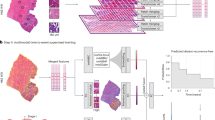
Abstract
Precision in the diagnosis of diverse central nervous system (CNS) tumor types is crucial for optimal treatment. DNA methylation profiles, which capture the methylation status of thousands of individual CpG sites, are state-of-the-art data-driven means to enhance diagnostic accuracy but are also time consuming and not widely available. Here, to address these limitations, we developed Deep lEarning from histoPathoLOgy and methYlation (DEPLOY), a deep learning model that classifies CNS tumors to ten major categories from histopathology. DEPLOY integrates three distinct components: the first classifies CNS tumors directly from slide images (‘direct model’), the second initially generates predictions for DNA methylation beta values, which are subsequently used for tumor classification (‘indirect model’), and the third classifies tumor types directly from routinely available patient demographics. First, we find that DEPLOY accurately predicts beta values from histopathology images. Second, using a ten-class model trained on an internal dataset of 1,796 patients, we predict the tumor categories in three independent external test datasets including 2,156 patients, achieving an overall accuracy of 95% and balanced accuracy of 91% on samples that are predicted with high confidence. These results showcase the potential future use of DEPLOY to assist pathologists in diagnosing CNS tumors within a clinically relevant short time frame.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
DBTA data, including WSIs, tumor types and demographics, are available on EBRAINS (https://ebrains.eu; https://doi.org/10.25493/WQ48-ZGX). Registration and acceptance of terms and data use agreements are required for access. CBTN WSIs and demographic data can be accessed via a request form at the Children Brain Tumor Network (https://cbtn.org/). Requests for data collected in-house, including the NCI and NCI–Prospective cohorts, should be directed to the corresponding authors. Each request will be assessed individually within 15 business days to ensure adherence to intellectual property rights and patient privacy standards.
Code availability
The DEPLOY code is available for academic research purposes via Zenodo at https://doi.org/10.5281/zenodo.8381774 (ref. 57).
References
Louis, D. N. et al. The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro. Oncol. 23, 1231–1251 (2021).
Bruner, J. M., Inouye, L., Fuller, G. N. & Langford, L. A. Diagnostic discrepancies and their clinical impact in a neuropathology referral practice. Cancer 79, 796–803 (1997).
Wu, Z. et al. Impact of the methylation classifier and ancillary methods on CNS tumor diagnostics. Neuro. Oncol. 24, 571–581 (2022).
Capper, D. et al. DNA methylation-based classification of central nervous system tumours. Nature 555, 469–474 (2018).
Pratt, D., Sahm, F. & Aldape, K. DNA methylation profiling as a model for discovery and precision diagnostics in neuro-oncology. Neuro. Oncol. 23, S16–S29 (2021).
Liu, Y. et al. A deep learning system for differential diagnosis of skin diseases. Nat. Med. 26, 900–908 (2020).
Lu, M. Y. et al. Data-efficient and weakly supervised computational pathology on whole-slide images. Nat. Biomed. Eng. 5, 555–570 (2021).
Campanella, G. et al. Clinical-grade computational pathology using weakly supervised deep learning on whole slide images. Nat. Med. 25, 1301–1309 (2019).
Esteva, A. et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature 542, 115–118 (2017).
Kalra, S. et al. Pan-cancer diagnostic consensus through searching archival histopathology images using artificial intelligence. NPJ Digit. Med. 3, 31 (2020).
Lu, M. Y. et al. AI-based pathology predicts origins for cancers of unknown primary. Nature 594, 106–110 (2021).
Li, Z. et al. Vision transformer-based weakly supervised histopathological image analysis of primary brain tumors. iScience 26, 105872 (2023).
Coudray, N. et al. Classification and mutation prediction from non-small cell lung cancer histopathology images using deep learning. Nat. Med. 24, 1559–1567 (2018).
Kather, J. N. et al. Deep learning can predict microsatellite instability directly from histology in gastrointestinal cancer. Nat. Med. 25, 1054–1056 (2019).
