Abstract
The ability of a plant parasitic nematode to infect and reproduce within a host plant depends on its genotype and the environmental conditions before and during infection. We studied the culturing conditions of the root lesion nematode Pratylenchus neglectus to produce inoculum for plant infection tests. Nematodes were either cultivated on carrot calli for different periods or directly isolated from the roots of the host plants. After infection of wheat and barley plants in the greenhouse, nematodes were quantified by RT-qPCR and by visual counting of the nematodes. We observed drastically reduced infection rates after long-term (> 96 weeks) cultivation on carrot callus. In contrast, fresh isolates from cereal roots displayed much higher pathogenicity. We recommend using root lesion nematodes cultivated on carrot calli no longer than 48 weeks to guarantee uniform infection rates.
Similar content being viewed by others
Introduction
Root lesion nematodes (RLN) of the genus Pratylenchus are the world's third most significant plant parasitic nematodes (PPN) after root-knot and cyst nematodes1. Four species causing high yield losses in a wide range of crops (P. crenatus, P. neglectus, P. penetrans, and P. thornei) are abundant in different climate zones around the globe. In temperate regions, P. neglectus and P. thornei are major threats to cereal production. They often occur as a mixed population2,3,4, primarily feeding on plant roots. Upon infection, necrotic lesions are formed that can reduce the plant's ability to absorb nutrients, which, in combination with secondary infections by other pathogens, can result in stunted growth, wilting, and reduced yield1,2,5. The severity of Pratylenchus infestations depends on the nematode species' composition, crop rotations, environmental conditions, and the nematode's pathogenicity2.
A standard inoculum is used for testing large plant populations during plant breeding. Appropriate methods for cultivating Pratylenchus spp. were established by Mountain6 for peach and tobacco. Since then, the monoxenic culture on carrot calli has been commonly used2,6. The published protocols differ only by flame or chemical sterilization of carrot discs or the chemicals used for surface sterilization of nematodes (HgCl2 or streptomycin sulfate) and their concentration7,8,9,10.
The cultivation on carrot calli aims to multiply nematodes by maintaining their genotype and pathogenicity. The pathogenicity is composed of fitness and virulence11, defined as the nematode's capacity to initiate a host-parasite interaction and its ability to inflict damage2. Fitness refers to the reproduction rate in a given environment. Virulence, in this respect, has been defined as the severity of infection, whereas aggressiveness refers to the ability of a nematode to compete with other nematodes or parasites for resources within the host11,12,13. The pathogenicity of Pratylenchus species is influenced by their virulence and fitness, by the host's vigorousness, e.g., due to nutrient supply, and by edaphic factors such as soil texture and temperature, and the interaction with the microbiome2,11,12,14,15,16,17,18. Although no Pratylenchus spp. races have been reported so far, host compatibility and pathogenicity in vegetable and fruit tree crops vary between isolates from different geographical regions2,19,20.
The interaction between nematodes and their microbiomes has been well-studied in the past years21,22,23,24,25,26,27. Dirksen, et al.22 discovered a species-rich bacterial community associated with Caenorhabditis elegans inside its pharynx (oesophagus), dominated by Proteobacteria such as Enterobacteriaceae of the genera Pseudomonas, Stenotrophomonas, Ochrobactrum, and Sphingomonas. The associated microbiome can have a direct impact on host development by supplying food and necessary metabolic compounds28 or by working as a stimulus for morphogenesis and/or pathogenicity22,26,27,29. Thus, the bacterial community can improve the nematode's fitness, especially under stress conditions. Another study with P. neglectus found that various bacterial species attached to the cuticle influenced the interactions with the host plant. In particular, the presence of Rothia sp. decreased the penetrating efficiency and pathogenicity in barley roots26.
This project aimed to assess the pathogenicity of P. neglectus after cultivation under different environments. For infection of cereal roots, we used nematodes cultivated on carrot callus and barley and wheat roots. Low infection rates suggested that long-term cultivation on carrot callus drastically reduced the pathogenicity of root lesion nematodes. This study demonstrates that infection rates can be severely flawed by inappropriate inoculum. Thus, the results are vital for breeding resistant cereal varieties.
Results
We aimed to investigate the nematode's pathogenicity after growth under different environments. We suspected extended pre-culture might decrease pathogenicity. Therefore, P. neglectus was grown under monoxenic culture conditions on carrot callus and quasi-natural conditions on barley and wheat roots. The monoxenic carrot culture (CC1, CC2, and CC3) isolates were obtained after cultivation between > 96 and 48 weeks. The LHP isolate was maintained for 48 weeks on living host plants and extracted directly from barley and wheat roots infected with P. neglectus. The PR1 to 4 isolates were also obtained from infected barley and wheat roots with CC1 to 3 and LHP isolates (Fig. 1 and Table 2).
Experimental design and production of P. neglectus inoculum from different origins. The susceptible barley variety Valentina and the susceptible wheat variety Machete were utilized. The experiments were performed twice, with 30 biological replicates. The nematode isolates were obtained between > 96 to 48 weeks on carrot callus (isolates CC1, CC2, CC3) or from barley and wheat roots (LHP and PR1, PR2, PR3, PR4) (Table 2). Ten days after sowing, the plants were inoculated with 1000 nematodes and grown in the greenhouse under long-day conditions (16 h of light) at 23 °C during the day and 18 °C at night. The plants were harvested eight weeks after inoculation. Non-inoculated plants were used as controls.
