Abstract
Infections by drug-resistant microorganisms are a threat to global health and antimicrobial peptides are considered to be a new hope for their treatment. Temporin-WY2 was identified from the cutaneous secretion of the Ranidae frog, Amolops wuyiensis. It presented with a potent anti-Gram-positive bacterial efficacy, but its activity against Gram-negative bacteria and cancer cell lines was unremarkable. Also, it produced a relatively high lytic effect on horse erythrocytes. For further improvement of its functions, a perfect amphipathic analogue, QUB-1426, and two lysine-clustered analogues, 6K-WY2 and 6K-1426, were synthesised and investigated. The modified peptides were found to be between 8- and 64-fold more potent against Gram-negative bacteria than the original peptide. Additionally, the 6K analogues showed a rapid killing rate. Also, their antiproliferation activities were more than 100-fold more potent than the parent peptide. All of the peptides that were examined demonstrated considerable biofilm inhibition activity. Moreover, QUB-1426, 6K-WY2 and 6K-1426, demonstrated in vivo antimicrobial activity against MRSA and E. coli in an insect larvae model. Despite observing a slight increase in the hemolytic activity and cytotoxicity of the modified peptides, they still demonstrated a improved therapeutic index. Overall, QUB-1426, 6K-WY2 and 6K-1426, with dual antimicrobial and anticancer functions, are proposed as putative drug candidates for the future.
Similar content being viewed by others
Introduction
Inappropriate handling of antibiotics has already resulted in the rapid selection, creation and dissemination of multi-drug resistant bacteria, which pose a serious danger to world health1. The story of penicillin can serve as a useful cautionary tale about how quickly drug resistance can emerge. In 1943, during World War 2, penicillin was first administered to patients. However, the first strain of Staphylococcus that was resistant to penicillin was discovered in 19482. According to the WHO, 10 million fatalities and a 2.5% to 3.5% decline in yearly GDP might occur from antibiotic resistance if it continues to spread at its current rate for 25 years3,4.
The WHO has classified some multidrug-resistant bacteria as "priority pathogens" or "ESKAPE pathogens," (including Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species), which pose a major risk to worldwide health5. The six ESKAPE pathogens include four Gram-negative bacteria. These bacteria have historically been more difficult to kill due to the structure of their cell walls and their increased density of drug efflux pumps. Thus, the quest for anti-Gram-negative bacterial agents is particularly important and indeed pressing5.
Antimicrobial peptides (AMPs) are now gaining popularity as alternative antimicrobial agents for treating infections that are resistant to antibiotics due to their unique modes of broad spectrum action and rapid killing of bacteria. Many peptide-based bactericides are currently available on the market such as Bacitracin, Dalbavancin, Colistin and Polymyxin B6.
Polymyxin was regarded as the last line treatment for the multidrug-resistant Gram-negative pathogen infections, in particular for Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacterales. Natural polymyxins were originally isolated from the Gram-positive, spore-producing bacterium, Paenibacillus polymyxa (also known as Bacillus polymyxa). They were first discovered in the 1940s and applied in the 1950s, but soon were abandoned for their nephrotoxicity and neurotoxicity7. Research on polymyxin was put on hold until recently. Several clinical trials have been carried out to attempt to overcome the toxicity and clinical shortage of polymyxins, and many polymyxin-like lipopeptides have been discovered7. Polymyxins initially target the lipid A of LPS on the outer membrane of Gram-negative bacteria. The amphipathicity of polymyxins is crucial for subsequent cell membrane penetration8. They are efficient in treating Gram-negative, typically antibiotic-resistant bacteria-related illnesses of the gastrointestinal and circulatory systems.
Temporins are one of the biggest AMP families yet found and have been well studied. Temporin peptides are ultra-short peptides, normally with 10–14 amino acids, forming a typical α-helical structure, with a C-terminal amidation without a Rana box, and normally have a better function against Gram-positive bacteria and the biofilms9,10,11,12. Their ultra-short length and potent function has made them a common focus for research.
This investigation led to the discovery of Temporin-WY2, a novel peptide from the skin secretion of the frog, Amolops wuyiensis. By using solid-phase peptide synthesis, reverse-phase HPLC, and MALDI-TOF mass spectrometry, this novel peptide was synthesized and purified. Moreover, three modified analogues were designed in an attempt to provide the peptide with multiple functions. The antibacterial activity of the peptide was assessed using a variety of bioassays and in addition, cancer cell lines were employed to assess putative antiproliferation activity. Antimicrobial efficacy evaluation in insect larvae models was conducted to study the potential in vivo effects of the peptides.
Results
Molecular cloning of Temporin-WY2
Using Dynabeads® mRNA Direct Kit, the mRNA in the Amolops wuyiensis frog skin secretions were successfully isolated. Then the isolated mRNA was applied as the template to amplify and construct the cDNA library. Following with the 3′-RACE PCR, the desired cDNA fragments were isolated, purified and analysed. The results from the DNA sequencing and alignment indicated that, the open reading frame (ORF) of the novel peptide consisted of 62 amino acid residues, including a signal peptide of 22 amino acid residues, a following spacer peptide containing 25 amino acid residues, and a mature peptide containing 13 residues (Fig. 1).
Subsequent peptide alignment with related peptides in the database was achieved by use of the NCBI-BLAST programme (https://www.ncbi.nlm.nih.gov/, accessed on 30 Oct 2021). After sequencing, the sequences of mature peptides and the open reading frames, were submitted to the BLASTp tool. Five sequences were found in the alignment of the open reading frame, which had high identities ranging from 72.58 to 85.48%. No peptide was found with a similarity of 100%, which meant Temporin-WY2 was a novel peptide. The peptide with the highest similarity belongs to the Temporin family, which meant Temporin-WY2 belongs to this family (Fig. 2).
Alignment of ORF amino acid sequences of precursors of Temporin-WY2, temporin-1KM, temporin-1GY, preprobrevinin-1SSd and antimicrobial peptide precursor. The precursors each contain four domains: (1) a putative N-terminal signal peptide region; (2) an acidic “spacer” peptide region; (3) a dibasic residue pro-peptide convertase cleavage site; (4) a mature peptide region. The asterisks indicate identical amino acid residues. The cDNA sequence of the Temporin-WY2 precursor has been deposited in the NCBI Database under an accession number: OQ847420.
Physicochemical properties analyses, secondary structure prediction and confirmation
The sequence of the novel peptide, Temporin-WY2, was established as: FLPFLASLALKIL-NH2. It was regarded as the template for further peptide modification. Peptide modification was performed to enhance the antimicrobial and anticancer activity of the peptide, as well as to reduce its haemolytic action. The other three peptides were designed knowing that amphipathicity, charge, and hydrophobicity are critical parameters that have important impacts on antimicrobial activity. To elucidate the influence of those factors on antimicrobial activity, QUB-1426 was designed. Compared with parent peptide, Temporin-WY2, QUB-1426 showed a lower hydrophobicity of 0.878H as the result of the substitution of a phenylalanine residue with a lysine residue in position four. Also, it had a long cluster of hydrophilic and hydrophobic residues due to the position swapping between the alanine residue on position eight and the leucine on position nine, and between the alanine residue on position six and the leucine on position ten. 6K-WY2 and 6K-1426 contain two lysine cluster sequences formed by adding six lysine residues at the N-terminus of Temporin-WY2 and QUB-1426.
The peptide sequences are listed in the table below. The purified peptides are identified by analytical HPLC and MALDI-TOF MS Spectrum (Suppl. Fig 1-8). The physicochemical properties were accessed from the Heliquest programme. Temporin-WY2 has the highest hydrophobicity of 1.092H whereas 6K-1426 has the lowest hydrophobicity of 0.288H. On the contrary, 6K-1426 has the highest number of positive charges of + 9, as the result of the six lysine residue cluster, while Temporin-WY2 has the least. The hydrophobic moment is a parameter that reflects the amphipathicity of the peptide. QUB-1426 with the highest hydrophobic moment revealed the highest amphipathicity among all the tested peptides (Table 1).
Secondary structure prediction of Temporin-WY2 and its analogues was conducted by using the PEP-FOLD3 programme. Both results indicated that all the peptides tended to have an α-helix structure (Fig. 3).
