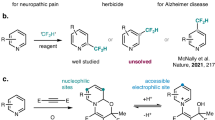
Abstract
Macrocyclic peptides represent promising scaffolds for chemical tools and potential therapeutics. Synthetic methods for peptide macrocyclization are often hampered by C-terminal epimerization and oligomerization, leading to difficult scalability. While chemical strategies to circumvent this issue exist, they often require specific amino acids to be present in the peptide sequence. Herein, we report the characterization of Ulm16, a peptide cyclase belonging to the penicillin-binding protein-type class of thioesterases that catalyze head-to-tail macrolactamization of nonribosmal peptides. Ulm16 efficiently cyclizes various nonnative peptides ranging from 4 to 6 amino acids with catalytic efficiencies of up to 3 × 106 M−1 s−1. Unlike many previously described homologs, Ulm16 tolerates a variety of C- and N-terminal amino acids. The crystal structure of Ulm16, along with modeling of its substrates and site-directed mutagenesis, allows for rationalization of this wide substrate scope. Overall, Ulm16 represents a promising tool for the biocatalytic production of macrocyclic peptides.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Ulm16 12–440 coordinates and processed diffraction data have been deposited into the PDB under the accession code 8FEK. Other structures used in this paper are available at the PDB (6KSU and 6KSV). Protein sequences used in this study are from the NCBI database: Ulm16 (accession ATU31793.1), CppA (QQY97180.1), PenA (WP_158102277), SurE (BBZ90014.1), MppK (AAU34204.1), Lon18 (QUJ09165.1), FlkO (AGI87381.1), WolJ (UNO41476.1) and DsaJ (AJW76712.1). PDB files of AlphaFold models generated this for this study are publicly available and can be accessed using the following DOI: https://doi.org/10.6084/m9.figshare.24467026. The data that support the findings of this study are available within the main text and its Supplementary Information files. Data are also available from the corresponding author upon request.
References
Driggers, E. M., Hale, S. P., Lee, J. & Terrett, N. K. The exploration of macrocycles for drug discovery—an underexploited structural class. Nat. Rev. Drug Discov. 7, 608–624 (2008).
Huang, Y., Wiedmann, M. M. & Suga, H. RNA display methods for the discovery of bioactive macrocycles. Chem. Rev. 119, 10360–10391 (2019).
Tsomaia, N. Peptide therapeutics: targeting the undruggable space. Eur. J. Med. Chem. 94, 459–470 (2015).
Malde, A. K., Hill, T. A., Iyer, A. & Fairlie, D. P. Crystal structures of protein-bound cyclic peptides. Chem. Rev. 119, 9861–9914 (2019).
White, C. J. & Yudin, A. K. Contemporary strategies for peptide macrocyclization. Nat. Chem. 3, 509–524 (2011).
Sarojini, V., Cameron, A. J., Varnava, K. G., Denny, W. A. & Sanjayan, G. Cyclic tetrapeptides from nature and design: a review of synthetic methodologies, structure, and function. Chem. Rev. 119, 10318–10359 (2019).
Du, L., Risinger, A. L., King, J. B., Powell, D. R. & Cichewicz, R. H. A potent HDAC inhibitor, 1-Alaninechlamydocin, from a Tolypocladium sp. induces G2/M cell cycle arrest and apoptosis in MIA PaCa-2 cells. J. Nat. Prod. 77, 1753–1757 (2014).
Pérez-Victoria, I. et al. Isolation and structural elucidation of cyclic tetrapeptides from Onychocola sclerotica. J. Nat. Prod. 75, 1210–1214 (2012).
Steele, J. A., Uchytil, T. F., Durbin, R. D. & Rich, D. H. Chloroplast coupling factor 1: a species-specific receptor for tentoxin. Proc. Natl Acad. Sci. USA 73, 2245–2248 (1976).