Fu, Y. et al. Pan-cancer computational histopathology reveals mutations, tumor composition and prognosis. Nat. Cancer 1, 800–810 (2020).
Qu, H. et al. Genetic mutation and biological pathway prediction based on whole slide images in breast carcinoma using deep learning. NPJ Precis. Oncol. 5, 87 (2021).
Schaumberg, A. J., Rubin, M. A. & Fuchs, T. J. H&E-stained whole slide image deep learning predicts SPOP mutation state in prostate cancer. Preprint at bioRxiv https://doi.org/10.1101/064279 (2018).
Tsou, P. & Wu, C.-J. Mapping driver mutations to histopathological subtypes in papillary thyroid carcinoma: applying a deep convolutional neural network. J. Clin. Med. Res. 8, 1675 (2019).
Chang, P. et al. Deep-learning convolutional neural networks accurately classify genetic mutations in gliomas. Am. J. Neuroradiol. 39, 1201–1207 (2018).
Kim, R. H. et al. A deep learning approach for rapid mutational screening in melanoma. Preprint at bioRxiv https://doi.org/10.1101/610311 (2020).
Chen, M. et al. Classification and mutation prediction based on histopathology H&E images in liver cancer using deep learning. NPJ Precis. Oncol. 4, 1–7 (2020).
Ghaffari, L. N. et al. Benchmarking weakly-supervised deep learning pipelines for whole slide classification in computational pathology. Med. Image Anal. 79, 102474 (2022).
Nasrallah, M. P. et al. Machine learning for cryosection pathology predicts the 2021 WHO classification of glioma. Med 4, 526–540.e4 (2023).
Wang, Y. et al. Predicting molecular phenotypes from histopathology images: a transcriptome-wide expression-morphology analysis in breast cancer. Cancer Res. 81, 5115–5126 (2021).
Schmauch, B. et al. A deep learning model to predict RNA-Seq expression of tumours from whole slide images. Nat. Commun. 11, 3877 (2020).
Alsaafin, A., Safarpoor, A., Sikaroudi, M., Hipp, J. D. & Tizhoosh, H. R. Learning to predict RNA sequence expressions from whole slide images with applications for search and classification. Nat. Commun. Biol. 6, 304 (2023).
Hoang, D.-T. et al. Prediction of cancer treatment response from histopathology images through imputed transcriptomics. Preprint at bioRxiv https://doi.org/10.1101/2022.06.07.495219 (2023).
He, B. et al. Integrating spatial gene expression and breast tumour morphology via deep learning. Nat. Biomed. Eng. 4, 827–834 (2020).
Monjo, T., Koido, M., Nagasawa, S., Suzuki, Y. & Kamatani, Y. Efficient prediction of a spatial transcriptomics profile better characterizes breast cancer tissue sections without costly experimentation. Sci. Rep. 12, 4133 (2022).
Levy-Jurgenson, A., Tekpli, X., Kristensen, V. N. & Yakhini, Z. Spatial transcriptomics inferred from pathology whole-slide images links tumor heterogeneity to survival in breast and lung cancer. Sci. Rep. 10, 18802 (2020).
Zheng, H., Momeni, A., Cedoz, P.-L., Vogel, H. & Gevaert, O. Whole slide images reflect DNA methylation patterns of human tumors. NPJ Genom. Med. 5, 11 (2020).
Roetzer-Pejrimovsky, T. et al. The Digital Brain Tumour Atlas, an open histopathology resource. Sci. Data 9, 55 (2022).
Lilly, J. V. et al. The children’s brain tumor network (CBTN)—accelerating research in pediatric central nervous system tumors through collaboration and open science. Neoplasia 35, 100846 (2023).
Unruh, D. et al. Methylation and transcription patterns are distinct in IDH mutant gliomas compared to other IDH mutant cancers. Sci. Rep. 9, 8946 (2019).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Iorio, F. et al. Pathway-based dissection of the genomic heterogeneity of cancer hallmarks’ acquisition with SLAPenrich. Sci. Rep. 8, 6713 (2018).