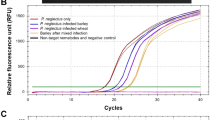
As a result, all morphological features aligned with the common descriptors of P. neglectus (data not shown). Moreover, species identity was determined by the species-specific primer combination Neg1. The PCR yielded the expected 234 bp amplicon (Supplementary Fig. 1A). The RT-qPCR assay showed the expected amplification curves and a single melting peak at 81.5 °C (Supplementary Fig. 1B, C)30.
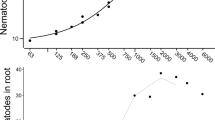
The roots were harvested eight weeks after infection. The number of nematodes per barley and wheat plant after infection with CC and LHP isolates ranged from 708.24 ± 83.46 to 1853.29 ± 72.4 (Pf/Pi from 0.71 to 1.85) and from 576.07 ± 86.23 to 1831.08 ± 96.2 (Pf/Pi from 0.58 to 1.83), respectively (Fig. 2; Table 1). After infection with PR isolates, the number of nematodes ranged from 926.38 ± 54.13 to 1894.23 ± 83.07 (Pf/Pi from 0.93 to 1.89) and from 779.62 ± 65.09 to 1853.99 ± 55.65 (Pf/Pi from 0.78 to 1.85), respectively. The plants inoculated with short-term isolates from carrot callus (CC3) and cereal roots (LHP, PR2, PR3, and PR4) displayed the highest Pf/Pi ratios. In contrast, plants infected with long-term isolates from carrot callus (CC1, CC2, and PR1) displayed the lowest Pf/Pi ratios (Fig. 2, number of nematodes; Table 1).
Results from greenhouse infection tests with wheat and barley to measure the effect of nematode pre-cultivation on the pathogenicity. Plants were inoculated with 1000 nematodes (Pi) from different P. neglectus isolates (CC1, CC2, CC3, LHP, and PR1, PR2, PR3, PR4, see Fig. 1 and Table 2) and harvested eight weeks later. For the RT-qPCR detection assay, the primer combination Neg1 was used. Black and red dots are the individual and mean values, respectively. The upper and lower quartiles are separated by the median (horizontal line). Blue triangles represent outliers. Error bars represent the standard error of the mean from biological replicates. An ANOVA test (p < 0.05) was performed, and significant differences between groups were calculated by a Tukey test (p < 0.05). Different letters above error bars represent groups based on significance (A–H: barley; a–h: wheat).
Then, we measured the abundance of nematode DNA within the infected roots using our recently established RT-qPCR detection assay. The quantification cycle (Cq) values for barley and wheat plants infected with CC and LHP ranged from 23.54 ± 0.7 to 17.85 ± 0.21 and from 23.36 ± 0.4 to 17.83 ± 0.2, respectively. The Cq values after infection with PR isolates were strikingly lower, ranging from 23.24 ± 0.2 to 17.71 ± 0.46 for barley and 23.2 ± 0.19 to 17.63 ± 0.40 for wheat (Fig. 2, Cq values; Table 1). These results correspond with previous findings that showed a negative correlation between the number of nematodes and the quantification cycle (Cq) values30. In conclusion, nematodes pre-cultured on carrot calli were less infective than those grown on cereal roots. We expected a correlation between infection rates and plant growth. Therefore, we measured the biomass and chlorophyll content eight weeks after inoculation (Supplementary Fig. 2). We reasoned that isolates with higher pathogenicity resulted in heavy infections, causing biomass and chlorophyll content reductions. As a result, the biomass and the chlorophyll content varied greatly in response to different isolates. The root and shoot weight of plants infected with short-term carrot callus and cereal-grown isolates (CC3, LHP, PR2, PR3, and PR4) was significantly lower, as compared to plants infected with isolates long-grown on carrot callus (CC1, CC2, and PR1), (Fig. 2). Likewise, the chlorophyll content of plants infected with short-term carrot callus and cereal-grown isolates (CC3, LHP, PR2, PR3, and PR4) was significantly lower, as compared to plants infected with isolates long grown on carrot callus (CC1, CC2, and PR1) (Fig. 2, chlorophyll content). Thus, there was a negative correlation between the number of nematodes per root and biomass/chlorophyll content.
Discussion
The multiplication and maintenance of root lesion nematodes is challenging because they do not form durable stages like cyst nematodes. We assessed different pre-cultivation methods to produce inoculum for plant infection experiments. Long-term cultivation on carrot callus drastically reduced the nematode's pathogenicity, i.e., its ability to infect and multiply within a host plant. In contrast, the pathogenicity of the nematodes was substantially higher after pre-culture on host plants. The infection rates were negatively correlated with plant vigor and chlorophyll content.
What can be the reason for the low pathogenicity after long-term cultivation on carrot callus? It has been known that the nutritional status of the host edaphic factors such as soil texture, moisture, temperature, and interactions with microorganisms can significantly impact the pathogenicity of root lesion nematodes2,14,15,17,18. In our experiments, we kept all factors constant except the pre-culture conditions. Unless not caused by a single virulence gene, pathogenicity can vary greatly in response to environmental factors.
