the humble soya bean into a powerful green glue
Turning the humble soya bean into a powerful green glue
Chemists have borrowed a strategy from plant cell walls to produce a high-strength wood adhesive.
-
Article |
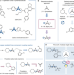 Copper-mediated synthesis of drug-like bicyclopentanes
Copper-mediated synthesis of drug-like bicyclopentanes
A one-step, three-component radical coupling of [1.1.1]propellane by a photoredox reaction mediated by a copper catalyst produces drug-like bicyclopentanes.
- Xiaheng Zhang
- , Russell T. Smith
- & David W. C. MacMillan
-
Article |
 Synthesis of rare sugar isomers through site-selective epimerization
Synthesis of rare sugar isomers through site-selective epimerization
Various rare sugars that cannot be isolated from natural sources are synthesized using light-driven epimerization, a process which may find application in other synthetic scenarios.
- Yong Wang
- , Hayden M. Carder
- & Alison E. Wendlandt
-
News & Views |
 Robots, hominins and superconductors: 10 remarkable papers from 2019
Robots, hominins and superconductors: 10 remarkable papers from 2019
Highlights from News & Views published this year.
-
Article |
 Lactonization as a general route to β-C(sp3)–H functionalization
Lactonization as a general route to β-C(sp3)–H functionalization
Pd-catalysed β-C(sp3)–H lactonization of aliphatic acids enabled by a mono-N-protected β-amino acid and tert-butyl hydrogen peroxide is reported, achieving high β-position selectivity without the use of a directing group.
- Zhe Zhuang
- & Jin-Quan Yu
-
Article |
 Site-specific allylic C–H bond functionalization with a copper-bound N-centred radical
Site-specific allylic C–H bond functionalization with a copper-bound N-centred radical
A Cu-bound nitrogen-centred radical is used to control site-specific and enantioselective allylic C–H cyanations of molecules with synthetic and medicinal relevance, such as tri- and tetrasubstituted alkenes.
- Jiayuan Li
- , Zhihan Zhang
- & Guosheng Liu
-
News & Views |
 Double-click enables synthesis of chemical libraries for drug discovery
Double-click enables synthesis of chemical libraries for drug discovery
Operationally simple chemical reactions, termed click reactions, are widely used in many scientific fields. A streamlined synthesis of compounds called azides looks set to expand the role of click chemistry still further.
- Joseph J. Topczewski
- & En-Chih Liu
-
Letter |
 Modular click chemistry libraries for functional screens using a diazotizing reagent
Modular click chemistry libraries for functional screens using a diazotizing reagent
A ‘click’ reaction is developed for the simple and rapid formation of azides from primary amines, and is used to prepare a library of over 1,200 azides for subsequent use in the existing triazole annulation click reaction.
- Genyi Meng
- , Taijie Guo
- & Jiajia Dong
-
Article |
 Metal-free directed sp2-C–H borylation
Metal-free directed sp2-C–H borylation
A method for the site-selective C–H borylation of arenes and heteroarenes is described, in which BBr3 acts as both a reagent and a catalyst.
- Jiahang Lv
- , Xiangyang Chen
- & Zhuangzhi Shi
-
Letter |
 A 16-step synthesis of the isoryanodane diterpene (+)-perseanol
A 16-step synthesis of the isoryanodane diterpene (+)-perseanol
The chemical synthesis of (+)-perseanol, a diterpene with potent insecticidal properties, is reported.
- Arthur Han
- , Yujia Tao
- & Sarah E. Reisman
-
Research Highlight |
 A reaction used by chemists worldwide goes green
A reaction used by chemists worldwide goes green
Thanks to a catalyst, a crucial chemical process no longer requires an explosive ingredient.
-
News & Views |
 Fluorinated compounds present opportunities for drug discovery
Fluorinated compounds present opportunities for drug discovery
A practical method has been devised to synthesize compounds that have previously been almost impossible to make. The combination of fluorine atoms and amide groups in the compounds might be useful in pharmaceuticals.
- Jonathan Clayden
-
Research Highlight |
 ‘Robochemist’ concocts its own compounds
‘Robochemist’ concocts its own compounds
Teamwork between neural networks and a swivelling robot churns out new compounds.
-
Article |
 Holistic prediction of enantioselectivity in asymmetric catalysis
Holistic prediction of enantioselectivity in asymmetric catalysis
A workflow for deriving statistical models of one set of reactions that can be used to predict related reactions is presented, facilitating catalyst and enantioselective reaction development.
- Jolene P. Reid
- & Matthew S. Sigman
-
News & Views |
 Holistic models of reaction selectivity
Holistic models of reaction selectivity
Computational models that predict the selectivity of reactions are typically accurate for only a specific reaction type and a narrow range of reaction components. A more general model has now been reported.
- Per-Ola Norrby
-
Letter |
 Peptide ligation by chemoselective aminonitrile coupling in water
Peptide ligation by chemoselective aminonitrile coupling in water
Prebiotic peptide formation is achieved through chemoselective, high-yielding ligation of α-aminonitriles in water, showing selectivity for α-peptide coupling and tolerance of all proteinogenic amino acid residues.
