Abstract
Hyperferritinaemia is a common laboratory finding that is often associated with metabolic dysfunction and fatty liver. Metabolic hyperferritinaemia reflects alterations in iron metabolism that facilitate iron accumulation in the body and is associated with an increased risk of cardiometabolic and liver diseases. Genetic variants that modulate iron homeostasis and tissue levels of iron are the main determinants of serum levels of ferritin in individuals with metabolic dysfunction, raising the hypothesis that iron accumulation might be implicated in the pathogenesis of insulin resistance and the related organ damage. However, validated criteria for the non-invasive diagnosis of metabolic hyperferritinaemia and the staging of iron overload are still lacking, and there is no clear evidence of a benefit for iron depletion therapy. Here, we provide an overview of the literature on the relationship between hyperferritinaemia and iron accumulation in individuals with metabolic dysfunction, and on the associated clinical outcomes. We propose an updated definition and a provisional staging system for metabolic hyperferritinaemia, which has been agreed on by a multidisciplinary global panel of expert researchers. The goal is to foster studies into the epidemiology, genetics, pathophysiology, clinical relevance and treatment of metabolic hyperferritinaemia, for which we provide suggestions on the main unmet needs, optimal design and clinically relevant outcomes.
Similar content being viewed by others
Introduction
Ferritin is the main intracellular iron storage protein and is a biomarker of iron stores and inflammation. An increased serum concentration of ferritin is a common biochemical finding, with a prevalence of 5.9–19.0% in apparently healthy individuals, depending on ethnicity1. As ferritin is the main protein responsible for iron storage in the body, it represents a valuable biomarker of total body iron in iron overload conditions such as haemochromatosis, where it is used to guide iron-reduction therapy1. Ferritin is also an acute phase reactant, and levels of ferritin are often increased in chronic inflammatory conditions, such as in individuals with insulin resistance and metabolic dysfunction (that is, the presence of metabolic alterations, including type 2 diabetes mellitus and/or obesity or at least two features typically associated with insulin resistance (increased visceral adiposity, atherogenic dyslipidaemia, arterial hypertension or hyperinsulinaemia))2. Serum levels of ferritin have been associated with the amount of hepatic lipid accumulation, the severity of insulin resistance and features of metabolic dysfunction, even in the absence of inflammation3,4,5,6,7,8,9,10,11,12,13. Insulin resistance and metabolic dysfunction have also been linked to an iron overload syndrome featuring hyperferritinaemia and accumulation of iron predominantly in non-parenchymal liver cells (Kupffer cells); this syndrome shares epidemiological, genetic and biochemical features with hyperferritinaemia related to metabolic dysfunction14. This condition, featuring hyperferritinaemia related to metabolic dysfunction, was initially named insulin resistance-associated hepatic iron overload syndrome and was subsequently defined as dysmetabolic iron overload syndrome (DIOS) or dysmetabolic hyperferritinaemia14,15,16,17. Unlike patients with haemochromatosis, patients with DIOS have reasonably preserved production of hepcidin, the iron regulatory hormone that is secreted by the liver in response to iron and inflammation14,15,16. The coexistence of various definitions (with some, for example, DIOS, requiring the histological demonstration of non-parenchymal tissue iron overload and others not, and using different criteria to define metabolic dysfunction) has so far posed severe limitations on the comparability of the results and conclusions of studies published in this field.
Individuals with hyperferritinaemia related to metabolic dysfunction, that here we name ‘metabolic hyperferritinaemia’ (MHF), show a variable degree of iron stores in the body, which are partly correlated with serum levels of ferritin14. In individuals with MHF, iron stores in the body range from normal to a moderate iron overload, usually below levels reached in haemochromatosis14. Indeed, elevated serum levels of ferritin can develop in individuals with metabolic alterations in the presence of risk factors for iron accumulation (for example, genetic risk variants, male sex and older age) and are associated with normal transferrin saturation14. Therefore, the spectrum of iron metabolism alterations in individuals with MHF ranges from minor biochemical changes to tissue iron accumulation, which might lead to iron-related organ damage14.
Previous categorization attempts focused on the most severe forms of MHF, such as DIOS, in which hyperferritinaemia is also associated with tissue iron overload, and were based on heterogeneous and unclear criteria to define iron accumulation as related to metabolic dysfunction. Historically, the diagnosis DIOS was based on histological demonstration of iron accumulation in Kupffer cells14,15,16. Liver histology has been pivotal to support the diagnosis of this condition in patients with clinically relevant iron overload. However, given its invasive nature, liver biopsy has limited the assessment of iron overload in clinical practice and in research studies. Progress in imaging techniques now enables the accurate non-invasive estimation of tissue concentrations of iron by MRI (Supplementary Box 1). MRI can approximate the cellular predominance of iron accumulation by assessing levels of iron in the spleen and liver. Accumulating evidence suggests that the hepatic and total body content of iron in patients with metabolic dysfunction correlates with serum levels of ferritin6,7,18,19,20,21,22. Importantly, most, but not all, studies23 have shown that in patients with marked hyperferritinaemia despite the absence of acute inflammation, a considerable increase in hepatic and body iron stores is typically seen19,24. However, the amount of iron accumulation rarely reaches thresholds that define haemochromatosis and other primary iron disorders, in which excess iron is sufficient to drive progressive organ damage6,7,18,19,20,21. The pattern of iron overload is also distinct in DIOS and haemochromatosis, with iron overload predominantly in macrophages being typical of DIOS25.
Methodology
Given the high prevalence of hyperferritinaemia in clinical practice, combined with a growing understanding of the underlying pathophysiology and clinical implications, an updated definition and grading system is warranted. The overall goal of this Consensus Statement was therefore to provide a proposal for a more accurate diagnosis and classification of MHF, to be validated by prospective studies, to provide suggestions on the design of these studies and to highlight some of the main unmet research needs in the field. Owing to the current absence of robust evidence to support the recommendation to screen for and diagnose MHF and then to treat this condition with a specific approach (such as iron depletion), the current Consensus Statement is mainly directed at clinicians who work in tertiary referral centres and clinical and basic researchers. The updated MHF definition will enable larger collaborative studies on the clinical implications, pathophysiology and therapy of this condition, although it will still require prospective validation and further refinement. The long-term goal is the improvement of clinical management of patients with MHF. On the basis of current evidence, we also provide expert recommendations for clinicians on the diagnosis, management, follow-up and treatment of this condition (presented in the Supplementary material).
Consensus was reached by a multidisciplinary global panel of expert researchers with an interest in MHF from five continents and 14 countries working in the fields of iron metabolism, clinical endocrinology, hepatology, radiology and haematology. The panel was selected to include the main research groups active in the field and to cover all of the main disciplines involved and the various geographical areas. We considered corresponding authors of papers related to MHF and representatives of the main regional scientific societies. The initial 15 statements (Supplementary Table 1) were drafted by the two corresponding authors after an initial review of the literature; the corresponding authors drafted the results of this literature review, developed written proposals and wrote the first manuscript draft. Statements relevant for the clinicians were marked as ‘clinically relevant’ (CR). Written feedback was received from all the authors, and this feedback was discussed through personal meetings, online teleconferences and e-mails during the coronavirus 2019 (COVID-19) pandemic.
