Abstract
Study design:
Prospective multicenter cohort study.
Objectives:
To compare the neurological recovery and functional outcomes between traumatic central cord syndrome (TCCS) patients and motor incomplete tetraplegic patients.
Setting:
European Multicenter Study of human spinal cord injury.
Methods:
In 248 traumatic motor incomplete tetraplegics, initial phase (0–15 days) American Spinal Injury Association (ASIA) impairment grading, upper and lower extremity motor scores (UEMS and LEMS), upper and lower sensory scores and chronic phase (6 or 12 months) neurological outcomes were analyzed. In addition, chronic phase self-care and indoor mobility Spinal Cord Independence Measure (SCIM) items were studied. Tetraplegics were subdivided into three groups: (1) non-TCCS group (UEMS⩾LEMS), (2) intermediate-TCCS group (UEMS=(1–9 points)
Results:
A total of 89 non-TCCS subjects (AIS D, n=28), 62 int-TCCS (AIS D, n=43) and 97 TCCS (AIS D, n=80) subjects were analysed. Although minimal significant differences in chronic phase LEMS and UEMS outcomes were identified between TCCS and non-TCCS patients after stratification by the AIS grade, our data showed no significant differences in functional upper and lower extremity outcomes at 6 or 12 months post-injury.
Conclusion:
The AIS grading system, and not the diagnosis TCCS, continues to be the best available prognostic parameter for neurological and functional outcomes in motor incomplete tetraplegics. The authors recommend that for future outcome studies in motor incomplete tetraplegia, patients should not be selected based on, or stratified by, the diagnosis TCCS.
Similar content being viewed by others
Introduction
The traumatic central cord syndrome (TCCS) is a clinical diagnosis that was first described by Schneider et al. in 1954.1 The TCCS is characterized by the following: (1) a disproportionate impairment (weakness and reduced function) of the upper limbs compared with the lower limbs, (2) neurogenic bladder dysfunction and (3) varying degrees of sensory loss at and below the level of lesion.1 Out of these three clinical characteristics, the first is generally considered to be the most typical and important one.2, 3
It has been hypothesized that TCCS patients have a favorable recovery pattern compared with other motor incomplete tetraplegics.4, 5, 6, 7 Although several studies have compared TCCS with other spinal cord injury syndromes, such as the Brown-Séquard Syndrome,4, 8, 9 no study has compared the neurological and functional recovery between TCCS and other motor incomplete tetraplegic patients. The assumed superior recovery of TCCS patients has also been expressed by an international panel of spinal cord injury (SCI) experts convened by the International Campaign for Cures of Spinal Cord Injury Paralysis.10
In the first two parts2, 3 of this three-paper series, we demonstrated that a wide variety of definitions of TCCS are employed among both researchers and physicians. In part 1, a pragmatic analysis of 312 pooled TCCS subjects included in previous studies showed that the average difference in motor strength between the upper extremity motor score (UEMS) and the lower extremity motor score (LEMS) was 10.5 points based on the Medical Research Council Scale.2 In part 2, a questionnaire survey among physicians showed that the majority of physicians considered this difference in motor score of at least 10 points as an acceptable cut-off criterion for scientific purposes. Nonetheless, there were a number of physicians who favored assigning the diagnosis TCCS in SCI patients with 1–9 motor points difference between the UEMS and LEMS.3
The introduction of the diagnostic TCCS criterion in part 2 of this three-paper series, that is, ‘a minimal difference of 10 motor score points between the upper and lower extremities, in favor of the lower extremities’, enabled us to investigate the hypothesized differences in recovery patterns between subgroups of motor incomplete tetraplegics with use of a face valid and reproducible criterion. The objective of this study therefore was to compare the neurological recovery and functional outcomes between motor incomplete tetraplegic patients with (1) equal or less motor strength in the lower extremities (non-TCCS), (2) 1–9 motor points more in the lower extremities (intermediate TCCS) and (3) 10 motor points or more in the lower extremities compared with the upper extremities (TCCS).
