Abstract
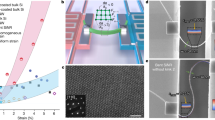
Monolayer graphene with nanometre-scale pores, atomically thin thickness and remarkable mechanical properties provides wide-ranging opportunities for applications in ion and molecular separations1, energy storage2 and electronics3. Because the performance of these applications relies heavily on the size of the nanopores, it is desirable to design and engineer with precision a suitable nanopore size with narrow size distributions. However, conventional top-down processes often yield log-normal distributions with long tails, particularly at the sub-nanometre scale4. Moreover, the size distribution and density of the nanopores are often intrinsically intercorrelated, leading to a trade-off between the two that substantially limits their applications5,6,7,8,9. Here we report a cascaded compression approach to narrowing the size distribution of nanopores with left skewness and ultrasmall tail deviation, while keeping the density of nanopores increasing at each compression cycle. The formation of nanopores is split into many small steps, in each of which the size distribution of all the existing nanopores is compressed by a combination of shrinkage and expansion and, at the same time as expansion, a new batch of nanopores is created, leading to increased nanopore density by each cycle. As a result, high-density nanopores in monolayer graphene with a left-skewed, short-tail size distribution are obtained that show ultrafast and ångström-size-tunable selective transport of ions and molecules, breaking the limitation of the conventional log-normal size distribution9,10. This method allows for independent control of several metrics of the generated nanopores, including the density, mean diameter, standard deviation and skewness of the size distribution, which will lead to the next leap in nanotechnology.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data needed to evaluate the conclusions herein are present in the article.
Code availability
The Python codes used in nanopore evolution simulation and Raman mapping are available at https://doi.org/10.5281/zenodo.10085248 (ref. 55).
References
Epsztein, R., DuChanois, R. M., Ritt, C. L., Noy, A. & Elimelech, M. Towards single-species selectivity of membranes with subnanometre pores. Nat. Nanotechnol. 15, 426–436 (2020).
Zuo, P. et al. Near-frictionless ion transport within triazine framework membranes. Nature 617, 299–305 (2023).
Bai, J. W., Zhong, X., Jiang, S., Huang, Y. & Duan, X. F. Graphene nanomesh. Nat. Nanotechnol. 5, 190–194 (2010).
O’Hern, S. C. et al. Selective ionic transport through tunable subnanometer pores in single-layer graphene membranes. Nano Lett. 14, 1234–1241 (2014).
Yang, Y. B. et al. Large-area graphene-nanomesh/carbon-nanotube hybrid membranes for ionic and molecular nanofiltration. Science 364, 1057–1062 (2019).
Huang, S. et al. Millisecond lattice gasification for high-density CO2- and O2-sieving nanopores in single-layer graphene. Sci. Adv. 7, eabf0116 (2021).
O’Hern, S. C. et al. Nanofiltration across defect-sealed nanoporous monolayer graphene. Nano Lett. 15, 3254–3260 (2015).
Hsu, K. et al. Multipulsed millisecond ozone gasification for predictable tuning of nucleation and nucleation-decoupled nanopore expansion in graphene for carbon capture. ACS Nano 15, 13230–13239 (2021).
Cheng, C., Iyengar, S. A. & Karnik, R. Molecular size-dependent subcontinuum solvent permeation and ultrafast nanofiltration across nanoporous graphene membranes. Nat. Nanotechnol. 16, 989–995 (2021).
Shen, L. et al. Highly porous nanofiber-supported monolayer graphene membranes for ultrafast organic solvent nanofiltration. Sci. Adv. 7, eabg6263 (2021).
Thanh, N. T. K., Maclean, N. & Mahiddine, S. Mechanisms of nucleation and growth of nanoparticles in solution. Chem. Rev. 114, 7610–7630 (2014).
