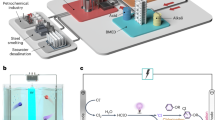
Abstract
Coagulation is one of the most common treatment processes for the removal of contaminants from water, representing ‘the first line of defence’ for drinking water safety. However, the macroscopic and descriptive theories of trivalent metal-based coagulation have limited the optimization of its performance in the removal of natural organic matter species, which are major precursors of hazardous disinfection by-products. In this study, we have extended the coagulation theory to the functional group level, highlighting the fundamental importance of η-H2O and η-OH groups on aluminium precipitates, and finding that the selectivity for natural organic matter during coagulation is determined by their functional groups. Drawing upon the fundamental characteristics of coagulants and organic substances, this study has elucidated their behaviour during the coagulation process and offers valuable theoretical insights to guide future applications of coagulation in practical settings.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information.
References
Gan, Y., Zhang, L. & Zhang, S. The suitability of titanium salts in coagulation removal of micropollutants and in alleviation of membrane fouling. Water Res. 205, 117692 (2021).
Cheng, X. et al. Coupling sodium percarbonate (SPC) oxidation and coagulation for membrane fouling mitigation in algae-laden water treatment. Water Res. 204, 117622 (2021).
Cai, G. et al. Control for chlorine resistant spore forming bacteria by the coupling of pre-oxidation and coagulation sedimentation, and UV-AOPs enhanced inactivation in drinking water treatment. Water Res. 219, 118540 (2022).
Du, P. et al. Regulated-biofilms enhance the permeate flux and quality of gravity-driven membrane (GDM) by in situ coagulation combined with activated alumina filtration. Water Res. 209, 117947 (2022).
Yu, W., Graham, N. J. D. & Fowler, G. D. Coagulation and oxidation for controlling ultrafiltration membrane fouling in drinking water treatment: application of ozone at low dose in submerged membrane tank. Water Res. 95, 1–10 (2016).
Jiang, J.-Q. The role of coagulation in water treatment. Curr. Opin. Chem. Eng. 8, 36–44 (2015).
Zhang, X., Graham, N., Xu, L., Yu, W. & Gregory, J. The influence of small organic molecules on coagulation from the perspective of hydrolysis competition and crystallization. Environ. Sci. Technol. 55, 7456–7465 (2021).
Su, Z. et al. Discovery of welcome biopolymers in surface water: improvements in drinking water production. Environ. Sci. Technol. 55, 2076–2086 (2021).
An, G. et al. Deprotonation and aggregation of Al13 under alkaline titration: a simulating study related to coagulation process. Water Res. 203, 117562 (2021).
Li, M. et al. Effects of pre-oxidation on residual dissolved aluminum in coagulated water: a pilot-scale study. Water Res. 190, 116682 (2021).
Liu, B. et al. Coagulation behavior of polyaluminum–titanium chloride composite coagulant with humic acid: a mechanism analysis. Water Res. 220, 118633 (2022).
Jarvis, P. et al. Comparison of coagulation performance and floc properties using a novel zirconium coagulant against traditional ferric and alum coagulants. Water Res. 46, 4179–4187 (2012).
Mattson, S. Cataphoresis and the electrical neutralization of colloidal material. J. Phys. Chem. 32, 1532–1552 (1928).
Abeysinghe, S., Unruh, D. K. & Forbes, T. Z. Crystallization of Keggin-type polyaluminum species by supramolecular interactions with disulfonate anions. Cryst. Growth Des. 12, 2044–2051 (2012).
Bi, S., Wang, C., Cao, Q. & Zhang, C. Studies on the mechanism of hydrolysis and polymerization of aluminum salts in aqueous solution: correlations between the ‘core-links’ model and ‘cage-like’ Keggin-Al13 model. Coord. Chem. Rev. 248, 441–455 (2004).
Tang, H., Xiao, F. & Wang, D. Speciation, stability, and coagulation mechanisms of hydroxyl aluminum clusters formed by PACl and alum: a critical review. Adv. Colloid Interface Sci. 226, 78–85 (2015).
Banfield, J. F., Welch, S. A., Zhang, H. Z., Ebert, T. T. & Penn, R. L. Aggregation-based crystal growth and microstructure development in natural iron oxyhydroxide biomineralization products. Science 289, 751–754 (2000).
Yukselen, M. A. & Gregory, J. The reversibility of floc breakage. Int. J. Miner. Process. 73, 251–259 (2004).
Jin, X., Liao, R., Zhang, T. & Li, H. Theoretical insights into the dimerization mechanism of aluminum species at two different pH conditions. Inorg. Chim. Acta 520, 120311 (2021).
