Abstract
Information on the associations of testosterone levels with abdominal muscle volume and density in men is limited, while the role of estradiol and SHBG on these muscle characteristics are unclear. Therefore, this study aimed to investigate the association between fasting serum sex hormones and CT-derived abdominal muscle area and radiodensity in adult men. Conducted as a cross sectional observational study using data from the Multi-Ethnic Study of Atherosclerosis, our analyses focused on a community-based sample of 907 men aged 45–84 years, with 878 men having complete data. CT scans of the abdomen were interrogated for muscle characteristics, and multivariable linear regressions were used to test the associations. After adjustment for relevant factors, higher levels of both total testosterone and estradiol were associated with higher abdominal muscle area (1.74, 0.1–3.4, and 1.84, 0.4–3.3, respectively). In the final analyses, levels of total testosterone showed a positive association, while an inverse relationship was observed for SHBG with abdominal muscle radiodensity (0.3, 0.0–0.6, and − 0.33, − 0.6 to − 0.1, respectively). Our results indicate a complex association between sex hormones and abdominal muscle characteristics in men. Specifically, total testosterone and estradiol were associated with abdominal muscle area, while only total testosterone was associated with muscle radiodensity and SHBG was inversely associated with muscle radiodensity.
Clinical Trial: NCT00005487
Similar content being viewed by others
Introduction
Abdominal obesity is linked to a higher risk of cardiometabolic disorders and mortality1. However, recent evidence suggests the quality and quantity of abdominal muscles may also play an important role in cardiometabolic health2. In this regard, abdominal muscle radiodensity, measured in Hounsfield Units (HU), reflects the function of muscle tissue and the degree of fat infiltration/fibrosis3. Of note, growing evidence suggests that abdominal muscle radiodensity is inversely associated with cardiovascular events and mortality in men2.
Earlier studies showed testosterone induces muscle fiber hypertrophy and increases the number of myonuclear cells by regulation of protein synthesis, satellite cells and stem cell proliferation, in addition to stimulation of the myogenic lineage and inhibition of the adipogenic cell line by activating androgen receptors (ARs)4,5. It is known that testosterone exerts regulatory control over pivotal proteins engaged in molecular processes such as glycolysis, glycogen synthesis as well as lipid and cholesterol metabolism6. Moreover, in men with testosterone deficiency, replacement therapy improves muscle volume, strength and quality of life7. However, it is unknown whether endogenous testosterone concentrations have different effects on different abdominal muscle functional groups. In addition, the effects of estradiol on muscle characteristics in men are unclear. In this regard, Russel and Colleagues reported in their review, no clear effect of estradiol on muscle mass or strength in men8. Yet, other studies have shown that estradiol could play a key role in regulating abdominal adiposity but is also directly associated with the volume of lean mass in men and appears to prevent expansion of adiposity8,9,10.
While there is some evidence to suggest that both testosterone and estradiol play an important role in the regulation of muscle function and volume, the nature of this relationship is not yet well established. Moreover, the bioactive role of sex hormone-binding globulin (SHBG) is still limited. Given this, we examined the cross-sectional associations between sex hormones and abdominal muscle characteristics in a large multi-ethnic sample of middle-aged and older men and hypothesized that higher estradiol and testosterone levels would be associated with greater abdominal muscle area, while testosterone would also be associated with higher abdominal muscle radiodensity.
Results
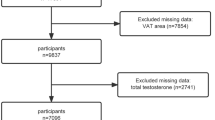
Baseline characteristics of the study population are presented in Table 1. The mean age was 61.6 years. The majority of participants where non-Hispanic White (42%), followed by Hispanic/Latino (27%), African American (17%) and Chinese American (14%). On average, men were overweight with a mean BMI of 27.6 kg/m2. The participants reported an average of 12 h a week of physical activity. Moreover, 42% of participants were hypertensive, 13% stated active cigarette smoking, 15% had diabetes mellitus, and 24% were taking a cholesterol-lowering medication. The mean total testosterone level was 15 nmol/L.
Association between sex hormones and abdominal muscle area
Total testosterone was significantly associated with total abdominal muscle area in the first model (B = 1.39, 95% CI 0.0–2.8, p = 0.05), which was accentuated with further adjustment (Model 2: 1.81, 0.2–3.5, p = 0.03; Model 3: 1.74, 0.1–3.3 p = 0.01) (Table 2). No significant associations were found between total testosterone and abdominal stabilizing muscle area (Table 3), while the associations were significant in all models for abdominal locomotor muscle area (Table 4).
Levels of estradiol were significantly associated with total abdominal muscle area in all three models: Model 1 (2.14, 0.8–3.6, p < 0.01), Model 2 (1.97, 0.6–3.4, p < 0.01), Model 3 (1.84, 0.4–3.3, p = 0.01) with similar results for abdominal locomotor and abdominal stabilizing muscle area (Tables 3 and 4).
No significant associations were found between levels of free testosterone and total abdominal muscle areas (Table 2), as well as stabilizing. However, significant associations were presented between free testosterone and locomotor area in model 1 & model 2(0.38, 0.0–0.7, p = 0.04, 0.37, 0.0–0.7, p = 0.04) with borderline significance in model 3 (0.34, − 0.0, 0.7, p = 0.05), respectively (Tables 2, 3, 4). Positive, although non-significant associations were found for SHBG with abdominal muscle areas (Tables 2, 3, 4).