Recent studies on Caenorhabditis elegans and nematodes of the genus Pristionchus and Strongyloides indicate several examples of phenotypic changes due to environmental stimuli. However, these studies primarily concentrated on the concept of phenotypic plasticity, which is the ability of an organism to change its phenotypes in response to external stimuli31,32,33,34,35,36,37,38,39,40. For example, Susoy, et al.41 described a surprising instance of macro-evolutionary scale diversification without genetic diversity. Symbiotic nematodes of the genus Pristionchus accumulated polyphenism with up to five distinct adult morphotypes per species after colonizing individual figs, which require transmission by pollinating wasps (Ceratosolen spp.). In response to diverse environmental cues, P. pacificus can generate either a bacterial feeding or a predatory mouth morph42. Igreja and Sommer43 discovered that sulfatases and sulfotransferases play a role in C. elegans and P. pacificus development and their behavior in different environments.
Aging processes could affect pathogenicity. Several studies have shown that aging in nematodes may directly influence microbiota and indirectly alter other characteristics, including pathogenicity. Cabreiro and Gems44 demonstrated that C. elegans in monoxenic culture could exhibit dysbiosis, which is defined as a state of microbial imbalance in the gut that leads to host dyshomeostasis and involves physiological changes in the bacteria caused by extrinsic factors (e.g., environmental stressors, growth conditions) or intrinsic factors (e.g., aging, either of the host or the bacterial population in the gut lumen). Several studies showed that during the development of C. elegans, bacteria primarily serve as a food source throughout development because the pharyngeal grinder completely crushes them. In young adults, bacteria that have escaped the action of the grinder grow and develop a community inside particular areas of the gut lumen, where they remain symbionts. As the nematodes age, bacteria growing in the intestinal lumen of the nematode become harmful to the host44,45,46,47,48,49. Klass50 demonstrated that a change in temperature or food composition might affect the lifespan of C. elegans at any stage in the life cycle. Interestingly, the findings revealed that parental age and life duration have a minor influence on the progeny's life span.
Genetic changes might alter the pathogenicity of the nematode. Sexual reproduction increases adaptability and heterogeneity in PPN populations. Accordingly, virulent genotypes may result from sexual multiplication51. However, P. neglectus is a monosexual species reproducing only by mitotic parthenogenesis52. Also, mutations cannot explain the varying pathogenicities in our experiments because the isolates could regain pathogenicity after growing on a host plant, which cannot be explained by genetic changes.
We reason that the tripartite pathogen-microbiota-plant interactions are likely to influence pathogenicity. PPNs are associated with intricate microbial communities22,23,24,53,54,55,56. Changes in native microbiota can alter the nematode's lifetime and survival rate. Dirksen, et al.22 showed that bacterial communities such as Pseudomonas, Stenotrophomonas, Ochrobactrum, and Sphingomonas improve the fitness of C. elegans under both normal and stressed situations. Furthermore, they showed that several Proteobacteria may enter the C. elegans gut and that an Ochrobactrum isolate can survive in the intestines under stressful conditions.
Host-microbiota-pathogen interactions in C. elegans have been studied in artificially generated tripartite interactions and by studying native microbiota. Stevens, et al.57 showed that Enterococcus faecalis in C. elegans behaves as a defensive mutualist supporting host survival in the presence of pathogenic bacteria such as Staphylococcus aureus. Their findings indicated that direct and indirect tripartite interactions would most likely occur. Non-competitive effects, such as immunopathology, immunosuppression, and microbiota-mediated tolerance, considerably affect competitive interactions27,57. Elhady, et al.23 investigated the nematode-associated microbiome between infective stages of Meloidogyne incognita and Pratylenchus penetrans, which attaches to the nematode's cuticle or surface coat. They found that Betaproteobacteria, Bacilli, Actinobacteria, and the fungal genera Malassezia, Aspergillus, and Cladosporium were most abundant. The composition of microbial communities on the nematode's surface depended on the soil's microbial community. The highly unique attachment of microorganisms to PPN infective stages in soil suggested an ecological purpose for this association and may be associated with soil suppressiveness towards them. Nuaima26 reported that populations of P. neglectus from different geographical regions differed in their soil bacterial communities adhering to their cuticles, such as Rothia sp. These bacteria decreased the nematode's ability to penetrate the host plant. We reason that long-term cultivation on carrot callus alters the microbiome of P. neglectus. In contrast, the cultivation on host plants, such as barley, maintains a microbiome needed for the infection.
Nematodes may use microbial metabolites or exploit host defense depletion and changing environments to trigger infection27,57. Metabolites produced by the microbiota may serve as an energy supply, allowing for enhanced virulence, fast development, and higher pathogenicity27,57. Hayes, et al.58 reported that hatching of the mouse parasite Trichuris muris was triggered by bacterial surface structures known as fimbriae58. King, et al.59 reported that Enterococcus faecalis living within Caenorhabditis elegans protects the nematode against infection by a more virulent pathogen. A similar study on the native microbiota of C. elegans showed that native microbiota might produce necessary nutrients for C. elegans and alter the nematode’s fitness60. MacNeil61 reported how native microbiota alters the development, body size, and fertility of Pristionchus pacificus by stimulating the neuronal expression of TGF-β ligands. Another study showed that the native microbiota provides nutrients that dramatically promote the life history of P. pacificus by regulating a neuroendocrine peptide in sensory neurons62.