Materials and methods
A total of 18 European SCI centres prospectively collected clinical data of complete and incomplete traumatic SCI patients between 2002 and 2009. Patients referred to one of the 18 SCI centers were enrolled consecutively into the European Multicenter Study on Human Spinal Cord Injury (EM-SCI; www.emsci.org) database. The data are gathered to establish a multicenter basis for future therapeutic interventions in human spinal cord injury. Data within the EM-SCI are collected at five time intervals: at the initial phase (that is, within the first 15 days post injury) and 1, 3, 6 and 12 months after the injury. Clinical assessments in the EM-SCI are conducted by trained neurological and rehabilitation physicians having at least 1-year experience in examining patients with SCI. The study protocols were approved by the local ethics committees and the subjects gave their informed consent before entering the study protocol.
Study population
Patients were included in the study if they had a motor incomplete traumatic tetraplegia (C2-T1) injury (American Spinal Injury Association (ASIA) Impairment Scale (AIS) C or D)11 assessed within the first 2 weeks post-injury. The motor incomplete tetraplegia were subdivided into three study groups: (1) patients with equal or less Medical Research Council Scale points in the LEMS compared with the UEMS (non-TCCS group), (2) patients with 1–9 points more in the LEMS compared with the UEMS (intermediate or int-TCCS group) and (3) patients with 10 points or more in the LEMS compared with the UEMS (TCCS group).3
Patients with a severe cognitive impairment, peripheral nerve lesion, incomplete database record, non-traumatic spinal cord lesion, polyneuropathy or craniocerebral injury were not included. Accompanying polyneuropathy independent of SCI was excluded by medical history and by means of measuring the ulnar and tibial nerve conduction velocity.
Neurological examination
Neurological examinations were conducted according to the International Standards for the Neurological Classification of Spinal Cord Injury and the extent of incomplete tetraplegia was graded by AIS.12 Only patients with completely conducted neurological examinations within the first 2 weeks post-injury were included for analysis. The scores of the UEMS and the LEMS were calculated. Each of the two motor score subscales consist of a total of five bilateral key muscles innervated by myotomes C5-T1 (UEMS) and L2-S1 (LEMS).12 To evaluate the sensory scores of the upper and lower extremities we subdivided and calculated the light touch and pin-prick scores into upper scores (dermatomes C4-T1) and lower scores (dermatomes L1-S4-5). The level of injury and AIS were determined on the basis of the ASIA protocol.
Functional outcomes
The Spinal Cord Independence Measure (SCIM) is an instrument that focuses on performing everyday tasks, and captures the disability as well as the impact of disability on the patient's overall medical condition and comfort.13 The SCIM II14, 15 consists of three main categories, namely, (1) self-care, (2) respiration and sphincter management and (3) mobility. The functional outcomes in the chronic phase focused on the self-care items that test upper extremity function (SCIM II items 1, 2a, 3a and 4) and ambulation using the mobility indoors (SCIM II item 12). This approach of testing ambulation by using mobility indoors, has been applied in a previous study.16
In addition, the SCIM scores of items 1, 2a, 3a, 4 and 12 were converted to dichotomous outcomes, that is, able to perform the function independently or not. Patients who needed fully assisted oral feeding, a gastrostomy, parenteral feeding or were able to eat food but unable to hold a cup, were scored as dependent ‘feeding’ group (SCIM II item 1). Patients who required total or partial assistance in upper-body bathing, were scored as dependent ‘upper-body bathing’ group (SCIM II item 2a). Patients who required total or partial assistance in upper-body dressing, were scored as dependent ‘upper-body dressing’ group (SCIM II item 3a). Patients who required total assistance, performed only one task or performed some tasks using adaptive devices but needed help to put on/take off devices, were scored as dependent ‘grooming’ group (SCIM II item 4). Patients who required total assistance for their mobility, partial assistance to operate a manual wheelchair, a manual wheelchair without assistance or supervision while walking, were grouped and scored as dependent ‘mobility indoors’ group (SCIM II item 12).
Statistics
Descriptive statistics on age, gender and AIS were used to provide general information about the study population. Analyses on AIS, gender (χ2 analysis) and age (Student's t-test) were performed to identify possible differences between the three study groups.