Associations between sex hormones and abdominal muscle radiodensities
Total testosterone was significantly associated with total abdominal muscle radiodensity in Models 2 and 3, but not in Model 1 (Model 1: 0.04, − 0.2 to 0.3, p = 0.79; Model 2; 0.32, 0.1–0.7, p = 0.04; Model 3: 0.34, 0.0–0.6, p = 0.04) (Table 2). Similar results were found for radiodensity of stabilizing muscles (Table 3), but not for locomotor muscle (Table 4).
No significant associations were found between free testosterone and abdominal muscle radiodensities in fully adjusted models (Tables 2, 3, 4).
No significant associations were found between levels of estradiol and total abdominal and stabilizing muscle radiodensity (Tables 2, 3), but there was a borderline significant association between estradiol and abdominal locomotor muscle radiodensity ((Model 1: 0.27, − 0.0 to 0.6, p = 0.09: Model 2: 0.28, − 0.0 to 0.6, p = 0.07; Model 3: 0.26, − 0.0 to 0.6, p = 0.09) (Table 4).
Higher SHBG levels were associated with a lower radiodensity of abdominal muscle in all models (Model 1: − 0.35, − 0.6 to − 0.1, p = 0.02: Model 2: − 0.35, − 0.6 to − 0.1, p = 0.02; Model 3: − 0.34, − 0.6 to − 0.1, p = 0.02) (Table 2). The results were similar for abdominal stabilizing and locomotor muscles (Table 3).
Associations between sex hormones and abdominal muscle area indexes
A significant association was found in all models for total testosterone and TAMAi (Model 1: B = 0.10, 0.0–0.2, p < 0.01, Model 2: 0.11, 0.1–0.2, p < 0.01, Model 3: 0.10, 0.1–0.2, p < 0.01) (Table 2). That is, in fully adjusted models, one SD increase in testosterone levels resulted with an increase of 0.10 cm2/(weight/height2) in abdominal muscle area index. Similar relationships were observed between total testosterone and abdominal locomotor and stabilizing muscle area indices (Tables 3, 4).
Estradiol was found to be significantly associated with total abdominal muscle index (TAMAi) in model 1 (B = 0.10, 0.0–0.1, p = 0.03), which was borderline significant in Models 2 and 3 (Model 2: 0.05, − 0.0 to 0.1, p = 0.06, Model 3: 0.05, − 0.0 to 0.1, p = 0.06). Significant associations were found for estradiol and abdominal locomotor muscle area index in all three models but not for abdominal stabilizing muscle area index (Tables 3 and 4).
Significant associations were shown for free testosterone with total, stabilizing and abdominal muscle area index in fully adjusted models (0.08, 0.0, 0.1, p = 0.008, 0.05, 0.0–0.1, p = 0.03, 0.02, 0.0–0.04, p = 0.02), respectively. Non-significant associations were found between levels of SHBG and abdominal muscle area indexes in both models 2 and 3 (Tables 2, 3, 4).
Significant negative correlations were presented between visceral fat and total testosterone (r = − 0.22, p < 0.001), and total abdominal muscle radiodensity (r = − 0.40, p < 0.001), and SHBG (− 0.18, p < 0.001), while a positive association was found for total abdominal muscle area (r = 0.11, p < 0.001) and estradiol (0.02, p = 0.6). Furthermore, the association between visceral fat and abdominal muscle area and radiodensity, significant associations were found (B = 0.03 p = 0.011, B = − 0.03 p < 0.001, respectively) even after adjustment for sex hormones.
Discussion
Our study presents novel findings on the associations between sex hormones and SHBG and abdominal muscles in men. First, our results indicate that increases in serum levels of total testosterone and estradiol were associated with significant increases in abdominal muscle mass in men. In fact, our data indicate that the associations were stronger for estradiol than total testosterone with abdominal muscle mass to include both stabilizing and locomotor muscles. Second, significant associations were found between higher levels of total testosterone and abdominal muscle radiodensities. Third, our study presented a significant negative association between SHBG and abdominal muscle radiodensity.
A significant association was found between total testosterone and total abdominal muscle area in all models, with similar associations presented for abdominal locomotor muscle area but not for abdominal stabilizing muscle area. A plausible explanation of these differences might be that locomotor muscle contains a greater number of types II myofibers, a more dynamic and power related muscle type11. Type I myofibrils, which are predominantly found in the abdominal stabilizing muscles, have shown to be rather associated with endurance and higher lipid content11,12,13,14. Even though some studies have shown that supraphysiologic levels of testosterone increase both type I and II myofibers equally, other studies have reported testosterone affects maximal voluntary strength rather than endurance5,15. Another potential explanation to different associations between total testosterone and abdominal muscle groups could be from earlier studies, observing that administration of testosterone caused dose-dependent and region-specific changes in muscle mass16. This could potentially indicate that testosterone levels act differently strong, dependent on body region, type of muscle fibers and proportion of ectopic fat.
