Abstract
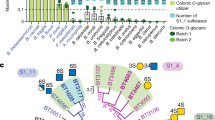
Sulfated glycans are ubiquitous nutrient sources for microbial communities that have coevolved with eukaryotic hosts. Bacteria metabolize sulfated glycans by deploying carbohydrate sulfatases that remove sulfate esters. Despite the biological importance of sulfatases, the mechanisms underlying their ability to recognize their glycan substrate remain poorly understood. Here, we use structural biology to determine how sulfatases from the human gut microbiota recognize sulfated glycans. We reveal seven new carbohydrate sulfatase structures spanning four S1 sulfatase subfamilies. Structures of S1_16 and S1_46 represent novel structures of these subfamilies. Structures of S1_11 and S1_15 demonstrate how non-conserved regions of the protein drive specificity toward related but distinct glycan targets. Collectively, these data reveal that carbohydrate sulfatases are highly selective for the glycan component of their substrate. These data provide new approaches for probing sulfated glycan metabolism while revealing the roles carbohydrate sulfatases play in host glycan catabolism.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Code availability
No new code was developed or compiled in this study.
Change history
05 August 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41589-022-01132-1
References
Sarrazin, S., Lamanna, W. C. & Esko, J. D. Heparan sulfate proteoglycans. Cold Spring Harb. Perspect. Biol. 3, a004952 (2011).
Soares da Costa, D., Reis, R. L. & Pashkuleva, I. Sulfation of glycosaminoglycans and its implications in human health and disorders. Annu. Rev. Biomed. Eng. 19, 1–26 (2017).
Luis, A. S. et al. A single sulfatase is required to access colonic mucin by a gut bacterium. Nature 598, 332–337 (2021).
Bloom, S. M. et al. Commensal Bacteroides species induce colitis in host-genotype-specific fashion in a mouse model of inflammatory bowel disease. Cell Host Microbe 9, 390–403 (2011).
Johansson, M. E., Larsson, J. M. & Hansson, G. C. The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host–microbial interactions. Proc. Natl Acad. Sci. USA 108, 4659–4665 (2011).
Field, C. B., Behrenfeld, M. J., Randerson, J. T. & Falkowski, P. Primary production of the biosphere: integrating terrestrial and oceanic components. Science 281, 237–240 (1998).
Chen, J. et al. Laminarin, a major polysaccharide in stramenopiles. Mar. Drugs 19, 576 (2021).
Hettle, A. G. et al. Insights into the κ/ι-carrageenan metabolism pathway of some marine Pseudoalteromonas species. Commun. Biol. 2, 474 (2019).
Reisky, L. et al. A marine bacterial enzymatic cascade degrades the algal polysaccharide ulvan. Nat. Chem. Biol. 15, 803–812 (2019).
Zhang, Q. et al. Chemical characteristics of a polysaccharide from Porphyra capensis (Rhodophyta). Carbohydr. Res. 340, 2447–2450 (2005).
Ponce, N. M. A. & Stortz, C. A. A comprehensive and comparative analysis of the fucoidan compositional data across the Phaeophyceae. Front. Plant Sci. 11, 556312 (2020).
Panggabean, J. A. et al. Antiviral activities of algal-based sulfated polysaccharides. Molecules 27, 1178 (2022).
Pereira, L. Carrageenans: Sources and Extraction Methods, Molecular Structure, Bioactive Properties and Health Effects (Nova Science Publishers, 2016).
Cartmell, A. et al. How members of the human gut microbiota overcome the sulfation problem posed by glycosaminoglycans. Proc. Natl Acad. Sci. USA 114, 7037–7042 (2017).
Tuncil, Y. E. et al. Reciprocal prioritization to dietary glycans by gut bacteria in a competitive environment promotes stable coexistence. mBio 8, e01068-17 (2017).
Raghavan, V. & Groisman, E. A. Species-specific dynamic responses of gut bacteria to a mammalian glycan. J. Bacteriol. 197, 1538–1548 (2015).
