Abstract
Human syncytin-1 and suppressyn are cellular proteins of retroviral origin involved in cell–cell fusion events to establish the maternal–fetal interface in the placenta. In cell culture, they restrict infections from members of the largest interference group of vertebrate retroviruses, and are regarded as host immunity factors expressed during development. At the core of the syncytin-1 and suppressyn functions are poorly understood mechanisms to recognize a common cellular receptor, the membrane transporter ASCT2. Here, we present cryo-electron microscopy structures of human ASCT2 in complexes with the receptor-binding domains of syncytin-1 and suppressyn. Despite their evolutionary divergence, the two placental proteins occupy similar positions in ASCT2, and are stabilized by the formation of a hybrid β-sheet or ‘clamp’ with the receptor. Structural predictions of the receptor-binding domains of extant retroviruses indicate overlapping binding interfaces and clamping sites with ASCT2, revealing a competition mechanism between the placental proteins and the retroviruses. Our work uncovers a common ASCT2 recognition mechanism by a large group of endogenous and disease-causing retroviruses, and provides high-resolution views on how placental human proteins exert morphological and immunological functions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Structural models of ASCT2WT–SYCY1RBD at 2.62 Å and 3.5 Å resolution, ASCT2CO–SUPYNRBD and ASCT2WT–Nb469 complexes have been deposited to the PDB with accession codes 8OUH, 8OUJ, 8OUI and 8OUD, respectively, and the corresponding cryo-EM maps have been deposited in the Electron Microscopy Data Bank (EMDB) under accession numbers EMD-17192, EMD-17194, EMD-17193 and EMD-17189. Materials are available upon reasonable request. Source data are provided with this paper.
References
Robbins, J. R. & Bakardjiev, A. I. Pathogens and the placental fortress. Curr. Opin. Microbiol. 15, 36–43 (2012).
Mi, S. et al. Syncytin is a captive retroviral envelope protein involved in human placental morphogenesis. Nature 403, 785–789 (2000).
Blond, J. L. et al. An envelope glycoprotein of the human endogenous retrovirus HERV-W is expressed in the human placenta and fuses cells expressing the type D mammalian retrovirus receptor. J. Virol. 74, 3321–3329 (2000).
Mallet, F. et al. The endogenous retroviral locus ERVWE1 is a bona fide gene involved in hominoid placental physiology. Proc. Natl Acad. Sci. USA 101, 1731–1736 (2004).
Lavialle, C. et al. Paleovirology of ‘syncytins’, retroviral env genes exapted for a role in placentation. Philos. Trans. R. Soc. Lond. B Biol. Sci. 368, 20120507 (2013).
Blond, J. L. et al. Molecular characterization and placental expression of HERV-W, a new human endogenous retrovirus family. J. Virol. 73, 1175–1185 (1999).
Langbein, M. et al. Impaired cytotrophoblast cell–cell fusion is associated with reduced Syncytin and increased apoptosis in patients with placental dysfunction. Mol. Reprod. Dev. 75, 175–183 (2008).
Lee, X. et al. Downregulation of placental syncytin expression and abnormal protein localization in pre-eclampsia. Placenta 22, 808–812 (2001).
Bolze, P. A., Mommert, M. & Mallet, F. Contribution of syncytins and other endogenous retroviral envelopes to human placenta pathologies. Prog. Mol. Biol. Transl. Sci. 145, 111–162 (2017).
Cheynet, V. et al. Synthesis, assembly, and processing of the Env ERVWE1/syncytin human endogenous retroviral envelope. J. Virol. 79, 5585–5593 (2005).
Chang, C., Chen, P. T., Chang, G. D., Huang, C. J. & Chen, H. Functional characterization of the placental fusogenic membrane protein syncytin. Biol. Reprod. 71, 1956–1962 (2004).
Lavillette, D. et al. The envelope glycoprotein of human endogenous retrovirus type W uses a divergent family of amino acid transporters/cell surface receptors. J. Virol. 76, 6442–6452 (2002).
Hogan, V. & Johnson, W. E. Unique structure and distinctive properties of the ancient and ubiquitous gamma-type envelope glycoprotein. Viruses 15, 274 (2023).
Henzy, J. E. & Johnson, W. E. Pushing the endogenous envelope. Philos. Trans. R. Soc. Lond. B Biol. Sci. 368, 20120506 (2013).
Ruigrok, K. et al. X-ray structures of the post-fusion 6-helix bundle of the human syncytins and their functional implications. J. Mol. Biol. 431, 4922–4940 (2019).