Interestingly, when adjustment was made for SHBG, lifestyle factors and time from baseline to CT in Model 2, a negative confounding was observed between total testosterone and both total abdominal muscle area and radiodensity. This underscores the significance of implementing additional adjustment, as the presence of visceral fat may obscure the true relationship between sex hormones and muscle composition. Furthermore, when adjustments were made for cardiometabolic disorders, similar associations were found. This could potentially suggest that the observed associations were less influenced by underlying metabolic conditions or were masked by prior adjustments.
Free testosterone was positively associated with total abdominal muscle area and radiodensity although, significance was found for locomotor muscle area in model 1 and 2 with borderline significance in model 3. Total and free testosterone was positively associated with TAMAi, abdominal stabilizing and locomotor muscle area indexes. In concurrence with our findings, Han et al. reported similar outcomes presented between total testosterone and abdominal muscle area index in men17. However, no adjustments were made for SHBG.
In our study, total testosterone was associated with increased total and stabilizing abdominal muscle radiodensities, independent of confounding factors.
Our results suggest that total testosterone is significantly associated with the degree of radiodensity of abdominal muscles, including muscle size. Total testosterone mainly includes SHBG-bound testosterone which has long been assumed to be inactive. However, recent experimental studies have shown the endocytic Megalin receptor, found in human skeletal myocytes, transports SHBG-bound testosterone and estradiol into cells18,19. This could indicate total testosterone may have an active role in cell regulation and muscle activity.
Our study found that higher levels of estradiol were significantly associated with higher levels of all abdominal muscle areas. In men, a great quantity of estradiol originates from the aromatase conversion of testosterone, which correlates with fat mass, activating estradiol receptors (ERs) in muscles20,21. This potentially suggests that testosterones` impact on muscles partly operates indirectly through its conversion to estradiol. Estradiol has earlier been found to play a key role in regulation of myokines, i.e., skeletal muscle proteins, with critical functions associated with exercise-related benefits and inflammation regulation in tissues22. Similarly, in a study on elderly Swedish men, estradiol, and not testosterone, was associated with lean mass measured with DXA scans9.
Estradiol was positively but non-significantly associated with total and stabilizing muscle radiodensities, while a borderline-significant association was shown with locomotor muscle radiodensity. Studies have reported higher concentrations of estradiol receptors in skeletal muscles of men engaged in greater endurance trainings as well as that supplementation of estradiol increases lipid utilization in skeletal muscles, increasing strength23,24. Whether there is region-specific dose-dependent effects is unknown. However, differences in associations between different sex hormone concentrations and various muscle radiodensities may stem from distinct mechanisms of action mediated by activation of androgen receptors or estrogen receptors in skeletal muscles. It has been demonstrated in animal experiments, that these receptors activate different genes, potentially accounting for the observed differences25.
The bioactive role of SHBG is still debated. An inverse association has been shown between SHBG levels and insulin resistance and metabolic syndrome48. Other studies have found a positive association between SHBG and inflammatory cytokines, low protein diet and hip fractures in elderly even after adjustment for sex hormones26,27. Our study showed an inverse association between SHBG and all abdominal muscle densities. Other results have reported an inverse relationship between SHBG and lean muscles measured by dual-energy x-ray absorptiometry (DXA) scans9. Furthermore, Yuki et al. reported SHBG levels were significantly higher in the group of individuals diagnosed with sarcopenia compared to the normal group28. In agreement with our findings, SHBG has been reported to have a significant inverse association with muscle strength in elderly men29. One plausible cause of the negative associations between SHBG and abdominal muscle radiodensities could be that an increase in SHBG concentrations may influence the binding capacity and magnitude of available free testosterone and could suggest a partial explanation to some of the weaker association found for other sex hormones30.
We imply the importance of investigating the interplay among sex hormones, SHBG and muscle tissue composition, given prior findings suggesting that both muscle area and radiodensity exert notable influences on survival among men31.
This study has a number of strengths, including usage of data from a large and diverse cohort, detailed sampling of information with validated instruments as well as standardized sampling of blood specimens according to guidelines32. Furthermore, by assessing muscle composition with CT, we were able to indirectly estimate its quality. In earlier studies, radiological slices of the lumbar region are approved when assessment of total muscle volumes are made33. However, our study does have some limitations. First, radioimmunoassay technique (RIA) was used to measure sex hormones and SHBG. This has been described to be less precise than mass spectrometry in the measurement of sex-hormone levels34. Studies have found that it could potentially result in difficulties in distinguishing eugonadal from mildly hypogonadal males35. Furthermore, levels of sex hormones may be affected by the presence of several cross-reacting steroids36. Second, free testosterone was calculated and not directly measured which has been shown to overestimate levels compared with laboratory measured free testosteronex37. While the Sodergard method has previously been described as one of the most common methods for calculating free testosterone in endocrinology literature, it has limitations, including a predefined albumin concentration, higher estimates compared to other algorithms and accurate mainly when competing steroids to binding sites are limited and normal levels of SHBG are involved38,39. Furthermore, the Sodergard method presents concordant results to the Vermuelen algorithm and its association constant was validated when compared to results of calculations with a gold standard technique38. Third, the sex hormones were measured at visit 1 whilst CT scans were made at visit 2 and visit 3. We partially addressed this limitation by adjusting for the time from baseline to CT scan as a confounder in model 2 and model 3. Also, the energy levels of the X-ray beams differed between the CT-scans (120–140 kVp) which is a further limitation. In addition, measurements of physical activity and sedentary behavior were self-reported.
