Abstract
Study design:
This study was designed as an experimental study (trial).
Objectives:
To verify the effects of the association between conventional pharmacological treatment and osteopathic manipulative treatment (OMT) for chronic pain management in spinal cord injury (SCI).
Setting:
This study was carried out at Spinal Unit, Ospedale Niguarda Ca’ Granda, Milan, Italy. Istituto Superiore di Osteopatia, Milan, Italy.
Methods:
We enrolled 47 patients with SCI, 26 with pain of both nociceptive and neuropathic origin, and 21 with pure neuropathic pain. In all, 33 patients had a complete spinal cord lesion (ASIA level A) and 14 had incomplete lesion (ASIA level B, C and D). The patients were subdivided in a pharmacological group (Ph), a pharmacological osteopathic (PhO) group and a osteopathic (Os) group. The verbal numeric scale (VNS) was used at various time intervals to evaluate treatment outcomes.
Results:
Ph patients reached a 24% improvement in their pain perception, assessed by the VNS scale after 3 weeks of treatment, whereas Os patients reached a 16% improvement in their pain perception for the same weeks. Both treatments per se failed to induce further improvements at later time points. In contrast, the combination of the two approaches yielded a significantly better pain relief both in patients with nociceptive or pure neuropathic pain in the PhO group.
Conclusions:
Our results suggest the OMT is a feasible approach in patients in whom available drugs cannot be used. Moreover, a benefit can be expected by the association of OMT in patients treated according to existing pharmacological protocols.
Similar content being viewed by others
Introduction
Pain in patients with spinal cord injury (SCI) is a common occurrence, with an incidence ranging between 65 and 80% of the subjects. One-third of these patients experienced severe pain. Despite pain, patients developed allodynia and hyperalgesia.1 Symptoms potentially originate in any moment of the patient history influencing patient psychological and social functioning.2, 3 It affects quality of life, causes a substantial morbidity, with worsening of the disability and reduced involvement in rehabilitation programs.4
The International Association of the Study of Pain established a ‘Spinal Cord Injury Pain Task Force’ to classify pain in the SCI. They identified two main types of pain: nociceptive and neuropathic2, 5 (Table 1).
The first study on the pathophysiological mechanism of pain associated to SCI dates back to 1950.6 Since then, the literature has produced evidence based mostly on the assumption that pain arises from changes in damaged nerve roots or changes in the brain.1, 7 Recent observations reveal a role for mediators of inflammation, such as cytokines and prostaglandins, in sustaining chronic central neuropathic pain.1 Furthermore, the perception of pain may be exacerbated by the loss of spinal and thalamic inhibition and the structural reorganization of inputs in the dorsal horn of the spinal cord.8
Several therapeutic strategies are used, including pharmacological treatment (analgesics, opioids and non-steroidal anti-inflammatory drugs). Management of pain in these patients is difficult and complete recovery is rare.9
Osteopathic manipulative treatment (OMT) is efficacious for the relief of chronic pain related to osteoarthritis and/or inflammatory conditions.10 OMT functions on the induction of small molecules, including endocannabinoids, endogenous opioids and serotonin, which have a non-redundant role on the inflammation and pain associated to the somatic dysfunction.11 In particular, myofascial release is associated to enhanced levels of β-endorphin and conversely reduced generation of serotonin and arachidonoylethanolamide.12 These combined effects are possibly associated to a virtuous circle that maintains the benefit caused by OMT.12 Clinical trials on OMT in patients with SCI-related pain, to the best of our knowledge, have not been published.
The objective of this study was to verify the efficacy of combining OMT and pharmacological therapies on the control chronic pain in patients with SCI.