Sugimoto, J., Sugimoto, M., Bernstein, H., Jinno, Y. & Schust, D. A novel human endogenous retroviral protein inhibits cell–cell fusion. Sci. Rep. 3, 1462 (2013).
Sugimoto, J. et al. Suppressyn localization and dynamic expression patterns in primary human tissues support a physiologic role in human placentation. Sci. Rep. 9, 19502 (2019).
Sugimoto, J. et al. Could the human endogenous retrovirus-derived syncytialization inhibitor, suppressyn, limit heterotypic cell fusion events in the decidua? Int. J. Mol. Sci. 22, 10259 (2021).
Rasko, J. E., Battini, J. L., Gottschalk, R. J., Mazo, I. & Miller, A. D. The RD114/simian type D retrovirus receptor is a neutral amino acid transporter. Proc. Natl Acad. Sci. USA 96, 2129–2134 (1999).
Tailor, C. S., Nouri, A., Zhao, Y., Takeuchi, Y. & Kabat, D. A sodium-dependent neutral-amino-acid transporter mediates infections of feline and baboon endogenous retroviruses and simian type D retroviruses. J. Virol. 73, 4470–4474 (1999).
Sommerfelt, M. A. & Weiss, R. A. Receptor interference groups of 20 retroviruses plating on human cells. Virology 176, 58–69 (1990).
Ponferrada, V. G., Mauck, B. S. & Wooley, D. P. The envelope glycoprotein of human endogenous retrovirus HERV-W induces cellular resistance to spleen necrosis virus. Arch. Virol. 148, 659–675 (2003).
Sinha, A. & Johnson, W. E. Retroviruses of the RDR superinfection interference group: ancient origins and broad host distribution of a promiscuous Env gene. Curr. Opin. Virol. 25, 105–112 (2017).
Stromberg, K. et al. Characterization of exogenous type D retrovirus from a fibroma of a macaque with simian AIDS and fibromatosis. Science 224, 289–282 (1984).
Daniel, M. D. et al. A new type D retrovirus isolated from macaques with an immunodeficiency syndrome. Science 223, 602–605 (1984).
Grange, Z. L. et al. Ranking the risk of animal-to-human spillover for newly discovered viruses. Proc. Natl Acad. Sci. USA 118, e2002324118 (2021).
Wozniakowski, G., Frant, M. & Mamczur, A. Avian reticuloendotheliosis in chickens—an update on disease occurrence and clinical course. J. Veterin. Res. 62, 257–260 (2018).
Cheynet, V., Oriol, G. & Mallet, F. Identification of the hASCT2-binding domain of the Env ERVWE1/syncytin-1 fusogenic glycoprotein. Retrovirology 3, 41 (2006).
Frank, J. A. et al. Evolution and antiviral activity of a human protein of retroviral origin. Science 378, 422–428 (2022).
Kanai, Y. et al. The SLC1 high-affinity glutamate and neutral amino acid transporter family. Mol. Asp. Med 34, 108–120 (2013).
Wahi, K. & Holst, J. ASCT2: a potential cancer drug target. Expert Opin. Ther. Targets 23, 555–558 (2019).
Liu, Y. et al. The role of ASCT2 in cancer: a review. Eur. J. Pharmacol. 837, 81–87 (2018).
Garaeva, A. A. et al. Cryo-EM structure of the human neutral amino acid transporter ASCT2. Nat. Struct. Mol. Biol. 25, 515–521 (2018).
Yu, X. et al. Cryo-EM structures of the human glutamine transporter SLC1A5 (ASCT2) in the outward-facing conformation. eLife https://doi.org/10.7554/eLife.48120 (2019).
Yernool, D., Boudker, O., Jin, Y. & Gouaux, E. Structure of a glutamate transporter homologue from Pyrococcus horikoshii. Nature 431, 811–818 (2004).
Canul-Tec, J. C. et al. Structure and allosteric inhibition of excitatory amino acid transporter 1. Nature 544, 446–451 (2017).
Reyes, N., Ginter, C. & Boudker, O. Transport mechanism of a bacterial homologue of glutamate transporters. Nature 462, 880–885 (2009).
Crisman, T. J., Qu, S., Kanner, B. I. & Forrest, L. R. Inward-facing conformation of glutamate transporters as revealed by their inverted-topology structural repeats. Proc. Natl Acad. Sci. USA 106, 20752–20757 (2009).