The host plant’s physiological status can also influence the nematode’s population structure, sex ratio, and likely pathogenicity63,64,65. In cyst and root-knot nematodes, the sex differentiation is epigenetically determined to some extent, and under conditions of crowding or poor nutrition, the frequency of males increases64,65,66. An adequate diet is essential for the nematode’s development as it supplies the necessary components for cell growth, chemical energy to drive cellular processes, and vital nutrients that the organism cannot produce in sufficient quantities67,68. Emerya, et al.69 demonstrated that smaller, less productive plants could reduce soil nematodes’ abundance and density by decreasing the belowground biomass. Differences in the plant’s nutritional status can also change belowground resources and influence the composition of soil nematode communities64,70. Cortois, et al.70 used the shoot carbon-to-nitrogen (C/N) ratio to measure resource quality. They reported that the PPN abundance is influenced more by resource quality than quantity. In another study, the soybean cyst nematode, Heterodera glycines, while capable of penetrating specific plant species roots, failed to reproduce successfully if faced with unfavorable conditions, possibly due to the plant’s nutritional status67,71. Moreover, the metabolite composition of the host plant has also been analyzed. Viketoft, et al.72 observed significant variations in nematode community diversity and composition among species within the same functional groups, such as grasses, legumes, or forbs, potentially due to variances in plant metabolites.
Our data demonstrate that the pathogenicity of P. neglectus is restored after infection on a living host such as barley. Therefore, we propose interrupting P. neglectus cultivation on carrot callus by host plant infections to prevent pathogenicity decline (Fig. 1). This is mainly relevant for plant breeding, where standardized inoculum is needed for selecting resistant genotypes among large segregating populations.
Moreover, the pathogenicity of root lesion nematodes depending on growth under different environments deserves further investigation. In vitro and in vivo experiments with different P. neglectus isolates and microbial communities can reveal details about the interaction between microbiome and nematode57,73.
Materials and methods
Plant material and greenhouse infection tests
The barley cultivar Valentina and the wheat cultivar Machete were used in this study. Both are susceptible to P. neglectus74,75,76. Three hundred plants of each variety were inoculated with different isolates of P. neglectus with 30 repetitions in each treatment and repeated twice in a completely randomized design, essentially as described by Keil, et al.76 and Fatemi, et al.30. Plants were grown in the greenhouse in plastic cylindrical tubes 4 cm in diameter and 15 cm in height, filled with heat-sterilized sand (Probau® Quarzsand eco, grain size: 0.1–0.4 mm) under long-day conditions (16 h light) at 23 °C during the day and 18 °C at night with supplemental light (Son-T Agro 400W, Koninklijke Philips Electronics N.V., Eindhoven, The Netherlands). Plants were irrigated twice a week from the bottom of the tubes with a nutrient solution (Supplementary Table 1), as described by Marshall and Ellis77. The nutrient solution was supplied from a 100-L tank and renewed monthly to avoid changes in nutrient concentrations.
Plants were inoculated with different P. neglectus isolates that had been cultured under different conditions. In each treatment, 1000 nematodes from specific isolates were used as an initial inoculum after visual counting under a stereo microscope.
Nematode culture conditions
The Pratylenchus neglectus population PnGLS4 originating from farmer’s fields near Groß Lüsewitz, Germany, was kindly provided by the Institute for Epidemiology and Pathogen Diagnostics, Julius Kühn-Institute, Braunschweig, Germany, as described in30. P. neglectus isolates were produced on carrot calli and living host plants (Table 2). The nematodes were multiplied on carrot calli as a monoxenic culture7,9. Carrot discs were surface sterilized by heating and kept in the dark at 25 ± 1 °C for one week. Then, nematodes were sterilized with streptomycin sulfate (0.1%), and 200 nematodes at different stages of development were placed on each disc. Each Petri dish containing one carrot disc was sealed with parafilm and stored in the dark at 25 °C. The carrot disc cultures were checked every two weeks for nematode reproduction and any contamination with fungi and bacteria under a stereo microscope. On average, the multiplied nematodes were transferred to a new carrot disc every three months. Finally, the nematodes were extracted from carrot discs using a misting chamber30,76.
Nematodes were cultivated on carrot callus (cultivation on Carrot Callus, CC) for more than 96 weeks (CC1), 96 weeks (CC2), and 48 weeks (CC3). LHP contained nematodes extracted directly from Living Host Plants, and non-inoculated plants were used as controls. Alternatively, after eight weeks of cultivation on plant roots, nematodes were obtained from barley and wheat roots (cultivation on Plant Roots, PR) infected with CC1, CC2, CC3, or LHP isolates. Before inoculation, nematodes were counted, and their mobility was inspected under a binocular microscope (Fig. 1 and Table 2). Before inoculation, each isolate was examined under the microscope to measure morphological characters and the demanian indexes2,78.
Phenotypic measurements
Leaf chlorophyll contents were determined by a Dualex instrument (Force A, Paris, France) described by Casa, et al.79. Dry shoot and root weight were measured using a precision scale after harvesting the plant samples and allowing them to dry completely to determine their respective biomass.