We only evaluated abdominal muscle area and radiodensity, and therefore, our findings may not be applicable to peripheral muscles. Finally, this study design was observational and cross-sectional, which is prone to residual confounding, as well as temporal and selection biases.
Conclusion
In this analysis, we demonstrate a positive association between total testosterone levels with abdominal muscle area and radiodensity, whereas estradiol showed a similar strong association with abdominal muscle area but not radiodensity. Additionally, SHBG was significantly and inversely associated with abdominal radiodensity although a negative trend was presented for abdominal muscle index. These results suggest the relevance of sex hormone levels to maintain muscle mass and density with advancing age.
Material and methods
Study design and study population
At baseline (2000 to 2002), 6814 adult men and women between 45–84 years that were free of clinical cardiovascular disease were recruited into the Multi-Ethnic Study of Atherosclerosis (MESA). Participants were enrolled from six US communities (New York [NY], Baltimore [MD], Chicago [IL], Los Angeles [CA], Twin Cities [MI] and Winston-Salem [NC]). Approximately 38% were Non-Hispanic White, 28% African American, 23% Hispanic American, and 11% Chinese American.
Data collection
Details on the MESA cohort methods have been published40. Briefly, trained staff performed specimen blood draws and processing of venous blood samples, blood pressure measurements and all interviews. Using standard procedures, fasting blood samples were processed and stored at − 80 °C41.
Information on lifestyle factors, medications and co-morbidities were gathered using validated questionnaires. Race/ethnicity was self-reported at baseline according to 2000 US Census criteria. All individuals treated with sex hormones at baseline were excluded from the study. Physical activity (hours/week) and sedentary behavior (hours/week) were measured by using a comprehensive, semiquantitative questionnaire42. Current medication use was assessed according to standardized questionnaires43. Hypertension was defined as a systolic blood pressure above 140 mmHg and/or a diastolic above 90 mmHg or taking a blood pressure lowering medication. Diabetes mellitus was defined as self-reported diabetes, fasting glucose levels according to the American Diabetes Association or use of glucose lowering medications44,45. Measurement of high-sensitivity C-reactive protein (hsCRP), a marker of systemic inflammation, has previously been described41,46.
Computed tomography for body composition
A random subset of 1970 participants (946 men) at visits 2 and 3 (2002 to 2005) were enrolled in an ancillary study obtaining abdominal computed tomography (CT) scans. The original aim of investigating CT-scans of the abdominal cavity was to measure the abdominal aortic calcium. Recently, approved data from the CT-scans were used to assess information on muscle composition such as muscle radiodensity and muscle area, which were then interrogated for abdominal muscle area, abdominal radiodensity, visceral adipose tissue and subcutaneous fat tissue. At two clinical sites (Northwestern University, University of California Los Angeles) electron-beam CT scanner (Imatron C-150) was used while at the remaining clinical sites (Columbia University, Wake Forest University, and University of Minnesota) multi-detector CT scanners (Sensation 64 GE lightspeed, Siemens S4 Volume Zoom, and Siemens Sensation 16) were used. A 35 cm field of view was used. In cases where a cut off was seen, imputation methods such as doubled values with repeat measure t-tests on a random sample, regression equations and ruler lines were used to estimate the areas. CT scans were set at a collimation of 3 mm with a slice thickness of 6 mm. In total, two cross-sectional slices were taken at L2/L3, L3/L4 and L4/L5 intervertebral disc spaces, constituting a total of six slices. Approximately half of the participants underwent CT scans at visit two and the other half at visit three.
Assessment of abdominal muscles and adipose tissue were obtained from these CT scans (Fig. 1a, b) and have been described earlier31. Using a semi-automated method, measurement of total tissue, lean muscle, and adipose tissue were assessed in each slice using Medical Imaging Processing Analysis and Visualization (MIPAV) software version 4.1.2 (National Institutes of Health, Bethesda, Maryland). Abdominal tissue was categorized by Hounsfield units (HU) with − 190 to − 30 HU assessed as adipose tissue, − 30 to − 0 HU defined as mixed connective tissue or undefined, while values 0 to 100 HU were set as muscle tissue47,48. Abdominal muscle area and adipose tissue area were calculated by summing the number of pixels while muscle radiodensity was defined by average HU value measured within that muscle’s corresponding fascial plane. Research staff responsible for analyzing CT scans were blinded to participants’ clinical information. The inter- and intra-rater reliability of measurements for total abdominal area as well as measurements for all muscle groups was 0.99 and 0.93 to 0.98, respectively28.