Cheng, H. Y., Ning, M. X., Chen, D. K. & Ma, W. T. Interactions between the gut microbiota and the host innate immune response against pathogens. Front. Immunol. 10, 607 (2019).
McNeil, N. I. The contribution of the large intestine to energy supplies in man. Am. J. Clin. Nutr. 39, 338–342 (1984).
Goodman, A. L. et al. Identifying genetic determinants needed to establish a human gut symbiont in its habitat. Cell Host Microbe 6, 279–289 (2009).
Li, H. et al. The outer mucus layer hosts a distinct intestinal microbial niche. Nat. Commun. 6, 8292 (2015).
Tsai, H. H., Dwarakanath, A. D., Hart, C. A., Milton, J. D. & Rhodes, J. M. Increased faecal mucin sulphatase activity in ulcerative colitis: a potential target for treatment. Gut 36, 570–576 (1995).
Alipour, M. et al. Mucosal barrier depletion and loss of bacterial diversity are primary abnormalities in paediatric ulcerative colitis. J. Crohns Colitis 10, 462–471 (2016).
Hickey, C. A. et al. Colitogenic Bacteroides thetaiotaomicron antigens access host immune cells in a sulfatase-dependent manner via outer membrane vesicles. Cell Host Microbe 17, 672–680 (2015).
Barbeyron, T. et al. Matching the diversity of sulfated biomolecules: creation of a classification database for sulfatases reflecting their substrate specificity. PLoS ONE 11, e0164846 (2016).
Hanson, S. R., Best, M. D. & Wong, C. H. Sulfatases: structure, mechanism, biological activity, inhibition, and synthetic utility. Angew. Chem. Int. Ed. Engl. 43, 5736–5763 (2004).
Hettle, A. G. et al. The molecular basis of polysaccharide sulfatase activity and a nomenclature for catalytic subsites in this class of enzyme. Structure 26, 747–758 (2018).
Terrapon, N., Lombard, V., Gilbert, H. J. & Henrissat, B. Automatic prediction of polysaccharide utilization loci in Bacteroidetes species. Bioinformatics 31, 647–655 (2015).
Ndeh, D. et al. Metabolism of multiple glycosaminoglycans by Bacteroides thetaiotaomicron is orchestrated by a versatile core genetic locus. Nat. Commun. 11, 646 (2020).
Wei, W., Ninonuevo, M. R., Sharma, A., Danan-Leon, L. M. & Leary, J. A. A comprehensive compositional analysis of heparin/heparan sulfate-derived disaccharides from human serum. Anal. Chem. 83, 3703–3708 (2011).
Sidhu, N. S. et al. Structure of sulfamidase provides insight into the molecular pathology of mucopolysaccharidosis IIIA. Acta Crystallogr. D Biol. Crystallogr. 70, 1321–1335 (2014).
von Bulow, R. et al. Defective oligomerization of arylsulfatase A as a cause of its instability in lysosomes and metachromatic leukodystrophy. J. Biol. Chem. 277, 9455–9461 (2002).
Robb, C. S. et al. Metabolism of a hybrid algal galactan by members of the human gut microbiome. Nat. Chem. Biol. 18, 501–510 (2022).
Juers, D. H. et al. A structural view of the action of Escherichia coli (lacZ) β-galactosidase. Biochemistry 40, 14781–14794 (2001).
Helbert, W. et al. Discovery of novel carbohydrate-active enzymes through the rational exploration of the protein sequences space. Proc. Natl Acad. Sci. USA 116, 6063–6068 (2019).
Lapebie, P., Lombard, V., Drula, E., Terrapon, N. & Henrissat, B. Bacteroidetes use thousands of enzyme combinations to break down glycans. Nat. Commun. 10, 2043 (2019).
Pudlo, N. A. et al. Diverse events have transferred genes for edible seaweed digestion from marine to human gut bacteria. Cell Host Microbe 30, 314–328 (2022).