Capper, D. et al. Practical implementation of DNA methylation and copy-number-based CNS tumor diagnostics: the Heidelberg experience. Acta Neuropathol. 136, 181–210 (2018).
Nasrallah, M. L. P., Desai, A., O’Rourke, D. M., Surrey, L. F. & Stein, J. M. A dual-genotype oligoastrocytoma with histologic, molecular, radiological and time-course features. Acta Neuropathol. Commun. 8, 115 (2020).
Ellison, D. W. et al. Histopathological grading of pediatric ependymoma: reproducibility and clinical relevance in European trial cohorts. J. Negat. Results Biomed. 10, 7 (2011).
van den Bent, M. J. Interobserver variation of the histopathological diagnosis in clinical trials on glioma: a clinician’s perspective. Acta Neuropathol. 120, 297–304 (2010).
Pickles, J. C. et al. DNA methylation-based profiling for paediatric CNS tumour diagnosis and treatment: a population-based study. Lancet Child Adolesc. Health 4, 121–130 (2020).
Priesterbach-Ackley, L. P. et al. Brain tumour diagnostics using a DNA methylation-based classifier as a diagnostic support tool. Neuropathol. Appl. Neurobiol. 46, 478–492 (2020).
Lujan, G. et al. Dissecting the business case for adoption and implementation of digital pathology: a white paper from the digital pathology association. J. Pathol. Inform. 12, 17 (2021).
Evans, A. J., Vajpeyi, R., Henry, M. & Chetty, R. Establishment of a remote diagnostic histopathology service using whole slide imaging (digital pathology). J. Clin. Pathol. 74, 421–424 (2021).
Williams, B. J., Bottoms, D., Clark, D. & Treanor, D. Future-proofing pathology part 2: building a business case for digital pathology. J. Clin. Pathol. 72, 198–205 (2019).
Ho, J. et al. Can digital pathology result in cost savings? A financial projection for digital pathology implementation at a large integrated health care organization. J. Pathol. Inform. 5, 33 (2014).
Hanna, M. G. et al. Implementation of digital pathology offers clinical and operational increase in efficiency and cost savings. Arch. Pathol. Lab. Med. 143, 1545–1555 (2019).
Koelsche, C. et al. Sarcoma classification by DNA methylation profiling. Nat. Commun. 12, 498 (2021).
Jurmeister, P. et al. DNA methylation-based classification of sinonasal tumors. Nat. Commun. 13, 7148 (2022).
Hackeng, W. M. et al. Genome methylation accurately predicts neuroendocrine tumor origin: an online tool. Clin. Cancer Res. 27, 1341–1350 (2021).
Leitheiser, M. et al. Machine learning models predict the primary sites of head and neck squamous cell carcinoma metastases based on DNA methylation. J. Pathol. 256, 378–387 (2022).
Jurmeister, P. et al. Machine learning analysis of DNA methylation profiles distinguishes primary lung squamous cell carcinomas from head and neck metastases. Sci. Transl. Med. 11, eaaw8513 (2019).
Aryee, M. J. et al. Minfi: a flexible and comprehensive Bioconductor package for the analysis of Infinium DNA methylation microarrays. Bioinformatics 30, 1363–1369 (2014).
Zhou, W., Laird, P. W. & Shen, H. Comprehensive characterization, annotation and innovative use of Infinium DNA methylation BeadChip probes. Nucleic Acids Res. 45, e22 (2017).
Moran, S., Arribas, C. & Esteller, M. Validation of a DNA methylation microarray for 850,000 CpG sites of the human genome enriched in enhancer sequences. Epigenomics 8, 389–399 (2016).
Fang, Z., Liu, X. & Peltz, G. GSEApy: a comprehensive package for performing gene set enrichment analysis in Python. Bioinformatics 39, btac757 (2023).
DEPLOY: an integrated deep learning model for predicting DNA methylation and tumor types from H&E images. Zenodo https://doi.org/10.5281/zenodo.8381774 (2024).