(a) Presents an axial slice from the lumbar region, including abdominal adipose and muscle tissue. Abdominal tissue was categorized by Hounsfield units (HU), with − 190 to − 30 HU assessed as adipose tissue, − 30 to − 0 HU defined as mixed connective tissue, while values 0 to 100 HU were set as lean muscle47,48. Abdominal muscle area and adipose tissue area were calculated by summing the number of pixels, while muscle radiodensity was defined by average HU value measured within that muscle’s corresponding fascial plane. The abdominal muscles were further categorized into stabilizing muscle groups (rectus abdominis, oblique muscle groups and paraspinal muscles) and locomotor muscle groups (psoas muscles). Subcutaneous adipose tissue was determined as adipose tissue in the subcutaneous area, whilst visceral adipose tissue was determined as fat tissue in the visceral cavity, excluding intermuscular fat. (b) Presents a sagittal slice from the lumbar region. 6 transverse cross section slices of data were analyzed; slice 0 is located at the L4/L5 vertebral junction and slice 1 is the immediately superior and adjacent to slice 0. Slice 2 is located at the L3/L4 junction with slice 3 superior and adjacent to slice 2. Slice 4 is located at the L2/L3 vertebral junction with slice 5 superior and adjacent to slice 4. CT scans were set at a collimation of 3 mm with a slice thickness of 6 mm.
Visceral adipose tissue was determined as fat tissue in the visceral cavity, excluding intermuscular fat. Four abdominal muscle groups were assessed bilaterally. Area and radiodensity of the oblique and paraspinal muscle group, and rectus abdominis comprised the muscles of stabilization, while the psoas muscles were the locomotor group. Combined area and radiodensity of muscles of stabilization and locomotion were defined as total abdominal muscle area (TAMA) and radiodensity (TAMD), respectively.
Adjustment for body mass index (BMI, kg/m2) was made for abdominal muscle areas and were defined as abdominal muscle indexes (total abdominal muscle area index (TAMAi) (TAMAi = TAMA/BMI), stabilizing muscle area index (TSMA/BMI) and locomotor muscle area index (TLMA/BMI)17.
For this analysis we excluded participants with missing data from CT scans, anthropometric measurements, endogenous sex hormones, lifestyle factors, co-morbidities, and medication use. The final sample size included 878 men (Fig. 2).
Assessment of endogenous sex hormones
Measurement of endogenous sex hormone levels have previously been described49,50. In brief, total testosterone was measured using radioimmunoassay kits. Sex hormone binding globulin (SHBG) concentration was assessed by Chemiluminescent enzyme immunometric assay (Immulite kits, Diagnostic Products Corporation, Los Angeles, CA). A ~ 10% blind pool was obtained to assess quality control serum. The coefficients of variation for total testosterone and SHBG were 12.3% and 9.0%, respectively51. Estradiol was measured using an ultra-sensitive radioimmunoassay kit (Diagnostic System Laboratories, Webster, TX) with a coefficient of variation of 10.5%51. Free testosterone( nmol/L) was calculated according to the method of Södergård52.
Statistical analysis
Continuous variables were reported as means and standard deviations (SD) while categorical variables were shown as frequencies and percentages. Abdominal muscle areas/radiodensities/indexes were the outcome variables. Total, locomotor and stabilizing abdominal muscles showed normal distributions. As such, multivariable linear regression models were conducted to assess the associations between levels of sex hormones and abdominal muscles. The outcome was defined as the change in HU for abdominal muscle radiodensity and square centimeters for abdominal muscle area for each 1-SD increment change in levels of testosterone, estradiol and SHBG.
Model 1 adjusted for age, race/ethnicity, and visceral adipose tissue. Model 2 included variables from model 1 and SHBG (when investigating the associations of total testosterone and estradiol), CRP, physical activity, sedentary behavior, cigarette smoking, alcohol use, and time from baseline to CT. Model 3 included variables from Model 2 with further adjustment for hypertension, diabetes mellitus, dyslipidemia, use of oral steroids or cholesterol/thyroid/hypertension/glucose lowering/ medication. No adjustment was made for SHBG when free testosterone was the independent variable.
A two-tailed p-value < 0.05 was considered as statistically significant. Analyses were conducted using IBM SPSS Statistics, version 29.
Ethical considerations
The MESA study protocol was approved by the Institutional Review Board at the Johns Hopkins University Hospital, University of California Los Angeles, University of Minnesota, Wake Forest University Hospital, Northwestern University Hospital, and Columbia University. All methods were performed in accordance with the relevant guidelines and regulations as set by the approving institutions in a standardized manner. Written informed consent was given by all participants40.
Data availability
The supporting data for the conclusions drawn in this study can be obtained from the MESA committee, but access is subject to certain restrictions as they were utilized under license for the present study and are not publicly accessible. Nevertheless, interested parties can request access to the data directly from the authors, pending approval from the MESA committee. https://www.mesa-nhlbi.org.
References
Zhang, C., Rexrode, K. M., van Dam, R. M., Li, T. Y. & Hu, F. B. Abdominal obesity and the risk of all-cause, cardiovascular, and cancer mortality. Circulation 117(13), 1658–1667. https://doi.org/10.1161/CIRCULATIONAHA.107.739714 (2008).
Miljkovic, I., Vella, C. A. & Allison, M. Computed tomography-derived myosteatosis and metabolic disorders. Diabetes Metab. J. 45(4), 482–491. https://doi.org/10.4093/dmj.2020.0277 (2021).