Saito, T. et al. CJ-15, 208, a novel kappa opioid receptor antagonist from a fungus, Ctenomyces serratus ATCC15502. J.Antibiotics 55, 847–854 (2002).
Chung, B. K. W., White, C. J., Scully, C. C. G. & Yudin, A. K. The reactivity and conformational control of cyclic tetrapeptides derived from aziridine-containing amino acids. Chem. Sci. 7, 6662–6668 (2016).
Skropeta, D., Jolliffe, K. A. & Turner, P. Pseudoprolines as removable turn inducers: tools for the cyclization of small peptides. J. Org. Chem. 69, 8804–8809 (2004).
Alcaro, M. C. et al. On-resin head-to-tail cyclization of cyclotetrapeptides: optimization of crucial parameters. J. Pept. Sci. 10, 218–228 (2004).
Meutermans, W. D. F. et al. Difficult macrocyclizations: new strategies for synthesizing highly strained cyclic tetrapeptides. Org. Lett. 5, 2711–2714 (2003).
Vidović, N. et al. Chloride-assisted peptide macrocyclization. Org. Lett. 22, 2129–2134 (2020).
Jing, X. & Jin, K. A gold mine for drug discovery: strategies to develop cyclic peptides into therapies. Med. Res. Rev. 40, 753–810 (2020).
Abdalla, M. A. Medicinal significance of naturally occurring cyclotetrapeptides. J. Nat. Med. 70, 708–720 (2016).
Li, Y.-H. et al. Putative nonribosomal peptide synthetase and cytochrome P450 genes responsible for tentoxin biosynthesis in Alternaria alternata ZJ33. Toxins 8, 234 (2016).
Witte, T. E., Villeneuve, N., Boddy, C. N. & Overy, D. P. Accessory chromosome-acquired secondary metabolism in plant pathogenic fungi: the evolution of biotrophs into host-specific pathogens. Front. Microbiol. 12, 664276 (2021).
Xu, H.-M. et al. Tataricins A and B, two novel cyclotetrapeptides from Aster tataricus, and their absolute configuration assignment. Tetrahedron Lett. 54, 1380–1383 (2013).
Ma, G.-L. et al. Biosynthesis of tasikamides via pathway coupling and diazonium-mediated hydrazone formation. J. Am. Chem. Soc. 144, 1622–1633 (2022).
Kohli, R. M., Walsh, C. T. & Burkart, M. D. Biomimetic synthesis and optimization of cyclic peptide antibiotics. Nature 418, 658–661 (2002).
Trauger, J. W., Kohli, R. M., Mootz, H. D., Marahiel, M. A. & Walsh, C. T. Peptide cyclization catalysed by the thioesterase domain of tyrocidine synthetase. Nature 407, 215–218 (2000).
Horsman, M. E., Hari, T. P. A. & Boddy, C. N. Polyketide synthase and non-ribosomal peptide synthetase thioesterase selectivity: logic gate or a victim of fate? Nat. Prod. Rep. 33, 183–202 (2016).
Gao, X. et al. Cyclization of fungal nonribosomal peptides by a terminal condensation-like domain. Nat. Chem. Biol. 8, 823–830 (2012).
Hoyer, K. M., Mahlert, C. & Marahiel, M. A. The iterative gramicidin S thioesterase catalyzes peptide ligation and cyclization. Chem. Biol. 14, 13–22 (2007).
Kuranaga, T. et al. Total synthesis of the nonribosomal peptide surugamide B and identification of a new offloading cyclase family. Angew. Chem. 130, 9591–9595 (2018).
Zhou, Y. et al. Investigation of penicillin binding protein (PBP)-like peptide cyclase and hydrolase in surugamide non-ribosomal peptide biosynthesis. Cell Chem. Biol. 26, 737–744.e4 (2019).
Thankachan, D. et al. A trans-acting cyclase offloading strategy for nonribosomal peptide synthetases. ACS Chem. Biol. 14, 845–849 (2019).
