Featured
-
-
Article
| Open Access Control of stereogenic oxygen in a helically chiral oxonium ion
Control of stereogenic oxygen in a helically chiral oxonium ion
The design, synthesis and characterization of a helically chiral triaryloxonium ion is reported, which is an example of a chiral non-racemic and configurationally stable molecule in which the oxygen atom is the sole stereogenic centre.
- Owen Smith
- , Mihai V. Popescu
- & Martin D. Smith
-
Article |
 Enantioselective transition-metal catalysis via an anion-binding approach
Enantioselective transition-metal catalysis via an anion-binding approach
Chiral hydrogen-bond donors bind anions of organometallic catalysts to achieve enantiocontrol and reaction-rate enhancement through ion pairing together with other non-covalent interactions.
- John M. Ovian
- , Petra Vojáčková
- & Eric N. Jacobsen
-
Article |
 RETRACTED ARTICLE: Intrinsically unidirectional chemically fuelled rotary molecular motors
RETRACTED ARTICLE: Intrinsically unidirectional chemically fuelled rotary molecular motors
A homochiral rotary molecular motor shows autonomous unidirectional rotation around a single bond driven by a chemical fuel.
- Ke Mo
- , Yu Zhang
- & Depeng Zhao
-
Article |
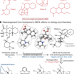 Synthesis and target annotation of the alkaloid GB18
Synthesis and target annotation of the alkaloid GB18
A synthetic route to GB18, an alkaloid from the bark of hallucinogenic Galbulimima sp., is developed, enabling its identification as an antagonist of κ- and μ-opioid receptors.
- Stone Woo
- & Ryan A. Shenvi
-
Article |
 Enantioselective synthesis of ammonium cations
Enantioselective synthesis of ammonium cations
Enantioselective supramolecular recognition allows for the asymmetric synthesis of nitrogen stereocentres, providing chiral ammonium cations in a dynamic crystallization process.
- Mark P. Walsh
- , Joseph M. Phelps
- & Matthew O. Kitching
-
Article |
 Holistic prediction of enantioselectivity in asymmetric catalysis
Holistic prediction of enantioselectivity in asymmetric catalysis
A workflow for deriving statistical models of one set of reactions that can be used to predict related reactions is presented, facilitating catalyst and enantioselective reaction development.
- Jolene P. Reid
- & Matthew S. Sigman
-
Letter |
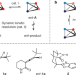 Catalytic deracemization of chiral allenes by sensitized excitation with visible light
Catalytic deracemization of chiral allenes by sensitized excitation with visible light
Photochemical deracemization through irradiation with visible light in the presence of a chiral sensitizer enables the direct formation of single enantiomers from a racemic mixture of the same compound.
- Alena Hölzl-Hobmeier
- , Andreas Bauer
- & Thorsten Bach
-
Review Article |
 Enhancing the potential of enantioselective organocatalysis with light
Enhancing the potential of enantioselective organocatalysis with light
This Review discusses recent developments in the combination of organocatalysis and photochemistry for the activation of molecules, which has enabled previously inaccessible reaction pathways and influenced many fields of chemical research.
- Mattia Silvi
- & Paolo Melchiorre
-
Letter |
 Synergy of synthesis, computation and NMR reveals correct baulamycin structures
Synergy of synthesis, computation and NMR reveals correct baulamycin structures
Experimental and computed nuclear magnetic resonance data and an iterative synthetic strategy have revealed the correct structures of the baulamycins, potentially important antimicrobial compounds, allowing them to be chemically synthesized.
- Jingjing Wu
- , Paula Lorenzo
- & Varinder K. Aggarwal
-
Letter |
 Catalytic allylic oxidation of internal alkenes to a multifunctional chiral building block
Catalytic allylic oxidation of internal alkenes to a multifunctional chiral building block
Non-symmetric internal alkenes are selectively converted into allylic functionalized products with high stereoselectivity and regioselectivity.
- Liela Bayeh
- , Phong Q. Le
- & Uttam K. Tambar
-
Letter |
 Forming all-carbon quaternary stereogenic centres in acyclic systems from alkynes
Forming all-carbon quaternary stereogenic centres in acyclic systems from alkynes
The synthesis of an all-carbon quaternary stereocentre in an acyclic system is reported; the chemical transformation involves the formation of two new stereogenic centres—including the challenging all-carbon quaternary one in an aldol adduct—via a combined carbometalation–oxidation reaction, giving a stereodefined trisubstituted enolate.
- Yury Minko
- , Morgane Pasco
- & Ilan Marek
-
News & Views |
 Catalysis in tight spaces
Catalysis in tight spaces
An acid has been found to catalyse the formation of a common chemical group, the spiroacetals, and to control which mirror-image isomer of the group is made. The key to success is the acid's bulky molecular structure. See Letter p.315
- Noah Z. Burns
- & Eric N. Jacobsen
-
Letter |
 Reconfigurable self-assembly through chiral control of interfacial tension
Reconfigurable self-assembly through chiral control of interfacial tension
Molecular chirality can be used to control interfacial tension in multi-component mixtures of chiral molecules, and tuning the chirality makes it possible to produce and manipulate self-assembling complex chiral structures.
- Thomas Gibaud
- , Edward Barry
- & Zvonimir Dogic
-
News & Views |
 Amide bonds made in reverse
Amide bonds made in reverse
Amide bonds connect the amino acids in proteins and occur in many other useful molecules. An amide-forming reaction that turns the conventional approach on its head offers a practical way of making these bonds.
- Karl Scheidt
-
Research Highlights |
Organic chemistry: Catalysts cooperate

 Symmetry breaking and chiral amplification in prebiotic ligation reactions
Symmetry breaking and chiral amplification in prebiotic ligation reactions