- Pierre Canavelli
- , Saidul Islam
- & Matthew W. Powner
-
Matters Arising |
 Reinvestigation of a robotically revealed reaction
Reinvestigation of a robotically revealed reaction
- Jonathan K. Sader
- & Jeremy E. Wulff
-
Letter |
 RETRACTED ARTICLE: Living annulative π-extension polymerization for graphene nanoribbon synthesis
RETRACTED ARTICLE: Living annulative π-extension polymerization for graphene nanoribbon synthesis
A solution-based living annulative π-extension polymerization technique produces graphene nanoribbons with simultaneous control over their length, width and edges, resulting in the synthesis of graphene nanoribbon block copolymers.
- Yuuta Yano
- , Nobuhiko Mitoma
- & Kenichiro Itami
-
Technology Feature |
 Automation: Chemistry shoots for the Moon
Automation: Chemistry shoots for the Moon
A new class of chemical instrumentation seeks to alleviate the tedium and complexity of organic syntheses.
- Katharine Sanderson
-
News & Views |
 Molecular-decoration technique offers boost to medicinal chemists
Molecular-decoration technique offers boost to medicinal chemists
The ability to attach a variety of chemical groups to one position in a molecule facilitates the search for compounds that have useful properties. Reactions have been reported that could transform how chemists do this.
- Eric M. Ferreira
-
Letter |
 Site-selective and versatile aromatic C−H functionalization by thianthrenation
Site-selective and versatile aromatic C−H functionalization by thianthrenation
A highly site-selective aromatic C–H functionalization reaction that does not require a particular directing group or arene substitution pattern provides functionalized arenes that can participate in various transformations.
- Florian Berger
- , Matthew B. Plutschack
- & Tobias Ritter
-
Letter |
 Enzyme-catalysed [6+4] cycloadditions in the biosynthesis of natural products
Enzyme-catalysed [6+4] cycloadditions in the biosynthesis of natural products
Enzymatic catalysis of pericyclic [6+4] cycloaddition reactions to form ten-membered rings is observed during biosynthesis of the macrocyclic antibiotic streptoseomycin, and the mechanism of these transformations is established.
- Bo Zhang
- , Kai Biao Wang
- & Hui Ming Ge
-
Letter |
 Deacylative transformations of ketones via aromatization-promoted C–C bond activation
Deacylative transformations of ketones via aromatization-promoted C–C bond activation
Aromatization-driven C–C bond activation through iridium/phosphine catalysis enables deacylative transformations in various ketone substrates.
- Yan Xu
- , Xiaotian Qi
- & Guangbin Dong
-
Letter |
 Photoredox catalysis using infrared light via triplet fusion upconversion
Photoredox catalysis using infrared light via triplet fusion upconversion
Photoredox transformations are achieved with infrared light by using triplet fusion upconversion that converts infrared into visible light, enabling the use of photoredox chemistry on larger scales and through barriers that are impenetrable by visible light.
- Benjamin D. Ravetz
- , Andrew B. Pun
- & Luis M. Campos
-
Letter |
 Desymmetrization of cyclohexanes by site- and stereoselective C–H functionalization
Desymmetrization of cyclohexanes by site- and stereoselective C–H functionalization
Unactivated cyclohexane derivatives can be desymmetrized by site- and stereoselective C–H functionalization using carbene-insertion chemistry.
- Jiantao Fu
- , Zhi Ren
- & Huw M. L. Davies
-
Career Q&A |
 Meet the scientific-database developer
Meet the scientific-database developer
A product manager develops resources that help users to save time and increase productivity.
- Nikki Forrester
-
Nature Index |
 Strong spending compounds chemistry prowess
Strong spending compounds chemistry prowess
The discipline’s historic prominence in China is underpinned by its crucial value to industrial processes. Committed funding sees it leading the way in emerging areas, such as nanomaterials.
- Hepeng Jia
-
Letter |
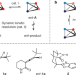 Catalytic deracemization of chiral allenes by sensitized excitation with visible light
Catalytic deracemization of chiral allenes by sensitized excitation with visible light
Photochemical deracemization through irradiation with visible light in the presence of a chiral sensitizer enables the direct formation of single enantiomers from a racemic mixture of the same compound.
- Alena Hölzl-Hobmeier
- , Andreas Bauer
- & Thorsten Bach
-
News & Views |
 An exciting tool for asymmetric synthesis
An exciting tool for asymmetric synthesis
A catalytic process driven by visible light converts a mixture of mirror-image isomers of compounds called allenes to a single mirror-image isomer — opening up avenues of research for synthetic chemistry.
- Cheng Yang
- & Yoshihisa Inoue
-
Nature Podcast |
 Podcast: The art of performing science, and chiral chemistry
Podcast: The art of performing science, and chiral chemistry
Join Benjamin Thompson and Shamini Bundell for this week's science updates.