Methods
Subjects
In all, 47 patients were recruited on informed consent from the Spinal Unit of the Niguarda Ca’ Granda Hospital, in Milan, inclusion criteria were an age between 18 and 60 years, and a lesion level between C5 and L5 evaluated with standard neurological classification of SCI (ASIA impairment scale A, B, C, D and E).13 Only patients with a traumatic SCI that has been stable at least for the last 6 months were recruited; no further exclusion criteria based on the time since the initial SCI were used. In all, 26 patients were selected by a medical team composed by anesthetists and physiatrists: 19 had paraplegia and 7 tetraplegia, 16 had neuropathic pain (10 below level lesion and 6 at the level lesion) and 10 had nociceptive pain above level lesion. In all, 21 patients had a complete spinal cord lesion (ASIA level A) and 5 had an incomplete lesion (ASIA level B). In all, 21 patients with pure neuropathic pain were also recruited. Diagnosis was carried out using the leeds assessment of neuropathic symptoms and signs (LANSS) scale.14 A total of 12 patients had a complete spinal cord lesion (ASIA level A) and 9 had an incomplete lesion (3 ASIA level B, 4 ASIA level C and 2 ASIA level D). In all, 14 patients had pain below level lesion and 7 had at the level lesion. Scores between 6 and 10, measured with verbal numeric scale (VNS) at the beginning of the study (time T0), had to be obtained. VNS15 is a validated scale to measure the intensity of pain with numbers between 0 and 10, with 0 representing absence of pain and 10 the worst possible pain experienced by the patient. The type of pain (nociceptive versus neuropathic) was determined by expert clinicians (anesthetists): clinical examination was carried out, comprising classification of SCI-related pain (pain description according to type, region presumed mechanism), pain perception (intensity, sensory description) and pain interference (impact on daily life/activity). All patients were treated with anti-depressant. Osteopathic treatments were carried out at the Istituto Superiore di Osteopatia, Milano, Italy.
Study
In all, 26 patients were randomized into three groups. The three groups were evaluated eight times, that is, after 1 (T1), 3 (T2), 8 (T3), 12 (T4), 13 (T5), 16, (T6), 20 (T7) and 24 (T8) weeks from the beginning of the study. In the first group, patients underwent pharmacological treatment for the entire study period (pharmacological group (Ph)). In the second group, patients underwent pharmacological treatment until improvement stabilization. Thereafter, they were treated with both drugs and OMT (pharmacological osteopathic group (PhO)). In the third group, patients were treated with OMT only for the entire study period (osteopathic group (Os)).
The 21 patients with pure neuropathic pain were randomized into the three groups (Ph, Os and PhO). The three groups were evaluated after 1 (T1), 3 (T2) and 8 (T3) weeks of treatment.
Pharmacological treatment protocol
The pharmacological treatment of patients in groups Ph and PhO was decided by the anesthetists depending on the origin of pain. The groups were matched for the fraction of patients experiencing nociceptive or neuropathic pain; neuropathic pain was treated with pregabalin (600 mg per day). Nociceptive pain was treated with non-steroidal anti-inflammatory drug (paracetamol 2–4 g per day).
Osteopathical treatment protocol
The OMT protocol was designed for each patient on the basis of the somatic dysfunctions identified during objective exam, as described.11 Myofascial release, strain–counterstrain, muscle energy, soft tissue and cranial sacral approach were used in any visit. We did not use high velocity low amplitude (thrust) because of the potential side effects in patients with SCI, such as vertebral fractures, surgical fixation, spasticity and flaccidity.
The OMT was repeated once a week during the first month, once every fortnight during the second month, once during the third month, for a total of seven treatments. Duration of each treatment session was of 45 min. The patients were treated by an osteopathic physician.
Statistical analysis
Data analysis was carried out using SPSS (version 17.0, Magnetic Media, Milano, Italy). To verify group homogeneity and estimate variance equality among the three groups Levene's test was performed at the time T0. A repeated-measures analysis of variance was used to compare the treatment efficacy in time and among the groups (Ph, O and PhO).