Consensus on the 15 recommendations was subsequently tested formally by means of an initial Delphi procedure conducted by online questionnaires on 1 June 2022. The data collection period ranged for 4 weeks. The round 1 survey contained five domains with five-point Likert-type categories for respondents to indicate their level of agreement with the statements (that is, ‘Agree’, ‘Somewhat agree’, ‘Neither agree nor disagree’, ‘Somewhat disagree’ or ‘Disagree’). In the first round, respondents who agreed or somewhat agreed with a statement could provide comments or suggest edits, while those who disagreed or somewhat disagreed needed to explain why. Results are reported in detail in Supplementary Table 1. Agreement by ≥75% of participants was required to enable a recommendation, which was achieved for all proposals during the first Delphi round. However, further discussion was undertaken by e-mail to report the results of round 1 and the comments and disagreements in round 1, which were taken into consideration in the final version of the manuscript. For the Delphi process, we assigned each statement and recommendation a grade to indicate the level of agreement using the grading system used in other Delphi studies, in which ‘U’ denotes unanimous (100%) agreement; ‘A’, 90–99% agreement; ‘B’, 78–89% agreement; and ‘C’, 67–77% agreement26.
The study was designed, and the recommendations finalized by L.V. and E.C.. The first manuscript draft was written by L.V., E.C., D.Mc., B.H., A. Pagani, D.P., L.S. and F.V., and was reviewed for important intellectual content by L.A.A., H.H., S.A., E.B., E.A.T., J.-M.F.-R., J.D.R., P.P. and H.Z.. All authors developed the recommendations, reviewed the first manuscript draft, participated in the Delphi consensus process, reviewed and approved the final manuscript.
Recommendations
Epidemiology and risk factors
R1
Insulin resistance and features of the metabolic dysfunction are associated with specific alterations of iron metabolism regulation, which are epidemiologically linked with organ damage and clinical outcomes (U).
Not all patients with metabolic dysfunction or fatty liver present with increased serum levels of ferritin, suggesting that those with MHF constitute a subset with distinct risk factors, pathophysiology and clinical outcomes, possibly deserving specific management. Box 1 outlines the main genetic and acquired conditions associated with hyperferritinaemia and accumulation of iron in the body in individuals with metabolic dysfunction, fatty liver and insulin resistance.
Serum levels of ferritin were first described to be linked to insulin resistance in 1998 (ref. 27). The severity of insulin resistance3,4,5,6,7,8,9,10,11,28 and the severity of fatty liver disease, but not circulating biomarkers of inflammation, have been reported as determinants of MHF6,7,18,19,20,21,29,30. By contrast, genetic variants associated with iron metabolism, rather than those that affect lipid handling in the liver, were associated with increased serum levels of ferritin and MHF in patients with fatty liver31. This observation is in line with findings that indicate that serum levels of ferritin reflect hepatic and body iron stores more closely than the severity of liver lipid accumulation in individuals with metabolic dysfunction6,7,18,19,20,21. Furthermore, male sex, older age and moderate alcohol intake contribute to iron accumulation in fatty liver disease. This evidence is based on liver histology and MRI evaluations of cohorts of patients with fatty liver or multiple metabolic alterations6,7,18,19,20,21. Therefore, in patients with insulin resistance but without acute inflammatory conditions, severe alcohol abuse or poorly controlled diabetes mellitus, serum levels of ferritin are mainly determined by dysregulation of iron metabolism and next by the severity of insulin resistance.
In a large prospective cohort of middle-age healthy men conducted in South Korea, elevated serum levels of ferritin were independently associated with development of the metabolic syndrome during the 5-year follow-up period32. Furthermore, in a large prospective study on European cases of incident type 2 diabetes mellitus, increased serum levels of ferritin were associated with an increased risk of type 2 diabetes mellitus, even among individuals with no overt inflammation, liver disease, high alcohol consumption or obesity33.
Data from the past 25 years in European populations suggest that serum concentrations of ferritin are associated with insulin resistance and metabolic dysfunction, even in individuals without fatty liver or with liver levels of iron that are within the reference range3,4,5,6,7,8,9,10,11,27. However, in individuals with high serum concentrations of ferritin, the presence of fatty liver might indicate an increased risk of insulin resistance and the metabolic syndrome, whereas the presence of hepatic iron overload might indicate an increased risk of hyperglycaemia34. These data are in line with genetic evidence from the past decade that body stores of iron have a causal role in determining liver disease and the development of type 2 diabetes mellitus in the general population35,36,37.
By means of Mendelian randomization as a robust epidemiological approach to analyse the causal estimation of fatty liver disease associated with metabolic dysfunction (also known as metabolic dysfunction-associated fatty liver disease (MAFLD)) as an outcome using genetic variants, a study published in 2022 that included European individuals showed that the genetically predicted increase in liver levels of iron was associated with an increased risk of fatty liver disease. Although they need confirmation, these data support a causal association between deposition levels of iron in the liver and metabolic dysfunction38.
Pathogenesis
R2
The pathophysiology of this alteration of iron metabolism regulation seems to be triggered by lipotoxicity in the presence of permissive environmental and genetic determinants, but additional studies are required to clarify the contribution of subclinical inflammation and the underlying mechanisms and implications (U).
Mechanism of iron accumulation
The pathogenesis of MHF is multifaceted, relating to the effect of common genetic variants on iron homeostasis, as well as hepatic, intestinal and adipose tissue factors. Systemic iron homeostasis is maintained through the hepcidin–ferroportin axis. Hepcidin is a liver peptide hormone, and the expression of hepcidin is upregulated by iron and inflammation. Hepcidin controls iron influx into the bloodstream from duodenal enterocytes and macrophages by binding, occluding and inducing the degradation of the iron exporter ferroportin. As a consequence of the increased hepcidin secretion, iron accumulates in cells expressing ferroportin, mainly macrophages and, to a lesser extent, hepatocytes. Genetic iron overload disorders (such as haemochromatosis) are most frequently caused by reduced hepcidin synthesis or impaired iron export39. In patients with MHF, hepcidin release in response to iron stores and the ability of hepcidin to downregulate intestinal iron absorption are generally preserved40. However, a subtle alteration in iron fluxes has been reported, whereby excess fatty acids have been linked to a reduced ability of hepcidin to limit intestinal iron absorption, while simultaneously increasing hepatic iron uptake and tissue deposition40,41,42,43.
Interestingly, patients with type 2 diabetes mellitus without clinical signs of inflammation show MHF with reduced hepcidin levels and increased systemic levels of iron44. Furthermore, the development of type 2 diabetes mellitus might be associated with impaired hepcidin release induced by hyperinsulinaemia45. In patients with metabolic dysfunction and hyperferritinaemia, body stores of iron have been associated with high dietary iron intake46 and a distinct microbiome profile47. In patients with MHF or obesity, development of mild iron accumulation in hepatocytes can lead to hepcidin upregulation and restrains further iron absorption48,49,50. Preserved regulation of hepcidin would favour the preferential retention of iron in liver macrophages (Kupffer cells), where ferroportin is highly expressed31,51,52. Within this context, the presence of inherited variants associated with impaired hepcidin would favour the development of more severe iron accumulation and DIOS (Table 1).
Reduced expression of the ferroxidase ceruloplasmin, which cooperates with ferroportin for iron export in several cell lineages, including hepatocytes, might also favour iron accumulation in the liver. Furthermore, low-frequency inherited genetic variants of the CP gene (which encodes ceruloplasmin) that determine a mild functional impairment of ceruloplasmin were associated with MHF and more severe liver disease in patients with fatty liver disease31.