Bergmann, R. B. & Bill, A. On the origin of logarithmic-normal distributions: an analytical derivation, and its application to nucleation and growth processes. J. Cryst. Growth 310, 3135–3138 (2008).
Wang, J. T., Park, J. H., Lu, A. Y. & Kong, J. Electrical control of chemical vapor deposition of graphene. J. Am. Chem. Soc. 144, 22925–22932 (2022).
Bohdansky, J. A universal relation for the sputtering yield of monatomic solids at normal ion incidence. Nucl. Instrum. Methods Phys. Res. 2, 587–591 (1984).
Kim, H. et al. Activation energy paths for graphene nucleation and growth on Cu. ACS Nano 6, 3614–3623 (2012).
Wang, Z. J. et al. Direct observation of graphene growth and associated copper substrate dynamics by in situ scanning electron microscopy. ACS Nano 9, 1506–1519 (2015).
Pollard, A. J. et al. Quantitative characterization of defect size in graphene using Raman spectroscopy. Appl. Phys. Lett. 105, 253107 (2014).
Lucchese, M. M. et al. Quantifying ion-induced defects and Raman relaxation length in graphene. Carbon 48, 1592–1597 (2010).
Zan, R., Ramasse, Q. M., Bangert, U. & Novoselov, K. S. Graphene reknits its holes. Nano Lett. 12, 3936–3940 (2012).
Rodenbucher, C. et al. Local surface conductivity of transition metal oxides mapped with true atomic resolution. Nanoscale 10, 11498–11505 (2018).
Wang, L. D. et al. Fundamental transport mechanisms, fabrication and potential applications of nanoporous atomically thin membranes. Nat. Nanotechnol. 12, 509–522 (2017).
Yuan, Z. et al. Predicting gas separation through graphene nanopore ensembles with realistic pore size distributions. ACS Nano 15, 1727–1740 (2021).
Herm, Z. R. et al. Separation of hexane isomers in a metal-organic framework with triangular channels. Science 340, 960–964 (2013).
Lu, Y. Q. et al. Monolayer graphene membranes for molecular separation in high-temperature harsh organic solvents. Proc. Natl Acad. Sci. USA 118, e2111360118 (2021).
Esfandiar, A. et al. Size effect in ion transport through angstrom-scale slits. Science 358, 511–513 (2017).
Huang, S. Q. et al. In situ nucleation-decoupled and site-specific incorporation of Å-scale pores in graphene via epoxidation. Adv. Mater. 34, 2206627 (2022).
Yin, Y. & Alivisatos, A. P. Colloidal nanocrystal synthesis and the organic–inorganic interface. Nature 437, 664–670 (2005).
Abe, S., Capek, R. K., De Geyter, B. & Hens, Z. Tuning the postfocused size of colloidal nanocrystals by the reaction rate: from theory to application. ACS Nano 6, 42–53 (2012).
Li, Z. Y. et al. Three-dimensional atomic-scale structure of size-selected gold nanoclusters. Nature 451, 46–48 (2008).
Yao, Y. G. et al. Carbothermal shock synthesis of high-entropy-alloy nanoparticles. Science 359, 1489–1494 (2018).
Zhang, S. C. et al. Arrays of horizontal carbon nanotubes of controlled chirality grown using designed catalysts. Nature 543, 234–238 (2017).
Li, X. S. et al. Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 324, 1312–1314 (2009).
Zhang, Z. M., Chen, S. & Liang, Y. Z. Baseline correction using adaptive iteratively reweighted penalized least squares. Analyst 135, 1138–1146 (2010).
Bendiab, N. et al. Unravelling external perturbation effects on the optical phonon response of graphene. J. Raman Spectrosc. 49, 130–145 (2018).
Jang, D., Idrobo, J. C., Laoui, T. & Karnik, R. Water and solute transport governed by tunable pore size distributions in nanoporous graphene membranes. ACS Nano 11, 10042–10052 (2017).