DNA isolation, PCR, and real-time quantitative PCR
After harvesting, root samples were divided into two groups; one half was placed in a freeze dryer for total DNA isolation and RT-qPCR, and the other half was placed in a misting chamber to extract the nematodes and for visual counting using a stereo microscope. Total DNA was isolated from freeze-dried infected roots. RT-qPCR was performed with the Rad CFX ConnectTMOptics Module Real-time PCR detection system using the species-specific Neg1 primer combination described by Fatemi, et al.30.
Statistical analyses
The RT-qPCR data were analyzed using the Bio-Rad CFX Manager software version 3.1. The amplification efficiency (E) was calculated from the slope of the quantification cycle (Cq) values (y-axis) and log picograms (log pg) of DNA (x-axis) using the equation E = (10(1/–m) – 1) × 100, where m is the slope80. The reproduction rates, Pf/Pi values, were calculated by dividing the final number of nematodes (Pf) by the number of nematodes in the initial inoculum. The ANOVA was performed with the "Agricolae" program package in R Studio software, version 4.1.0., and significant differences between groups were calculated by a Tukey test (p < 0.05).
Data availability
The authors declare that data of this study are available from this manuscript and its supplementary information files. Upon request, more data, information, and materials used in this study are available from the corresponding authors. All methods were carried out following relevant guidelines. The materials used for this study were purchased through standard commercial paths, and no special permits were required.
References
Jones, J. T. et al. Top 10 plant-parasitic nematodes in molecular plant pathology. Mol. Plant Pathol. 14, 946–961. https://doi.org/10.1111/mpp.12057 (2013).
Castillo, P. & Vovlas, N. Pratylenchus (Nematoda: Pratylenchidae): Diagnosis, Biology, Pathogenicity and Management). Vol. 6 (Brill, 2007).
Smiley, R. W., Gourlie, J. A., Yan, G. & Rhinhart, K. E. L. Resistance and tolerance of landrace wheat in fields infested with Pratylenchus neglectus and P. thornei. Plant Dis. 98, 797–805. https://doi.org/10.1094/PDIS-10-13-1069-RE (2014).
Dababat, A. A. et al. Association analysis of resistance to cereal cyst nematodes (Heterodera avenae) and root-lesion nematodes (Pratylenchus neglectus and P. thornei) in CIMMYT advanced spring wheat lines for semi-arid conditions. Breed Sci. 66, 692–702. https://doi.org/10.1270/jsbbs.15158 (2016).
Whish, J. P. M., Thompson, J. P., Clewett, T. G., Lawrence, J. L. & Wood, J. Pratylenchus thornei populations reduce water uptake in intolerant wheat cultivars. Field Crops Res. 161, 1–10. https://doi.org/10.1016/j.fcr.2014.02.002 (2014).
Mountain, W. B. Nematology. (eds J. N. Sasser & W. R. Jenkins). 422–425 (Chapel Hill, 1960).
Moody, E. H., Lownsbery, B. F. & Ahmed, J. M. Culture of the root-lesion nematode Pratylenchus vulnus on carrot discs. J. Nematol. 5, 225–226 (1973).
Boisseau, M. & Sarah, J. L. In vitro rearing of Pratylenchidae nematodes on carrot discs. Fruits 63, 307–310. https://doi.org/10.1051/fruits:2008028 (2008).
Kagoda, F., Coyne, D. L., Mbiru, E., Derera, J. & Tongoona, P. Monoxenic culture of Pratylenchus zeae on carrot discs. Nematol. Mediterr. 38, 107–108 (2010).
Coyne, D. L., Nicol, J. M. & Claudius-Cole, B. Practical Plant Nematology: A Field and Laboratory Guide. 2nd Ed. 96 (SP-IPM Secretariat, International Institute of Tropical Agriculture (IITA), 2014).
Shaner, G., Stromberg, E. L., Lacy, G. H., Barker, K. R. & Pirone, T. P. Nomenclature and concepts of pathogenicity and virulence. Annu. Rev. Phytopathol. 30, 47–66. https://doi.org/10.1146/annurev.py.30.090192.000403 (1992).
Thomas, S. R. & Elkinton, J. S. Pathogenicity and virulence. J. Invertebr. Pathol. 85, 146–151. https://doi.org/10.1016/j.jip.2004.01.006 (2004).
Shapiro-Ilan, D. I., Fuxa, J. R., Lacey, L. A., Onstad, D. W. & Kaya, H. K. Definitions of pathogenicity and virulence in invertebrate pathology. J. Invertebr. Pathol. 88, 1–7. https://doi.org/10.1016/j.jip.2004.10.003 (2005).
Endo, B. Y. Pathogenesis of nematode-infected plants. Annu. Rev. Phytopathol. 13, 213–238 (1975).
Wallace, M. K., Rust, R. H., Hawkins, D. M. & MacDonald, D. H. Correlation of edaphic factors with plant-parasitic nematode population densities in a forage field. J. Nematol. 25, 642–653 (1993).
McDonald, A. H. & Van den Berg, E. Effect of watering regimen on injury to corn and grain sorghum by Pratylenchus species. J. Nematol. 25, 654–658 (1993).