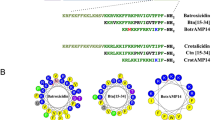
Predicted 3D secondary structures of the peptides from PEP-FOLD3 (above) and Helical wheel projection of the peptides (bottom) [(a) Temporin-WY2, (b) QUB-1426, (c) 6K-WY2, (d) 6K-1426]. In the predicted 3D structure figures, the N-terminus and C-terminus of the peptides were marked in blue and red, respectively. In the helical wheel projections, the C-terminus and N-terminus are indicated by “C” and “N”, respectively. The arrow in the middle of the circle indicates the hydrophobic direction formed by amino acids with high hydrophobicity. The yellow group represents the hydrophobic surface. The grey group represents the neutral amino acids. The blue group represents the polar amino acid residues.
The helical wheel projection was created by the Heliquest programme (Fig. 3). QUB-1426 presented a cluster of hydrophobic residues on the hydrophobic side owing to the position swapping between the alanine residue at position eight and the leucine at position nine, and between the alanine residue at position six and the leucine at position ten, compared with template peptide, Temporin-WY2.
The secondary structure confirmation was carried out by circular dichroism study. In 10 mL of NH4Ac, the aqueous environment is replicated. The results indicated that Temporin-WY2 presented an α-helical structure in an aqueous environment and that QUB-1426, 6K-WY2 and 6K-1426, tended to form random coil structures in this environment (Fig. 4a). The 50% TFE mimics the membrane environment. All the peptides tended to form an α-helical structure when contacting the bacteria cell membrane (Fig. 4b). A structural transformation can be found in QUB-1426, 6K-WY2 and 6K-1426, from random coil to α-helical structure, when in contact with the bacterial cell membrane.
Antimicrobial activity assessment
The outcomes of the MIC and MBC evaluation indicated that all peptides showed potent antimicrobial activity against most tested microorganisms. Both Temporin-WY2 and QUB-1426 showed stronger bactericidal effects against Gram-positive bacteria than against Gram-negative bacteria. The effective concentrations of Temporin-WY2 for Gram-positive bacteria ranged from 2 to 4 µM and the MBC values ranged from 2 to 8 µM. The bactericidal activity against MRSA (ATCC 12493) was the most potent among all the Gram-positive bacteria. As for QUB-1426, all the MIC and MBC values against the tested Gram-positive bacteria were 2 μM, with a two- to fourfold improvement after modification when targeting S. aureus (ATCC CRM 6538), MRSA (BAA 1707) and E. faecalis (NCTC 12697) in the MIC or MBC concentrations. Compared with Temporin-WY2, two 6K variants performed marginally less effectively. The MIC values of these two peptides were two- to eightfold higher than Temporin-WY2. The MIC and MBC values of 6K-WY2 ranged from 4 to 16 μM, and 8 to 16 μM for 6K-1426. The positive control polymyxin B showed antimicrobial efficacy at high concentrations of 128 μM and greater, and almost lost its function against two MRSA strains and E. faecalis.
When applied to Gram-negative bacteria, the situation was completely reversed. The parent peptide, Temporin-WY2, showed limited inhibition of the bacteria. The MIC value of Temporin-WY2 was 32 μM against E. coli, and greater than 128 μM against P. aeruginosa and K. pneumoniae. To promote its function on Gram-negative bacteria, a perfect amphipathicity variant, and two lysine cluster variants were designed. After modification, the antimicrobial activities were much better and among all the analogues, 6K-1426 showed the best activity against Gram-negative bacteria, with MIC values from 2 to 8 μM. The advances in treating K. pneumoniae and P. aeruginosa for 6K-WY2 and 6K-1426 were significant, but still had an 8- to 16-fold gap compared with polymyxin B.
The clinically-isolated strains of bacteria are multidrug-resistant and are usually harder to treat. For the MRSA (B038 V1S1 A) strain, Temporin-WY2 and QUB-1426 still maintained powerful efficacies, with both MIC and MBC values of 2 μM, while the two 6K variants presented with a slight reduction. For the E. coli (BAA 2340) strain, all the tested peptides displayed either equal or even better bioactivity. The two clinically-isolated P. aeruginosa strains, on the other hand, appeared obstinate and difficult to treat. PAO1 strain was derived from the original PAO strain which presented chloramphelicol resistance. And PA14 strain was isolated from the burn wound which is more virulent strain compared with PAO1 13. QUB-1426 almost lost its function, from a MIC value of 16 μM against the normal strain of P. aeruginosa, to 64 μM and greater than 128 μM, for PAO1 and PA14. 6K-WY2, on the contrary, this peptide demonstrated an equal or even better function on PA14 and PAO1. The MIC value of 6K-1426 against PA14 was 16 μM, displaying a twofold decrease compared to P. aeruginosa (ATCC 9027) strain, but the inhibition concentration against PAO1 was fourfold greater than that against the normal strain. The disparity between polymyxin and the test peptide in the killing of clinical strains was obvious. Polymyxin B demonstrated a steady efficacy on the clinically-isolated Gram-negative strains, 8- to 32-fold stronger than the tested peptides (Table 2).
MBIC/MBEC assay
To further explore the biological activities of these peptides, antibiofilm activities were investigated. All the tested peptides presented a remarkable ability for preventing the formation of Gram-positive bacterial biofilm with MBIC values ranging from 2 to 16 μM. The Gram-negative bacterial biofilm formation also was effectively inhibited by the QUB-1426, 6K-WY2 and 6K-1426, which shared a comparable trend with the MIC values. Although the peptides had a considerable inhibitory activity on biofilm formation, their capacity to eradicate existing biofilm was quite modest. Only Temporin-WY2 demonstrated a biofilm eradication function in Gram-positive bacteria, while 6K-WY2 and 6K-1426 had some E. coli biofilm eradication capacity (Table 3).
Condition sensitivity assays
The salt sensitivity of peptides determines whether they can sustain their function in a physiological environment. Four clinically isolated bacterial strains were applied in this study.
For MRSA B038, Temporin-WY2 and QUB-1426 were selected for the salt sensitivity evaluation. The salt sensitivity of Temporin-WY2 against MRSA B038 established a low salt sensitivity, and the MIC and MBC values were sustained unchanged in a saline environment. QUB-1426 also presented a low salt sensitivity only exhibiting a slight reduction of its bactericidal efficacy in the zinc and iron salt environments (Table 4).
When targeting E. coli (2340) cells, calcium ions seemed to have a great impact on the bactericidal activity of QUB-1426 as the MIC and MBC values increased eightfold compared to those in the absence of Ca2+. QUB-1426 also had a relatively high sensitivity to sodium and magnesium ions, with twofold increases in MIC values. On the contrary, the ionic environment seemed to have a positive impact on the bactericidal efficacy of the two 6K variants. Variable improvements were found in both their bacteriostatic and bactericidal properties. Worth noticing, was that the positive control, polymyxin B, showed salt sensitivity in some ionic environments such as those with potassium, magnesium, zinc and iron. Especially in the presence of Fe3+, a four –fold decrease in activity was demonstrated when targeting E. coli (Table 5).
For the PA14 strain, calcium seemed to have a great impact on the peptides’ function. All the tested antimicrobial agents showed a two- to fourfold lower effectiveness in CaCl2. For 6K-WY2, it presented a low salt sensitivity in the remaining six salt environments and even had better bioactivity in Na+. A similar situation was found with 6K-1426. As for polymyxin B, the monovalent sodium ions and also ammonium ions were demonstrated to have a negative impact on its function, resulting in a doubling of the MIC value when targeting PA14 (Table 6). When targeting PAO1, both 6K-WY2, 6K-1426 and polymyxin B, demonstrated a high salt sensitivity, with various degrees of reduction in antimicrobial activity (Table 7).
The development of antimicrobial peptides into clinical use is hampered by their stability when presented in a physiological environment such as human serum 14. In this study, two clinically isolated bacteria MRSA (B038 V1S1 A) and E. coli (BAA 2340) were selected to study the peptides’ sensitivity in human serum.
The parent peptide Temporin-WY2 exhibited the highest sensitivity to the human when targeted MRSA B038 and E. coli 2340. Its antimicrobial function was 16 times weaker when treated with MRSA in the serum environment and lost its function when targeted E. coli. After modification, compared to Temporin-WY2, QUB-1426 exhibited reduced sensitivity to human serum, but it still showed a decrease in antibacterial activity against both tested strains. For MRSA B038, its MIC and MBC values have increased by 2 and 4 times. When targeting E. coli 2340, the function of QUB-1426 was compromised 8 and 16 times on MIC and MBC values, respectively. The 6K analogues demonstrated a very low sensitivity to human serum when targeted MRSA B038 and E. coli 2340. The MIC and MBC values of both peptides had a twofold decrease when targeted MRSA. For E. coli 2340, the antimicrobial function of 6K-WY2 had a slight improvement and 6K-1426 maintained its capability (Table 8).