Verma, S. et al. Identification and engraftment of new bacterial strains by shotgun metagenomic sequence analysis in patients with recurrent Clostridioides difficile infection before and after fecal microbiota transplantation and in healthy human subjects. PLoS ONE 16, e0251590 (2021).
Roche, P. et al. Molecular basis of symbiotic host specificity in Rhizobium meliloti: nodH and nodPQ genes encode the sulfation of lipo-oligosaccharide signals. Cell 67, 1131–1143 (1991).
Varki, A. et al. Symbol nomenclature for graphical representations of glycans. Glycobiology 25, 1323–1324 (2015).
Das, T. M., Rao, C. P. & Kolehmainen, E. Synthesis and characterisation of N-glycosyl amines from the reaction between 4,6-O-benzylidene-d-glucopyranose and substituted aromatic amines and also between 2-(o-aminophenyl)benzimidazole and pentoses or hexoses. Carbohydr. Res. 334, 261–269 (2001).
Byrne, D. P., London, J. A., Eyers, P. A., Yates, E. A. & Cartmell, A. Mobility shift-based electrophoresis coupled with fluorescent detection enables real-time enzyme analysis of carbohydrate sulfatase activity. Biochemical J. 478, 735–748 (2021).
Labourel, A. et al. Structural and functional analysis of glycoside hydrolase 138 enzymes targeting chain A galacturonic acid in the complex pectin rhamnogalacturonan II. J. Biol. Chem. 294, 7711–7721 (2019).
Byrne, D. P. et al. cAMP-dependent protein kinase (PKA) complexes probed by complementary differential scanning fluorimetry and ion mobility-mass spectrometry. Biochemical J. 473, 3159–3175 (2016).
Kabsch, W. Xds. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Evans, P. Scaling and assessment of data quality. Acta Crystallogr. D Biol. Crystallogr. 62, 72–82 (2006).
Evans, P. R. An introduction to data reduction: space-group determination, scaling and intensity statistics. Acta Crystallogr. D Biol. Crystallogr. 67, 282–292 (2011).
Long, F., Vagin, A. A., Young, P. & Murshudov, G. N. BALBES: a molecular-replacement pipeline. Acta Crystallogr. D Biol. Crystallogr. 64, 125–132 (2008).
McCoy, A. J. Solving structures of protein complexes by molecular replacement with Phaser. Acta Crystallogr. D Biol. Crystallogr. 63, 32–41 (2007).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr 66, 486–501 (2010).
Murshudov, G. N. et al. REFMAC5 for the refinement of macromolecular crystal structures. Acta Crystallogr. D Biol. Crystallogr. 67, 355–367 (2011).
Lebedev, A. A. et al. JLigand: a graphical tool for the CCP4 template-restraint library. Acta Crystallogr. D Biol. Crystallogr. 68, 431–440 (2012).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Potterton, L. et al. CCP4i2: the new graphical user interface to the CCP4 program suite. Acta Crystallogr. D Struct. Biol. 74, 68–84 (2018).
Collaborative Computational Project The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D Biol. Crystallogr. 50, 760–763 (1994).
Katoh, K., Misawa, K., Kuma, K. & Miyata, T. MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 30, 3059–3066 (2002).
Clamp, M., Cuff, J., Searle, S. M. & Barton, G. J. The Jalview Java alignment editor. Bioinformatics 20, 426–427 (2004).
Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313 (2014).
Felsenstein, J. Evolutionary trees from DNA sequences: a maximum likelihood approach. J. Mol. Evol. 17, 368–376 (1981).
Le, S. Q. & Gascuel, O. An improved general amino acid replacement matrix. Mol. Biol. Evol. 25, 1307–1320 (2008).
Felsenstein, J. Confidence limits on phylogenies: an approach using the Bootstrap. Evolution 39, 783–791 (1985).