Marin, M., Lavillette, D., Kelly, S. M. & Kabat, D. N-linked glycosylation and sequence changes in a critical negative control region of the ASCT1 and ASCT2 neutral amino acid transporters determine their retroviral receptor functions. J. Virol. 77, 2936–2945 (2003).
Cirri, E. et al. Consensus designs and thermal stability determinants of a human glutamate transporter. eLife https://doi.org/10.7554/eLife.40110 (2018).
Martinez-Molledo, M., Nji, E. & Reyes, N. Structural insights into the lysophospholipid brain uptake mechanism and its inhibition by syncytin-2. Nat. Struct. Mol. Biol. 29, 604–612 (2022).
Fass, D. et al. Structure of a murine leukemia virus receptor-binding glycoprotein at 2.0 angstrom resolution. Science 277, 1662–1666 (1997).
Barnett, A. L., Wensel, D. L., Li, W., Fass, D. & Cunningham, J. M. Structure and mechanism of a coreceptor for infection by a pathogenic feline retrovirus. J. Virol. 77, 2717–2729 (2003).
McCarthy, K. R. et al. Structure of the receptor binding domain of EnvP(b)1, an endogenous retroviral envelope protein expressed in human tissues. mBio https://doi.org/10.1128/mBio.02772-20 (2020).
Holm, L. DALI and the persistence of protein shape. Protein Sci. 29, 128–140 (2020).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Hotzel, I. Deep-time structural evolution of retroviral and filoviral surface envelope proteins. J. Virol. 96, e0006322 (2022).
Boudker, O., Ryan, R. M., Yernool, D., Shimamoto, K. & Gouaux, E. Coupling substrate and ion binding to extracellular gate of a sodium-dependent aspartate transporter. Nature 445, 387–393 (2007).
Garaeva, A. A., Guskov, A., Slotboom, D. J. & Paulino, C. A one-gate elevator mechanism for the human neutral amino acid transporter ASCT2. Nat. Commun. 10, 3427 (2019).
Benton, D. J. et al. Receptor binding and priming of the spike protein of SARS-CoV-2 for membrane fusion. Nature 588, 327–330 (2020).
Blaise, S., de Parseval, N., Benit, L. & Heidmann, T. Genomewide screening for fusogenic human endogenous retrovirus envelopes identifies syncytin 2, a gene conserved on primate evolution. Proc. Natl Acad. Sci. USA 100, 13013–13018 (2003).
Esnault, C. et al. A placenta-specific receptor for the fusogenic, endogenous retrovirus-derived, human syncytin-2. Proc. Natl Acad. Sci. USA 105, 17532–17537 (2008).
Shaik, M. M. et al. Structural basis of coreceptor recognition by HIV-1 envelope spike. Nature 565, 318–323 (2019).
Gong, X. et al. Structural insights into the Niemann-Pick C1 (NPC1)-mediated cholesterol transfer and Ebola infection. Cell 165, 1467–1478 (2016).
Yan, R. et al. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 367, 1444–1448 (2020).
Garibsingh, R. A. et al. Rational design of ASCT2 inhibitors using an integrated experimental-computational approach. Proc. Natl Acad. Sci. USA 118, e2104093118 (2021).
Iwaki, T., Figuera, M., Ploplis, V. A. & Castellino, F. J. Rapid selection of Drosophila S2 cells with the puromycin resistance gene. Biotechniques 35, 482–484 (2003).
Backovic, M. & Krey, T. Stable Drosophila cell lines: an alternative approach to exogenous protein expression. Methods Mol. Biol. 1350, 349–358 (2016).
Pardon, E. et al. A general protocol for the generation of nanobodies for structural biology. Nat. Protoc. 9, 674–693 (2014).
Cabantous, S., Terwilliger, T. C. & Waldo, G. S. Protein tagging and detection with engineered self-assembling fragments of green fluorescent protein. Nat. Biotechnol. 23, 102–107 (2005).
Guo, H. et al. Electron-event representation data enable efficient cryoEM file storage with full preservation of spatial and temporal resolution. IUCrJ 7, 860–869 (2020).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Punjani, A., Zhang, H. & Fleet, D. J. Non-uniform refinement: adaptive regularization improves single-particle cryo-EM reconstruction. Nat. Methods 17, 1214–1221 (2020).
Sanchez-Garcia, R. et al. DeepEMhancer: a deep learning solution for cryo-EM volume post-processing. Commun. Biol. 4, 874 (2021).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Varadi, M. et al. AlphaFold protein structure database: massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. 50, D439–D444 (2022).