Role of excess iron in tissue damage and insulin resistance
Although the role of iron overload in determining liver disease (liver fibrosis progression and hepatocellular carcinoma) and pancreatic β-cell failure is established in haemochromatosis39, the potential effect of dysregulation of iron metabolism on the pathogenesis of dyslipidaemia and insulin resistance is less clear. A detailed discussion of the complex relationship between iron accumulation, activation of the BMP–SMAD signalling pathway, modulation of lipid metabolism and development of liver disease is presented in Supplementary Box 2. This process might involve the facilitation of ferroptosis as well as of other forms of cell death in hepatocytes and other liver cells53. Accumulation of iron in macrophages in the liver has been associated with more severe liver damage in patients with fatty liver disease20,54, compared with patients without iron accumulation in these cells. By catalysing the formation of reactive oxygen species (ROS), excess iron favours subclinical inflammation, which contributes to insulin resistance by directly downregulating insulin receptor expression and signalling and by worsening of alterations of glucose and lipid metabolism10,44,55,56, fibrogenesis and carcinogenesis14. Unlike in patients with haemochromatosis, whose macrophages are typically iron depleted because of hepcidin deficiency, iron accumulation in macrophages and hepatic stellate cells has been associated with a pro-inflammatory and pro-fibrotic response57,58. Moreover, excess fatty acids and lipotoxicity predispose to inflammation and type 2 diabetes mellitus by inducing macrophage iron accumulation via induction of FTH1, which encodes the ferritin H subunit (which has iron oxidase activity)59.
In addition to insulin resistance and dyslipidaemia, ferritin levels and levels of iron stores in the liver have also been associated with the build-up of iron in adipose tissue, which leads to insulin resistance, impaired adiponectin secretion and altered endocrine function in mouse models and in patients55,56,60, as well as impaired regulation of amino acid and phospholipid metabolism in patients with fatty liver61,62. Indeed, ferroportin is also expressed in adipocytes, where it can be targeted by hepcidin to exert its endocrine function55. Insulin resistance and the expansion of adipose tissue modulate the expression of iron-related genes, such as transferrin receptor 1 (TFRC) and FTH1 in adipose tissue in patients with metabolic alterations63,64. Increased iron levels in adipocytes, such as in conditions of high levels of hepcidin or adipose tissue-specific FPN deletion in experimental models, negatively regulate expression of the genes that encode leptin and adiponectin55,65. This mechanism might contribute to the development of obesity and insulin resistance. The crucial role of adipose levels of iron in the development of insulin resistance was supported by evidence demonstrating that adipose-specific genetic variants that decreased expression of the iron export protein ferroportin led to insulin resistance and obesity66. Although the exact mechanism remains to be elucidated, paracrine trafficking of iron between adipocytes and macrophages in adipose tissue is altered by high-fat diet feeding in mice, which contributes to changes in macrophage polarization57,67. Iron accumulation might also be directly involved in facilitating vascular damage, including by epigenetic mechanisms (Supplementary Box 3).
In line with the concept of multi-level crosstalk between the adipose tissue and the gut in controlling caloric and nutrient influx, low iron levels in adipose tissue, as a result of constitutive or inducible adipocyte-specific transferrin receptor 1 (TFRC) deficiency or adeno-associated virus-mediated overexpression of the iron exporter ferroportin in mature adipocytes, protects mice from high-fat-diet-induced metabolic disorders68. In such preclinical models, reduced cellular levels of iron in adipocytes have been associated with the restriction of lipid absorption from enterocytes following high-fat-diet feeding via as yet unidentified signals and mechanisms68. However, it should be noted that transferrin receptor 1 is required for browning of adipocytes, thermogenesis and protection against insulin resistance, through the regulation of intracellular iron metabolism and mitochondrial function69.
Within cells, ferritin is involved in iron storage; therefore, increased ferritin synthesis has a potentially protective function by limiting the production of free radicals in redox biology and inducing the expression of anti-inflammatory cytokines in immune responses70. By contrast, experimental work in mice and cellular models suggests that ferritin might also act as pro-inflammatory molecule (reviewed in70). Consistent with this finding, in a retrospective study in patients with haemochromatosis, serum levels of ferritin were a better predictor of fibrosis stage than iron content in the liver, sex, steatosis or alcohol intake, which suggests that ferritin might be involved in fibrosis instead of simply acting as a passive indicator of iron storage71. Whether ferritin is a bystander or a mediator of pathological processes in patients with metabolic dysfunction deserves further investigation.
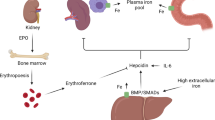
A working model of the mechanism underlying altered iron metabolism that facilitates iron accumulation in the tissues of patients with MHF is presented in Fig. 1.
Hepatic lipid accumulation (steatosis) and alterations of lipid metabolism in the liver are conditions associated with systemic insulin resistance, and they lead to lipotoxicity and local inflammation, inducing the synthesis of ferritin, a molecule with antioxidant activity. Within this context, excess levels of lipids and iron in the diet lead to increased circulating levels of iron (Fe), especially in male individuals with a permissive genetic background. Owing to the presence of subclinical inflammation downregulating ferroportin 1, iron is accumulated not only in hepatocytes, but also in Kupffer cells and hepatic stellate cells, triggering hepatocellular damage, ferroptosis, inflammation and fibrogenesis. This process triggers progressive liver disease, but spillover of iron from the liver might also worsen insulin resistance in adipose tissue and impair the secretion of appetite-controlling peptides, thereby facilitating the progression to type 2 diabetes mellitus and its complications. The figure represents the main pathophysiological pathways leading to metabolic hyperferritinaemia in patients with metabolic dysfunction (not all the extrahepatic and intrahepatic interactions between lipid, glucose and iron metabolism are depicted).
Definition and diagnosis of MHF
R3
We propose serum levels of ferritin as the most accurate and available biomarker to non-invasively capture and grade the presence of the aforementioned iron metabolism alteration (namely MHF), associated with glucose and lipid metabolism dysregulation and with hepatic lipid accumulation (A) (CR).
There are four reasons why we suggest that the diagnosis of MHF and grading of the severity of MHF should be established according to the serum concentrations of ferritin. First, the serum level of ferritin is an almost universally available and inexpensive biomarker, compared with MRI-based tissue iron quantification. Second, there is well-established standardization of measurements for serum levels of ferritin72. Third, evidence shows that in the absence of acute inflammation, serum levels of ferritin reflect the severity of iron stores or iron overload6,7,18,19,20,21. Fourth, serum levels of ferritin can also reflect alterations in iron metabolism related to lipotoxicity and subclinical chronic inflammation that are correlated with tissue damage independently of iron stores14. We named the condition MHF without referring to alterations of iron metabolism and accumulation to reflect this choice.
R4
We propose to define this condition as MHF, and to grade its severity according to serum levels of ferritin thresholds (grades 1–3), which will need prospective validation and optimization. When possible, serum levels of ferritin should be evaluated after at least 3 months of lifestyle changes (U) (CR).
R5
As criteria for metabolic dysfunction, we propose those matching the definition of MAFLD, with the following modifications: inclusion of the presence of fatty liver among the criteria, exclusion of those with biochemical signs of overt inflammation and of heavy alcohol intake (A) (CR).
The proposed diagnostic criteria for MHF are reported in Boxes 2 and 3. To provide a consistent and comprehensive conceptual framework, the definition of metabolic dysfunction was modelled after that used to diagnose MAFLD73. We propose to grade the severity of iron metabolism alterations from 1 to 3 according to the serum concentration of ferritin and, when available, hepatic concentration of iron (Boxes 2 and 3; note that haemochromatosis, persistently increased transferrin saturation and some cancers exclude the diagnosis of MHF). We suggest re-evaluation of serum levels of ferritin after at least 3 months of lifestyle counselling, including reduction of alcohol intake and optimization of pharmacological therapy, when this approach is feasible in clinical practice or research studies. This suggestion is justified by the fact that serum levels of ferritin are partially responsive to lifestyle modifications74,75, which highlights the concept that the alterations in iron metabolism seen in MHF might reflect lipotoxicity as well as iron stores. Lifestyle counselling should be personalized and usually includes advice on weight loss, dietary composition, cessation of alcohol intake and increasing physical activity. MHF grading needs to be validated for the ability to stratify the risk of clinical events; this validation will help to standardize outcome reports in epidemiological studies and improve stratification in therapeutic studies.