Killing time assays
The killing time kinetic curves reveals the bactericidal rate of the peptides. The results indicated that QUB-1426, 6K-WY2 and 6K-1426, could kill S. aureus at their MIC concentrations within 60, 60, and 90 min, respectively. Temporin-WY2 did not present a significant inhibition of the bacteria at its MIC, which could be correlated to its MBC value. At twofold MIC, the bactericidal rate of all the tested peptides advanced dramatically. QUB-1426, 6K-WY2 and 6K-1426 achieved complete killing within 5, 10 and 10 min, showing a significant concentration-related bactericidal trend. Temporin-WY2 started to achieve 100% killing at twofold MIC in 30 min. At fourfold MIC, the killing rate of Temporin-WY2 and 6K-1426 continued to decrease, achieving complete killing in 5 min (Fig. 5).
As for the E. coli killing curve, Temporin-WY2 and QUB-1426 presented a time-consuming killing trend, which achieved thorough killing at twofold MIC in 120 min and at fourfold MIC, in 90 min, respectively. Nonetheless, 6K-1426 possessed an extraordinary killing rate with a MIC value within 5 min. Moreover, 6K-WY2 achieved an instantaneous killing action at its MIC. When the concentration increased to fourfold MIC, 6K-1426 also achieved an instantaneous killing effect (Fig. 6).
SYTOX green permeability assays
The evaluation of membrane permeability activity of peptides on the bacterial cell membrane was conducted by using SYTOX green permeability assays. The S. aureus membrane permeability rate treated with Temporin-WY2 and QUB-1426 is time-related and concentration-related, achieving 100% permeability at fourfold MIC in 30 min for Temporin-WY2 and at MIC concentration in about 100 min. 6K-WY2 and 6K-1426, however, did not show a concentration-related membrane permeability trend and presented a lower permeability percentage at higher concentrations. The permeability rate of 6K-1426 was lower than 90% even at its peak permeability (Fig. 7).
Kinetics of membrane permeabilization of Temporin-WY2, QUB-1426, 6K-WY2 and 6K-1426 against S. aureus (ATCC 6538) at 1 ×, 2 × and 4 × MIC. The percentage of membrane permeabilization was measured using bacterial cells treated with melittin. The error bars in the graphs around mean data points indicated the standard error of the mean (SEM) for each set of data in the nine replicates from three experiments.
As for their action on the E. coli cell membrane, all the peptides demonstrated a significant concentration-dependent membrane permeability rate. The membrane rupture rates of Temporin-WY2 and QUB-1426 were relatively slow. At the highest concentration, Temporin-WY2 completed the membrane permeability in 60 min. The permeability rate of QUB-1426 at fourfold MIC reached a peak value in 30 min and was soon reduced afterwards (Fig. 8).
Kinetics of membrane permeabilization of Temporin-WY2, QUB-1426, 6K-WY2 and 6K-1426 against E. coli (NCTC 8739) at 1 ×, 2 × and 4 × MIC. The percentage of membrane permeabilization was measured using bacterial cells treated with melittin. The error bars in the graphs around mean data points indicated the standard error of the mean (SEM) for each set of data in the nine replicates from three experiments.
In vivo antimicrobial efficacy evaluation
Temporin-WY2 and its analogues exhibited outstanding in vitro antimicrobial activity against MRSA (B038 V1S1 A) and E. coli (BAA 2340). Therefore, an in vivo antimicrobial activity evaluation was carried out using the waxworm infection model. As shown in Fig. 9a, treatment with Temporin-WY2 at a concentration of 3 mg/kg resulted in a gradual decrease in survival rate, ultimately leading to a 20% survival rate within 168 h. At twofold and fourfold concentrations, the final survival rate of the larvae was 80%, and the larvae survived longer at twofold concentration. It is worth noting that polymyxin did not exhibit satisfactory in vivo antimicrobial activity against MRSA and only resulted in a 60% survival rate after treatment. The situation was opposite when applied to QUB-1426, as the survival rates of the larvae showed a concentration-related trend. Increasing the dosing concentration resulted in a higher survival rate of the larvae. At the highest concentration of 12 mg/kg, the larvae had a 100% survival rate (Fig. 9b).
The percentage of survival in waxworms infected with MRSA (B038 V1S1 A) (a,b); E. coli (BAA 2340) (c,d and e) and peptides (f and g). The larvae were treated with 10 μM of Temporin-WY2 (3, 6, 9 mg/kg); QUB-1426 (3, 6, 9 mg/kg) for MRSA and QUB-1426 (6, 12, 24 mg/kg); 6K-WY2 (9, 18, 36 mg/kg) and 6K-1426 (18, 36, 72 mg/kg) for E. coli. Sterile phosphate-buffered saline (PBS) and 15 mg/kg of polymyxin were applied as negative control and positive control respectively. In the toxicity evaluation, the waxworms were treated with 10 μM of peptides in varies concentrations.
For the in vivo anti-E. coli study, QUB-1426, 6K-WY2 and 6K-1426 were selected due to their potent in vitro anti-E. coli efficacy. At both one- and twofold concentrations, QUB-1426 showed similar in vivo antibacterial activity, culminating in 80% survival of the worms. At the twofold concentration of 12 mg/kg, the survival time of the worms was significantly longer than the dosage concentration of 6 mg/kg. Both one- and twofold concentrations presented a comparable in vivo efficacy compared with positive control polymyxin, while the larvae exhibited a longer survival time when treated with polymyxin. When the concentration increased to fourfold, the survival rate decreased speedily, presenting an 80% mortality in 96 h (Fig. 9c). Regarding 6K-WY2, at a twofold concentration, the peptide exhibited the most potent in vivo efficacy, with an 80% survival rate. However, as the concentration increased or decreased, the in vivo efficacy reduced to varying degrees, with survival rates of 20% and 60% at one- and fourfold concentrations, respectively (Fig. 9d). On the other hand, 6K-1426 presented the weakest in vivo anti-E. coli efficacy among the three tested peptides. The results indicated that when treated with one- and fourfold MIC concentrations of the peptide, the larvae were completely killed. However, at a twofold concentration, the peptide demonstrated the strongest in vivo efficacy, with a 60% survival rate within 168 h (Fig. 9e).
Analysing the peptide in vivo toxicity in the waxworm model is an efficient way to evaluate toxicity. The result indicated that, the larvae demonstrated a 100% survival rate when treated with all concentrations of Temporin-WY2 (Fig. 9f). A concentration-dependent trend in the toxicity of QUB-1426 was evident, with consistent overall mortality rates observed at concentrations of onefold, twofold, and fourfold MIC, but with varying survival times for the larvae. However, the mortality rate sharply increased at a concentration of 24 mg/kg (Fig. 9f). For 6K-1426, a significant concentration-dependent change in peptide toxicity was observed, with a 20% and 40% increase in the mortality rate of the worms observed as the concentration doubled (Fig. 9g).
MTT anti-proliferation activity
The evaluation of antiproliferation activity was conducted using three cancer cell lines including NCI-H838, U251MG, MCF-7 (Fig. 10). In summary, Temporin-WY2 showed the weakest antiproliferation action among all the tested peptides. All the modified peptides had a dramatically increased antiproliferation function. The IC50 values of all the modified peptides were reduced significantly (Table 9).
Effects on proliferation of human cancer cell lines with Temporin-WY2 (red), QUB-1426 (orange), 6K-WY2 (green), and 6K-1426 (blue). Cancer cell line H838; MCF-7and U251MG were selected in this study. A range of peptide concentrations, from 10−9 M to 10−4 M, were applied to the cancer cells. Triton X-100 (0.1%) was selected as the positive control. Utilizing normalised dose–response analysis, the curves were fitted. The error bar displays the standard deviation and means across three separate studies.