Acknowledgements
This project has received funding from the European Union’s Horizon 2020 research and innovation program under the Marie Skłodowska-Curie grant agreement number 748336 and Wilhelm och Martina Lundgrens Vetenskapsfond (2020.3597) awarded to A.S.L. Additional funding sources include The European Research Council ERC (694181), the Knut and Alice Wallenberg Foundation (2017.0028), Swedish Research Council (2017-00958) awarded to G.C.H., the National Institute of Health (R01 DK118024 and DK125445 and U01AI095473) award to E.C.M. and G.C.H. and the Academy of Medical Sciences/Wellcome Trust through the Springboard Grant (SBF005\1065 163470) awarded to A.C. We acknowledge access to the SOLEIL and Diamond Light sources via both the University of Liverpool and Newcastle University BAGs (proposals mx21970 and mx18598, respectively). We thank the staff of DIAMOND and SOLEIL and members of the Liverpool’s Molecular biophysics group for assistance with data collection. We are also grateful for E. Corre’s help regarding bioinformatics analyses (ABIMS platform, Station Biologique de Roscoff, France).
Author information
Authors and Affiliations
Contributions
A.S.L. and A.C. conceived and designed experiments. A.C., A.S.L. and E.C.M. wrote the draft manuscript. A.S.L. and A.C. cloned, expressed and purified sulfatases and performed the enzymatic assays. A.C., D.P.B. and J.A.L. performed and analyzed kinetic and binding experiments. E.A.Y. and J.A.L. performed labeling and NMR experiments. A.C. and A.B. performed structural biology experiments. M.C. and T.B. performed sulfatase phylogenetic analyses. J.C. and N.G.K. performed glycan analyses. G.S.A.W. performed light scattering experiments and size determination. A.C., A.S.L., G.C.H. and E.C.M. supervised and provided funding for the project. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks Jan-Hendrick Hehemann, Nicolas Terrapon and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Electron density maps of extracted ligands.
The 2mFobs-Fc maps are shown contoured at 1σ for all substrates and products co-crystallised with their respective sulfatase.
Extended Data Fig. 2 Biophysical, Specificity and phylogenetic analysis of BT19183S-GlcNAc and S1_46.
a, Radial version of the phylogenetic tree of representative sulfatases from subfamily S1_46. The tree comprises a total of 564 sequences with 250 being Firmicutes; 156 are Bacteroidetes; 54 are Actinobacteria; 25 are Proteobacteria; 20 are Lentisphaerae. For clarity all labels and sequence accession codes have been omitted. The annotations next to the colour code reveal the presence or absence of conserved residues crucial for substrate recognition by BT19183S-GlcNAc (acc-code Q8A6G6) in the following order: Y94, N174, R327 and Y408. These residues are invariant in HGM Bacteroidetes, whilst in Firmicutes from the HGM, the Y408 equivalent is not conserved. The residues are coloured as following: black means an equivalent residue is present; a grey and bold letter at any position means that the corresponding residue is replaced by that amino acid; a grey, bold and italic letter at any position means that the equivalent position is replaced by any type of amino acid; a bold grey letter followed by one-letter codes in parentheses indicates that the equivalent position can be substituted by any of those amino acids; the dash at the Y408-equivalent position indicates that no equivalent amino acid can be deduced from the multiple alignment. Branches of the same colour have the corresponding pattern in common. The red filled circle designates the sequence of the S1_46 sulfatase from B. thetaiotaomicron (See Supplementary Fig. 2 for full tree). b, Thin layer chromatography analysis of BT19183S,6S-GlcNAc versus 3S-glucosamine, 3 S,6S-glucosamine, and 3S,6S-N-acetylglucosamine, and QSI_25163S,6S-GlcNAc versus 3S-glucosamine. 3 S,6S-glucosamine, and 3S,6S-N-acetylglucosamie. All assays described were performed for 48 h at 37 °C, containing 6 mM substrate and 5 μM (BT19183S-GlcNAc) or 100 μM enzyme (QSI_25163S,6S-GlcNAc) and 3 mM HEPES pH 7.0, 45 mM NaCl and 5 mM CaCl2.