R6
Given the initial evidence that in patients with stable MHF, serum levels of ferritin might be associated with iron accumulation in tissue, we suggest the non-invasive estimation of iron concentration in the liver by MRI in clinical studies. This approach can be considered, when available, in pathophysiological studies and in clinical practice in patients with high serum levels of ferritin (grade 2, but in particular, grade 3) and/or additional clinical risk factors for iron overload. When available, iron concentration in tissues should have priority over serum levels of ferritin for grading MHF (A) (CR).
After staging of fibrosis by non-invasive approaches, as recommended by clinical practice guidelines for fatty liver disease76, quantification of hepatic levels of iron by MRI, if available, can be used to stage MHF as a complementary approach to serum levels of ferritin to better characterize iron metabolism and tissue iron stores in clinical research studies. We also suggest the non-invasive evaluation of tissue levels of iron for patients with grade 2, but especially grade 3, MHF (based on serum levels of ferritin) in clinical practice. Grade 3 MHF corresponds to DIOS, and requires non-invasive assessment of increased iron stores in the liver to enable the clinical diagnosis. In addition, precise non-invasive criteria are needed to diagnose DIOS. Non-invasive assessment of the iron content of the liver using MRI, whenever possible, is also suggested for patients with grade 2 MHF based on serum levels of ferritin. An R2* value of 70–140 s−1 would assign the patient to grade 2 MHF (that is, dysmetabolic iron accumulation) or a value of >140 s−1 for grade 3 MHF (that is, DIOS). Iron accumulation in the liver is quantified according to expert recommendations and available clinical practice guidelines on non-invasive quantification of iron content in the liver for radiologists, as reported in Supplementary Box 1 (refs. 77,78,79). Although the serum levels of ferritin and MRI signal cut-offs for grading MHF are provisional and the correlation between serum levels of ferritin and R2* is not universally validated, we propose to identify three levels of iron accumulation for a granular stratification. Estimation of iron stores by T2 or R2 relaxometry is an acceptable alternative when locally available. When MRI is not available, in patients with coexistent liver disease, quantification of liver levels of iron could be determined by direct iron measurement in histological tissue samples.
R7
We propose to define the presence of DIOS in patients with MHF and increased hepatic iron stores, as evaluated by R2* >140 s−1 or direct evidence of increased hepatic iron stores (U) (CR).
R8
Liver biopsy is not required for the diagnosis of MHF or DIOS, unless otherwise indicated for the management of associated liver disease or for specific research purposes (U) (CR).
We propose that the acronym DIOS be kept for individuals with evidence of severe serum and hepatic iron load, as summarized in Table 1. Evidence from large population studies suggests that a predisposition to even a mild degree of iron accumulation can facilitate organ damage, such as cirrhosis36,80. Determining the relative contribution of inflammation or iron overload remains a major challenge in assessment of patients with MHF. We believe that liver biopsy is not required for the diagnosis of DIOS, unless otherwise indicated for the management of associated liver disease or for specific research purposes.
The most frequent differential diagnoses to be considered in clinical practice are listed in Supplementary Table 2. The suggested diagnostic and staging work-up for patients with suspected MHF is reported in Supplementary Table 3.
Clinical management
R9
The clinical management should be focused on lifestyle factors associated with increased risk of cardiometabolic risk factors (such as caloric intake and dietary patterns, alcohol intake, fructose and salt intake and sedentary lifestyle) and the pharmacological control of cardiovascular risk factors (U) (CR).
The association between serum levels of ferritin and the risk of cardiometabolic diseases is reported in Supplementary Table 4. Increased serum concentrations of ferritin and hepatic iron stores have been associated with the risk of type 2 diabetes mellitus, cardiovascular damage and several phenotypes related to liver disease (inflammation, fibrosis and hepatocellular carcinoma; evidence detailed and referenced in Supplementary Table 4). This finding highlights the unmet need to assess for and treat cardiovascular risk factors in patients with MHF. The promotion of a healthy lifestyle, a balanced diet low in processed foods, regular exercise and limited alcohol consumption are the cornerstone of the clinical management of this condition.
R10
In patients with MHF and DIOS, iron depletion therapy should be considered as an experimental therapy to be tested in well-powered controlled trials (U) (CR).
Several studies have suggested that increased stores of iron in the body might have a causal role in determining organ damage and that iron stores could be a modifiable risk factor in patients with metabolic disorders. The main results of controlled studies testing the effect of iron depletion in patients with hyperferritinaemia associated with metabolic dysfunction are shown in Supplementary Table 5. Case–control studies suggested that phlebotomy might decrease insulin resistance in patients with fatty liver and metabolic dysfunction81,82. Case–control studies also highlighted an improvement in liver damage (detected by liver enzymes and histology) in people with hyperferritinaemia who underwent and maintained iron depletion83,84. Furthermore, iron depletion was associated with a reduced risk of cancer in patients with peripheral arterial vascular disease, mostly people who smoke and had severe atherosclerosis85. However, in a randomized trial of patients with DIOS, iron depletion did not improve insulin resistance in the short term but was associated with reduced levels of liver enzymes (that is, alanine aminotransferase and aspartate transaminase)75. A meta-analysis, which was limited by the low number and heterogeneity of the studies considered (in terms of selection criteria, baseline iron stores, outcomes and duration of observation), concluded that in patients with nonalcoholic fatty liver disease (NAFLD) with or without DIOS, iron depletion had only a minor effect on the improvement of alanine aminotransferase levels, but did not affect insulin resistance and liver histology compared with lifestyle changes alone86. Therefore, outside clinical studies, iron depletion can be considered only in patients with confirmed severe hepatic iron accumulation (grade 3) associated with steatohepatitis or clinically significant liver fibrosis unresponsive to therapy.
The recommended clinical management for MHF based on the currently available evidence is reported in Fig. 2.
The diagnosis, based on the evaluation of inclusion and exclusion criteria, is followed by staging of iron accumulation (by ferritin and if available by MRI) and of organ damage. In addition to the current therapy for metabolic dysfunction, iron depletion therapy can be considered for patients with the most severe iron stores or within clinical trials. Iron stores are then monitored non-invasively.
The role of blood donation
R11
Blood donation is not contraindicated in individuals with MHF with controlled cardiovascular risk factors, in the absence of organ damage and of other contraindications to phlebotomy (U) (CR).
Approximately 250 mg of iron are removed with each 450 ml blood donation, which accounts for about 30% of the average iron stores in the liver in men and nearly 80% in women. With regular blood donation, individuals usually reach stability of iron balance at a lower level of body iron stores than individuals who do not donate blood regularly87. On this basis, as the proposal of the iron–heart hypothesis on cardiovascular disease in the early 1980s88, several studies have been conducted to evaluate whether regular blood donation can counteract iron toxicity and cardiometabolic complications by preventing iron overload and cardiometabolic complications. In the general population, frequent blood donors (defined as 2–10 donations per year) had increased insulin sensitivity, decreased insulin secretion and significantly lower serum levels of ferritin than non-donors, with both groups having similar blood concentrations of haematocrit and haemoglobin89.