Bepler, T. et al. Positive-unlabeled convolutional neural networks for particle picking in cryo-electron micrographs. Nat. Methods 16, 1153–1160 (2019).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Mirdita, M. et al. ColabFold: making protein folding accessible to all. Nat. Methods 19, 679–682 (2022).
Acknowledgements
IECB cryo-EM imaging facility is acknowledged for support in cryo-EM sample screening and initial data acquisition, and the EMBL-Heidelberg Cryo-Electron Microscopy Service Platform for support in image acquisition. R. Assal for preliminary protein production and construct screening. This research was funded by the European Research Council (ERC) under the European Union Horizon 2020 Program (grant no. 771965), with additional contributions from the Institut National du Cancer (INCA grant 2017-44), IDEX Senior Chair Universite de Bordeaux and Region Nouvelle-Aquitaine (grant no. 8166910), all to N.R., Instruct-ERIC (PID6507), as part of the European Strategy Forum on Research Infrastructures (ESFRI), and the Research Foundation-Flanders (FWO) for nanobody discovery. A. Lundqvist for technical assistance during nanobody discovery. The Swiss National Science Foundation (SNSF) (PP00P3_198903 and 310030_207974 to CP) supported the work performed by the group of C.P. F.A.R. and M.B. acknowledge funding from grant ANR-10-LABX-62-IBEID.
Author information
Authors and Affiliations
Contributions
S.K., J.C.C.-T. and A.K. performed protein purification, cryo-EM sample preparation and image analysis, as well as atomic model building and refinement. S.K. and M.I.V. performed binding assays, and M.I.V. performed cell fusion assays, as well as optimized, screened and produced the HERV-derived proteins. M.I.V. and J.C.C.-T. produced the proteoliposomes. A.B.C. and C.P. performed and analyzed transport and electrophysiology assays with proteoliposomes and binders. M.B. and F.A.R. contributed to the design and development of soluble HERV constructs. E.P. and J.S. designed and performed nanobody development. N.R. analyzed functional and cryo-EM data, as well as structures. All authors contributed to the preparation of the manuscript. N.R. conceived and supervised the project.
Corresponding authors
Ethics declarations
Competing interests
A.K. is currently an employee of the Integrated Drug Discovery, Sanofi R&D, France. All other authors declare no competing interests.
Peer review
Peer review information
Nature Structural & Molecular Biology thanks Winfried Weissenhorn and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available. Primary Handling Editor: Katarzyna Ciazynska, in collaboration with the Nature Structural & Molecular Biology team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
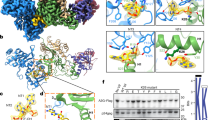
Extended Data Fig. 1 ASCT2 consensus design.
a, Amino acid sequence alignment of ASCT2WT and ASCT2CO. b, Cell-cell fusion assay based on split-GFP. ASCT2CO shows similar levels of syncytin-1 mediated cell-cell fusion than ASCT2WT. Purified SYCY1∆439 and suppressyn constructs recognize ASCT2WT and inhibit syncytin-1 mediated cell-cell fusion. The syncytin-1 mutant C186S precludes cell fusion and it is used as a negative control to account for background fluorescence. c, Reconstituted ASCT2CO yields nearly 5-fold higher radioactive substrate uptake in proteoliposomes compared to ASCT2WT. In b, and c bars depict averages of at least three biologically independent experiments, and error bars represent SEM. Circles represent values from individual experiments.
Extended Data Fig. 2 ASCT2WT-SYCY1-∆G469 and ASCT2CO-suppressyn complexes.
a, SEC profile of the ASCT2CO-suppressyn complex. b, SEC profile of the ASCT2WT-SYCY1∆469 complex. c, Binding curves of suppressyn (triangles) and SYCY1∆469 (circles) to ASCT2CO in detergent solution. Empty symbols depict the averages of 3 biologically independent experiments, and error bars represent SEM. Solid lines are fits of the Hill equation with APPKD and Hill coefficient values of 1 μM and 1.7, respectively, for SYCY1∆469, and 0.2 μM and 1.5, respectively, for suppressyn.
Extended Data Fig. 3 ASCT2WT-SYCY1RBD cryo-EM data processing pipeline.
a, Representative EM micrograph. 15,376 micrographs were taken in total. b, Gallery of representative 2D class-averages. c, 3D classes from ab initio classification. d, Local refined map and Fourier Shell Correlation (FSC) plot with FSC threshold at 0.143. e, Viewing direction distribution plot. f, The 2.6 Å map (overall resolution) is coloured based on local resolution with the scale bar in angstroms. g, 3D ab-initio class after reprocessing 640k particles to improve the density of the third tranD. h, Local refined map and corresponding FSC plot with threshold at 0.143. i, Viewing direction distribution plot. j, The 3.5 Å map (overall resolution) is coloured based on local resolution with the scale bar in angstroms.