Extended Data Fig. 3 Activity and stability analysis of S1_16 sulfatases and their mutant variants.
a, DSF analysis of the effects of galactose and N-acetylgalactosamine on thermostability, with a positive-shift indicative of substrate binding. b, Normalised DSF melt curves of BT30574S-Gal/GalNAc and BT37964S-Gal/GalNAc. c, DSF melt curves of purified monomer and dimer species (left), monomer species in the presence of galactose and N-acetylgalactosamine (middle), or dimer species in the presence of galactose and N-acetylgalactosamine (right). d, Thin layer chromatography (TLC) analysis of wild-type (WT) and mutant S1_16 sulfatases. Asterisks are placed above lanes where sulfatase activity is observed. e, High pressure anion exchange chromatography (HPAEC) of WT and mutants. A grey block highlights the desulfated product. Both TLC and HPAEC reactions utilised 6 mM substrate and 1 μM enzyme, except for W109A variants where 10 μM was used, with 3 mM HEPES, 45 mM NaCl, and 5 mM CaCl2. Reactions incubated at 37 °C for 48 h. Control represents the substrate incubated in same conditions without adding enzyme. Experiments are technical triplicates and error bars represent SEM.
Extended Data Fig. 4 Radial phylogenetic tree of representative sulfatases from subfamily S1_16.
The tree comprises a total of 1368 sequences of which 854 are Bacteroidetes; 211 are Planctomycetes; 107 are Kiritimatiellaeota; 64 are Verrucomicrobia; 53 are Lentisphaerae. For clarity all labels and sequence accession codes have been omitted. The annotations next to the colour code reveal the presence or absence of conservation of the critical residues in substrate recognition by BT30574S-Gal/GalNAc (acc-code Q8A397) in the following order: W109, H182, and H423. Sequences coded by orange branches contain an additional W332 present in BT37964S-Gal/GalNAc (acc-code Q8A171) but absent in other sequences. For simplification the residue numbers have been omitted, except for W332. The residues are coloured as following: black means an equivalent amino acid is present; a grey and bold letter at any position means that the corresponding residue is replaced by that amino acid; a grey and italic letter at any position means that the equivalent position is replaced by any type of amino acid; a bold grey letter followed by one-letter codes in parentheses indicates that the equivalent position can be substituted by any of those amino acids; the dash at the H-equivalent position indicates that no equivalent amino acid can be deduced from the multiple alignment. When two patterns are indicated separated by a comma (that is W - H, W H -) both have been given the same colour code. Branches having the same colour have the corresponding pattern in common. Red filled circles designate sequences of S1_16 sulfatases from B. thetaiotaomicron (See Supplementary Fig. 4 for full tree).
Extended Data Fig. 5 Analysis of the activity and stability of BT16246S-Gal/GalNAc and its mutant variants.
a, Thin layer chromatography (TLC) analysis of wild-type (WT) BT16246S-Gal/GalNAc and its mutants. Asterisks are placed above lanes where activity is observed. b, High pressure anion exchange chromatography (HPAEC) of wild-type BT16246S-Gal/GalNAc WT and its mutants. The desulfated product is highlighted by a grey box. All TLC (a) HPAEC (b) reactions utilised 6 mM substrate and 5 μM enzyme, with 3 mM HEPES, 45 mM NaCl, and 5 mM CaCl2. Reactions incubated for 48 h at 37 °C. Control represents the substrate incubated in same conditions without adding enzyme. c, DSF analysis showing relative thermostability of BT16246S-Gal/GalNAc mutant proteins with respect to the WT enzyme. d, DSF analysis of the effects of alanine scanning on the ability of BT16246S-Gal/GalNAc to bind galactose, with the Tm of the protein shown above the bar. The experiments were performed using 5 μM of protein and 324 mM of galactose in 100 mM BTP pH 7.0 and 150 mM NaCl. Experiments are technical triplicates and error bars represent SEM.