A large cohort study from the USA has reported a protective effect (OR 0.67) of blood donation in terms of cardiovascular morbidity in non-smoking men90. The benefit of donation was greater in those with higher serum levels of LDL cholesterol than in those with lower levels, whereas no significant effect of blood donation was seen in women. This finding is in agreement with the iron–cardiovascular disease hypothesis, which postulates that the protective effect of phlebotomy would be more prominent in men, who have higher iron load than women88. Furthermore, an 88% reduced risk of myocardial infarction was also observed in a prospective epidemiological study from eastern Finland, which included 2,862 men aged 42–60 years (153 donors and 2,529 non-donors), who were followed up for an average of 9 years91. However, the results of these studies could have been biased by the selection of fairly healthy people for blood donation, the so-called healthy donor effect92. Subsequent studies adjusted for the healthy donor effect gave conflicting results, which further underscores the difficulties of drawing conclusions from observational and cohort studies on this topic93,94,95,96,97. Unfortunately, a controlled randomized clinical trial on the effects of blood donation on cardiometabolic morbidity and mortality seems to be almost impossible to conduct, and the question will have to be addressed by alternative study designs.
Future perspectives
Further recommendations
R12
Additional studies are required to define the specific genetic and environmental risk factors for MHF and DIOS development (U).
R13
Additional studies are required to define the correlation between serum levels of ferritin and hepatic iron content determined by MRI in patients with MHF (U).
R14
Additional studies are required to investigate whether MHF and/or mild tissue iron accumulation in the liver, adipose tissue and other organs are causally involved in the pathogenesis of insulin resistance, liver disease and other chronic degenerative conditions associated with MHF (U).
The main future challenges in the field are reported in Supplementary Table 6. We list specific items related to the definition, diagnosis, epidemiology, genetics, pathophysiology and treatment, including suggestions on the optimal study design that might be fostered by the implementation and prospective validation of a common definition of MHF. The main goals from a pathophysiological point of view will be to clarify what determines serum levels of ferritin and iron accumulation in individuals with metabolic dysfunction, and to clarify the relationships between alterations in iron metabolism, lipotoxicity, inflammation, insulin resistance and organ damage. From a clinical perspective, the goal should be to determine whether assessment of serum levels of ferritin and iron stores in the body has clinically meaningful additive prognostic value and whether reducing iron stores protects against organ-specific complications and improves clinical outcomes in individuals with metabolic dysfunction.
R15
Clinical studies to evaluate iron depletion in patients with MHF or DIOS should consider, as main outcomes, biomarkers that are closely linked to clinical events and take into account the perceived quality of life. These outcomes should be assessed after an adequate duration of follow-up, at least 3 months after achievement of iron depletion in the active arm (A).
Further multicentre randomized studies with simpler inclusion criteria and longer follow-up than those currently available, and eventually evaluation of hard clinical outcomes (that are more closely associated with major clinical events and/or mortality than liver enzymes or insulin levels), are necessary to draw more reliable conclusions. In particular, we make several proposals (as reported in Supplementary Table 6). MHF should be stratified by severity in randomized controlled studies. Serum concentrations of ferritin after at least 3 months of lifestyle change should be among the inclusion criteria of these studies. Iron depletion protocols and methods for iron maintenance should be standardized, and the possible use of supportive therapies (for example, folate supplementation) should be clarified. Outcomes should be examined at least at 3 months after achievement of iron store normalization. Observational studies in blood donors that control for donation frequency and propensity score should be considered. The incidence of type 2 diabetes mellitus, major cardiovascular events, cancer and liver events (such as progression of liver fibrosis) should be considered as outcomes. Sustained modification of diet and lifestyle habits remains the first therapeutic interventions in patients with MHF, together with control of cardiovascular risk factors by pharmacological approaches when necessary17.
Conclusions
Although not yet universally recognized as a distinct clinical entity, MHF and its disease stages might identify a subset of individuals in whom alterations of carbohydrate and lipid metabolism extend into a disrupted iron homeostasis, possibly triggered by insulin resistance, lipotoxicity and subclinical chronic systemic inflammation. Compared with patients with metabolic dysfunction without MHF, these individuals seem to be at higher risk of type 2 diabetes mellitus and of hepatic, cardiovascular and neoplastic diseases, independently of classic risk factors. Conflicting data suggest the possibility that iron accumulation in tissues is causally implicated in the pathogenesis of these cardiometabolic diseases. Although this hypothesis requires evaluation in robust clinical studies, the potential to reduce the complications of metabolic disorders by achieving and maintaining a state of near iron depletion deserves further investigation. As iron deficiency is associated with fatigue and impaired quality of life, and with worse outcomes in patients with heart failure98 and type 2 diabetes mellitus99, these studies must include an assessment of patient-related outcome measures, and symptoms of heart failure should be assessed at baseline. In this context, non-invasive multi-organ MRI evaluation could enable the simultaneous assessment of lipid levels in the liver, inflammation, fibrosis and hepatic and splenic iron concentration, as well as cardiac function98.
However, there are two major obstacles that hinder the design and implementation of randomized controlled clinical trials in this field. The first obstacle is the difficulty in finding sufficient funding for conventional iron depletion by phlebotomy, and the second one is the impossibility of running blind studies together with the presence of many confounders by using phlebotomy. There are two strategies to overcome the difficulties in conducting randomized clinical trials. The first one is to invest more in basic and translational research in the field, aiming to better define the molecular pathways that underlie MHF and pharmacological approaches to improve organ damage by modulating iron metabolism. The second one is to set up a robust research framework and generate new data to create a strong rationale for supporting these studies by public funding. In this regard, an apparent weakness (that is, the current necessity to rely on phlebotomy to achieve normalization of iron stores) might turn into a compelling argument for public health systems to support research evaluating this approach. Indeed, there is currently a global shortage of blood donations and blood products that has been worsened by the COVID-19 pandemic. Expanding the pool of blood donors, while assessing whether regular donation might protect against the development of cardiovascular, liver and neoplastic disorders, might also help to overcome this threat to public health100. We believe that the current Consensus Statement enables an important first step in this direction.
Change history
14 December 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41574-023-00940-2
References
Adams, P. C. et al. Hemochromatosis and iron-overload screening in a racially diverse population. N. Engl. J. Med. 352, 1769–1778 (2005).
Zhou, B., Liu, S. & Yuan, G. Combined associations of serum ferritin and body size phenotypes with cardiovascular risk profiles: a Chinese population-based study. Front. Public Health 9, 550011 (2021).
Wrede, C. E. et al. Association between serum ferritin and the insulin resistance syndrome in a representative population. Eur. J. Endocrinol. 154, 333–340 (2006).
Jehn, M., Clark, J. M. & Guallar, E. Serum ferritin and risk of the metabolic syndrome in U.S. adults. Diabetes Care 27, 2422–2428 (2004).
Zelber-Sagi, S., Nitzan-Kaluski, D., Halpern, Z. & Oren, R. NAFLD and hyperinsulinemia are major determinants of serum ferritin levels. J. Hepatol. 46, 700–707 (2007).
Haap, M. et al. Insulin sensitivity and liver fat: role of iron load. J. Clin. Endocrinol. Metab. 96, E958–E961 (2011).
Valenti, L. et al. Serum ferritin levels are associated with vascular damage in patients with nonalcoholic fatty liver disease. Nutr. Metab. Cardiovasc. Dis. 21, 568–575 (2011).
Manco, M. et al. Early interplay of intra-hepatic iron and insulin resistance in children with non-alcoholic fatty liver disease. J. Hepatol. 55, 647–653 (2011).
Zheng, X. et al. Hepatic iron stores are increased as assessed by magnetic resonance imaging in a Chinese population with altered glucose homeostasis. Am. J. Clin. Nutr. 94, 1012–1019 (2011).
Wlazlo, N. et al. Iron metabolism is associated with adipocyte insulin resistance and plasma adiponectin: the cohort on diabetes and atherosclerosis maastricht (CODAM) study. Diabetes Care 36, 309–315 (2013).