Melakeberhan, H. Pathogenicity of Pratylenchus penetrans, Heterodera glycines, and Meloidogyne incognita on soybean genotypes. J. Nematol. 30, 93–99 (1998).
Sikder, M. M. & Vestergard, M. Impacts of root metabolites on soil nematodes. Front. Plant Sci. 10, 1792. https://doi.org/10.3389/fpls.2019.01792 (2020).
Pinochet, J., Cenis, J. L., Fernandez, C., Doucet, M. & Marull, J. Reproductive fitness and random amplified polymorphic DNA variation among isolates of Pratylenchus vulnus. J. Nematol. 26, 271–277 (1994).
France, R. A. & Brodie, B. B. Differentiation of two New York isolates of Pratylenchus penetrans based on their reaction on potato. J. Nematol. 27, 339–345 (1995).
Adam, M., Westphal, A., Hallmann, J. & Heuer, H. Specific microbial attachment to root knot nematodes in suppressive soil. Appl. Environ. Microbiol. 80, 2679–2686 (2014).
Dirksen, P. et al. The native microbiome of the nematode Caenorhabditis elegans: Gateway to a new host–microbiome model. BMC Biol. 14, 38. https://doi.org/10.1186/s12915-016-0258-1 (2016).
Elhady, A. et al. Microbiomes associated with infective stages of root-knot and lesion nematodes in soil. PloS one 12, e0177145. https://doi.org/10.1371/journal.pone.0177145 (2017).
Topalovic, O. & Heuer, H. Plant–nematode interactions assisted by microbes in the rhizosphere. Curr. Issues Mol. Biol. 30, 75–88. https://doi.org/10.21775/cimb.030.075 (2019).
Topalovic, O., Elhady, A., Hallmann, J., Richert-Pöggeler, K. R. & Heuer, H. Bacteria isolated from the cuticle of plant-parasitic nematodes attached to and antagonized the root-knot nematode Meloidogyne hapla. Sci. Rep. https://doi.org/10.1038/s41598-019-47942-7 (2019).
Nuaima, R. H. The difference in the bacterial attachment among Pratylenchus neglectus populations and its effect on the nematode infection. Microorganisms https://doi.org/10.3390/microorganisms10081524 (2022).
Armitage, S. A., Genersch, E., McMahon, D. P., Rafaluk-Mohr, C. & Rolff, J. Tripartite interactions: how immunity, microbiota and pathogens interact and affect pathogen virulence evolution. Curr. Opin. Insect Sci. 50, 100871. https://doi.org/10.1016/j.cois.2021.12.011 (2022).
Nicholson, J. K. et al. Host–gut microbiota metabolic interactions. Science 336, 1262–1267. https://doi.org/10.1126/science.1223813 (2012).
McFall-Ngai, M. J. & Ruby, E. G. Symbiont recognition and subsequent morphogenesis as early events in an animal–bacterial mutualism. Science. 1254, 1491–1494 (1991).
Fatemi, E., Melzer, S. & Jung, C. DNA-based assessment of root lesion nematode infections in cereal roots. Sci. Rep. 13, 12602. https://doi.org/10.1038/s41598-023-39559-8 (2023).
Gardner, M. P., Viney, M. E. & Gems, D. Handbook for Models of Human Aging. 229–240 (Academic Press, 2006).
McGaughran, A. & Sommer, R. J. Natural variation in cold tolerance in the nematode Pristionchus pacificus: The role of genotype and environment. Biol. Open 3, 832–838. https://doi.org/10.1242/bio.20148888 (2014).
Sikkink, K. L., Reynolds, R. M., Ituarte, C. M., Cresko, W. A. & Phillips, P. C. Rapid evolution of phenotypic plasticity and shifting thresholds of genetic assimilation in the nematode Caenorhabditis remanei. G3 Genes|Genomes|Genet. Early Online 4, 1103–1112. https://doi.org/10.1534/g3.114.010553 (2014).
Wilecki, M., Lightfoot, J. W., Susoy, V. & Sommer, R. J. Predatory feeding behaviour in Pristionchus nematodes is dependent on phenotypic plasticity and induced by serotonin. J. Exp. Biol. 218, 1306–1313. https://doi.org/10.1242/jeb.118620 (2015).
Susoy, V., Ragsdale, E. J., Kanzaki, N. & Sommer, R. J. Rapid diversification associated with a macroevolutionary pulse of developmental plasticity. eLife https://doi.org/10.7554/eLife.05463 (2015).
Diaz, S. A. & Viney, M. The evolution of plasticity of dauer larva developmental arrest in the nematode Caenorhabditis elegans. Ecol. Evolut. 5, 1343–1353. https://doi.org/10.1002/ece3.1436 (2015).
Tandonnet, S. & Pires-da Silva, A. Phenotypic plasticity and developmental innovations in nematodes. Curr. Opin. Genet. Dev. 39, 8–13. https://doi.org/10.1016/j.gde.2016.05.018 (2016).
Susoy, V. & Sommer, R. J. Stochastic and conditional regulation of nematode mouth-form dimorphisms. Front. Ecol. Evol. https://doi.org/10.3389/Fevo.2016.00023 (2016).