We performed three main analyses on the study groups (see Figure 1). The first analysis was performed to demonstrate neurological impairment of motor incomplete tetraplegics as assessed within the first 2 weeks post-injury (‘initial descriptive analysis’). The second analysis was performed to demonstrate any differences between initial neurological findings (<15 days post-injury), and neurological and functional outcomes (6 or 12 months post-injury) in incomplete tetraplegic patients (‘outcome analysis’). Finally, those patients with a complete set of follow-up measurements (1, 3, and 6 or 12 months) were analysed to demonstrate the neurological recovery over time (‘longitudinal analysis’). In both the ‘outcome analysis’ and ‘longitudinal analysis’, 6-months follow-up measurements were used for analysis in patients with missing chronic phase (12 months) follow-up measurements.17, 18
Differences in mean ASIA scores between the three study groups were calculated using Student's t-tests. The differences in the number of patients who were capable of performing the SCIM II items 1, 2a, 3a or 4 independently were calculated using χ2 or Fisher's exact tests as appropriate between the three study groups. The differences were considered statistically significant at P<0.05. Spearman correlation coefficients were calculated for comparisons between 6 and 12 months UEMS and LEMS outcomes in the ‘outcome analysis’. In addition, the agreement between the 6 and 12 months outcomes of the dichotomous SCIM scores were calculated using kappa statistic (κ). Data were analysed using SPSS software (version 16.0, SPSS, Chicago, IL, USA).
Results
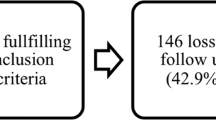
Among the 1733 traumatic SCI patients within the EM-SCI database, 248 (14%) met the study inclusion criteria, see Figure 1. Of the 248 patients, there were 89 (36%) non-TCCS patients, 62 (25%) int-TCCS and 97 (39%) TCCS patients. The patient characteristics of each study group are presented in Table 1. The non-TCCS group consisted of a significantly higher proportion of AIS grade C patients compared with patients in both the int-TCCS and TCCS groups (P<0.001). Conversely, the int-TCCS and TCCS group consisted of a significantly higher proportion of AIS grade D patients compared with patients in the non-TCCS group (P<0.001). As the number of subjects with an AIS grade C and D appeared not to be equally distributed within the three study groups, we decided to stratify each study group by the AIS grading system in all of the three main analyses. Thus, in each of the three study groups two additional subgroups, consisting of AIS grade C and D patients, were evaluated. No significant differences in age were identified between the subgroups after stratification by the AIS grading system. In addition, the analysis within every subgroup showed no differences in age between AIS grade C and AIS grade D patients.
Initial descriptive analysis
The neurological measurements assessed within the first 2 weeks post-injury were available in 248 motor incomplete tetraplegics. In the majority of the patients (72%) the NLI was situated at level C4 or C5 (see Table 2). Only four motor incomplete tetraplegics (2%) had a NLI caudal to level C6. Table 2 clearly illustrates that stratification by the AIS grading system has a bigger impact on the mean UEMS at each NLI compared with the categorization by the TCCS descriptors. Nonetheless, compared with non-TCCS subjects, TCCS subjects had lower UEMSs at each NLI within both the AIS grade C and D strata. A clear association between a more caudal NLI and higher UEMS scores was not identified. Because of the small sample sizes, some of the descriptive associations were not tested statistically.
Table 3 covers the initial neurological subscores of the motor incomplete tetraplegics. Compared with the mean UEMS of 22.2 in TCCS subjects, the initial UEMS in non-TCCS and int-TCCS groups, was significantly higher (25.9 (P=0.032) and 28.8 (P=0.001), respectively). Compared with the mean LEMS of 40.6 in TCCS subjects, the initial LEMS in non-TCCS and int-TCCS groups, was significantly lower (11.2 (P<0.001) and 33.0 (P<0.001), respectively). The differences in UEMS and LEMS hold true also after stratification by the AIS grading system.
With regard to the initial upper extremity sensory scores, no statistically significant differences were observed between the three study groups (see Table 3). Compared with AIS grade C non-TCCS patients, AIS grade C TCCS subjects had significantly higher pin-prick scores for the lower extremities (P<0.05). This difference was not observed in the light touch sensation scores in AIS grade C patients. Compared with AIS grade D non-TCCS patients, AIS grade D TCCS subjects had significantly higher pin-prick and light touch scores for the lower extremities (P<0.05).
Outcome analysis
A complete record of 12 months post-injury neurological and functional measurements was available in 121 patients (49%). In 21 patients (9%) with absent 12 months post-injury neurological and/or functional measurements, 6-months follow-up measurements were available and used for analysis. The detailed distribution of available 6 and 12 months UEMS/LEMS measurements among the 142 motor tetraplegics are presented in Supplementary Table 1. Strong and significant correlations (P<0.001) were observed between 6 and 12 months UEMS (Spearman correlation coefficients: 0.92), LEMS (Spearman correlation coefficients: 0.89) and SCIM (κ: 0.64–0.88) outcome measures in 102 patients with complete 6 and 12 month measurements.