Chen, X. B. et al. Tunable pore size from sub-nanometer to a few nanometers in large-area graphene nanoporous atomically thin membranes. ACS Appl. Mater. Interfaces 13, 29926–29935 (2021).
Wang, S. F., Mahalingam, D., Sutisna, B. & Nunes, S. P. 2D-dual-spacing channel membranes for high performance organic solvent nanofiltration. J. Mater. Chem. A 7, 11673–11682 (2019).
Wang, C. X. et al. Aromatic porous polymer network membranes for organic solvent nanofiltration under extreme conditions. J. Mater. Chem. A 8, 15891–15899 (2020).
Cheng, X. Q. et al. Building additional passageways in polyamide membranes with hydrostable metal organic frameworks to recycle and remove organic solutes from various solvents. ACS Appl. Mater. Interfaces 9, 38877–38886 (2017).
Dai, J. et al. Fabrication and characterization of a defect-free mixed matrix membrane by facile mixing PPSU with ZIF-8 core-shell microspheres for solvent-resistant nanofiltration. J. Membrane Sci. 589, 117261 (2019).
Ong, C. et al. Green synthesis of thin-film composite membranes for organic solvent nanofiltration. ACS Sustain. Chem. Eng. 8, 11541–11548 (2020).
Wang, A. Q. et al. Heterostructured MoS2 quantum dot/GO lamellar membrane with improved transport efficiency for organic solvents inspired by the Namib Desert beetle. J. Membrane Sci. 650, 120402 (2022).
Liang, B. et al. Microporous membranes comprising conjugated polymers with rigid backbones enable ultrafast organic-solvent nanofiltration. Nat. Chem. 10, 961–967 (2018).
Echaide-Gorriz, C., Sorribas, S., Tellez, C. & Coronas, J. MOF nanoparticles of MIL-68(Al), MIL-101(Cr) and ZIF-11 for thin film nanocomposite organic solvent nanofiltration membranes. RSC Adv. 6, 90417–90426 (2016).
Xu, Y. C., Tang, Y. P., Liu, L. F., Guo, Z. H. & Shao, L. Nanocomposite organic solvent nanofiltration membranes by a highly-efficient mussel-inspired co-deposition strategy. J. Membrane Sci. 526, 32–42 (2017).
Xu, S. J., Shen, Q., Chen, G. E. & Xu, Z. L. Novel β-CD@ZIF-8 nanoparticles-doped poly(m-phenylene isophthalamide) (PMIA) thin-film nanocomposite (TFN) membrane for organic solvent nanofiltration (OSN). ACS Omega 3, 11770–11787 (2018).
Darvishmanesh, S. et al. Novel polyphenylsulfone membrane for potential use in solvent nanofiltration. J. Membrane Sci. 379, 60–68 (2011).
Jimenez-Solomon, M. F., Song, Q. L., Jelfs, K. E., Munoz-Ibanez, M. & Livingston, A. G. Polymer nanofilms with enhanced microporosity by interfacial polymerization. Nat. Mater. 15, 760–767 (2016).
Kandambeth, S. et al. Selective molecular sieving in self-standing porous covalent-organic-framework membranes. Adv. Mater. 29, 1603945 (2017).
Mahalingam, D. K., Wang, S. F. & Nunes, S. P. Stable graphene oxide cross-linked membranes for organic solvent nanofiltration. Ind. Eng. Chem. Res. 58, 23106–23113 (2019).
Hai, Y. Y. et al. Thin film composite nanofiltration membrane prepared by the interfacial polymerization of 1,2,4,5-benzene tetracarbonyl chloride on the mixed amines cross-linked poly(ether imide) support. J. Membrane Sci. 520, 19–28 (2016).
Navarro, M. et al. Thin-film nanocomposite membrane with the minimum amount of MOF by the Langmuir–Schaefer technique for nanofiltration. ACS Appl. Mater. Interfaces 10, 1278–1287 (2018).