De Marco, D. et al. Muscle area and density assessed by abdominal computed tomography in healthy adults: Effect of normal aging and derivation of reference values. J. Nutr. Health Aging 26(2), 243–246. https://doi.org/10.1007/s12603-022-1746-3 (2022).
Singh, R., Artaza, J. N., Taylor, W. E., Gonzalez-Cadavid, N. F. & Bhasin, S. Androgens stimulate myogenic differentiation and inhibit adipogenesis in C3H 10T1/2 pluripotent cells through an androgen receptor-mediated pathway. Endocrinology 144(11), 5081–5088. https://doi.org/10.1210/en.2003-0741 (2003).
Sinha-Hikim, I. et al. Testosterone-induced increase in muscle size in healthy young men is associated with muscle fiber hypertrophy. Am. J. Physiol. Endocrinol. Metab. 283(1), E154–E164. https://doi.org/10.1152/ajpendo.00502.2001 (2002).
Kelly, D. M. & Jones, T. H. Testosterone: A metabolic hormone in health and disease. J. Endocrinol. 217(3), R25-45. https://doi.org/10.1530/joe-12-0455 (2013).
Srinivas-Shankar, U. et al. Effects of testosterone on muscle strength, physical function, body composition, and quality of life in intermediate-frail and frail elderly men: A randomized, double-blind, placebo-controlled study. J. Clin. Endocrinol. Metab. 95(2), 639–650. https://doi.org/10.1210/jc.2009-1251 (2010).
Russell, N. & Grossmann, M. MECHANISMS IN ENDOCRINOLOGY: Estradiol as a male hormone. Eur. J. Endocrinol. 181(1), R23–R43. https://doi.org/10.1530/eje-18-1000 (2019).
Vandenput, L. et al. Serum estradiol is associated with lean mass in elderly Swedish men. Eur. J. Endocrinol. 162(4), 737–745. https://doi.org/10.1530/eje-09-0696 (2010).
Finkelstein, J. S. et al. Gonadal steroids and body composition, strength, and sexual function in men. N. Engl. J. Med. 369(11), 1011–1022. https://doi.org/10.1056/NEJMoa1206168 (2013).
Baracos, V. E. Psoas as a sentinel muscle for sarcopenia: A flawed premise. J Cachexia Sarcopenia Muscle. 8(4), 527–528. https://doi.org/10.1002/jcsm.12221 (2017).
Häggmark, T. & Thorstensson, A. Fibre types in human abdominal muscles. Acta Physiol. Scand. 107(4), 319–325. https://doi.org/10.1111/j.1748-1716.1979.tb06482.x (1979).
Goodpaster, B. H. et al. Attenuation of skeletal muscle and strength in the elderly: The Health ABC Study. J. Appl. Physiol. (1985) 90(6), 2157–2165. https://doi.org/10.1152/jappl.2001.90.6.2157 (2001).
He, J., Watkins, S. & Kelley, D. E. Skeletal muscle lipid content and oxidative enzyme activity in relation to muscle fiber type in type 2 diabetes and obesity. Diabetes 50(4), 817–823. https://doi.org/10.2337/diabetes.50.4.817 (2001).
Herbst, K. L. & Bhasin, S. Testosterone action on skeletal muscle. Curr. Opin. Clin. Nutr. Metab. Care 7(3), 271–277. https://doi.org/10.1097/00075197-200405000-00006 (2004).
Woodhouse, L. J. et al. Dose-dependent effects of testosterone on regional adipose tissue distribution in healthy young men. J. Clin. Endocrinol. Metab. 89(2), 718–726. https://doi.org/10.1210/jc.2003-031492 (2004).
Han, S. et al. Testosterone is associated with abdominal body composition derived from computed tomography: A large cross sectional study. Sci. Rep. 12(1), 22528. https://doi.org/10.1038/s41598-022-27182-y (2022).
Hammes, A. et al. Role of endocytosis in cellular uptake of sex steroids. Cell 122(5), 751–762. https://doi.org/10.1016/j.cell.2005.06.032 (2005).
Poole, C. N., Roberts, M. D., Dalbo, V. J., Sunderland, K. L. & Kerksick, C. M. Megalin and androgen receptor gene expression in young and old human skeletal muscle before and after three sequential exercise bouts. J. Strength Condition. Res. 25(2), 309–317. https://doi.org/10.1519/JSC.0b013e318202e45d (2011).
Matsumine, H., Hirato, K., Yanaihara, T., Tamada, T. & Yoshida, M. Aromatization by skeletal muscle. J. Clin. Endocrinol. Metab. 63(3), 717–720. https://doi.org/10.1210/jcem-63-3-717 (1986).
Barros Rodrigo, P. A. & Gustafsson, J. -Å. Estrogen receptors and the metabolic network. Cell Metab. 14(3), 289–299. https://doi.org/10.1016/j.cmet.2011.08.005 (2011).
Velez, L. M. et al. Genetic variation of putative myokine signaling is dominated by biological sex and sex hormones. Elife 11, e76887. https://doi.org/10.7554/eLife.76887 (2022).