Upper extremities
Compared with the initial UEMS, the chronic phase UEMS improved considerably in all of the three study groups (see Table 4). After stratification by the AIS grading system, the mean improvement of non-TCCS patients with an AIS grade C was 16 motor points. Compared with these non-TCCS patients, int-TCCS and TCCS patients with an AIS grade C had a significantly greater improvement of motor points in 6 or 12 months (25.2 (P<0.01) and 29.7 (P<0.001), respectively). In addition, with a mean improvement of 20.1 versus 10.0 motor points, AIS grade D TCCS patients gained significantly (P<0.001) more motor points than non-TCCS AIS grade D patients.
Except for a significant difference in AIS grade D patients (P=0.033), no significant differences were observed in the UEMS between the three study groups after 6 or 12 months (see Table 4). However, when the AIS grade C and D strata were compared within each of the three study groups clear differences in neurological outcomes were found. On average, AIS grade C patients had approximately 10 upper extremity motor points less in the chronic phase than AIS grade D patients.
Lower extremities
As for the upper extremities, the LEMS also improved considerably over time in the three subgroups, especially in the non-TCCS patients (see Table 5). After stratification by the AIS grading system, the mean improvement of non-TCCS patients with an AIS grade C was 26.1 motor points. Compared with these non-TCCS patients, int-TCCS and TCCS patients with an AIS grade C had a non-significantly different improvement of motor points in 6 or 12 months (26.2 and 19.6, respectively). In addition, the mean improvement of non-TCCS patients with an AIS grade D was 20.1 motor points. Compared with the non-TCCS patients, int-TCCS and TCCS patients with an AIS grade D gained significantly less motor points in 6 or 12 months (9.8 (P<0.001) and 5.2 (P<0.001) motor points, respectively).
However, after 6 or 12 months, non-TCCS patients had a significantly worse (P<0.001) LEMS compared with TCCS patients in the total subgroup. Nonetheless, after stratification by the AIS grading system, no differences were found between the AIS grade C patients of the three study groups. However, a significant mean difference of 3.2 motor points (P<0.001) was observed between AIS grade D non-TCCS (LEMS: 45.4) and AIS grade D TCCS (LEMS: 48.6) patients.
Functional outcomes
No clear differences were observed between the three study groups for upper-extremity function. Although AIS grade C patients were more dependent in assistance in self-care components compared with AIS grade D patients within each of the three study groups, no significant differences were observed (see Table 4).
For the ambulation outcomes, the non-TCCS group showed a significantly greater proportion of patients (P<0.001) unable to ambulate independently compared with the TCCS group. However, this statistical relation completely disappeared after stratification by the AIS grading system. Although the majority of AIS grade D patients (>93%) were able to walk independently after 6 months post injury, approximately half of the AIS grade C patients were able to do so (see Table 5).
Longitudinal analysis
A complete record of 12 months post-injury neurological and functional measurements was available in 95 patients (28%). In 16 patients (7%) with absent 12 months post-injury measurements, 6-months follow-up measurements were available and used for analysis. The detailed distribution of available 6 and 12 months UEMS/LEMS measurements among the 111 motor tetraplegics are presented in Supplementary Table 2. In Figure 2, the mean UEMS of each (stratified) study group is plotted against the timing of the follow-up assessment. After stratification by the AIS grading system, the three study groups are typically ordered from the non-TCCS group with relatively high initial mean UEMSs to the TCCS group with relatively low initial mean UEMS's. It is only after 6 months to 1 year when neurological outcomes of the three study groups approach each other. Figure 2 also demonstrates that the relation between the AIS grades and neurological outcomes appears to be stronger than the relation between the three study groups (TCCS, int-TCCS and non-TCCS) and neurological outcomes.
In Figure 3, the mean LEMS of each (stratified) study group is plotted against the timing of the follow-up assessment. In contrast to Figure 2, the three study groups are now typically ordered from the TCCS group with relatively high initial mean LEMSs to the non-TCCS with relatively low initial mean LEMSs. At three months post injury, the largest proportion of the total motor recovery was regained.