The VNS evaluations were analyzed first at the times T0–T1–T2–T3–T4 and then at the times T5–T6–T7–T8. At the end, Tukey's honestly significantly different test was used in multiple comparisons at the times T5–T6–T7–T8 to evaluate which group significantly differed from the others. P<0.05 was considered statistically significant and P<0.01 highly statistically significant.
Results
In all, 26 patients, 22 males and 4 females, with a mean age of 36±9.95 years and a lesion time equal to 4±4.47 years were recruited. In all, 21 patients had a complete spinal cord lesion (ASIA A) and 5 had incomplete lesion (ASIA B; Table 2).
The mean VNS scores were 8.70±0.67 for Ph, 8.60±0.84 for PhO and 8.92±0.92 for Os at T0 (Table 3). The three groups of patients were, therefore, homogenous for pain perception at the time of enrollment (P=0.62).
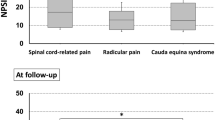
The repeated-measures analysis of variance at times T0–T1–T2–T3–T4 revealed a progressive reduction of pain perception in all the three groups of patients (P<0.01). The VNS scores were at T1 between 6.85±0.82 in the Ph group, 7.10±0.87 in the PhO group and 8.17±1.17 in the Os group. At T3 the mean VNS scores further reduced to 5.50±1.08 in the Ph group, 5.70±0.82 in the PhO group and 6.17±0.75 in the Os group. Results are shown in Table 4. The three treatments yielded similar rates of improvements, as demonstrated by the analysis of variance analysis comparing the time intervals T0–T4 (P=0.26). However, as shown in Figure 1, the pharmacological treatment (Ph and PhO groups) resulted in a faster improvement than the OMT (Os group).
The combined treatment with drugs and OMT (PhO) started when we failed to detect the significant differences in VNS scores at two consecutive time points: this occurred between T3 and T4 (P=0.57; Table 5). Patients in PhO group yielded a significant better pain relief at T5–T6–T7 compared with patients in the other groups (P=0.04 versus Ph and P=0.03 versus Os). At the same times, VNS values in the Ph and Os groups remained constant, without any significant difference (P=0.88). The PhO group VNS scores were 5.65±0.75 and 3.80±1.14 at T5 and T7, respectively (Figure 2). Later (T8), we failed to detect any further improvement (T7 3.8±1.14; T8 3.70±1.06; P=0.28). Table 6 reports the actual VNS data.
To further evaluate the efficacy of the combination between pharmacological therapy and OMT in patients with pure neuropathic pain, we enrolled 21 patients, selected after evaluation with the LANSS scale. Characteristics of the patients are depicted in the Table 2. There were 17 males and 4 females, with a mean age of 41±13.36 years and a lesion time equal to 8±2.57 years. A total of 13 patients had a complete spinal cord lesion (ASIA level A) and 9 had an incomplete lesion (3 ASIA level B; 4 ASIA level C and 2 ASIA level D).
The mean VNS scores at T0 were 6.35±0.90 for Ph, 6.50±1.08 for PhO and 6.75±1.04 for Os (Table 7), the three groups of patients were homogenous for pain perception at the time of enrollment (P=0.75).
The repeated-measures analysis of variance at times T0–T1–T2–T3 revealed a progressive reduction of pain perception in the three groups of patients (P<0.01). The VNS scores at T1 were 5.50±1.22 in the Ph group, 4.78±1.35 in the PhO group and 6.31±0.96 in the Os group. At T3 the mean VNS scores further decreased to 3.92±0.67 in the Ph group, 2.92±0.67 in the PhO group and 4.62±0.74 in the Os group. Results are shown in Table 7. The three treatments yielded similar rates of improvements, as demonstrated comparing the results obtained at T0–T3 (P=0.26). Patients in PhO group experienced a significantly higher pain relief compared with patients in the other groups (P=0.05 versus Ph and P=0.001 versus Os). At the same time points, VNS values in the Ph and Os groups had a similar improvement, without any significant difference. The PhO group VNS scores were as 4.78±1.35 and 2.92±0.67 at T1 and T3, respectively.