Acknowledgements
This work was partially supported by grant DP190103402 from the Australian Research Council (D.-T.H and E.A.S.) and by the Intramural Research Program of the National Institutes of Health, NCI and Center for Cancer Research (E.D.S., R.T., Z.A., E.M.C., H.L., E.R. and K.A). E.D.S. is supported by the NCI T2I Fellowship. This work utilized the super computational resources of the Australian National Computational Infrastructure, the Australian National University Merit Allocation Scheme and National Institutes of Health High Performance Computing (Biowulf cluster).
Author information
Authors and Affiliations
Contributions
K.A. and E.R. conceived and designed the study. D.-T.H. and E.D.S. developed the DEPLOY models. D.-T.H. and E.D.S. conducted data analysis and validation. M.P.N. generated data and figures for mapping tumor diagnostic class heterogeneity. K.A. and M.P.N. provided expertise in neuropathology. R.T., Z.A. and O.S. generated and collected the NCI datasets. R.T. normalized methylation data. O.S., E.M.C. and H.L. provided assistance with computing and data visualization. E.A.S. interpreted the results and provided feedback on the study. K.A. and E.R. supervised the study. D.-T.H., E.D.S., M.P.N., E.R. and K.A. wrote the paper with the assistance and feedback from all the other co-authors.
Corresponding authors
Ethics declarations
Competing interests
D.-T.H., E.D.S., E.R. and K.A. are listed as inventors on a provisional patent (application no. 63/626,277, United States, 2024) filed based on the methodology outlined in this study. E.R. is a co-founder of Medaware, Metabomed and Pangea Biomed (divested from the latter). E.R. serves as a non-paid scientific consultant to Pangea Biomed under a collaboration agreement between Pangea Biomed and the NCI. The other authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editors: Lorenzo Righetto and Saheli Sadanand, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Pathway enrichment analysis of predicted beta values across brain tumor types not covered by the ten predefined categories.
a, This panel broadens the scope of Fig. 2f, incorporating brain tumor types that are not included in the ten predefined types. Values inside the grid denote the mean of the scaled normalized enrichment score for the specific tumor type and pathway combination. Analyses based on actual and predicted beta values from the NCI cohort are displayed on the right and left, respectively (Methods). b, This panel follows the format of (a) but focuses on gene body methylation. It extends the analysis from Fig. 2g to include 31 additional brain tumor types and two control tissue signatures not covered by the ten predefined categories. c, The bar plots display the Pearson correlation coefficients, which quantify the agreement between DEPLOY’s predicted and actual average enrichment scores across various brain cancer types in the NCI cohort (n = 31 tumor types). The upper section of the panel presents correlations for promoter methylation, whereas the lower section addresses gene body methylation. Error bars indicate the 95% confidence intervals calculated using Fisher transformation and centers correspond to the computed correlation values. This panel focuses on brain tumor types that fall outside the scope of the ten predefined tumor types. For correlations associated with these predefined tumor types, refer to Fig. 2h.
Extended Data Fig. 2 Micro-averaged precision-recall curves and the area under the precision-recall curve (AUPRC) values on each cohort.
Results are depicted for the four models: Demographic (light blue), Direct (orange), Indirect (green), and Integrated (DEPLOY, dark blue). Error bands represent the 95% confidence intervals for each model, calculated through 1,000 bootstrapping iterations and centered on each model’s computed values.
Extended Data Fig. 3 Overall (micro-averaged) accuracy and balanced accuracy for all the external datasets together.
a-b, results for all samples (n = 2,156 patients). c-d, results for samples with high prediction scores (above 0.39; n = 1,407 patients). Error bars denote the 95% confidence intervals derived from bootstrapping. For every case, the top-1 and top-2 accuracies of the integrated model are significantly higher than those of each individual component (P-values < 0.001, according to one-sided bootstrap test, see Supplementary Table 3).
Extended Data Fig. 4 Performance of the combination of the direct and demographic models, and the combination of the indirect and demographic models.