Yang, Q. et al. Ultrathin graphene-based membrane with precise molecular sieving and ultrafast solvent permeation. Nat. Mater. 16, 1198–1202 (2017).
Luo, D. et al. The wet-oxidation of a Cu(111) foil coated by single crystal graphene. Adv. Mater. 33, e2102697 (2021).
Wang, J. et al. Code source files for the paper entitled “Cascaded compression of size distribution of nanopores in monolayer graphene”. Zenodo https://doi.org/10.5281/zenodo.10085248 (2023).
Acknowledgements
J.W., J.-H.P. and J.K. acknowledge the financial support from the Air Force Office of Scientific Research (AFOSR) Multi-University Research Initiative FA9550-22-1-0166 and the US Army Research Office (ARO) under grant no. W911NF-18-1-0431. C.C. is the recipient and acknowledges the financial support from the Australian Research Council Australian Future Fellowship Award (FT210100364) funded by the Australian Government. A.-Y.L., J.-H.P. and J.K. acknowledge the US ARO through the Institute for Soldier Nanotechnologies at MIT, under cooperative agreement no. W911NF-18-2-0048. X.Z., T.Z. and J.K. acknowledge the support by the US Department of Energy (DOE), Office of Science, Basic Energy Sciences (BES) under award DE-SC0020042, which provided STEM and other characterizations for the samples. Y.H., G.G. and B.S. acknowledge the support from Welch Foundation (C-2065-20210327). CAFM imaging was performed in part in the MIT.nano Characterization Facilities on the Cypher VRS enabled by the DURIP award (N000142012203). STEM imaging was conducted as part of a user project at the Center for Nanophase Materials Sciences (CNMS), which is a US DOE, Office of Science User Facility at Oak Ridge National Laboratory. The views and conclusions contained herein are those of the authors and should not be interpreted as necessarily representing the official policies or endorsements of the US Government.
Author information
Authors and Affiliations
Contributions
J.K. supervised the project. J.W. and J.K. conceived the experiments. J.W. conducted the theoretical analysis and simulations. J.W. established the LPCVD system with electrodes and prepared the nanoporous graphene film. C.C. and R.K. conceived and designed the nanofiltration and diffusion experiments and contributed to the design of the project. C.C. performed the nanofiltration and diffusion experiments. J.W. performed the Raman and CAFM characterization. X.Z., J.C.I., J.W., G.G., B.S. and Y.H. performed the STEM and HRTEM characterization. A.-Y.L. and J.W. wrote the Raman fitting program. B.G.S. and S.J.J. performed the STM characterization. J.-H.P., T.Z., H.W., X.J., L.J., L.-J.L. and R.K. participated in the characterization and discussion. J.W., C.C. and J.K. wrote the manuscript; all authors read and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.W., C.C. and J.K. are co-inventors on a patent application (provisional filling number 63/502,064) related to the research presented in this paper.
Peer review
Peer review information
Nature thanks Ling Liu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 In situ deposition of Cu particles on the graphite electrode for in situ sputter.
a, Schematic of electrode reactions and enhanced copper evaporation in the CVD chamber. The surface electrochemical reaction changes Cu to Cu(OH)2, which is very easy to evaporate. Then Cu(OH)2 particles would be reduced to Cu on the graphite electrode in the atmosphere of H2. b, A scanning electron microscopy image of the graphite electrode surface after Cu particle deposition. c, The corresponding energy-dispersive spectroscopy mapping of Cu of the graphite electrode in b. The red dots indicate the deposited Cu particles. Scale bars, 5 μm. d–f, Illustration of whether sputter-etching happens or not with different types of electrode: graphite (d), graphite deposited with Cu particles (e) and flat Cu covered by graphene film (f). g–i, The corresponding Raman spectrum of the graphene film after applying electric field with the anode materials shown in d–f. It proves that the in situ sputter results in the presence of exposed Cu particles at the anode. With more area of exposed Cu, the sputter effect would be stronger.