CCK-8 cell proliferation assay
The cell viability of human primary epidermal keratinocytes (HaCaT cells) was assessed using the CCK8 cell counting kit. The parent peptide, Temporin-WY2, exhibited moderate cytotoxicity to normal cells, with cell viability reduced to 50% at a concentration of 60.12 μM (Fig. 11). Despite the overall elevated toxicity, at the effective bactericidal concentration of Temporin-WY2 against S. aureus (C = 2 μM), the peptide exhibited very low toxicity, with cell viability close to 100%. Among all the tested peptides, QUB-1426 demonstrated the lowest cytotoxicity, maintaining cell viability above 50% at all tested concentrations. Despite exhibiting increased hemolytic activity and cytotoxicity, they still demonstrated a high therapeutic index against S. aureus.
Effects on proliferation of human primary epidermal keratinocytes with Temporin-WY2 (red), QUB-1426 (orange), 6K-WY2 (green), and 6K-1426 (blue). Human primary epidermal keratinocytes (HaCaT cells) were selected in this study. A range of peptide concentrations, from 10−9 M to 10−4 M, were applied to the cancer cells. Utilizing normalised dose–response analysis, the curves were fitted. The error bar displays the standard deviation and means across three separate studies.
Haemolytic activity
The haemolytic activity of the peptides was evaluated by their lysis of horse erythrocytes. Despite their potent haemolytic activity at high concentrations, Temporin-WY2 and QUB-1426 still had less than 10% haemolytic activity at their MIC values against S. aureus. 6K-WY2 and 6K-1426 had a higher percentage of haemolysis at their effective concentration (Table 10). At their effective anti-S. aureus concentrations, 6K-WY2 and 6K-1426 possessed 17.78% and 36.56% haemolysis, respectively (Fig. 12).
Discussion
The inappropriate consumption of antibiotics has hastened the increase of the spread of multidrug-resistant pathogens which cause a series of problems such as hospitalization lengthening, morbidity and mortality increases 15. Alternative antimicrobial agents are doubtless needed to overcome this problem. Antimicrobial peptides with clear advantages including slower emergence of resistance, rapid killing, and effective biofilm control were considered competitive candidates for treating drug-resistant pathogens 16.
In this study, a novel peptide was identified from the skin secretion of Amolops wuyiensis skin secretion. Like the other antimicrobial peptides, its precursor signal peptide and the acidic pro-peptide domain are highly conserved, while the antimicrobial domain at the C-terminus is highly variable. Peptide alignment provided a clear relationship between the newly found peptide with other identified peptides in the database. The sequence with the highest similarity was found in the Temporin family (Temporin-WY1). So, the novel peptide most likely belongs to the Temporin family and was named Temporin-WY2.
Temporin-WY2 was rationally modified to obtain a superior function. Amphipathicity, charge, and hydrophobicity are critical parameters that have important impacts on antimicrobial activity 17,18. To elucidate the influence of those factors on antimicrobial activity, QUB-1426 was designed. Compared with the parent peptide, Temporin-WY2, QUB-1426 showed a lower hydrophobicity as the result of the substitution of a phenylalanine residue for a lysine residue. Also, it showed a perfect amphipathicity characteristic by amino acid position swapping to form a long cluster of hydrophilic and hydrophobic residues.
The lysine cluster sequence is a specific sequence that not only can increase a peptide’s antimicrobial function, especially against Gram-negative bacteria, but also can reduce the haemolytic activity dramatically19. These sequences were added at the N-terminal of Temporin-WY2 and QUB-1426 and two new modified analogues, 6K-WY2 and 6K-1426 were thus produced.
The secondary structure prediction revealed that all the peptides tended to form an α-helical structure, and the circular dichroism study further substantiated this conjecture. All the peptides possessed an α-helical structure in the 50% TFE environment. While in the aqueous environment, most tended to form a random coil structure. Structural change upon membrane contact is a common occurrence for linear short peptides lacking cysteine residues. They normally form a flexible coil structure in aqueous environments, undergoing a structural transformation in the presence of trifluoroethanol; sodium dodecyl sulphate (SDS) micelles; phospholipid vesicles and liposomes, or Lipid A 20. A great example was the antimicrobial peptide from the cathelicidin family—LL-37. According to previous research, LL-37 is in random-coil conformation in pure aqueous solution, but in settings such as trifluoroethanol and the presence of salts or lipid bilayer vesicles, often undergoes a secondary structure change into the helical structure 21.
Temporin-WY2 demonstrated a more effective bactericidal ability against Gram-positive bacteria, which is consistent with the antibacterial property of Temporin peptides 22. The modified peptide QUB-1426 showed identical antimicrobial activity against Gram-positive bacteria and 8- to 32-fold stronger antimicrobial activity against Gram-negative bacteria compared with Temporin-WY2. Several critical factors can influence the antimicrobial activity of the peptide (charge, hydrophobicity, amphipathicity, secondary structure) 23. This significant improvement might have been caused by the addition of one extra positive charge provided by the lysine residue after the substitution of phenylalanine residue into lysine at position three. Fernandez-Vidal 24 demonstrated that amphipathicity is more important than hydrophobicity for binding to microbial membranes. The amphipathic helical structure consisted of a non-polar face on one side, and a polar face on the other. This property gives the peptide an ideal structure to bind to bacterial cell membranes by the polar face in an aqueous environment, while the non-polar side faces the inside leaflet 25. Compared with Temporin-WY2, QUB-1426 has a higher hydrophobic moment which might make it has a stronger binding ability to the bacteria cell membrane, leading to a more potent antimicrobial activity.
An anchoring structure was put forward in He’s study 26. This unique structure consisted of a cluster of positively charged amino acids in the N-terminus of the peptide. It provided the peptide with an anchor-like structure which endowed a strong orientation to the cell membrane, and subsequently offered the long list of hydrophobic “tail” opportunities to penetrate the cell membrane. The prominent enhancement of the anti-Gram-negative activity and the instantaneous membrane penetrating of 6K-WY2 in E. coli, could be the best illustration.
However, this structure did not give priority to the anti-Gram-positive bacteria function. The possible reason might be caused by the different mechanisms when targeting Gram-positive bacteria and Gram-negative bacteria. The SYTOX green permeability assay revealed that peptides with relatively more potent activity to Gram-positive bacteria had a lower membrane penetrating rate at all the tested concentrations. There might be other mechanisms besides membrane targeting when Temporin-WY2 attacks Gram-positive bacteria.
In summary, in the antimicrobial aspect, all the tested peptides presented broader spectra of antimicrobial activities than polymyxin and showed both anti-Gram-positive and anti-Gram-negative efficacy. However, the polymyxin had a better function on the Gram-negative bacteria, reflecting the limitations of peptide function.
Maintaining function in the salty and human serum environment is a critical aspect that determines whether the peptide could be effective in our body. The salt sensitivity result pointed out that all the tested peptides could maintain their activity when targeting Gram-positive bacteria in saline. However, with E. coli, the salt ions had a significant influence on QUB-1426. Especially calcium ions. When targeting Gram-negative bacteria, some AMPs can quickly cross by a charge-exchange mechanism where the cationic peptides compete with Ca2+ and Mg2+ bound to lipopolysaccharide 25. The presence of positively charged cationic salts may affect this process leading to a significant decrease in their function.
The results of salt sensitivity applied on E. coli cells revealed that, when treating E. coli cells in a bivalent cations environment, the lysine rich peptides 6K-WY2 and 6K-1426 were unaffected in the saline environment. While a two- to eightfold functional decrease can be found in QUB-1426. The difference tendency of these three modified peptides may be attributed to their varying helicity. Park and colleagues demonstrated the peptides salt sensitivity were impacted by the helicity 27. The helical structures formed by peptides after contacting the membrane contribute to their stability, thus enhancing their resistance to salt and maintaining their structural integrity and antimicrobial function in saline solutions. CD spectra also demonstrate similar trends. The most salt-sensitive peptide, QUB-1426, exhibits the lowest helicity in the membrane environment, while the least sensitive peptide, 6K-WY2, displays the highest helicity.
An improved antimicrobial activity can be found for 6K-WY2 in the presence of sodium ions when targeting PA14 and PAO1. A similar situation can be found in Zhang’s study 28. It has been reported that P. aeruginosa is one of the most common pathogens found in cystic fibrosis (CF) patients and the elevated sodium level in the CF patient’s airway might inactivate some peptide functions. All the tested peptides in Zhang’s study demonstrated various weakened antimicrobial activities in the presence of 150 mM NaCl.