Serobyan, V. & Sommer, R. J. Developmental systems of plasticity and trans-generational epigenetic inheritance in nematodes. Curr. Opin. Genet. Dev. 45, 51–57. https://doi.org/10.1016/j.gde.2017.03.001 (2017).
Sommer, R. J. et al. The genetics of phenotypic plasticity in nematode feeding structures. Open Biol. https://doi.org/10.1098/rsob.160332 (2017).
Susoy, V. et al. Large-scale diversification without genetic isolation in nematode symbionts of figs. Sci. Adv. 2, e1501031. https://doi.org/10.1126/sciadv.1501031 (2016).
Dardiry, M., Piskobulu, V., Kalirad, A. & Sommer, R. J. Experimental and theoretical support for costs of plasticity and phenotype in a nematode cannibalistic trait. Evolut. Lett. 7, 48–57. https://doi.org/10.1093/evlett/qrac001 (2023).
Igreja, C. & Sommer, R. J. The role of sulfation in nematode development and phenotypic plasticity. Front. Mol. Biosci. 9, 838148. https://doi.org/10.3389/fmolb.2022.838148 (2022).
Cabreiro, F. & Gems, D. Worms need microbes too: Microbiota, health and aging in Caenorhabditis elegans. EMBO Mol. Med. 5, 1300–1310. https://doi.org/10.1002/emmm.201100972 (2013).
Gems, D. & Riddle, D. L. Genetic, behavioral and environmental determinants of male longevity in Caenorhabditis elegans. Genetics 154, 1597–1610. https://doi.org/10.1093/genetics/154.4.1597 (2000).
Garigan, D. et al. Genetic analysis of tissue aging in Caenorhabditis elegans: A role for heat-shock factor and bacterial proliferation. Genetics 161, 1101–1112. https://doi.org/10.1093/genetics/161.3.1101 (2002).
Kurz, C. L. et al. Virulence factors of the human opportunistic pathogen Serratia marcescens identified by in vivo screening. EMBO J. 22, 1451–1460. https://doi.org/10.1093/emboj/cdg159 (2003).
McGee, M. D. et al. Loss of intestinal nuclei and intestinal integrity in aging C. elegans. Aging Cell 10, 699–710. https://doi.org/10.1111/j.1474-9726.2011.00713.x (2011).
Portal-Celhay, C., Bradley, E. R. & Blaser, M. J. Control of intestinal bacterial proliferation in regulation of lifespan in Caenorhabditis elegans. BMC Microbiol. 12, 49. https://doi.org/10.1186/1471-2180-12-49 (2012).
Klass, M. R. Aging in the nematode Caenorhabditis elegans: Major biological and environmental factors influencing life span. Mech. Ageing Dev. 6, 413–429. https://doi.org/10.1016/0047-6374(77)90043-4 (1977).
Molinari, S. Nematodes: Morphology, Functions and Management Strategies (eds. Boeri, F. & Chung, J. A.). 59–82 (Nova Science, 2012).
Roman, J. & Triantaphyllou, A. C. Gametogenesis and reproduction of seven species of Pratylenchus. J. Nematol. 1, 357–362 (1969).
Elhady, A., Adss, S., Hallmann, J. & Heuer, H. Rhizosphere microbiomes modulated by pre-crops assisted plants in defense against plant-parasitic nematodes. Front. Microbiol. 9, 1133. https://doi.org/10.3389/fmicb.2018.01133 (2018).
Topalovic, O., Hussain, M. & Heuer, H. Plants and associated soil microbiota cooperatively suppress plant-parasitic nematodes. Front. Microbiol. 11, 313. https://doi.org/10.3389/fmicb.2020.00313 (2020).
Dahan, D., Preston, G. M., Sealey, J. & King, K. C. Impacts of a novel defensive symbiosis on the nematode host microbiome. BMC Microbiol. 20, 159. https://doi.org/10.1186/s12866-020-01845-0 (2020).
Elhady, A., Topalovic, O. & Heuer, H. Plants specifically modulate the microbiome of root-lesion nematodes in the rhizosphere, affecting their fitness. Microorganisms https://doi.org/10.3390/microorganisms9040679 (2021).
Stevens, E. J., Bates, K. A. & King, K. C. Host microbiota can facilitate pathogen infection. PLoS Pathog. 17, e1009514. https://doi.org/10.1371/journal.ppat.1009514 (2021).
Hayes, K. S. et al. Exploitation of the intestinal microflora by the parasitic nematode Trichuris muris. Science 328, 1391–1394. https://doi.org/10.1126/science.1187703 (2010).
King, K. C. et al. Rapid evolution of microbe-mediated protection against pathogens in a worm host. ISME J. 10, 1915–1924. https://doi.org/10.1038/ismej.2015.259 (2016).
Zimmermann, J. et al. The functional repertoire contained within the native microbiota of the model nematode Caenorhabditis elegans. ISME J. 14, 26–38. https://doi.org/10.1038/s41396-019-0504-y (2020).
MacNeil, L. T. Microbiomes: How a gut bacterium promotes healthier living in a nematode. Curr. Biol. 32, R428–R430 (2022).
Lo, W. S., Han, Z. D., Witte, H., Roseler, W. & Sommer, R. J. Synergistic interaction of gut microbiota enhances the growth of nematode through neuroendocrine signaling. Curr. Biol. 32, 2037. https://doi.org/10.1016/j.cub.2022.03.056 (2022).