Yu, W., Gregory, J. & Campos, L. C. Breakage and re-growth of flocs: effect of additional doses of coagulant species. Water Res. 45, 6718–6724 (2011).
Han, Q. et al. Trihalomethanes (THMs) precursor fractions removal by coagulation and adsorption for bio-treated municipal wastewater: molecular weight, hydrophobicity/hydrophily and fluorescence. J. Hazard. Mater. 297, 119–126 (2015).
Kim, H.-C. & Yu, M.-J. Characterization of natural organic matter in conventional water treatment processes for selection of treatment processes focused on DBPs control. Water Res. 39, 4779–4789 (2005).
Mecozzi, S., West, A. P. & Dougherty, D. A. Cation−π Interactions in simple aromatics: electrostatics provide a predictive tool. J. Am. Chem. Soc. 118, 2307–2308 (1996).
Ma, J. C. & Dougherty, D. A. The cation−π interaction. Chem. Rev. 97, 1303–1324 (1997).
Casey, W. H. Large aqueous aluminum hydroxide molecules. Chem. Rev. 106, 1–16 (2006).
Guo, K. et al. Highly efficient Al–Ti gel as a coagulant for surface water treatment: insights into the hydrolysate transformation and coagulation mechanism. Water Res. 221, 118826 (2022).
Yu, W.-Z. et al. Aggregation of nano-sized alum–humic primary particles. Sep. Purif. Technol. 99, 44–49 (2012).
Hopkins, D. C. & Ducoste, J. J. Characterizing flocculation under heterogeneous turbulence. J. Colloid Interface Sci. 264, 184–194 (2003).
Xiao, F., Ma, J., Yi, P. & Huang, J. C. H. Effects of low temperature on coagulation of kaolinite suspensions. Water Res. 42, 2983–2992 (2008).
Huguet, A. et al. Properties of fluorescent dissolved organic matter in the Gironde Estuary. Org. Geochem. 40, 706–719 (2009).
Gregory, J. & Nelson, D. W. Monitoring of aggregates in flowing suspensions. Colloids Surf. 18, 175–188 (1986).
Su, Z., Li, X. & Yang, Y. Regrowth ability and coagulation behavior by second dose: breakage during the initial flocculation phase. Colloids Surf. A 527, 109–114 (2017).
Zhang, B., Shan, C., Wang, S., Fang, Z. & Pan, B. Unveiling the transformation of dissolved organic matter during ozonation of municipal secondary effluent based on FT-ICR-MS and spectral analysis. Water Res. 188, 116484 (2021).
Johansson, G. The crystal structures of [Al2(OH)2(H2O)8](SO4)2·2H2O and [Al2(OH)2(H2O)8](SeO4)2·2H2O. Acta Chem. Scand. 16, 403–420 (1962).
Johansson, G., Lundgren, G., Sillén, L. & Sderquist, R. J. On the crystal structure of a basic aluminum sulfate and the corresponding selenate. Acta Chem. Scand. 14, 769–771 (1960).
Pascual-Cosp, J., Artiaga, R., Corpas-Iglesias, F. & Benítez-Guerrero, M. Synthesis and characterization of a new aluminium-based compound. Dalton Trans. 28, 6299–6308 (2009).
Wang, W., Wentz, K. M., Hayes, S. E., Johnson, D. W. & Keszler, D. A. Synthesis of the hydroxide cluster [Al13(μ3-OH)6(μ-OH)18(H2O)24]15+ from an aqueous solution.Inorg. Chem. 50, 4683–4685 (2011).
Wu, M., Yu, W., Qu, J. & Gregory, J. The variation of flocs activity during floc breakage and aging, adsorbing phosphate, humic acid and clay particles. Water Res. 155, 131–141 (2019).
Liu, M. & Yu, W. Surface chemical groups of flocs are key factors for the growth of flocs in sweep coagulation: a case study of surface occupation by humic acid. ACS ES T Eng. 2, 2301–2310 (2022).
Acknowledgements
This work was financially supported by the Key Research and Development Plan of the Chinese Ministry of Science and Technology (grant no. 2019YFC1906501, W.Y.) and Beijing Natural Science Foundation (grant no. JQ21032, W.Y.).
Author information
Authors and Affiliations
Contributions
W.Y., N.G. and M.E. designed the experiments. M.L. completed the main experiments and wrote the first draft of the paper. W.Y., N.G., J.G. and M.E. made the final revisions to the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Water thanks Gregory Korshin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Notes 1–6, Figs. 1–16, and Tables 1 and 2.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, M., Graham, N., Gregory, J. et al. Towards a molecular-scale theory for the removal of natural organic matter by coagulation with trivalent metals. Nat Water 2, 285–294 (2024). https://doi.org/10.1038/s44221-024-00212-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44221-024-00212-x