Neither in the mean UEMS nor in the mean LEMS were differences in recovery patterns found between the three study groups.
Discussion
In contrast to the general assumption that TCCS patients have a favorable neurological and functional outcome compared with other motor incomplete tetraplegics, this study demonstrates that the neurological and functional outcomes in motor incomplete tetraplegia cannot be simply attributed to the presence or absence of TCCS. We found that the severity of the initial neurological deficit, as expressed by the AIS, has a stronger impact on the prognosis of neurological and functional outcomes than categorization into TCCS or not. The presented data confirm that TCCS subjects are likely to have a less severe neurological deficit and therefore are often categorized as AIS grade D patients on admission.4, 19, 20, 21 These findings show that most differences between TCCS and non-TCCS patients dissolve when stratified by AIS grade. Although TCCS patients showed significantly higher rates of upper extremity motor strength recovery compared with non-TCCS patients, non-TCCS patients showed significantly higher rates of lower extremity motor strength recovery. These differences can be easily explained by ceiling effects in neurological recovery after traumatic SCI. On the basis of our results, we recommend that future outcome studies in patients with a traumatic motor incomplete tetraplegia use a stratification based on the AIS grading rather than the presence or absence of TCCS.10
In tetraplegic patients, recovery of arm and hand function is regarded as the most important clinical outcome.22 Although AIS grade D TCCS patients had significantly lower UEMS outcomes when compared with AIS grade D non-TCCS patients, no significant differences in the functional upper extremity independence were found between the two groups. Vice versa, whereas AIS grade D TCCS patients had significantly higher LEMS outcomes when compared with AIS grade D non-TCCS patients, no significant difference in independent ambulation was found between the two groups. Therefore, the clinical relevance of the only two identified statistical differences in outcomes between the non-TCCS and TCCS patient groups—after stratification by AIS grade—is minimal.
In 1996, Waters et al.23 suggested that the recovery of strength in TCCS patients is comparable with that of other motor incomplete tetraplegic patients. They concluded, however, that a lower proportion of the nine TCCS patients were able to walk at least 150 feet compared with non-TCCS patients. The authors’ explanation for this unexpected finding was that the residual upper extremity weakness in TCCS patients restricted the use of assisted devices such as canes and crutches and therefore limited the ability to walk.23 In contrast, we found no differences in independent ambulation outcomes between TCCS and non-TCCS patients when stratified by AIS grades. The residual upper extremity weakness in TCCS patients therefore does not appear to have a negative influence on ambulation outcomes. In fact, the lower extremity strength outcomes in TCCS patients are comparable with, or even slightly better than in, non-TCCS patients.
Our results should be interpreted in the context of specific study limitations. First, several putative confounders such as treatment regimens, including administration of methylprednisolone, blood pressure augmentation and urgent spinal cord decompression, are not standardized within the EM-SCI consortium. Second, co-morbidities, rehabilitation programs and walking aids have not been registered in detail within the EM-SCI database. Third, the small sample sizes in the three study groups resulted in limited statistical power of the analyses. Fourth, as our study population was not corrected for other SCI syndromes, these other syndromes could have been absorbed into either TCCS, non-TCCS or both. Hayes et al.19 reported that many SCI patients defy a clear-cut classification because of a mixed presentation of two or more SCI syndromes. It remains unclear what the influence is of the other SCI syndromes on the neurological and functional outcome. However, the second most common SCI syndrome, the Brown-Séquard syndrome,4 showed a similar neurological and functional outcome when compared with other incomplete SCI patients.9, 24 Finally, although the use of dichotomized SCIM outcome measures could reduce the sensitivity of the analysis and has not yet been validated, the clinical relevance and utility of this method has been demonstrated in previous studies.16, 25
The strength of this study is that, to our knowledge, it is the first one that stratified TCCS patients by applying a quantitative and reproducible TCCS diagnostic criterion. Until now the diagnosis of TCCS was based on non-specific criteria and subjective interpretation of the neurological examination.1, 11 In earlier publications (part 1 and 2), we proposed a minimum of 10 motor points in favor of the lower extremities to diagnose TCCS for research purposes.2, 3 However, among physicians, two points of discussion originated along with the introduction of this diagnostic criterion.