Matsuda, K. et al. SurE is a trans -acting thioesterase cyclizing two distinct non-ribosomal peptides. Org. Biomol. Chem. 17, 1058–1061 (2019).
Matsuda, K. et al. Heterochiral coupling in non-ribosomal peptide macrolactamization. Nat. Catal. 3, 507–515 (2020).
Matsuda, K., Fujita, K. & Wakimoto, T. PenA, a penicillin-binding protein-type thioesterase specialized for small peptide cyclization. J. Ind. Microbiol. Biotechnol. 48, kuab023 (2021).
Fazal, A., Wheeler, J., Webb, M. E. & Seipke, R. F. The N-terminal substrate specificity of the SurE peptide cyclase. Org. Biomol. Chem. 20, 7232–7235 (2022).
Kobayashi, M., Fujita, K., Matsuda, K. & Wakimoto, T. Streamlined chemoenzymatic synthesis of cyclic peptides by non-ribosomal peptide cyclases. J. Am. Chem. Soc. 145, 3270–3275 (2023).
Fazal, A., Webb, M. E. & Seipke, R. F. The desotamide family of antibiotics. Antibiotics 9, 452 (2020).
Booth, T. J. et al. Bifurcation drives the evolution of assembly-line biosynthesis. Nat. Commun. 13, 3498 (2022).
Mirdita, M. et al. ColabFold: making protein folding accessible to all. Nat. Methods 19, 679–682 (2022).
Son, S. et al. Genomics-driven discovery of chlorinated cyclic hexapeptides Ulleungmycins A and B from a Streptomyces species. J. Nat. Prod. 80, 3025–3031 (2017).
Gao, D.-W. et al. A polyketide cyclase that forms medium-ring lactones. J. Am. Chem. Soc. 143, 80–84 (2021).
Bar-Even, A. et al. The moderately efficient enzyme: evolutionary and physicochemical trends shaping enzyme parameters. Biochemistry 50, 4402–4410 (2011).
Hostetler, M. A. et al. Synthetic natural product inspired cyclic peptides. ACS Chem. Biol. 16, 2604–2611 (2021).
Walsh, C. & Wencewicz, T. A. Antibiotics: Challenges, Mechanisms, Opportunities (ASM Press, 2016).
Dubus, A., Normark, S., Kania, M. & Page, M. G. P. The role of tyrosine 150 in catalysis of beta-lactam hydrolysis by AmpC beta.-lactamase from Escherichia coli investigated by site-directed mutagenesis. Biochemistry 33, 8577–8586 (1994).
McIntosh, J. A. et al. Circular logic: nonribosomal peptide-like macrocyclization with a ribosomal peptide catalyst. J. Am. Chem. Soc. 132, 15499–15501 (2010).
Ludewig, H. et al. Characterization of the fast and promiscuous macrocyclase from plant PCY1 enables the use of simple substrates. ACS Chem. Biol. 13, 801–811 (2018).
Nguyen, G. K. T. et al. Butelase-mediated cyclization and ligation of peptides and proteins. Nat. Protoc. 11, 1977–1988 (2016).
Hemu, X., Qiu, Y., Nguyen, G. K. T. & Tam, J. P. Total synthesis of circular bacteriocins by butelase 1. J. Am. Chem. Soc. 138, 6968–6971 (2016).
Nguyen, G. K. T. et al. Butelase 1: a versatile ligase for peptide and protein macrocyclization. J. Am. Chem. Soc. 137, 15398–15401 (2015).
Waterhouse, A. M., Procter, J. B., Martin, D. M. A., Clamp, M. & Barton, G. J. Jalview version 2—a multiple sequence alignment editor and analysis workbench. Bioinformatics 25, 1189–1191 (2009).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Terlouw, B. R. et al. MIBiG 3.0: a community-driven effort to annotate experimentally validated biosynthetic gene clusters. Nucleic Acids Res. 51, D603–D610 (2023).