Discussion
This study had the objective to verify the effects of the association between conventional pharmacological treatment and OMT for chronic pain management in SCI. At the beginning, the efficacy of OMT was compared with the efficacy of the pharmacological treatment. The results obtained clearly indicate that both drugs and OMT were effective on pain (P<0.001). Moreover, a direct comparison between the two treatments reveals a similar pain improvement, as no significant difference was detected after 12 weeks of treatment (P=0.57). The clinical relevance of significant difference in VNS scores, is an improvement of back and shoulder mobility. This is important for their wheelchair movement.
Therefore, OMT could represent a feasible treatment for pain management, in those patients that experience antiepileptic drug toxicity (gait unsteadiness, cerebellar toxicity, alterations of vestibular function, cognition, and blood pressure regulation with postural changes, ataxia and imbalance) or that cannot be treated with non-steroidal anti-inflammatory drugs because of the risk of bleeding or of hepatic toxicity.
An efficacy of OMT similar to that of non-steroidal anti-inflammatory drugs has been previously reported by Andersson et al.16 in patients with low back pain. To the best of our knowledge, no further comparison has been published; in particular, no evidence exists on the efficacy of OMT in SCI.
We also report that patients in whom pharmacological treatment was associated with OMT obtained a further significant improvement. Similar data were obtained in different clinical settings,17 including in particular the pain associated with surgery in women undergoing total abdominal hysterectomy.18 In these conditions, OMT associated with pharmacological treatment reduced the use of analgesics, anti-inflammatory, miorelaxation and physical therapies.
OMT causes changes in the release of pain endogenous biomarkers, such as opioids (endorphin and endocannabinoids), and serotonin with involvement of serotoninergic and noradrenergic descending tracts.12 A similar mechanism has also been proposed for the anti-hyperalgesia produced by joint manipulation.19 Moreover, the OMT results in an increase of concentration of circulating opioids, and this allows an improvement of the anti-nociceptive properties and analgesic effect of pain, a natural pain modulation.20 Therefore, a possible explanation for the efficacy of the treatment is an enhancement of the action of the administered opioids. However, we observe a definite improvement in patients that were not assuming exogenous opioids. The molecular bases of the results are so far largely unknown, even if a potential effect on endogenous opioid cannot be ruled out. Further studies are warranted to verify the effects of the combined treatment in SCI patients on the concentration of serotonin and endocannabinoids.
References
Hulsebosch CE, Hains BC, Crown ED, Carlton SM . Mechanisms of chronic central neuropathic pain after spinal cord injury. Brain Res Rev 2009; 60: 202–213.
Siddall PJ, McClelland JM, Rutkowski SB, Cousins MJ . A longitudinal study of the prevalence and characteristics of pain in the first 5 years following spinal cord injury. Pain 2003; 103: 249–257.
Nicholson Perry K, Nicholas MK, Middleton J, Siddall P . Psychological characteristics of people with spinal cord injury-related persisting pain referred to a tertiary pain management center. J Rehabil Res Dev 2009; 46: 57–67.
Ragnarsson KT . Management of pain in persons with spinal cord injury. J Spinal Cord Med 1997; 20: 186–199.
Widerstrom-Noga E, Biering-Sorensen F, Bryce T, Cardenas DD, Finnerup NB, Jensen MP et al. The international spinal cord injury pain basic data set. Spinal Cord 2008; 46: 818–823.
Pollock LJ, Brown M, Boshes B, Finkelman I, Chor H, Arieff AJ et al. Pain below the level of injury of the spinal cord. AMA Arch Neurol Psychiatry 1951; 65: 319–322.
Hains BC, Waxman SG . Sodium channel expression and the molecular pathophysiology of pain after SCI. Prog Brain Res 2007; 161: 195–203.