First, a minimum difference of 10 motor points was considered to be too high to diagnose TCCS in patients with a lower cervical level of injury. As an interesting additional demographic finding, the current study found that motor incomplete tetraplegics with a lower cervical level of injury are rare. Out of 248 patients, only four were diagnosed with a motor incomplete SCI at the NLI C7-T1. Therefore, the hypothesized limited (clinical) sensitivity of the TCCS criterion in patients with a lower cervical level of injury, as discussed in part 2, did not result in an underestimation of the number of TCCS subjects.
Second, a substantial number of physicians considered any (⩾1 motor points) difference between upper and lower extremity strength as an appropriate criterion to diagnose patients with TCCS. Although a minimum of 10 motor points was supported by the majority of the physicians in part 2, we decided to evaluate the outcomes of the so-called intermediate TCCS patient group as well.3 Nevertheless, as no apparent clinically relevant differences in neurological and functional outcomes were found between the non-TCCS and TCCS study groups after stratification by AIS grade C or D, investigation of the intermediate TCCS study group was not relevant.
In fact, the current study also challenges the scientific relevance of the applied diagnostic TCCS criterion itself. One should realize, however, that without the introduction of a face valid, quantitative and reproducible diagnostic TCCS criterion, we probably would not have been able to postulate and support the conclusions of this study. This three-paper series is unique in its kind because it evaluates, analyses and challenges the reproducibility and prognostic relevance of a commonly diagnosed SCI syndrome.4 This project clearly demonstrates that there is a need to revisit the scientific and clinical value of previously introduced concepts in SCI by applying a systematic and sound methodological approach.
Conclusion
The AIS grading system, and not the diagnosis TCCS, continues to be the most important prognostic parameter for neurological and functional outcomes in motor incomplete tetraplegics. On the basis of this study, in which a quantitative TCCS diagnostic criterion was applied, we recommend that for future outcome studies in traumatic motor incomplete tetraplegia, patients should not be stratified by the presence or absence of TCCS, but rather by the severity of the initial injury as quantified by the AIS grading system.
References
Schneider RC, Cherry G, Pantek H . The syndrome of acute central cervical spinal cord injury; with special reference to the mechanisms involved in hyperextension injuries of cervical spine. J Neurosurg 1954; 11: 546–577.
Pouw MH, van Middendorp JJ, van KA, Hirschfeld S, Veth RP et al. Diagnostic criteria of traumatic central cord syndrome. Part 1: a systematic review of clinical descriptors and scores. Spinal Cord 2010; 48: 652–656.
van Middendorp JJ, Pouw MH, Hayes KC, Williams R, Chhabra HS, Putz C et al. Diagnostic criteria of traumatic central cord syndrome. Part 2: a questionnaire survey among spine specialists. Spinal Cord 2010; 48: 657–663.
McKinley W, Santos K, Meade M, Brooke K . Incidence and outcomes of spinal cord injury clinical syndromes. J Spinal Cord Med 2007; 30: 215–224.
Roth EJ, Lawler MH, Yarkony GM . Traumatic central cord syndrome: clinical features and functional outcomes. Arch Phys Med Rehabil 1990; 71: 18–23.
Merriam WF, Taylor TK, Ruff SJ, McPhail MJ . A reappraisal of acute traumatic central cord syndrome. J Bone Joint Surg Br 1986; 68: 708–713.
Tow AM, Kong KH . Central cord syndrome: functional outcome after rehabilitation. Spinal Cord 1998; 36: 156–160.
Bosch A, Stauffer ES, Nickel VL . Incomplete traumatic quadriplegia. A ten-year review. JAMA 1971; 216: 473–478.
Wirz M, Zorner B, Rupp R, Dietz V . Outcome after incomplete spinal cord injury: central cord versus Brown-Sequard syndrome. Spinal Cord 2010; 48: 407–414.
Steeves JD, Lammertse D, Curt A, Fawcett JW, Tuszynski MH, Ditunno JF et al. Guidelines for the conduct of clinical trials for spinal cord injury (SCI) as developed by the ICCP panel: clinical trial outcome measures. Spinal Cord 2007; 45: 206–221.
Maynard Jr FM, Bracken MB, Creasey G, Ditunno Jr JF, Donovan WH, Ducker TB et al. International standards for neurological and functional classification of spinal cord injury. American Spinal Injury Association. Spinal Cord 1997; 35: 266–274.