Gilchrist, C. L. M. & Chooi, Y.-H. clinker & clustermap.js: automatic generation of gene cluster comparison figures. Bioinformatics 37, 2473–2475 (2021).
Jones, D. T., Taylor, W. R. & Thornton, J. M. The rapid generation of mutation data matrices from protein sequences. Bioinformatics 8, 275–282 (1992).
Kumar, S., Stecher, G., Li, M., Knyaz, C. & Tamura, K. MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 35, 1547–1549 (2018).
Schmidt, J. J. et al. A versatile chemoenzymatic synthesis for the discovery of potent cryptophycin analogs. ACS Chem. Biol. 15, 524–532 (2020).
Minor, W., Cymborowski, M., Otwinowski, Z. & Chruszcz, M. HKL-3000: the integration of data reduction and structure solution–from diffraction images to an initial model in minutes. Acta Crystallogr. D. Biol. Crystallogr. 62, 859–866 (2006).
Rossmann, M. G. The molecular replacement method. Acta Crystallogr. A 46, 73–82 (1990).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D. Biol. Crystallogr. 66, 213–221 (2010).
Liebschner, D. et al. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr. Sect. Struct. Biol. 75, 861–877 (2019).
Waterhouse, A. et al. Swiss-model: Homology modelling of protein structures and complexes. Nucleic Acids Res. 46, W296–W303 (2018).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D. Biol. Crystallogr. 66, 486–501 (2010).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D. Biol. Crystallogr. 66, 12–21 (2010).
Blanco-Canosa, J. B., Nardone, B., Albericio, F. & Dawson, P. E. Chemical protein synthesis using a second-generation n-acylurea linker for the preparation of peptide-thioester precursors. J. Am. Chem. Soc. 137, 7197–7209 (2015).
Acknowledgements
We thank A. Alwali and L. Wilbanks for their helpful discussions and aid with UPLC–MS and protein expression, respectively, C. Martinez-Brokaw and M. Hostetler for their advice on chemical synthesis and G. Buechel for her initial work on peptide synthesis. This work was funded by the National Institutes of Health (grant nos. 1R35GM138002 to E.I.P. and 1F31CA275390 to R.S.P.). Z.L.B. acknowledges the National Science Foundation for support under the Graduate Research Fellowship Program under grant no. DGE-1842166. We acknowledge the support from the Purdue Center for Cancer Research, NIH grant no. P30 CA023168.
Author information
Authors and Affiliations
Contributions
Z.L.B., C.N.E. and J.J.A. synthesized all the peptides used in this study. A.E. and C.N.E. performed nuclear magnetic resonance analyses of the peptides. Z.L.B., R.S.P. and H.M.R.-C. expressed the proteins. Z.L.B. performed the protein assays. R.S.P. performed the crystallography studies. Z.L.B. and R.S.P. performed the docking studies. Z.L.B., R.S.P., C.D. and E.I.P. conceived of the ideas and wrote the paper, with input from all authors. Project management and funding was the responsibility of C.D. and E.I.P.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks Hai Deng, Akimasa Miyanaga and Ryan Seipke for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Alpha fold models of predicted and validated PBP-TEs.
compared to the SurE Crystal structure (RMSD). The alpha fold model of SurE was used as a control and was compared to its known crystal structure. Highlighted in red is the loop region, which is shorter in the Ulm16 sequence, in yellow is the lipocalin loop previously hypothesized to play a role in substrate selectivity, and in orange is the sequence insertion only found in Ulm16 (residues 106-122).
Extended Data Fig. 2 Biosynthetic gene clusters of PBP-TEs that lack the loop region.
These biosynthetic gene clusters all share high homology to the ulleungmycin biosynthetic gene cluster and are predicted to produce natural products identical or very similar to the ulleungmycins.
Extended Data Fig. 3 Ulm peptide structure.