Extended Data Fig. 2 Optical images of the graphene film after in situ sputtering.
a, Optical image of the as-prepared nanoporous graphene on copper substrate. b, Optical image of the sample after being immersed in 80 °C water for 5 min. The contrast shows the wet-oxidation of different copper domains by water54, suggesting the presence of nanopores in the graphene film. c, Optical image of the sample after being oxidized in 150 °C ambient air for 2 min. The colour change indicates Cu oxidation owing to nanopores in graphene film. The arrow indicates a bilayer island that is not oxygen-permeable. Scale bars, 100 μm. d, Optical image of the whole sample after oxidation, in which only the indicated region was overlapped with the graphite electrode and sputtered.
Extended Data Fig. 3 Nanopore creation through in situ e-beam irradiation and STEM images of defects created by cascaded compression but with extended shrinking time in graphene film.
a, Random nucleation and linear expansion of two nanopores under in situ e-beam irradiation. The randomness of the size distribution comes from the fact that the nanopores are created at different starting times (that is, nucleation time), therefore, by the end of expansion time, the nanopores have different sizes. b–g, The STEM images of the in situ creation and expansion of two nanopores at various times. Scale bar, 1 nm. Voltage: 100 kV, current: 38 pA, temperature: 700 °C. h–k, STEM images of defects in graphene obtained by using extended regrowth time. h, A large topological defect with threefold symmetry. i, A divacancy defect. j, A linear 5–7 defect. k, A single-vacancy defect. The dotted circles indicate the area of the structural disorder. Scale bar, 0.5 nm.
Extended Data Fig. 4 Atomic characterization of nanopore density and size.
a, An STM image of 80 × 80 nm2. Corrugation with terrace width of about 20 nm was induced by the Cu substrate. Bright and dark dot features were observed in a large density compared with the conventional CVD graphene on Cu. b, Close-up STM image with atomic resolution for the area of the rectangle in a. Arrows indicated nanopore structures. c, Close-up STM image with atomic resolution for the area of the rectangle in b. The size of the nanopore is about 1 nm. The honeycomb structure of graphene is indicated with an inset of honeycomb cells. Nanopore density of 3.875 × 1012 cm−2 was counted in STM images, based on the characterization of the atomic resolution. d–g, CAFM images of pristine graphene and nanoporous graphene samples with different nanopore diameters. The acquirements of the nanopore area are indicated by the white outlines. The diameter of the nanopore is calculated by using \(d=2\sqrt{A/\pi }\), in which A is the area of the nanopore. h,i, The experimental diameter distribution of samples #822 and #835, respectively. j,k, The simulated diameter distribution of samples #822 and #835, respectively.
Extended Data Fig. 5 Nanofiltration experimental setup and results analysis.
a, Methanol solutions of Rose bengal was used as feed to measure membrane rejection. The feed volume was 35 ml and had a solute concentration of 0.01 mM. To maintain an approximately constant solute concentration in the feed during each measurement, the total permeate that flowed out of the pressure cell was controlled to be less than 3 ml. Vigorous stirring was maintained on the feed side to minimize concentration-polarization effects. The stirrer was suspended above the membrane using a built-in mechanism inside the HP4750 High-pressure Stirred Cell (Sterlitech, Inc.). b, The benchmark of relative tail deviation and density. c, Rejection versus permeance plot of various membrane materials for organic solvent nanofiltration of Rose bengal in methanol. The innate permeance of nanoporous graphene is calculated using the measured membrane permeance normalized by the support porosity and subtracting the support flow resistance (see Extended Data Table 1).