Extended Data Fig. 6 Radial phylogenetic tree of S1_15 showing the conservation of the galacto- recognition triad and N-acetyl-D-galactosamine specificity features.
The tree comprises a total of 1906 sequences of which 1424 are Bacteroidetes; 172 are Planctomycetes; 119 are Kiritimatiellaeota; 57 are Proteobacteria; 53 are Verrucomicrobia. The annotations next to the colour code concern the presence or absence of conservation of the BT16246S-Gal/GalNAc (acc-code Q8A7A1) indicated residues and in this order: I100, D170, R171, H220, K461 and A462. These residues are crucial in substrate recognition and D170, R171, and H220 represent the galacto-recognition triad within S1_15 subfamily. For simplification the residue numbers have been omitted. For example, an I in black means an equivalent isoleucine is present; a grey and bold letter at any position means that the corresponding residue is replaced by that amino acid; a grey and italic letter at any position means that the equivalent position can be replaced by any type of amino acid; a bold grey letter followed by one-letter codes in parentheses indicates that the equivalent position is substituted by any of those amino acids. Branches having the same colour have the corresponding pattern in common. For clarity all labels and sequence accession codes have been omitted. Red filled circles designate sequences of S1_15 sulfatases from B. thetaiotaomicron (See Supplementary Fig. 6 for full tree).
Extended Data Fig. 7 Conservation of the N-acetyl-D-galactosamine specificity features (Y463/W464) in S1_15 enzymes within PULs targeting chondroitin sulfate.
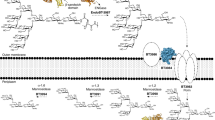
Schematic representation of PULs targeting chondroitin sulfate aligned by orthologues of BT33336S-GalNAc. Light green background shows orthologues with Y463/W464, a dark green background highlights orthologues with F463/W464, a light blue background highlights orthologues with H463/W464, and a purple background highlights orthologues with Q463/W464. The numbering used corresponds to the sequence of BT33336S-GalNAc. A red background highlights the presence of GH88 and S1_27 (an endo 4S-chrondroitin sulfatase), which is encoded by a discrete genetic region not always physically localised next to the core PUL. A black background highlights a core block observed in CS PULs containing BT33336S-GalNAc orthologues. HP (protein of unknown function), S1 (sulfatase S1 with the respective subfamily number superscript), GHXX (glycoside hydrolase with X representing the family number), PL (polysaccharide lyase), DUF (domain of unknown function), HTCS (hybrid two-component system), SusC (starch utilization system C-like), SusD (starch utilization system D-like), SGBP (surface glycan binding protein).
Extended Data Fig. 8 Analysis of the activity and stability of BT31776S-GlcNAc and mutant variants.
a, High pressure anion exchange chromatography (HPAEC) of wild-type BT31776S-GlcNAc wild-type (WT) and substituted variants. The produced product is highlighted by a grey box. b, Thin layer chromatography (TLC) analysis of WT BT31776S-GlcNAc and its mutants. Asterisks are placed above lanes where activity is observed. Both HPAEC (a) and TLC (b) reactions utilised 6 mM substrate and 5 μM enzyme, with 3 mM HEPES, 45 mM NaCl, and 5 mM CaCl2 over a 48 h period at 37 °C. Control represents the substrate incubated in same conditions without enzyme. c, DSF analysis showing relative thermostability of mutant proteins of BT31776S-GlcNAc in comparison to the WT enzyme. Experiments are technical triplicates and error bars represent SEM.
Extended Data Fig. 9 Radial phylogenetic tree of S1_11 showing the conservation of the substrate recognition triad and N-sulfate specificity features.