The Ulleungmycin A sequence was modified to incorporate only commercially available amino acids, resulting in a new sequence referred to as 'Ulm'. The changed amino acids are highlighted in grey, while the C-Terminal amino acid is highlighted in red and the N-Terminal amino acid is highlighted in blue.
Extended Data Fig. 4 Michaelis-Menten plots of Ulm16 kinetics for three peptide thioesters and alanine scans of the 'Ulm' peptide.
The names of the substrates used are indicated above each plot, and the enzyme concentrations employed are provided in the methods section. The data is summarized in of the main text. The plots represent the mean of triplicate experiments, and the error bars indicate the standard error of the mean (S.E.M).
Extended Data Fig. 5 Michaelis-Menten plots of Ulm16 and SurE kinetics for a common substrate (13).
The names of the substrates used are indicated above each plot, and the enzyme concentrations employed are provided in the methods section. The plots represent the mean of triplicate experiments, and the error bars indicate the standard error of the mean (S.E.M).
Extended Data Fig. 6 Michaelis-Menten plot of Ulm16 incubated with tetrapeptide 16.
The names of the substrates used are indicated above each plot, and the enzyme concentrations employed are provided in the methods section. The data is summarized in Table 1 of the main text. The plots represent the mean of triplicate experiments, and the error bars indicate the standard error of the mean (S.E.M).
Extended Data Fig. 7 Covalent docking of tetrapeptide 16 with SurE and comparison with Ulm16.
(a, b) Side-by-side of lowest MMGBSA score poses for SurE-16 (orange, A) and Ulm16-16 (blue, B). Peptide 16 is noticeably farther away from the hydrophobic pocket of the lipocalin domain in SurE due to differing angle of the lipocalin domain. (c) Overlay of Ulm16-16 (Sky blue-Light blue) and SurE-16 (Orange-Light Orange) highlights residues that we believe are key for binding and cyclization. Residues L284/L300 and D306/D297 are in the alpha beta hydrolase domain, and we hypothesize are key in binding the C-terminal D-amino acid. Residues R446/R431 and Y443/Y428 are in the lipocalin domain. We hypothesize that they help in cyclization of the peptide. The alpha beta hydrolase domain has been hidden for clarity. (d) Overlay of the C-terminal SNAC-D-Leu (orange) from the SurE substrate bound crystal structure (6SKV) and C-terminal covalently docked D-Tyr from peptide 16 (light orange). (e) Overlay of the C-Terminal SNAC-D-Leu (orange) from the SurE substrate bound crystal structure (6SKV) and C-terminal covalently docked D-Tyr from peptide 16 with Ulm16 (blue) showing they are occupying the same pocket.
Extended Data Fig. 8 Side-by-side comparison of top 5 lowest MMGBSA scoring peptides from covalent docking of 16 with SurE (light orange, A) and Ulm16 (light blue, B).
Peptide 16 exhibits limited access to the hydrophobic pocket within the lipocalin domain of SurE, resulting in a notable diversity of poses generated. Conversely, in the case of Ulm16, distinct conformations of the lipocalin domain facilitate a more proximal interaction with the hydrophobic pocket, leading to a predominant binding orientation consistently observed across outputs. Residues L286/L300 and D306/D297 are in the alpha-beta-hydrolase domain while Y443/Y428 is in the lipocalin domain.
Supplementary information
Supplementary Information
Supplementary Schemes 1 and 2, Tables 1–4, Notes 1 and 2, Figs. 1–106 and references.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Budimir, Z.L., Patel, R.S., Eggly, A. et al. Biocatalytic cyclization of small macrolactams by a penicillin-binding protein-type thioesterase. Nat Chem Biol 20, 120–128 (2024). https://doi.org/10.1038/s41589-023-01495-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-023-01495-z
This article is cited by
-
Chemoenzymatic tandem cyclization for the facile synthesis of bicyclic peptides
Communications Chemistry (2024)
-
Size matters
Nature Chemical Biology (2024)