As a competitive antimicrobial agent for clinical development, peptides have been widely studied. However, only a few peptide-based antimicrobial agents are available in the market. The biggest obstacle impeding the entry of antimicrobial peptides into the market is their susceptibility to proteolytic degradation within the body. The drug pharmacokinetics may be negatively impacted by their sensitivity, there by hindering the systemic application of antimicrobial peptides 29. Therefore, the sensitivity of peptides to the human serum is important to evaluate the peptides’ function. The condition sensitivity assay by human serum indicated that the parent peptide Temporin-WY2 did not maintain its function in the serum environment. While after modification, 6K-1426 demonstrated the lowest sensitivity to human serum. A previous study conducted by Stone and colleagues studied the six-lysine clustered peptides’ stability in human blood plasma 19. The experimental result indicated that the charge-cluster analogue 6K-F17 was stable for up to 24 h. The significant improvement in peptide stability might attribute to the electrostatic repulsion provided by the charge clustering in the N terminus of the peptide, which might push away the peptides from the binding site of the enzyme.
Killing kinetics is another important factor that decides whether the peptides could become antimicrobial candidates. When targeting S. aureus, the killing rate at their MIC values is not very satisfactory. However, at a higher concentration of twofold the MIC value, QUB-1426, 6K-WY2 and 6K-1426 presented a speedy bactericidal rate in 10 min, which is much better than the parent peptide. A similar situation was found when treating E. coli. 6K-WY2 and 6K-1426 showed a surprising killing rate, almost achieving instant killing at their MIC concentrations.
The in vivo antimicrobial activity might be different from the in vitro antimicrobial activity because of the impact of all kinds of physiological components. Galleria mellonella (the waxworm) served as the preliminary in vivo infection model and is widely used in the evaluation of in vivo antimicrobial efficacy evaluation. All worms treated with the MIC doses of the peptides showed varying degrees of death, confirming the influence of the internal environment on peptide function. However, at slightly higher concentrations, QUB-1426 still showed the best in vivo activity of all the peptides, with no mortality observed in the MRSA-infected worms within 7 days. Furthermore, the in vivo efficacy of Temporin-WY2 against MRSA and QUB-1426 and 6K-WY2 against E. coli was commendable, achieving a survival rate of 80% within a period of 7 days. The in vivo efficacy of the peptides is highly related to their cationicity and hydrophobicity 30. In Mishra’s study, the in vivo efficacies of the lysine-rich peptides were evaluated by the murine model. The result revealed that, the presence of extra positive charges could have assisted in the binding of peptides to host factors within the body, thereby preventing them from being used for bacterial elimination, which is consistent with the results of this study.
Biofilms, which are defined as adherent micro-communities, cause many public health problems 31. Eighty per cent of pathogen-forming biofilms are associated with persistent infections 32. They are hard to be treated under the protection of the extracellular matrix (EPS) which consists of proteins, exopolysaccharides, extracellular DNA and enzymes. Besides, different kinds of toleration mechanisms such as slow growth rate and alternative metabolisms, makes the treatment of biofilm even harder 33. Antimicrobial peptides are regarded as hopeful agents to solve this problem. In this study, 7 kinds of pathogens were selected in the evaluation of the anti-biofilm activity of the peptides. All the tested peptides show considerable activity in preventing the formation of Gram-positive bacterial biofilm. However, the inhibition of Gram-positive and Gram-negative bacteria had a significant difference and the eradication function of formed biofilm was almost lost. Although Gram-positive and Gram-negative bacteria biofilm showed a similar pattern in their micro-colony formation, the physiology, genomes and the components of the matrix are quite different 34. This might explain the differences in activity among different Gram types.
The improvement of the antiproliferation function is another achievement in this study. Antimicrobial peptides were considered as potential anticancer agents due to their similar mechanisms of antimicrobial and anticancer activity. The electrostatic attraction is the main driving force between AMPs and membranes. Cancer cells are more negatively charged because of phosphatidylserine (PS) and the O-glycosylated mucins35. The more potent electrical attraction between peptides and the cancer cell membrane provided the peptides with a more potent ability against the cancer cell membrane which determined the improvement of their antiproliferation activity. However, in the cytotoxicity evaluation, the 6K analogues demonstrated significantly increased toxicity to the primary cells, which needs to be addressed in future studies.
Haemolysis is an essential parameter to evaluate the peptides' cytotoxicity. Stone and his colleagues investigated six peptide analogues with the same compositions of amino acids but lysine residues scattered at different points in the sequence. The results indicated that the lysines clustered at the N-terminus demonstrated the lowest haemolysis 19. After six lysines were introduced in Temporin-WY2 and 6K-1426, the opposite situation was presented in our study. 6K analogues had more potent haemolytic activity. The key difference between these two studies was the peptides’ helical tendency. In Stone’s study, by clustering the lysine residues into the N-terminus, a decreased helical tendency could be found when contact with the bacterial cell membrane and the mammal cell membrane. However, in this study, by adding the lysine residues, the helical tendency which was also found in the CD spectrum. In Grieco’s study 36, the results indicated that hydrophobicity and helicity, rather than cationicity and amphipathicity, were the critical parameters that influence peptide toxicity. That might explain the reason for the increased haemolysis.
Conclusion
In this study, a novel antimicrobial peptide named, Temporin-WY2, was identified from the skin secretion of the frog, Amolops wuyiensis. Temporin-WY2 has remarkable antimicrobial activity against Gram-positive bacteria with moderate haemolysis at the effective concentration. However, the peptide did not exhibit a satisfactory anti-Gram-negative bacterial effect and anti-proliferation activity against cancer cells. After subsequent modification, three different peptide sequences were acquired. These were designated, QUB-1426, 6K-WY2 and 6K-1426. QUB-1426, 6K-WY2 and 6K-1426, had broad spectrum activity, high selectivity and fast killing rates, making them promising drug candidates for treating bacterial infections and potentially as anticancer agents.
Methods
Acquisition of skin secretion from Amolops wuyiensis
Adult Amolops wuyiensis frogs were obtained from Wuyi mountain, Wuyishan city, Fujian province, China through commercial sources (n = 5). The skin secretions were obtained through gentle electricalstimulation (5 V,100 Hz, and 140 ms pulse width), which is non-invasive and does not sacrifice the animals. The secretions were rinsed from the skin using deionised water and collected in a chilled beaker. The samples were immediately frozen in liquid nitrogen and lyophilised, then stored in a −20 ℃ freezer. The study was carried out following the guidelines of the UK Animal (Scientific Procedures) Act 1986, project license PPL 2694, issued by the Department of Health, Social Services and Public Safety, Northern Ireland. Procedures had been reviewed by the IACUC at Queen’s University Belfast and approved on 1st March 2011. All methods were carried out in accordance with relevant guidelines and regulations. This study was carried out in compliance with the ARRIVE guidelines 37.
Shotgun cloning of the precursor-encoding cDNA of the novel peptide
Lyophilised skin secretion was dissolved in lysis/binding buffer (Dynal Biotech, U.K.) (5 mg/mL). The mRNA was isolated using a Dynabeads® mRNA Direct Kit (Life Technologies, Oslo, Norway) following the manufacturer’s instructions. Then, the isolated mRNA was used as the template for building a full-length cDNA library by using a BD SMART RACE cDNA Amplification kit (BD Clontech UK). After the full-length cDNA sequence was obtained, 3′-RACE PCR was applied to amplify the desired cDNA by using nested universal primer A (NUP, 20 μM) (5′-AAGCAGTGGTATCAACGCAGAGT-3′) and a degenerate primer S2 (5′-CTAGTGATTATGTTCACCTCAAAGAAA-3′) (20 μM). After PCR, the products were analysed by gel-electrophoresis using a Benchtop UV Transilluminator (UVP, UK), purified by using E.Z.N.A® Tissue DNA kit (Omega, Norcross, UK) and ligated using the pGEM®-T Easy Vector (Promega, USA). After ligation, the recombinant DNA was transfected into the JM109 high-efficiency competent cells and selected based on the blue and white screening principle. The selected product was cloned, identified and purified. A BIG-DYE Terminator sequencing kit (Applied Biosystems, Foster City, CA, USA) was used for sequencing reactions, and the result was analyzed by an automated ABI 3700 DNA sequencer (Life Technologies, Paisley, UK). The peptide alignment was achieved using the NCBI-BLAST program (https://www.ncbi.nlm.nih.gov/, accessed on 30 Oct 2021).