Top-1 and top-2 accuracies obtained by the four models: Indirect, combination of Direct and Demographic (Direct+Demographic), combination of Indirect and Demographic (Indirect+Demographic), and Integrated model. a, The results are shown for all samples. Results for each cohort are organized separately from left to right: NCI (n = 1,796 patients), DBTA (n = 1,522 patients), CBTN (n = 348 patients), and NCI-Prospective (n = 286). b, Results for samples with prediction score above 0.39. Results for each cohort are organized separately from left to right: NCI (n = 1,193 patients), DBTA (n = 1,026 patients), CBTN (n = 212 patients), and NCI-Prospective (n = 169 patients) (b). Error bars denote the 95% confidence intervals derived from 1,000 bootstrapping iterations and the centers correspond to the computed accuracy values for each model.
Extended Data Fig. 5 Model performance on NCI cohort for each individual cancer type.
The one-versus-rest AUPRC, Precision, Recall, and F1 score for each brain tumor type achieved by demographic (light blue), direct (orange), indirect (red), and our integrated model (dark blue). The number of patients in each cancer type is shown in parentheses. For each metric indicated on the vertical axis, the error bars represent the 95% confidence intervals, derived from 1,000 bootstrapping iterations and the centers correspond to computed values of the respective metric.
Extended Data Fig. 6 Model performance on all the three external cohorts together for each individual cancer type.
The one-versus-rest AUPRC, Precision, Recall, and F1 score for each brain tumor type achieved by demographic (light blue), direct (orange), indirect (red), and our integrated model (dark blue). The number of patients in each cancer type is shown in parentheses. For each metric indicated on the vertical axis, the error bars represent the 95% confidence intervals, derived from 1,000 bootstrapping iterations and the centers correspond to computed values of the respective metric.
Extended Data Fig. 7 Model performance on DBTA cohort for each individual cancer type.
The one-versus-rest AUPRC, Precision, Recall, and F1 score for each brain tumor type achieved by demographic (light blue), direct (orange), indirect (red), and our integrated model (dark blue). The number of patients in each cancer type is shown in parentheses. For each metric indicated on the vertical axis, the error bars represent the 95% confidence intervals, derived from 1,000 bootstrapping iterations and the centers correspond to computed values of the respective metric.
Extended Data Fig. 8 Model performance on CBTN cohort for each individual cancer type.
The one-versus-rest AUPRC, Precision, Recall, and F1 score for each brain tumor type achieved by demographic (light blue), direct (orange), indirect (red), and our integrated model (dark blue). Notably, GBM, SE and O-IDH are not present in the CBTN cohort. The number of patients in each cancer type is shown in parentheses. For each metric indicated on the vertical axis, the error bars represent the 95% confidence intervals, derived from 1,000 bootstrapping iterations and the centers correspond to computed values of the respective metric.
Extended Data Fig. 9 Model performance on NCI-Prospective cohort for each individual cancer type.
The one-versus-rest AUPRC, Precision, Recall, and F1 score for each brain tumor type achieved by demographic (light blue), direct (orange), indirect (red), and our integrated model (dark blue). The number of patients in each cancer type is shown in parentheses. For each metric indicated on the vertical axis, the error bars represent the 95% confidence intervals, derived from 1,000 bootstrapping iterations and the centers correspond to computed values of the respective metric.
Extended Data Fig. 10 Balanced accuracies on each cohort for samples with high prediction scores (above 0.39).
Results for each cohort are organized separately from left to right: NCI (first column), DBTA (second column), CBTN (third column) and NCI-Prospective (fourth column). Error bars denote the 95% confidence intervals obtained via bootstrapping (1,000 iterations) and the centers correspond to the computed accuracy values for each model.
Supplementary information
Rights and permissions
About this article
Cite this article
Hoang, DT., Shulman, E.D., Turakulov, R. et al. Prediction of DNA methylation-based tumor types from histopathology in central nervous system tumors with deep learning. Nat Med (2024). https://doi.org/10.1038/s41591-024-02995-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41591-024-02995-8