The tree comprises a total of 2178 sequences of which 1190 are Bacteroidetes; 233 are Verrucomicrobia;184 are Planctomycetes;143 are Ascomycota (fungi); 100 are Actinobacteria.The annotations next to the colour code concern the presence or absence of conservation of the indicated residues and in this order: R290, W273, D385, R387 and H471. These residues are required for substrate recognition by BT46566S-GlcNAc/GlcNS (acc-code Q89YS5). D385, R387, and H471 represent the recognition triad, whilst the presence of W or R at positions 273 and 290, respectively, represent N-sulfate specificity features. Residue numbers have been omitted for simplicity. For example, an R in black means an equivalent arginine is present; a grey and bold letter at this position means that the corresponding residue is replaced by that amino acid; the grey and italic R at this position means that the R-equivalent position is replaced by any type of amino acid; a bold grey R followed by one-letter codes in parentheses indicates that the R-equivalent position can be substituted by any of those amino acids; the dash at the R-equivalent position indicates that no equivalent amino acid can be deduced from the multiple alignment. Branches having the same colour have the corresponding pattern in common. Red filled diamonds designate sequences of S1_11 sulfatases from B. thetaiotaomicron. All sequences in the specific branch that contains BT46566S-GlcNAc/GlcNS are found within a conserved heparan sulfate PUL. For clarity, all labels and sequence accession codes have been omitted (See Supplementary Fig 7 for full tree).
Extended Data Fig. 10 Conservation of the N-sulfate targeting features, W273/R290, in S1_11 enzymes within PULs targeting heparan sulfate.
PULs targeting heparan sulfate (HS) aligned by orthologues of BT46566S-GlcNAc/GlcNS. Orthologues of BT46566S-GlcNAc/GlcNS with W273/R290 and W273/Q290 are highlighted with a green and blue background, respectively. A black background highlights a core block observed in HS PULs containing BT46566S-GlcNAc/GlcNS orthologues. HP (protein of unknown function), S1 (sulfatase S1 with the respective subfamily number superscript), GHXX (glycoside hydrolase with X representing the family number), PL (polysaccharide lyase), DUF (domain of unknown function), HTCS (hybrid two-component system), SusC (starch utilization system C-like), SusD (starch utilization system D-like), SGBP (surface glycan binding protein), MFS (major facilitator superfamily), ROK (repressor, ORF, kinase superfamily).
Supplementary information
Supplementary Information
Supplementary Figs. 1–8 and Tables 1–10.
Source data
Source Data Fig. 3
Raw and processed kinetic data.
Source Data Fig. 4
Raw and processed kinetic data.
Source Data Fig. 5
Raw and processed DSF data.
Source Data Fig. 6
Raw and processed DSF data.
Source Data Extended Data Fig. 2
Unmodified TLC gels.
Source Data Extended Data Fig. 3
Unmodified TLC gels and DSF data.
Source Data Extended Data Fig. 5
Unmodified TLC gels and DSF data.
Source Data Extended Data Fig. 8
Unmodified TLC gels and DSF data.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luis, A.S., Baslé, A., Byrne, D.P. et al. Sulfated glycan recognition by carbohydrate sulfatases of the human gut microbiota. Nat Chem Biol 18, 841–849 (2022). https://doi.org/10.1038/s41589-022-01039-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-022-01039-x
This article is cited by
-
Quantifying the adaptive landscape of commensal gut bacteria using high-resolution lineage tracking
Nature Communications (2024)
-
Host–microbiome orchestration of the sulfated metabolome
Nature Chemical Biology (2024)
-
An expanded transcriptome atlas for Bacteroides thetaiotaomicron reveals a small RNA that modulates tetracycline sensitivity
Nature Microbiology (2024)
-
A bacterial sulfoglycosidase highlights mucin O-glycan breakdown in the gut ecosystem
Nature Chemical Biology (2023)
-
Engineering sulfonate group donor regeneration systems to boost biosynthesis of sulfated compounds
Nature Communications (2023)