-
News & Views |
 Practically simple reactions convert hydrocarbons to precious chemicals
Practically simple reactions convert hydrocarbons to precious chemicals
Operationally simple chemistry enables aliphatic carbon–carbon bonds — the ‘girders’ in the framework of many organic molecules — to be prepared from widely available hydrocarbons known as alkenes.
- James P. Morken
-
News |
 ‘Why didn’t we think to do this earlier?’ Chemists thrilled by speedy atomic structures
‘Why didn’t we think to do this earlier?’ Chemists thrilled by speedy atomic structures
Cross-disciplinary thinking was key to realizing the potential of electron diffraction to organic chemistry.
- Matthew Warren
-
Letter |
 Base-free nickel-catalysed decarbonylative Suzuki–Miyaura coupling of acid fluorides
Base-free nickel-catalysed decarbonylative Suzuki–Miyaura coupling of acid fluorides
This nickel-catalysed Suzuki–Miyaura coupling of aryl boronic acids with in-situ-generated acid fluorides does not require an exogenous base and is applicable to a range of base-sensitive boronic acids and bioactive carboxylic acids.
- Christian A. Malapit
- , James R. Bour
- & Melanie S. Sanford
-
Letter |
 Catalytic enantioconvergent coupling of secondary and tertiary electrophiles with olefins
Catalytic enantioconvergent coupling of secondary and tertiary electrophiles with olefins
Nickel-catalysed coupling of racemic alkyl electrophiles and olefins in the presence of a hydrosilane is achieved with good enantioselectivity and yield under very mild reaction conditions.
- Zhaobin Wang
- , Haolin Yin
- & Gregory C. Fu
-
Letter |
 Asymmetric α-arylation of amino acids
Asymmetric α-arylation of amino acids
This approach introduces an aryl substituent to the α-carbon of amino acids without a transition-metal catalyst, and uses the inherent chirality of the amino acid itself as the source of asymmetry.
- Daniel J. Leonard
- , John W. Ward
- & Jonathan Clayden
-
Letter |
 Multicomponent synthesis of tertiary alkylamines by photocatalytic olefin-hydroaminoalkylation
Multicomponent synthesis of tertiary alkylamines by photocatalytic olefin-hydroaminoalkylation
Complex tertiary alkylamines are prepared in one step from readily available amines, carbonyls and alkenes, via the visible-light-mediated reduction of in-situ-generated iminium ions to form alkyl-substituted α-amino radicals.
- Aaron Trowbridge
- , Dominik Reich
- & Matthew J. Gaunt
-
News & Views |
 Enzymes team up with light-activated catalysts
Enzymes team up with light-activated catalysts
Catalysts working in pairs can promote more-effective reactions than can the same catalysts used sequentially. The coupling of an enzyme with a light-activated catalyst offers great potential for organic synthesis.
- Nicholas J. Turner
-
News & Views |
 Reaction combination opens up 3D molecular diversity for drug discovery
Reaction combination opens up 3D molecular diversity for drug discovery
Cycloaddition reactions are powerful tools for synthesizing three-dimensional molecules, but their scope has been limited. A creative solution to this problem opens up opportunities for drug discovery.
- Wenbo Ye
- & Ang Li
-
Letter |
 Direct arylation of strong aliphatic C–H bonds
Direct arylation of strong aliphatic C–H bonds
Direct coupling of aliphatic C–H nucleophiles to aryl electrophiles is described, through the combination of light-driven polyoxometalate hydrogen atom transfer and nickel catalysis.
- Ian B. Perry
- , Thomas F. Brewer
- & David W. C. MacMillan
-
Letter |
 Building C(sp3)-rich complexity by combining cycloaddition and C–C cross-coupling reactions
Building C(sp3)-rich complexity by combining cycloaddition and C–C cross-coupling reactions
Combining cycloaddition and carbon–carbon cross-coupling offers a way of simplifying the enantioselective preparation of chemical building blocks, natural products and medicines such as the antipsychotic asenapine.
- Tie–Gen Chen
- , Lisa M. Barton
- & Phil S. Baran
-
Letter |
 Decarboxylative sp3 C–N coupling via dual copper and photoredox catalysis
Decarboxylative sp3 C–N coupling via dual copper and photoredox catalysis
The synergistic combination of copper catalysis and photoredox catalysis forms sp3 C–N bonds in a rapid, room-temperature coupling protocol with high efficiencies and regioselectivities and a broad substrate scope.
- Yufan Liang
- , Xiaheng Zhang
- & David W. C. MacMillan
-
Letter |
 Enantioselective remote meta-C–H arylation and alkylation via a chiral transient mediator
Enantioselective remote meta-C–H arylation and alkylation via a chiral transient mediator
Remote, enantioselective C–H activation reactions can be achieved by relaying ortho-C–H activation to the meta position, using a chiral norbornene as the mediator.
- Hang Shi
- , Alastair N. Herron
- & Jin-Quan Yu

 Preparation of cyclohexene isotopologues and stereoisotopomers from benzene
Preparation of cyclohexene isotopologues and stereoisotopomers from benzene