Bozzini, C. et al. Prevalence of body iron excess in the metabolic syndrome. Diabetes Care 28, 2061–2063 (2005).
Yu, L. et al. Association between serum ferritin and blood lipids: influence of diabetes and hs-CRP levels. J. Diabetes Res. 2020, 4138696 (2020).
Suarez-Ortegon, M. F. et al. Ferritin, metabolic syndrome and its components: a systematic review and meta-analysis. Atherosclerosis 275, 97–106 (2018).
Dongiovanni, P., Fracanzani, A. L., Fargion, S. & Valenti, L. Iron in fatty liver and in the metabolic syndrome: a promising therapeutic target. J. Hepatol. 55, 920–932 (2011).
Mendler, M. H. et al. Insulin resistance-associated hepatic iron overload. Gastroenterology 117, 1155–1163 (1999).
Riva, A. et al. Revaluation of clinical and histological criteria for diagnosis of dysmetabolic iron overload syndrome. World J. Gastroenterol. 14, 4745–4752 (2008).
Deugnier, Y., Bardou-Jacquet, E. & Laine, F. Dysmetabolic iron overload syndrome (DIOS). Presse Med. 46, e306–e311 (2017).
Stickel, F. & Hampe, J. Dissecting the evolutionary genetics of iron overload in non-alcoholic fatty liver disease. J. Hepatol. 53, 793–794 (2010).
Ryan, J. D. et al. Hepatic iron is the major determinant of serum ferritin in NAFLD patients. Liver Int. 38, 164–173 (2018).
Nelson, J. E. et al. Relationship between the pattern of hepatic iron deposition and histological severity in nonalcoholic fatty liver disease. Hepatology 53, 448–457 (2011).
Kowdley, K. V. et al. Serum ferritin is an independent predictor of histologic severity and advanced fibrosis in patients with nonalcoholic fatty liver disease. Hepatology 55, 77–85 (2012).
Buzzetti, E. et al. Evaluating the association of serum ferritin and hepatic iron with disease severity in non-alcoholic fatty liver disease. Liver Int. 39, 1325–1334 (2019).
Castiella, A. et al. Liver iron concentration in dysmetabolic hyperferritinemia: results from a prospective cohort of 276 patients. Ann. Hepatol. 19, 31–35 (2020).
Jezequel, C. et al. Both hepatic and body iron stores are increased in dysmetabolic iron overload syndrome. A case-control study. PLoS ONE 10, e0128530 (2015).
Deugnier, Y., Bardou-Jacquet, E. & Laine, F. Dysmetabolic iron overload syndrome. Bull. Acad. Natl Med. 200, 327–333 (2016).
Lazarus, J. V. et al. Advancing the global public health agenda for NAFLD: a consensus statement. Nat. Rev. Gastroenterol. Hepatol. 19, 60–78 (2022).
Fernandez-Real, J. M. et al. Serum ferritin as a component of the insulin resistance syndrome. Diabetes Care 21, 62–68 (1998).
Martinelli, N. et al. Increased serum hepcidin levels in subjects with the metabolic syndrome: a population study. PLoS ONE 7, e48250 (2012).
Yoneda, M. et al. Serum ferritin is a clinical biomarker in Japanese patients with nonalcoholic steatohepatitis (NASH) independent of HFE gene mutation. Dig. Dis. Sci. 55, 808–814 (2010).
Valenti, L. et al. Increased susceptibility to nonalcoholic fatty liver disease in heterozygotes for the mutation responsible for hereditary hemochromatosis. Dig. Liver Dis. 35, 172–178 (2003).
Corradini, E. et al. Ceruloplasmin gene variants are associated with hyperferritinemia and increased liver iron in patients with NAFLD. J. Hepatol. 75, 506–513 (2021).
Park, S. K., Ryoo, J. H., Kim, M. G. & Shin, J. Y. Association of serum ferritin and the development of metabolic syndrome in middle-aged Korean men: a 5-year follow-up study. Diabetes Care 35, 2521–2526 (2012).
Podmore, C. et al. Association of multiple biomarkers of iron metabolism and type 2 diabetes: the EPIC-InterAct study. Diabetes Care 39, 572–581 (2016).
Pitchika, A. et al. Hepatic steatosis and hepatic iron overload modify the association of iron markers with glucose metabolism disorders and metabolic syndrome. Liver Int. 41, 1841–1852 (2021).
Wang, X. et al. Genetic support of A causal relationship between iron status and type 2 diabetes: a mendelian randomization study. J. Clin. Endocrinol. Metab. 106, e4641–e4651 (2021).
Valenti, L. Uncovering the genetics of cirrhosis: new plots for the usual suspects. Liver Int. 40, 281–282 (2020).
Gan, W. et al. Association of TMPRSS6 polymorphisms with ferritin, hemoglobin, and type 2 diabetes risk in a Chinese Han population. Am. J. Clin. Nutr. 95, 626–632 (2012).
He, H. et al. Causal relationships between metabolic-associated fatty liver disease and iron status: two-sample Mendelian randomization. Liver Int. 42, 2759–2768 (2022).
Pietrangelo, A. Hereditary hemochromatosis: pathogenesis, diagnosis, and treatment. Gastroenterology 139, 393–408 (2010).
Rametta, R. et al. Hepcidin resistance in dysmetabolic iron overload. Liver Int. 36, 1540–1548 (2016).
Dongiovanni, P. et al. High fat diet subverts hepatocellular iron uptake determining dysmetabolic iron overload. PLoS ONE 10, e0116855 (2015).
Vecchi, C. et al. Gluconeogenic signals regulate iron homeostasis via hepcidin in mice. Gastroenterology 146, 1060–1069 (2014).
Hoki, T. et al. Increased duodenal iron absorption through up-regulation of divalent metal transporter 1 from enhancement of iron regulatory protein 1 activity in patients with nonalcoholic steatohepatitis. Hepatology 62, 751–761 (2015).
Altamura, S. et al. Iron aggravates hepatic insulin resistance in the absence of inflammation in a novel db/db mouse model with iron overload. Mol. Metab. 51, 101235 (2021).
Altamura, S. et al. Uncoupled iron homeostasis in type 2 diabetes mellitus. J. Mol. Med. 95, 1387–1398 (2017).
Avila, F. et al. Serum ferritin is associated with metabolic syndrome and red meat consumption. Oxid. Med. Cell Longev. 2015, 769739 (2015).
Mayneris-Perxachs, J. et al. Iron status influences non-alcoholic fatty liver disease in obesity through the gut microbiome. Microbiome 9, 104 (2021).
Ruivard, M., Laine, F. & Deugnier, Y. Iron absorption in nonalcoholic steatohepatitis and dysmetabolic iron overload syndrome. Hepatology 63, 1737–1738 (2016).
Trombini, P. et al. Hepcidin response to acute iron intake and chronic iron loading in dysmetabolic iron overload syndrome. Liver Int. 31, 994–1000 (2011).
Moreno-Navarrete, J. M. et al. Hepatic iron content is independently associated with serum hepcidin levels in subjects with obesity. Clin. Nutr. 36, 1434–1439 (2017).
Aigner, E. et al. Copper availability contributes to iron perturbations in human nonalcoholic fatty liver disease. Gastroenterology 135, 680–688 (2008).
Aigner, E. et al. A role for low hepatic copper concentrations in nonalcoholic fatty liver disease. Am. J. Gastroenterol. 105, 1978–1985 (2010).
Jiang, X., Stockwell, B. R. & Conrad, M. Ferroptosis: mechanisms, biology and role in disease. Nat. Rev. Mol. Cell Biol. 22, 266–282 (2021).