Peptide synthesis, purification and identification
Temporin-WY2 and its designed derivatives were synthesised by Solid Phase Peptide Synthesis (SPPS) in a Tribute™ automated solid-phase peptide synthesiser (Protein Technologies, Tucson, AZ, USA). All the weighed amino acids and the activator 2-(1H-benzotriazole-1-yl)-1,1,3,3-tetra methyluronium hexafluorophosphate (HBTU), along with Rink amide MBHA resin, were loaded into the machine then coupled. After the peptide was synthesised, the cleavage solution, which consisted of 94% Trifluoroacetic acid (TFA), 2% thioanisole (TIS), 2% 1,2-ethanedithiol (EDT) and 2% H2O was added to cleave the side chain protecting groups by stirring for 2 h. Then, the peptides were washed with diethyl ether three times and then dissolved in 15 mL of buffer A (0.05% TFA and 99.95% water) and 15 mL of buffer B (0.05% TFA, 19.95% water and 80% acetonitrile).
The synthesised crude peptides were purified by reverse-phase HPLC (LUNA C-5 preparation column, 250 × 10 mm, Phenomenex, UK) with a linear gradient from 100% buffer A (0.05/99.5 (v/v) TFA/water) and 0% buffer B (0.05/19.95/80.00 (v/v/v) TFA/water/acetonitrile) to 0% buffer A and 100% buffer B in 40 min at a flow rate of 1 mL/min. The masses of the peptides were verified by MALDI-TOF MS (Voyager DE, Perseptive Biosystems, Framingham, Ma, USA), using CHCA (10 mg/mL in 70% acetonitrile, 0.02% TFA and 30% water) as the matrix in a positive detection mode.
Physicochemical properties analyses, secondary structure prediction and confirmation
Physicochemical properties of Temporin-WY2 and its analogues were acquired via the Heliquest programme (https://heliquest.ipmc.cnrs.fr/cgi-bin/computparams.py, accessed on 11 Nov 2021). Secondary structure prediction was carried out using the PEP-FOLD3 program (https://bioserv.rpbs.univ-paris-diderot.fr/services/PEP-FOLD3/, accessed on 11 Nov 2021). The helical wheel projections were created by the Heliquest program (https://heliquest.ipmc.cnrs.fr/cgi-bin/computparams.py, accessed on 11 Nov 2021).
The confirmation of the secondary structure of the peptides was obtained using circular dichroism on a JASCO J815 CD spectrometer (Jasco, Essex, UK). Peptides were dissolved in 10 mM NH4Ac and 50% TFE (tetrafluoroethylene) (v/v, in 10 mM NH4Ac) at a concentration of 50 μM, which mimicked the water environment and the membrane environment, respectively. Samples were loaded into a 1 mm quartz cuvette and then analyzed at 20℃, at a scan range of 190–240 nm, a scan speed of 100 nm/min, 1 nm bandwidth and 0.5 nm data patch. The obtained data were further analyzed by the online tool, BeStSel (https://bestsel.elte.hu/index.php, accessed on 14 Nov 2022).
Minimum inhibitory concentration (MIC)/minimum bactericidal concentration (MBC) determinations
Seven pathogens and four clinically isolated strains were used in the antimicrobial assays. They were Gram-positive bacteria, Staphylococcus aureus (ATCC CRM 6538), methicillin-resistant Staphylococcus aureus (BAA 1707), methicillin-resistant Staphylococcus aureus (ATCC 12493), Enterococcus faecalis (NCTC 12697); Gram-negative bacteria, Escherichia coli (NCTC 8739), Pseudomonas aeruginosa (ATCC 9027), Klebsiella pneumoniae (ATCC 43816); clinically isolated strains, methicillin-resistant Staphylococcus aureus (B038 V1S1 A), Escherichia coli (BAA 2340), PAO1 and PA14.
The pathogens were cultured in the corresponding medium (Table 11) overnight at 37℃, then subcultured and diluted to 5 × 105 CFU/mL. Then the peptides were dissolved in dimethyl sulfoxide (DMSO) for a final concentration series from 1 to 128 µM. The positive control contained 20 μg/mL of norfloxacin. Peptide solution and diluted bacteria were loaded into the 96 well-plate and then incubated at culture temperature for 20–24 h under the guidence of previous research 38. The OD value (λ = 550 nm) of each well was detected using a Synergy HT plate reader (Biotech, USA). The results were analysed according to the formula: Viability (%) =\(\frac{{\text{A}}_{\text{S}}-{\text{A}}_{\text{B}}}{{\text{A}}_{\text{V}}-{\text{A}}_{\text{B}}}\) \(\times \) 100% (AS: absorbance of the sample; AB: absorbance of the blank control group; AV: absorbance of the vehicle control group). Afterwards, 10 µL of the solution in each clear well was loaded on an agar plate and cultured for 20 h. The lowest concentration without colony growth was regarded as the MBC.
Minimum biofilm inhibition concentration (MBIC)/minimum biofilm eradication concentration (MBEC) determination
Six types of bacteria were used for the measurement of the anti-biofilm activity of the peptides. Tryptic soy broth (TSB) broth with 1% glucose was used for Gram-positive bacteria biofilm culture, and Luria–Bertani (LB) broth with 1% glucose, was used for the culture of Gram-negative bacteria. The peptides were prepared as described before. The peptide solution and subcultured bacteria were mixed in 96 well-plates as described before, and cultured at 37℃ with a shear force of 200 rpm in a humid environment for 20–24 h. Afterwards, the biofilm was rinsed with 130 mL PBS twice before 100 µL of methanol was utilized for the biofilm stabilization. The detection of biofilm used 100 µL of 0.5% crystal violet as the staining dye for 15 min. Subsequently, the stained biofilm was further rinsed and dried overnight. Thirty per cent acetic acid was used for dissolving the stain and shaking for 15 min ensured that it was homogenous. Then the plate was read at 595 nm using a Synergy HT plate reader. The minimum concentration without biofilm formation was regarded as the MBIC value 39.
In the minimum biofilm eradication assay, the biofilm was first formed in the plate using the same method. Then, the forming biofilm was rinsed with PBS twice. One µL of peptide solution, together with 99 µL of corresponding broth medium, were loaded onto the plate before it was cultured in the culture environment described before. After 20–24 h of culturing, the biofilm was rinsed, stabilized and stained. The concentration of peptide causing complete eradication of biofilm was defined as the MBEC of the peptide.
Condition sensitivity assays
The determination of the conditions sensitivity assays of the peptides was performed in a MIC/MBC assay in an environment containing different physiological salts and 10% human serum as previously describe 27,40,41,42. Four clinically-isolated bacteria, were selected as the test bacteria in the salt sensitivity assay. And two clinically-isolated strains, MRSA (B038 V1S1 A) and E. coli (BAA 2340) were selected as the test bacteria in the human serum sensitivity assay. Bacterial cultures and peptide preparations were produced as described previously. Seven salt solutions at their physiological concentrations (150 mM NaCl, 4.5 mM KCl, 6 μM NH4Cl, 2.5 mM CaCl2, 1 mM MgCl2, 8 mM ZnCl2, and 4 mM FeCl3) and human serum were added into the culture broth to examine resultant antibacterial activities.
Time killing assay
The time-killing assay was carried out to acquire the killing kinetics of peptides. Two bacteria, S. aureus (ATCC 6538) and E. coli (NCTC 8739), were selected and cultured to obtained a final cell density of 105–106 CFU/mL and was incubated with peptides in different concentrations (1 × MIC, 2 × MIC, 4 × MIC). The mixed cocktail were drawn and diluted before dispatching into the agar plate at the selected time points (0, 5, 10, 20, 30, 60, 90 and 120 min) to count and record the number of living bacteria colonies after incubation at culture temperature for 20–24 h.
SYTOX green permeability assay
With regard to assessing the peptides’ disruptive effect on the bacterial cell membrane, the SYTOX green permeability assay was conducted. Bacteria were subcultured at 37 ℃ for 1.5 h, then collected at 4℃, 1000×g, and washed with 30 mL of 5% TSB (in 0.85% NaCl) solution twice. The collected bacteria were resuspended using 5% TSB solution to OD values of 0.68–0.72 for S. aureus, and 0.65–0.7 for E. coli at 590 nm. Subsequently, 50 μL of bacteria and 40 μL of peptide (concentrations = 1 × MIC, 2 × MIC, 4 × MIC), in conjunction with 10μL SYTOX green (50 μM in 5% TSB, Life Technologies, UK) were loaded into the black 96 well plates then read on a Synergy HT plate reader under the excitation and emission wavelengths of 485 nm and 528 nm, respectively, at 37℃ for 120 min (interval 5 min).