Gerke MB, Duggan AW, Xu L, Siddall PJ . Thalamic neuronal activity in rats with mechanical allodynia following contusive spinal cord injury. Neuroscience 2003; 117: 715–722.
Cardenas DD, Jensen MP . Treatments for chronic pain in persons with spinal cord injury: A survey study. J Spinal Cord Med 2006; 29: 109–117.
Licciardone JC, Brimhall AK, King LN . Osteopathic manipulative treatment for low back pain: a systematic review and meta-analysis of randomized controlled trials. BMC Musculoskelet Disord 2005; 6: 43.
Kuchera ML . Applying osteopathic principles to formulate treatment for patients with chronic pain. J Am Osteopath Assoc 2007; 107 (10 Suppl 6): ES28–ES38.
Degenhardt BF, Darmani NA, Johnson JC, Towns LC, Rhodes DC, Trinh C et al. Role of osteopathic manipulative treatment in altering pain biomarkers: a pilot study. J Am Osteopath Assoc 2007; 107: 387–400.
Maynard Jr FM, Bracken MB, Creasey G, Ditunno Jr JF, Donovan WH, Ducker TB et al. International Standards for Neurological and Functional Classification of Spinal Cord Injury. American Spinal Injury Association. Spinal Cord 1997; 35: 266–274.
Bennett M . The LANSS pain scale: the Leeds assessment of neuropathic symptoms and signs. Pain 2001; 92: 147–157.
Dixon JS, Bird HA . Reproducibility along a 10 cm vertical visual analogue scale. Ann Rheum Dis 1981; 40: 87–89.
Andersson GB, Lucente T, Davis AM, Kappler RE, Lipton JA, Leurgans S . A comparison of osteopathic spinal manipulation with standard care for patients with low back pain. N Engl J Med 1999; 341: 1426–1431.
Przewlocki R, Przewlocka B . Opioids in chronic pain. Eur J Pharmacol 2001; 429: 79–91.
Goldstein FJ, Jeck S, Nicholas AS, Berman MJ, Lerario M . Preoperative intravenous morphine sulfate with postoperative osteopathic manipulative treatment reduces patient analgesic use after total abdominal hysterectomy. J Am Osteopath Assoc 2005; 105: 273–279.
Skyba DA, Radhakrishnan R, Rohlwing JJ, Wright A, Sluka KA . Joint manipulation reduces hyperalgesia by activation of monoamine receptors but not opioid or GABA receptors in the spinal cord. Pain 2003; 106: 159–168.
McPartland JM, Giuffrida A, King J, Skinner E, Scotter J, Musty RE . Cannabimimetic effects of osteopathic manipulative treatment. J Am Osteopath Assoc 2005; 105: 283–291.
Acknowledgements
This study was not possible without the precious and professional aide of Patrizia Rovere-Querini and Angelo Manfredi. We also gratefully acknowledge the volunteers of Spinal Unit of A.O. Ospedale Niguarda Ca’ Granda, Milan, Italy that accepted to participate to this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Arienti, C., Daccò, S., Piccolo, I. et al. Osteopathic manipulative treatment is effective on pain control associated to spinal cord injury. Spinal Cord 49, 515–519 (2011). https://doi.org/10.1038/sc.2010.170
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2010.170
Keywords
This article is cited by
-
The CanPain SCI clinical practice guidelines for rehabilitation management of neuropathic pain after spinal cord injury: 2021 update
Spinal Cord (2022)
-
The CanPain SCI Clinical Practice Guidelines for Rehabilitation Management of Neuropathic Pain after Spinal Cord: Recommendations for treatment
Spinal Cord (2016)
-
Use of complementary and alternative medicine in persons with spinal cord injury in Switzerland: a survey study
Spinal Cord (2015)
-
Management of Neuropathic Pain Associated with Spinal Cord Injury
Pain and Therapy (2015)
-
Molecular targeting of NOX4 for neuropathic pain after traumatic injury of the spinal cord
Cell Death & Disease (2012)