Wiik, A. et al. Expression of oestrogen receptor alpha and beta is higher in skeletal muscle of highly endurance-trained than of moderately active men. Acta Physiol. Scand. 184(2), 105–112. https://doi.org/10.1111/j.1365-201X.2005.01433.x (2005).
Maher, A. C., Akhtar, M. & Tarnopolsky, M. A. Men supplemented with 17beta-estradiol have increased beta-oxidation capacity in skeletal muscle. Physiol Genomics 42(3), 342–347. https://doi.org/10.1152/physiolgenomics.00016.2010 (2010).
Svensson, J., Movérare-Skrtic, S., Windahl, S., Swanson, C. & Sjögren, K. Stimulation of both estrogen and androgen receptors maintains skeletal muscle mass in gonadectomized male mice but mainly via different pathways. J. Mol. Endocrinol. 45(1), 45–57. https://doi.org/10.1677/jme-09-0165 (2010).
Wang, Q. et al. Sex hormone-binding globulin associations with circulating lipids and metabolites and the risk for type 2 diabetes: Observational and causal effect estimates. Int. J. Epidemiol. 44(2), 623–637. https://doi.org/10.1093/ije/dyv093 (2015).
Osmancevic, A., Daka, B., Michos, E. D., Trimpou, P. & Allison, M. The association between inflammation, testosterone and SHBG in men: A cross-sectional Multi-Ethnic Study of Atherosclerosis. Clin. Endocrinol. (Oxf.) https://doi.org/10.1111/cen.14930 (2023).
Yuki, A. et al. Relationship between low free testosterone levels and loss of muscle mass. Sci. Rep. 3(1), 1818. https://doi.org/10.1038/srep01818 (2013).
Auyeung, T. W. et al. Testosterone but not estradiol level is positively related to muscle strength and physical performance independent of muscle mass: A cross-sectional study in 1489 older men. Eur. J. Endocrinol. 164(5), 811–817. https://doi.org/10.1530/eje-10-0952 (2011).
Narinx, N. et al. Role of sex hormone-binding globulin in the free hormone hypothesis and the relevance of free testosterone in androgen physiology. Cell Mol. Life Sci. 79(11), 543. https://doi.org/10.1007/s00018-022-04562-1 (2022).
Larsen, B. et al. Muscle area and density and risk of all-cause mortality: The Multi-Ethnic Study of Atherosclerosis. Metabolism 111, 154321. https://doi.org/10.1016/j.metabol.2020.154321 (2020).
Al-Sharefi, A. & Quinton, R. Current national and international guidelines for the management of male hypogonadism: Helping clinicians to navigate variation in diagnostic criteria and treatment recommendations. Endocrinol. Metab. (Seoul) 35(3), 526–540. https://doi.org/10.3803/EnM.2020.760 (2020).
Schweitzer, L. et al. What is the best reference site for a single MRI slice to assess whole-body skeletal muscle and adipose tissue volumes in healthy adults?. Am. J. Clin. Nutr. 102(1), 58–65. https://doi.org/10.3945/ajcn.115.111203 (2015).
Trost, L. W. & Mulhall, J. P. Challenges in testosterone measurement, data interpretation, and methodological appraisal of interventional trials. J. Sex Med. 13(7), 1029–1046. https://doi.org/10.1016/j.jsxm.2016.04.068 (2016).
Wang, C., Catlin, D. H., Demers, L. M., Starcevic, B. & Swerdloff, R. S. Measurement of total serum testosterone in adult men: Comparison of current laboratory methods versus liquid chromatography-tandem mass spectrometry. J. Clin. Endocrinol. Metab. 89(2), 534–543. https://doi.org/10.1210/jc.2003-031287 (2004).
Krasowski, M. D. et al. Cross-reactivity of steroid hormone immunoassays: Clinical significance and two-dimensional molecular similarity prediction. BMC Clin. Pathol. 14, 33. https://doi.org/10.1186/1472-6890-14-33 (2014).
Ly, L. P. & Handelsman, D. J. Empirical estimation of free testosterone from testosterone and sex hormone-binding globulin immunoassays. Eur. J. Endocrinol. 152(3), 471–478. https://doi.org/10.1530/eje.1.01844 (2005).
Vermeulen, A., Verdonck, L. & Kaufman, J. M. A critical evaluation of simple methods for the estimation of free testosterone in serum. J. Clin. Endocrinol. Metab. 84(10), 3666–3672. https://doi.org/10.1210/jcem.84.10.6079 (1999).
de Ronde, W. et al. Calculation of bioavailable and free testosterone in men: A comparison of 5 published algorithms. Clin Chem. 52(9), 1777–1784. https://doi.org/10.1373/clinchem.2005.063354 (2006).
Bild, D. E. et al. Multi-Ethnic Study of atherosclerosis: Objectives and design. Am. J. Epidemiol. 156(9), 871–881. https://doi.org/10.1093/aje/kwf113 (2002).
Bhatraju, P. K., Zelnick, L. R., Shlipak, M., Katz, R. & Kestenbaum, B. Association of soluble TNFR-1 concentrations with long-term decline in kidney function: The Multi-Ethnic Study of atherosclerosis. J. Am. Soc. Nephrol. 29(11), 2713–2721. https://doi.org/10.1681/asn.2018070719 (2018).