Extended Data Fig. 4 ASCT2CO-SUPYNRBD cryo-EM data processing pipeline.
a, Representative EM micrograph. 8,455 micrographs were taken in total. b, Gallery of representative 2D class-averages. c, 3D classes from ab initio classification. d, 3D class from heterogeneous refinement. e, Local refined map and Fourier Shell Correlation (FSC) plot with FSC threshold at 0.143. f, Viewing direction distribution plot. g, 3D-Flex reconstruction map and corresponding FSC plot with threshold at 0.143. h, The 3.6 Å map (overall resolution) is coloured based on local resolution with the scale bar in angstroms.
Extended Data Fig. 5 Cryo-EM density of ASCT2WT-SYCY1RBD and ASCT2CO-SUPYNRBD complexes.
a, Cryo-EM density corresponding to individual ASCT2WT and SYCY1RBD structural elements. b, Cryo-EM density corresponding to individual ASCT2CO and SUPYNRBD structural elements.
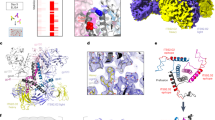
Extended Data Fig. 6 Alignments of endogenous exogenous retroviral RBDs.
a, Sequence alignment of the SYCY1RBD and SUPYNRBD. Disulfide bonds (S1, S2, and S3) and the secondary structural elements observed in the cryo-EM structures are marked above (SYCY1RBD) and below (SUPYNRBD) the alignment rows. b, Sequence alignment of the RBDs of simian retrovirus 3 (SRV3RBD), avian reticuloendotheliosis virus (REVRBD), simian type D retrovirus (RD114RBD), avian spleen necrosis virus (SNVRBD), and sycncytin-1. Disulfide bonds (S1, S2, and S3) and the secondary structural elements observed in the ASCT2WT-SYCY1RBD cryo-EM structure are marked.
Extended Data Fig. 7 Protease cleavage of GFP-ASCT2CO and Nb469 ITC binding.
a, Determination of ASCT2CO orientation in proteoliposomes. ASCT2CO N-terminal fused to GFP was reconstituted in proteoliposomes and incubated in the presence or absence of 3C-protease. In-gel fluorescence shows profiles of technical triplicates of non-cleaved and cleaved (inside−out orientation) GFP-ASCT2CO in proteoliposomes. b, Isothermal titration calorimetry analysis of Nb469 binding to ASTCCO at 25 °C in detergent solutions. The black line in the lower panel is the fit of the quadratic binding equation (“one site binding model”) with the following parameters: Nb469:ASCTCO-protomer=1.1; KD = 360 nM; ∆H = 6.3 kcal/mol; ∆S = 8.0 cal/mol/deg.
Extended Data Fig. 8 ASCT2WT-Nb469 cryo-EM data processing pipeline.
a, Representative EM micrograph with examples of individual picked particles (green circles). 4,344 micrographs were taken in total. b, Gallery of representative 2D class-averages. c, 3D classes from ab initio classification. d, Non-uniform refinement map. e, Non-uniform refinement map is coloured based on local resolution. f, Viewing direction distribution plot. g, Fourier Shell Correlation (FSC) plot of the non-uniform refinement with FSC threshold at 0.143.
Extended Data Fig. 9 Cryo-EM density of ASCT2WT-Nb469 complex.
a, Cryo-EM density around different structural elements of ASCT2CO. b Cryo-EM density around the complementary-determining regions of the Nb469. c, Cryo-EM density around the bound sodium ions (purple spheres), structural waters (red spheres), and L-ala substrate (green sticks). The tranD is shown as ribbon.
Supplementary information
Supplementary Information
Supplementary discussion, Fig. 1 and Tables 1 and 2.
Source data
Source Data Fig. 5
Experimental data points.
Source Data Extended Data Fig. 1
Experimental data points.
Source Data Extended Data Fig. 2
Experimental data points.
Source Data Extended Data Fig. 7
Experimental data points.
Source Data Extended Data Fig. 7
Unmodified gel.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khare, S., Villalba, M.I., Canul-Tec, J.C. et al. Receptor-recognition and antiviral mechanisms of retrovirus-derived human proteins. Nat Struct Mol Biol (2024). https://doi.org/10.1038/s41594-024-01295-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41594-024-01295-6