In vivo antimicrobial efficacy evaluation
The in vivo antimicrobial activity efficacy evaluation was conducted using a waxworm (Galleria mellonella) model. The larvae (Livefood UK Ltd., Rooks Bridge, UK) were weighed and sorted, and those with weights in a range from 225 to 275 mg were selected and placed in Petri dishes (n = 5 in each group). MRSA (B038 V1S1 A) and E. coli (BAA 2340) were selected for this study. The bacteria were subcultured in TSB broth at 37℃ for 1.5 h, then were collected in the bottom of the tube by centrifuging at 4 °C, 1000×g for 10 min. Afterwards, the bacteria were washed with PBS twice. The bacteria were resuspended and diluted to 1 × 107 CFU/mL for MRSA and 5 × 107 CFU/mL for E. coli before 10 μL of the diluted bacteria were injected into the larvae. The infected larvae were put into the incubator for 1 h at 37 °C,. When assessing MRSA, Temporin-WY2 and QUB-1426 were selected for study. The peptides were prepared at concentrations of 3, 6, and 12 mg/kg, respectively, according to their in vitro MIC values; when treating E. coli, QUB-1426, 6K-WY2 and 6K-1426 were selected for study. The peptides were prepared at 6, 12 and 24 mg/kg for QUB-1426; 9, 18 and 36 mg/kg for 6K-WY2 and 6K-1426. PBS was used as the negative control, and 15 mg/kg of polymyxin was used as the positive control. After exposure, 10 μL of the peptide and the control groups were injected into the larvae and then cultured at 37 °C,. In the in vivo toxicity evaluation, 10 μL of peptides were injected into the larvae to study the peptides’ cytotoxicity. The survival of larvae was recorded after 24, 48, 72, 96, 120, 144 and 168 h.
MTT anti-proliferation assay
The MTT assay is used to determine the cell metabolic activity based on the conversion of MTT into a water-insoluble formazan by living cells, to measure the anti-proliferation effects of the peptide.
Three cancer cell lines were chosen for this study: human non-small cell lung cancer cell line, NCI-H838 (ATCC-CRL-5844); human glioblastoma astrocytoma cell line, U251MG (ECACC-09063001) and human breast cancer cell line MCF-7, (ATCC-HTB-22). The cancer cells were seeded into 96 well plates at a cell density of 80,000 cells/mL and cultured for 20–24 h at 37℃. Then the cells were starved by replacing the complete growth medium with a serum-free medium and then cultured for 4 h. Peptides were dissolved to make series of final concentrations from 10–9 to 10–4 M in ddH2O. After starvation, 1 μL of the peptide with 99 μL of the serum-free medium were added to the plate and then cultured for 20–24 h. Ten μL of MTT (3-(4,5‐dimethylthiazol‐2‐yl)‐2,5‐ diphenyltetrazolium bromide, 5 mg/mL) were loaded into each well in the dark and incubated at 37℃. Subsequently, the liquid in each well was removed completely before 100 μL of DMSO were added to each well to dissolve the formazan. The plate was read at 570 nm using a Synergy HT plate reader.
CCK-8 cell proliferation assay
The CCK-8 (Cell counting kit-8) is used to determine the cell viability through the reduction of WST-8 by dehydrogenase in cells to form a water-soluble orange-colored formazan. The human primary epidermal keratinocytes HaCaT cell line was selected for this study. The cells were seeded into the 96 wells plate at a cell density of 60,000 cells/mL and cultured for 20–24 h in 37 °C to make sure all the cells adhered to the surface. Peptides were dissolved to make series of final concentrations from 10–9 to 10–4 M in ddH2O. And 100 μL of the peptides were loaded into the plate and cultured for 24 h. One hundred μL of 10% CCK8 was added into the plated after the original medium was removed. Subsequently, the plated was cultured for 2h before the absorbance was detected at 450nm using a Synergy HT plate reader.
Haemolysis assay
The erythrocytes from defibrinated horse blood were used to perform a haemolysis assay. Peptides were dissolved using PBS to achieve final concentrations of 1–128 μM. One hundred μL of peptide solution and blood cell suspension were mixed and incubated for two hours at 37 °C. After incubation, the sample was centrifuged at 930×g for 5 min before the supernatant was transferred into a 96 well plate. Positive control and negative controls were 100 μL 0.1% Triton X-100 and PBS, respectively. Degree of lysis of erythrocytes was indicated by the OD values of supernatants and was measured at 470 nm using an ELISA plate reader.
Statistical analysis
Statistical analysis of all bioactivity assays was performed by using Prism (GraphPad Software, USA). One-way ANOVA was used to analyse the statistical significance of the differences. The data points represent the mean of the independent experiments; the error bars represent the standard error (SEM) of the mean, ns = no significant difference, * = p < 0.5, ** = p < 0.1, *** = p < 0.01, **** = p < 0.001 (Supplementary Information).
Data availability
The Temporin-WY2 biosynthetic precursor-encoding cDNA is deposited in the GenBank Database under accession number: OQ847420.
References
Davies, J. & Davies, D. Origins and evolution of antibiotic resistance. Microbiol. Mol. Biol. Rev. 74, 417–433. https://doi.org/10.1128/mmbr.00016-10 (2010).
Browne, K. et al. A new era of antibiotics: The clinical potential of antimicrobial peptides. Int. J. Mol. Sci. https://doi.org/10.3390/ijms21197047 (2020).
Aminov, R. I. The role of antibiotics and antibiotic resistance in nature. Environ. Microbiol. 11, 2970–2988. https://doi.org/10.1111/j.1462-2920.2009.01972.x (2009).
Wang, C. H., Hsieh, Y. H., Powers, Z. M. & Kao, C. Y. Defeating antibiotic-resistant bacteria: Exploring alternative therapies for a post-antibiotic era. Int. J. Mol. Sci. https://doi.org/10.3390/ijms21031061 (2020).
De Oliveira, D. M. P. et al. Antimicrobial resistance in ESKAPE pathogens. Clin. Microbiol. Rev. https://doi.org/10.1128/cmr.00181-19 (2020).
Chen, C. H. & Lu, T. K. Development and challenges of antimicrobial peptides for therapeutic applications. Antibiotics https://doi.org/10.3390/antibiotics901002424 (2020).
Nang, S. C., Azad, M. A. K., Velkov, T., Zhou, Q. T. & Li, J. Rescuing the last-line polymyxins: Achievements and challenges. Pharmacol. Rev. 73, 679–728. https://doi.org/10.1124/pharmrev.120.000020 (2021).
Meredith, J. J., Dufour, A. & Bruch, M. D. Comparison of the structure and dynamics of the antibiotic peptide polymyxin B and the inactive nonapeptide in aqueous trifluoroethanol by NMR spectroscopy. J. Phys. Chem. B 113, 544–551. https://doi.org/10.1021/jp808379x (2009).
Abbassi, F. et al. Temporin-SHf, a new type of phe-rich and hydrophobic ultrashort antimicrobial peptide. J. Biol. Chem. 285, 16880–16892. https://doi.org/10.1074/jbc.M109.097204 (2010).
Mangoni, M. L., Grazia, A. D., Cappiello, F., Casciaro, B. & Luca, V. Naturally occurring peptides from rana temporaria: Antimicrobial properties and more. Curr. Top. Med. Chem. 16, 54–64. https://doi.org/10.2174/1568026615666150703121403 (2016).
Mishra, B. et al. Antibacterial, antifungal, anticancer activities and structural bioinformatics analysis of six naturally occurring temporins. Peptides. 106, 9–20. https://doi.org/10.1016/j.peptides.2018.05.011 (2018).
Casciaro, B. et al. The antimicrobial peptide Temporin G: Anti-biofilm, anti-persister activities, and potentiator effect of tobramycin efficacy against Staphylococcus aureus. Int. J. Mol. Sci. https://doi.org/10.3390/ijms21249410 (2020).
Grace, A., Sahu, R., Owen, D. R. & Dennis, V. A. Pseudomonas aeruginosa reference strains PAO1 and PA14: A genomic, phenotypic, and therapeutic review. Front. Microbiol. https://doi.org/10.3389/fmicb.2022.1023523 (2022).