Extended Data Fig. 6 The uniformity comparison of the size and density of the nanoporous graphene grown on copper foil.
a,b, The mapping of D to G ratio and 2D to G ratio of sample #809 after transfer to SiO2/Si, respectively. The dashed blue line in a shows a copper grain boundary before the transfer. c,d, The mapping of D to G ratio and 2D to G ratio of sample #817, respectively. The preparation condition of sample #817 involves adding an extra 1-min shrinkage after the same conditions of sample #809. As a result, the D to G ratio of #sample 817 reflects the density of the nanopores of sample #809, and the D to G ratio difference between samples #809 and #817 indicates the variation of the nanopore size of sample #809.
Extended Data Fig. 7 Diametric evolution towards the designed asymptotic value for cascaded compression based on our modelling.
a,b, Nanopore density versus diameter distribution after different cycles of compression. The starting distributions (0 cycles) (\({d}_{{\rm{peak}}}=0.2\,{\rm{nm}},\,\widetilde{\sigma }=0.4\)) in our experiments are also log-normal distributions and are the same for a and b. After several compression cycles, such a log-normal distribution becomes a left-skewed distribution with total density increasing. The designed asymptotic values (d∞) are 0.8 nm and 0.6 nm, respectively, for a and b by tuning the compression factor from 0.750 to 0.667. c,d, The corresponding plots of nanopore density versus diameter of a and b in logarithmic coordinates. The high nanopore density with a slope of 0.1 or 0.05 nm dec−1 indicates high selectivity and permeance in these nanoporous graphene membranes.
Extended Data Fig. 8 Simulation of the cases beyond the ideal linear model.
a, The evolution of density, mean diameter and RSD based on the conditions of sample #825 and extra nine times higher nucleation rate. b,c, The corresponding final spatial (b) and diametric (c) distributions of a. d, The evolution of density, mean diameter and RSD based on the conditions of sample #825 and extra 90 cycles of compression. e,f, The corresponding final spatial (e) and diametric (f) distributions of d. Scale bars in b and e, 5 nm. With much higher density, the fusion is notable, but the diameter distribution is still very narrow. g, The evolution of density, mean diameter and RSD based on a nonlinear diametric transformation as shown in equations (29) and (30). h,i, The corresponding final spatial (h) and diametric (i) distributions of g. Red and black dots in b, e and h indicate the created nanopores and 5–7 defects (or single/double vacancies), respectively. Scale bars, 10 nm.
Extended Data Fig. 9 Numeric calculation of the nanopore shrinkage.
a,b, The linear shrinkage dynamics of the nanopores when the adsorption rate of carbon source on the copper surface is much faster than the rate of carbon atoms attaching to the graphene lattice. b, Corresponding logarithmic scale of a. c,d, The exponential compression of the nanopores when the adsorption rate of carbon source on the copper surface is much slower. d, Corresponding logarithmic scale of c.
Extended Data Fig. 10 Raman mappings and scattering plots of the nanoporous graphene film.
a, Processing stages of the samples with different shrinkage time. The fitted shrinkage rate is 0.078 s−1. b–d, The Raman mapping and scattering plot of the sample after ten cycles of cascaded compression. e–g, The Raman mapping and scattering plot of the sample with extra 30 s shrinkage. The samples are transferred onto SiO2/Si substrate.
Supplementary information
Supplementary Information
This file includes Supplementary Notes 1–4, Supplementary Figs. 1–4 and Supplementary Tables 1 and 2.
Supplementary Video 1
The simulated time-resolved evolutions of the density, mean diameter and RSD of sample no. 825.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, J., Cheng, C., Zheng, X. et al. Cascaded compression of size distribution of nanopores in monolayer graphene. Nature 623, 956–963 (2023). https://doi.org/10.1038/s41586-023-06689-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-023-06689-y
This article is cited by
-
Self-similarity study based on the particle sizes of coal-series diatomite
Scientific Reports (2024)
-
Regulation mechanism of epsilon-negative monolayer graphene/CaCu3Ti4O12 metacomposites for boosting electromagnetic shielding
Advanced Composites and Hybrid Materials (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.