Handa, P. et al. Iron alters macrophage polarization status and leads to steatohepatitis and fibrogenesis. J. Leukoc. Biol. 105, 1015–1026 (2019).
Gabrielsen, J. S. et al. Adipocyte iron regulates adiponectin and insulin sensitivity. J. Clin. Invest. 122, 3529–3540 (2012).
Dongiovanni, P. et al. Dietary iron overload induces visceral adipose tissue insulin resistance. Am. J. Pathol. 182, 2254–2263 (2013).
Vinchi, F. et al. Hemopexin therapy reverts heme-induced proinflammatory phenotypic switching of macrophages in a mouse model of sickle cell disease. Blood 127, 473–486 (2016).
Gao, H. et al. Aberrant iron distribution via hepatocyte–stellate cell axis drives liver lipogenesis and fibrosis. Cell Metab. 34, 1201–1213 e1205 (2022).
Ikeda, Y. et al. Deletion of H-ferritin in macrophages alleviates obesity and diabetes induced by high-fat diet in mice. Diabetologia 63, 1588–1602 (2020).
Fernandez-Real, J. M., McClain, D. & Manco, M. Mechanisms linking glucose homeostasis and iron metabolism toward the onset and progression of type 2 diabetes. Diabetes Care 38, 2169–2176 (2015).
Stechemesser, L. et al. Metabolomic profiling identifies potential pathways involved in the interaction of iron homeostasis with glucose metabolism. Mol. Metab. 6, 38–47 (2017).
Mayneris-Perxachs, J., Mousa, A., Naderpoor, N., Fernandez-Real, J. M. & de Courten, B. Plasma phospholipids with long-chain polyunsaturated fatty acids and dihydroceramides at the crossroads of iron stores and insulin resistance. Mol. Nutr. Food Res. 64, e1901055 (2020).
Moreno-Navarrete, J. M. et al. Insulin resistance modulates iron-related proteins in adipose tissue. Diabetes Care 37, 1092–1100 (2014).
Segrestin, B. et al. Adipose tissue expansion by overfeeding healthy men alters iron gene expression. J. Clin. Endocrinol. Metab. 104, 688–696 (2019).
Gao, Y. et al. Adipocyte iron regulates leptin and food intake. J. Clin. Invest. 125, 3681–3691 (2015).
McClain, D. A. et al. Adipose tissue transferrin and insulin resistance. J. Clin. Endocrinol. Metab. 103, 4197–4208 (2018).
Winn, N. C., Volk, K. M. & Hasty, A. H. Regulation of tissue iron homeostasis: the macrophage “ferrostat”. JCI Insight 5, e132964 (2020).
Zhang, Z. et al. Adipocyte iron levels impinge on a fat-gut crosstalk to regulate intestinal lipid absorption and mediate protection from obesity. Cell Metab. 33, 1624–1639 e1629 (2021).
Li, J. et al. Transferrin receptor 1 regulates thermogenic capacity and cell fate in brown/beige adipocytes. Adv. Sci. 7, 1903366 (2020).
Kernan, K. F. & Carcillo, J. A. Hyperferritinemia and inflammation. Int. Immunol. 29, 401–409 (2017).
Wood, M. J., Crawford, D. H. G., Wockner, L. F., Powell, L. W. & Ramm, G. A. Serum ferritin concentration predicts hepatic fibrosis better than hepatic iron concentration in human HFE-haemochromatosis. Liver Int. 37, 1382–1388 (2017).
Garcia-Casal, M. N. et al. Performance and comparability of laboratory methods for measuring ferritin concentrations in human serum or plasma: a systematic review and meta-analysis. PLoS ONE 13, e0196576 (2018).
Eslam, M. et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J. Hepatol. 73, 202–209 (2020).
Fargion, S. et al. Hyperferritinemia, iron overload, and multiple metabolic alterations identify patients at risk for nonalcoholic steatohepatitis. Am. J. Gastroenterol. 96, 2448–2455 (2001).
Laine, F. et al. Metabolic and hepatic effects of bloodletting in dysmetabolic iron overload syndrome: a randomized controlled study in 274 patients. Hepatology 65, 465–474 (2017).
European Association for the Study of the Liver. EASL Clinical Practice Guidelines on non-invasive tests for evaluation of liver disease severity and prognosis - 2021 update. J. Hepatol. 75, 659–689 (2021).
Paisant, A., d’Assignies, G., Bannier, E., Bardou-Jacquet, E. & Gandon, Y. MRI for the measurement of liver iron content, and for the diagnosis and follow-up of iron overload disorders. Presse Med. 46, e279–e287 (2017).
Henninger, B., Alustiza, J., Garbowski, M. & Gandon, Y. Practical guide to quantification of hepatic iron with MRI. Eur. Radiol. 30, 383–393 (2020).
Henninger, B. et al. R2* relaxometry for the quantification of hepatic iron overload: biopsy-based calibration and comparison with the literature. Rofo 187, 472–479 (2015).
Chen, V. L., Chen, Y., Du, X., Handelman, S. K. & Speliotes, E. K. Genetic variants that associate with cirrhosis have pleiotropic effects on human traits. Liver Int. 40, 405–415 (2020).
Facchini, F. S. Effect of phlebotomy on plasma glucose and insulin concentrations. Diabetes Care 21, 2190 (1998).
Valenti, L. et al. Iron depletion by phlebotomy improves insulin resistance in patients with nonalcoholic fatty liver disease and hyperferritinemia: evidence from a case-control study. Am. J. Gastroenterol. 102, 1251–1258 (2007).
Valenti, L. et al. Venesection for non-alcoholic fatty liver disease unresponsive to lifestyle counselling—a propensity score-adjusted observational study. QJM 104, 141–149 (2011).
Valenti, L. et al. A randomized trial of iron depletion in patients with nonalcoholic fatty liver disease and hyperferritinemia. World J. Gastroenterol. 20, 3002–3010 (2014).
Zacharski, L. R. et al. Decreased cancer risk after iron reduction in patients with peripheral arterial disease: results from a randomized trial. J. Natl Cancer Inst. 100, 996–1002 (2008).
Murali, A. R., Gupta, A. & Brown, K. Systematic review and meta-analysis to determine the impact of iron depletion in dysmetabolic iron overload syndrome and non-alcoholic fatty liver disease. Hepatol. Res. 48, E30–E41 (2018).
Kiss, J. E. Laboratory and genetic assessment of iron deficiency in blood donors. Clin. Lab. Med. 35, 73–91 (2015).
Sullivan, J. L. Iron and the sex difference in heart disease risk. Lancet 1, 1293–1294 (1981).
Fernandez-Real, J. M., Lopez-Bermejo, A. & Ricart, W. Iron stores, blood donation, and insulin sensitivity and secretion. Clin. Chem. 51, 1201–1205 (2005).
Meyers, D. G. et al. Possible association of a reduction in cardiovascular events with blood donation. Heart 78, 188–193 (1997).
Salonen, J. T., Tuomainen, T. P., Salonen, R., Lakka, T. A. & Nyyssonen, K. Donation of blood is associated with reduced risk of myocardial infarction. The Kuopio ischaemic heart disease risk factor study. Am. J. Epidemiol. 148, 445–451 (1998).
Atsma, F., Veldhuizen, I., Verbeek, A., de Kort, W. & de Vegt, F. Healthy donor effect: its magnitude in health research among blood donors. Transfusion 51, 1820–1828 (2011).
Peffer, K., den Heijer, M., de Kort, W., Verbeek, A. L. M. & Atsma, F. Cardiovascular risk in 159 934 frequent blood donors while addressing the healthy donor effect. Heart 105, 1260–1265 (2019).