Sato, A. K., Viswanathan, M., Kent, R. B. & Wood, C. R. Therapeutic peptides: Technological advances driving peptides into development. Curr. Opin. Biotechnol. 17, 638–642. https://doi.org/10.1016/j.copbio.2006.10.002 (2006).
Medina, E. & Pieper, D. H. Tackling threats and future problems of multidrug-resistant bacteria. Curr. Top. Microbiol. Immunol. 398, 3–33. https://doi.org/10.1007/82_2016_492 (2016).
Magana, M. et al. The value of antimicrobial peptides in the age of resistance. Lancet Infect. Dis. 20, e216–e230. https://doi.org/10.1016/s1473-3099(20)30327-3 (2020).
Lee, T. H., Hall, K. N. & Aguilar, M. I. Antimicrobial peptide structure and mechanism of action: A focus on the role of membrane structure. Curr. Top. Med. Chem. 16, 25–39. https://doi.org/10.2174/1568026615666150703121700 (2016).
Zhu, X. et al. Characterization of antimicrobial activity and mechanisms of low amphipathic peptides with different α-helical propensity. Acta Biomater. 18, 155–167. https://doi.org/10.1016/j.actbio.2015.02.023 (2015).
Stone, T. A. et al. Positive charge patterning and hydrophobicity of membrane-active antimicrobial peptides as determinants of activity, toxicity, and pharmacokinetic stability. J. Med. Chem. 62, 6276–6286. https://doi.org/10.1021/acs.jmedchem.9b00657 (2019).
Gennaro, R. & Zanetti, M. Structural features and biological activities of the cathelicidin-derived antimicrobial peptides. Biopolymers. 55, 31–49. https://doi.org/10.1002/1097-0282(2000)55:1%3c31::Aid-bip40%3e3.0.Co;2-9 (2000).
Johansson, J., Gudmundsson, G. H., Rottenberg, M. E., Berndt, K. D. & Agerberth, B. Conformation-dependent antibacterial activity of the naturally occurring human peptide LL-37. J. Biol. Chem. 273, 3718–3724. https://doi.org/10.1074/jbc.273.6.3718 (1998).
Romero, S. M., Cardillo, A. B., Martínez Ceron, M. C., Camperi, S. A. & Giudicessi, S. L. Temporins: An approach of potential pharmaceutic candidates. Surg. Infect. (Larchmt). 21, 309–322. https://doi.org/10.1089/sur.2019.266 (2020).
Kumar, P., Kizhakkedathu, J. N. & Straus, S. K. Antimicrobial peptides: Diversity, mechanism of action and strategies to improve the activity and biocompatibility in vivo. Biomolecules. https://doi.org/10.3390/biom8010004 (2018).
Fernández-Vidal, M., Jayasinghe, S., Ladokhin, A. S. & White, S. H. Folding amphipathic helices into membranes: Amphiphilicity trumps hydrophobicity. J. Mol. Biol. 370, 459–470. https://doi.org/10.1016/j.jmb.2007.05.016 (2007).
Bechinger, B. & Gorr, S. U. Antimicrobial peptides: Mechanisms of action and resistance. J. Dent. Res. 96, 254–260. https://doi.org/10.1177/0022034516679973 (2017).
He, S., Stone, T. A. & Deber, C. M. Uncoupling amphipathicity and hydrophobicity: Role of charge clustering in membrane interactions of cationic antimicrobial peptides. Biochemistry. 60, 2586–2592. https://doi.org/10.1021/acs.biochem.1c00367 (2021).
Deslouches, B. et al. Activity of the de novo engineered antimicrobial peptide WLBU2 against Pseudomonas aeruginosa in human serum and whole blood: Implications for systemic applications. Antimicrob. Agents Chemother. 49, 3208–3216. https://doi.org/10.1128/aac.49.8.3208-3216.2005 (2005).
Zhang, L. et al. Antimicrobial peptide therapeutics for cystic fibrosis. Antimicrob. Agents Chemother. 49, 2921–2927. https://doi.org/10.1128/aac.49.7.2921-2927.2005 (2005).
Jenssen, H., Hamill, P. & Hancock, R. E. Peptide antimicrobial agents. Clin. Microbiol. Rev. 19, 491–511. https://doi.org/10.1128/cmr.00056-05 (2006).
Mishra, B., Lakshmaiah Narayana, J., Lushnikova, T., Wang, X. & Wang, G. Low cationicity is important for systemic in vivo efficacy of database-derived peptides against drug-resistant Gram-positive pathogens. Proc. Natl. Acad. Sci. USA 116, 13517–13522. https://doi.org/10.1073/pnas.1821410116 (2019).
Hall, C. W. & Mah, T. F. Molecular mechanisms of biofilm-based antibiotic resistance and tolerance in pathogenic bacteria. FEMS Microbiol. Rev. 41, 276–301. https://doi.org/10.1093/femsre/fux010 (2017).
Algburi, A., Comito, N., Kashtanov, D., Dicks, L. M. T. & Chikindas, M. L. Control of biofilm formation: Antibiotics and beyond. Appl. Environ. Microbiol. https://doi.org/10.1128/aem.02508-16 (2017).
Römling, U. & Balsalobre, C. Biofilm infections, their resilience to therapy and innovative treatment strategies. J. Intern. Med. 272, 541–561. https://doi.org/10.1111/joim.12004 (2012).
Ruhal, R. & Kataria, R. Biofilm patterns in gram-positive and gram-negative bacteria. Microbiol. Res. 251, 126829. https://doi.org/10.1016/j.micres.2021.126829 (2021).
Hoskin, D. W. & Ramamoorthy, A. Studies on anticancer activities of antimicrobial peptides. Biochim. Biophys. Acta. 1778, 357–375. https://doi.org/10.1016/j.bbamem.2007.11.008 (2008).
Grieco, P. et al. Alanine scanning analysis and structure-function relationships of the frog-skin antimicrobial peptide temporin-1Ta. J. Pept. Sci. 17, 358–365. https://doi.org/10.1002/psc.1350 (2011).
Percie du Sert, N. et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. J. Cerebral Blood Flow Metab. 40, 1769–1777. https://doi.org/10.1177/0271678X20943823 (2020).
Wu, C. L., Peng, K. L., Yip, B. S., Chih, Y. H. & Cheng, J. W. Boosting synergistic effects of short antimicrobial peptides with conventional antibiotics against resistant bacteria. Front. Microbiol. 12, 747760. https://doi.org/10.3389/fmicb.2021.747760 (2021).
Masihzadeh, S., Amin, M. & Farshadzadeh, Z. In vitro and in vivo antibiofilm activity of the synthetic antimicrobial peptide WLBU2 against multiple drug resistant Pseudomonas aeruginosa strains. BMC Microbiol. 23, 131. https://doi.org/10.1186/s12866-023-02886-x (2023).
Deslouches, B. et al. De novo generation of cationic antimicrobial peptides: Influence of length and tryptophan substitution on antimicrobial activity. Antimicrob. Agents Chemother. 49, 316–322. https://doi.org/10.1128/aac.49.1.316-322.2005 (2005).
Nguyen, L. T. et al. Serum stabilities of short tryptophan- and arginine-rich antimicrobial peptide analogs. PLOS ONE 5, e12684. https://doi.org/10.1371/journal.pone.0012684 (2010).
Ma, Y. et al. Generation of truncated derivatives through in silico enzymatic digest of peptide GV30 target MRSA both in vitro and in vivo. Comput. Struct. Biotechnol. J. 19, 4984–4996. https://doi.org/10.1016/j.csbj.2021.08.039 (2021).
Acknowledgements
This work was supported by the National Technology Foundation Project (U22A20152). The funder played no role in study design, data collection, analysis and interpretation of data, or the writing of this manuscript.
Author information
Authors and Affiliations
Contributions
LW, XC, XZ, MZ, FL and TC conceived the work. FL and XZ performed the experiments. JC, JZ and CM contributed materials. FL, TW and YJ analyzed the data. FL and XC wrote the first draft of the manuscript. CS, LW, XC, JZ and TC provided comments and corrections to the manuscript draft. All the authors have read and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liao, F., Ye, Z., Cheng, J. et al. Discovery and engineering of a novel peptide, Temporin-WY2, with enhanced in vitro and in vivo efficacy against multi-drug resistant bacteria. Sci Rep 14, 18769 (2024). https://doi.org/10.1038/s41598-024-67777-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-67777-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.