Davide, R. G. Influence of different crops on the dimensions of Meloidogyne arenaria isolated from fig. Proc. Helminthol. Soc. Washington 47, 80–84 (1980).
Yeates, G. W. Advances in Ecological Research. Vol. 17. 61–100 (Academic Press Inc., 1987).
Williamson, V. M. & Hussey, R. S. Nematode pathogenesis and resistance in plants. Plant Cell 8, 1735–1745. https://doi.org/10.1105/tpc.8.10.1735 (1996).
Triantaphyllou, A. C. Environmental sex differentiation of nematodes in relation to pest management. Annu. Rev. Phytopathol. 11, 441–462 (1973).
Goheen, S. C., Campbell, J. A. & Donald, P. Soybean—Pest Resistance. Vol. 1 (ed. El-Shemy, H. A.). 1–17 (Intech, 2013).
Zecic, A., Dhondt, I. & Braeckman, B. P. The nutritional requirements of Caenorhabditis elegans. Genes Nutr. 14, 15. https://doi.org/10.1186/s12263-019-0637-7 (2019).
Emerya, S. M., Reida, M. L. & Hacker, S. D. Soil nematodes differ in association with native and non-native dune building grass species. Appl. Soil Ecol. https://doi.org/10.1016/j.apsoil.2019.06.009 (2019).
Cortois, R. et al. Possible mechanisms underlying abundance and diversity responses of nematode communities to plant diversity. Ecosphere. https://doi.org/10.1002/ecs2.1719 (2017).
Riggs, R. D. Nonhost root penetration by soybean cyst nematode. J. Nematol. 19, 251–254 (1987).
Viketoft, M., Palmborg, C., Sohlenius, B., Huss-Danell, K. & Bengtsson, J. Plant species effects on soil nematode communities in experimental grasslands. Appl. Soil Ecol. 30, 90–103. https://doi.org/10.1016/j.apsoil.2005.02.007 (2005).
Mooser, C., Gomez de Aguero, M. & Ganal-Vonarburg, S. C. Standardization in host-microbiota interaction studies: challenges, gnotobiology as a tool, and perspective. Curr. Opin. Microbiol. 44, 50–60. https://doi.org/10.1016/j.mib.2018.07.007 (2018).
Taylor, S. P., Hollaway, G. J. & Hunt, C. H. Effect of field crops on population densities of Pratylenchus neglectus and P. thornei in southeastern Australia; part 1: P. neglectus. J. Nematol. 32, 591–599 (2000).
Williams, J. et al. Mapping of the root lesion nematode (Pratylenchus neglectus) resistance gene Rlnn1 in wheat. Theor. Appl. Genet. 104, 874–879. https://doi.org/10.1007/s00122-001-0839-3 (2002).
Keil, T., Laubach, E., Sharma, S. & Jung, C. Screening for resistance in the primary and secondary gene pool of barley against the root-lesion nematode Pratylenchus neglectus. Plant Breed. 128, 436–442. https://doi.org/10.1111/j.1439-0523.2009.01612.x (2009).
Marshall, B. & Ellis, R. P. Growth, yield and grain quality of barley (Hordeum vulgare L.) in response to nitrogen uptake-I. A low cost, controlled nutrient supply system. J. Exp. Bot. 49, 1049–1057. https://doi.org/10.1093/jexbot/49.323.1049 (1998).
Geraert, E. The Pratylenchidae of the World: Identification of the Family Pratylenchidae (Nematoda: Tylenchida). (Academia Press, 2013).
Casa, R., Castaldi, F., Pascucci, S. & Pignatti, S. Chlorophyll estimation in field crops: An assessment of handheld leaf meters and spectral reflectance measurements. J. Agric. Sci.-Camb. 153, 876–890. https://doi.org/10.1017/S0021859614000483 (2015).
Ginzinger, D. G. Gene quantification using real-time quantitative PCR: An emerging technology hits the mainstream. Exp. Hematol. 30, 503–512. https://doi.org/10.1016/S0301-472X(02)00806-8 (2002).
Acknowledgements
We appreciate the technical support by Bettina Rohardt, Verena Kowalewski, and Monika Bruisch. In addition, we thank Professor Johannes Hallmann (Julius Kühn Institute, Institute for Epidemiology and Pathogen Diagnostics, Braunschweig, Germany) for providing the source of nematode inoculum and Dr. Hans-Joachim Harloff for critically reading the manuscript. The authors sincerely thank Amirhossein Fatemi and Atieh Mir-Torabi for generously providing illustrations of the monoxenic cultural pathway. This study was partially funded by the German Federal Ministry of Education and Research (BMBF) (grant No. 031B0186A).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
E.F. designed and performed the experiments, followed by analyzing data. C.J. was the PI and supervised data analysis. E.F. and C.J. jointly wrote the manuscript and approved the final version. Correspondence and requests for materials should be addressed to C.J.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fatemi, E., Jung, C. Pathogenicity of the root lesion nematode Pratylenchus neglectus depends on pre-culture conditions. Sci Rep 13, 19642 (2023). https://doi.org/10.1038/s41598-023-46551-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-46551-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.