American Spinal Injury Association. American Spinal Injury Association: International Standards for Neurological Classification of Spinal Cord Injury, revised 2002. American Spinal Injury Association: Chicago, IL, 2002.
Itzkovich M, Tripolski M, Zeilig G, Ring H, Rosentul N, Ronen J et al. Rasch analysis of the Catz-Itzkovich spinal cord independence measure. Spinal Cord 2002; 40: 396–407.
Catz A, Itzkovich M, Steinberg F, Philo O, Ring H, Ronen J et al. The Catz-Itzkovich SCIM: a revised version of the Spinal Cord Independence Measure. Disabil Rehabil 2001; 23: 263–268.
Itzkovich M, Gelernter I, Biering-Sorensen F, Weeks C, Laramee MT, Craven BC et al. The Spinal Cord Independence Measure (SCIM) version III: reliability and validity in a multi-center international study. Disabil Rehabil 2007; 29: 1926–1933.
van Middendorp JJ, Hosman AJ, Pouw MH, van de MH . Is determination between complete and incomplete traumatic spinal cord injury clinically relevant? Validation of the ASIA sacral sparing criteria in a prospective cohort of 432 patients. Spinal Cord 2009; 47: 809–816.
Curt A, Van Hedel HJ, Klaus D, Dietz V . Recovery from a spinal cord injury: significance of compensation, neural plasticity, and repair. J Neurotrauma 2008; 25: 677–685.
Waters RL, Adkins RH, Yakura JS, Sie I . Motor and sensory recovery following incomplete tetraplegia. Arch Phys Med Rehabil 1994; 75: 306–311.
Hayes KC, Hsieh JT, Wolfe DL, Potter PJ, Delaney GA . Classifying incomplete spinal cord injury syndromes: algorithms based on the International Standards for Neurological and Functional Classification of Spinal Cord Injury Patients. Arch Phys Med Rehabil 2000; 81: 644–652.
Aito S, D’Andrea M, Werhagen L, Farsetti L, Cappelli S, Bandini B et al. Neurological and functional outcome in traumatic central cord syndrome. Spinal Cord 2007; 45: 292–297.
Uribe J, Green BA, Vanni S, Moza K, Guest JD, Levi AD . Acute traumatic central cord syndrome—experience using surgical decompression with open-door expansile cervical laminoplasty. Surg Neurol 2005; 63: 505–510.
Anderson KD . Targeting recovery: priorities of the spinal cord-injured population. J Neurotrauma 2004; 21: 1371–1383.
Waters RL, Adkins RH, Sie IH, Yakura JS . Motor recovery following spinal cord injury associated with cervical spondylosis: a collaborative study. Spinal Cord 1996; 34: 711–715.
Pouw MH, van de MH, van Middendorp JJ, Hirschfeld S, Thietje R, van Kampen A et al. Relevance of the diagnosis traumatic cervical Brown-Sequard-plus syndrome: an analysis based on the neurological and functional recovery in a prospective cohort of 148 patients. Spinal Cord 2010; 48: 614–618.
Van Hedel HJ . Gait speed in relation to categories of functional ambulation after spinal cord injury. Neurorehabil Neural Repair 2009; 23: 343–350.
Acknowledgements
This study was granted by the ‘Internationale Stiftung für Forschung in Paraplegie’ (IFP). We acknowledge the contribution of the SCI centers participating in the European Multicenter Spinal Cord Injury.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Spinal Cord website
Supplementary information
Rights and permissions
About this article
Cite this article
Pouw, M., van Middendorp, J., van Kampen, A. et al. Diagnostic criteria of traumatic central cord syndrome. Part 3: Descriptive analyses of neurological and functional outcomes in a prospective cohort of traumatic motor incomplete tetraplegics. Spinal Cord 49, 614–622 (2011). https://doi.org/10.1038/sc.2010.171
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2010.171
Keywords
This article is cited by
-
Central cord syndrome definitions, variations and limitations
Spinal Cord (2023)
-
Is it time to redefine or rename the term “Central Cord Syndrome”?
Spinal Cord (2021)
-
Evaluation of a ketogenic diet for improvement of neurological recovery in individuals with acute spinal cord injury: study protocol for a randomized controlled trial
Trials (2020)
-
Degenerative cervical myelopathy — update and future directions
Nature Reviews Neurology (2020)
-
The case for revisiting central cord syndrome
Spinal Cord (2020)