Ainsworth, B. E., Irwin, M. L., Addy, C. L., Whitt, M. C. & Stolarczyk, L. M. Moderate physical activity patterns of minority women: The Cross-Cultural Activity Participation Study. J. Womens Health Gend Based Med. 8(6), 805–813. https://doi.org/10.1089/152460999319129 (1999).
Psaty, B. M. et al. Assessing the use of medications in the elderly: Methods and initial experience in the Cardiovascular Health Study. The Cardiovascular Health Study Collaborative Research Group. J. Clin. Epidemiol. 45(6), 683–692. https://doi.org/10.1016/0895-4356(92)90143-b (1992).
Kramer, H. et al. Racial/ethnic differences in hypertension and hypertension treatment and control in the Multi-Ethnic Study of atherosclerosis (MESA). Am. J. Hypertens. 17(10), 963–970. https://doi.org/10.1016/j.amjhyper.2004.06.001 (2004).
Bertoni, A. G., Kramer, H., Watson, K. & Post, W. S. Diabetes and clinical and subclinical CVD. Glob Heart. 11(3), 337–342. https://doi.org/10.1016/j.gheart.2016.07.005 (2016).
Harhay, M. O. et al. Relationship of CRP, IL-6, and fibrinogen with right ventricular structure and function: The MESA-Right Ventricle Study. Int. J. Cardiol. 168(4), 3818–3824. https://doi.org/10.1016/j.ijcard.2013.06.028 (2013).
Aubrey, J. et al. Measurement of skeletal muscle radiation attenuation and basis of its biological variation. Acta Physiol. (Oxf.) 210(3), 489–497. https://doi.org/10.1111/apha.12224 (2014).
Goodpaster, B. H., Thaete, F. L. & Kelley, D. E. Composition of skeletal muscle evaluated with computed tomography. Ann. N. Y. Acad. Sci. 904, 18–24. https://doi.org/10.1111/j.1749-6632.2000.tb06416.x (2000).
Zhao, D. et al. Endogenous sex hormones and incident cardiovascular disease in post-menopausal women. J. Am. Coll. Cardiol. 71(22), 2555–2566. https://doi.org/10.1016/j.jacc.2018.01.083 (2018).
Michos, E. D. et al. Sex hormones, sex hormone binding globulin, and abdominal aortic calcification in women and men in the Multi-Ethnic Study of atherosclerosis (MESA). Atherosclerosis. 200(2), 432–438. https://doi.org/10.1016/j.atherosclerosis.2007.12.032 (2008).
Golden, S. H. et al. Endogenous sex hormones and glucose tolerance status in postmenopausal women. J. Clin. Endocrinol. Metab. 92(4), 1289–1295. https://doi.org/10.1210/jc.2006-1895 (2007).
Södergård, R., Bäckström, T., Shanbhag, V. & Carstensen, H. Calculation of free and bound fractions of testosterone and estradiol-17 beta to human plasma proteins at body temperature. J. Steroid. Biochem. 16(6), 801–810. https://doi.org/10.1016/0022-4731(82)90038-3 (1982).
Acknowledgements
The authors thank the other investigators, the staff, and the participants of the MESA study for their valuable contributions. A full list of participating MESA investigators and institutions can be found at http://www.mesa-nhlbi.org.
Funding
Open access funding provided by University of Gothenburg. The study was supported by The Local Research and Development Council Göteborg och Södra Bohuslän, the VGR Regional Research and Development Council Grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF-agreement ALFGBG-966255. Moreover, this research was supported by contracts 75N92020D00001, HHSN268201500003I, N01-HC-95159, 75N92020D00005, N01-HC-95160, 75N92020D00002, N01-HC-95161, 75N92020D00003, N01-HC-95162, 75N92020D00006, N01-HC-95163, 75N92020D00004, N01-HC-95164, 75N92020D00007, N01-HC-95165, N01-HC-95166, N01-HC-95167, N01-HC-95168 and N01-HC-95169 from the National Heart, Lung, and Blood Institute, and by grants UL1-TR-000040, UL1-TR-001079, and UL1-TR-001420 from the National Center for Advancing Translational Sciences (NCATS). The sex hormone ancillary study was supported by R01 HL074406 and R01 HL074338 from the National Heart, Lung, and Blood Institute. This paper has been reviewed and approved by the MESA Publications and Presentations Committee.
Author information
Authors and Affiliations
Contributions
A.O.: Conceptualization, Methodology, Formal Analysis, Writing—Original Review, M.A., B.D.: Conceptualization, Writing—Review and Editing, Supervision, Formal Analysis, Methodology, I.M., C.V., P.O., P.T.: Writing—Substantial Reviews, Interpretations and Edits. Each author has reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Osmancevic, A., Allison, M., Miljkovic, I. et al. Levels of Sex Hormones and Abdominal Muscle Composition in Men from The Multi-Ethnic Study of Atherosclerosis. Sci Rep 14, 16114 (2024). https://doi.org/10.1038/s41598-024-66948-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-66948-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.