Ascherio, A., Rimm, E. B., Giovannucci, E., Willett, W. C. & Stampfer, M. J. Blood donations and risk of coronary heart disease in men. Circulation 103, 52–57 (2001).
Germain, M. et al. Iron and cardiac ischemia: a natural, quasi-random experiment comparing eligible with disqualified blood donors. Transfusion 53, 1271–1279 (2013).
Peffer, K. et al. Donation intensity and metabolic syndrome in active whole-blood donors. Vox Sang. 109, 25–34 (2015).
Waldvogel-Abramovski, S. et al. Iron and transfusion medicine. Blood Rev. 27, 289–295 (2013).
Rizzo, C., Carbonara, R., Ruggieri, R., Passantino, A. & Scrutinio, D. Iron deficiency: a new target for patients with heart failure. Front. Cardiovasc. Med. 8, 709872 (2021).
Suarez-Ortegon, M. F. et al. Decreased iron stores are associated with cardiovascular disease in patients with type 2 diabetes both cross-sectionally and longitudinally. Atherosclerosis 272, 193–199 (2018).
Cohn, C. S. et al. How do I manage long-term blood component shortages in a hospital Transfusion service? Transfusion 60, 1897–1904 (2020).
Valenti, L. et al. HFE genotype, parenchymal iron accumulation, and liver fibrosis in patients with nonalcoholic fatty liver disease. Gastroenterology 138, 905–912 (2010).
Dongiovanni, P. et al. PCSK7 gene variation bridges atherogenic dyslipidemia with hepatic inflammation in NAFLD patients. J. Lipid Res. 60, 1144–1153 (2019).
Valenti, L. et al. The A736V TMPRSS6 polymorphism influences iron metabolism and hepatocellular damage, and modifies the expression of HFE mutations in patients with NAFLD. Hepatology 56, 830a–831a (2012).
Valenti, L. et al. Alpha 1-antitrypsin mutations in NAFLD: high prevalence and association with altered iron metabolism but not with liver damage. Hepatology 44, 857–864 (2006).
Rametta, R. et al. Impact of natural neuromedin-B receptor variants on iron metabolism. Am. J. Hematol. 95, 167–177 (2020).
McKinnon, E. J., Rossi, E., Beilby, J. P., Trinder, D. & Olynyk, J. K. Factors that affect serum levels of ferritin in Australian adults and implications for follow-up. Clin. Gastroenterol. Hepatol. 12, 101–108 e104 (2014).
Aigner, E. et al. Pathways underlying iron accumulation in human nonalcoholic fatty liver disease. Am. J. Clin. Nutr. 87, 1374–1383 (2008).
Valenti, L. et al. Definition of healthy ranges for alanine aminotransferase levels: a 2021 update. Hepatol. Commun. 5, 1824–1832 (2021).
Bedogni, G. et al. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 6, 33 (2006).
Mariani, R. et al. Prolonged exposure to welding fumes as a novel cause of systemic iron overload. Liver Int. 41, 1600–1607 (2021).
Acknowledgements
The authors thank R. Gualtierotti, Università degli Studi di Milano, for critical revision of the manuscript. The authors also acknowledge the support of the following grants. Ministero della Salute, Ricerca Finalizzata RF-2016-02364358, RC Rete cardiologica ‘CV PREVITAL’, Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico ‘Liver BIBLE’ (PR-0391), Innovative Medicines Initiative 2 joint undertaking of European Union’s Horizon 2020 research and innovation programme and EFPIA European Union (EU) Programme Horizon 2020 (under grant agreement No. 777377) for the project LITMUS, H2020 under grant agreement ‘101016726’, the European Union, programme ‘Photonics’ under grant agreement ‘101016726’ for L.V. Fondo Nacional de Ciencia y Tecnología de Chile (FONDECYT #1191145 to M.A.) and from the Agencia Nacional de Investigación y Desarrollo, ANID through ANID ACE 210009 grant for M.A. NIH (P30DK124723), and Veterans Administration (2I01 BX001140) for D.M. SFB1036, SFB1118 and DFG (FerrOs — FOR5146; SPP2306) as well as Marsilius Kolleg for M.U.M. FRRB-Early Career Award (1749055) for A.P. National Natural Science Foundation of China (82070588), High Level Creative Talents from Department of Public Health in Zhejiang Province (S2032102600032) for M.-H.Z. Swedish Cancer Society (170690) and Stockholm County Council (K2017-4579) for P.S.
Author information
Authors and Affiliations
Contributions
L.V., E.C. and D.P. contributed to all aspects of the manuscript. L.A.A., E.A., M.A., E.B.-J., E.B., J.-M.F.-R., D.G., H.H., K.K., G.L., F.L., K.M., A. Pietrangello, C.W.S., P.S, E.A.T. and H.Z. provided substantial contributions to the discussion of the content and reviewed and/or edited the manuscript before submission. S.A., B.H., D.M., M.U.M., P.P., J.D.R., L.S., A. Pagani and F.V. provided substantial contributions to the discussion of the content, wrote the manuscript and reviewed and/or edited the manuscript before submission. M.-H.Z. researched data for the article, provided substantial contributions to the discussion of the content and reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
L.V. has received speaking fees from MSD, Gilead, AlfaSigma and AbbVie, served as a consultant for Gilead, Pfizer, AstraZeneca, Novo Nordisk, Intercept Pharmaceuticals, Diatech Pharmacogenetics and Ionis Pharmaceuticals, and received research grants from Gilead. E.C. has received speaking fees from Vifor Pharma and Sanofi–Genzyme, and served as a consultant for Vifor Pharma and Kedrion Biopharma. L.A.A. has been on advisory boards for Pfizer, Novartis and Roche Diagnostics. E.A. has received speaking and/or consultancy fees from Sanofi, Gilead Sciences, Intercept Pharmaceuticals, Roche, Novartis, Amgen, Novo Nordisk, Alnylam Pharmaceuticals, Sanofi–Aventis, Vifor, Daiiki–Sanyo, Sobi, PharmGenetix, Takeda. E.B. advises on Gilead, Pfizer, Novo Nordisk, Intercept, Inventiva, MSD, Boehringer and received a research grant from Gilead. E.B.-J. received speaking fees from Gilead, AbbVie and Orphalan. H.H.’s institution has received research grants from AstraZeneca, EchoSens, Gilead, Intercept, MSD and Pfizer with H.H. as a PI. These are unrelated to the current study. K.K. advises, is on the speakers’ bureau for, and received grants from Gilead, Intercept, HighTide. K.K. consults for Altimmune, Roche and Boeringer Ingelheim. K.K. advises Assembly and Calliditas. K.K. is on the speakers’ bureau for AbbVie. K.K. received grants from Janssen, Allergan, Genfit, CymaBay, Novartis, Enanta, Protagonist, Pfizer, BMS, Celgene, Intercept, Madrigal and Viking. D.P. advises for, has received speaking fees or travel/research grants from Macopharma, Ortho Clinical Diagnostics, Grifols, Gilead, Terumo, Immucor, Diamed, Diatech Pharmacogenetics and Diasorin. J.D.R. received consulting fees from Pfizer, Alnylam, 5am Ventures, Bond Biosciences, Gilead, Kyowa Kirin. P.S. has received speaking fees from MSD, Eisai, Roche and Albireo. C.W.S. has received speaking fees from Gilead and Abbott. E.A.T. has served as a consultant for Alexion, Boehringer, Gilead, Intercept, Novo Nordisk, Orphalan and Pfizer. M.H.Z. has received speaking fees from Hisky Medical. F.V. receives research grants from Vifor, Pharmanutra and Silence Therapeutics. H.Z. has received speaking fees from AbbVie, BMS, Bayer, Gilead, Intercept, Eisai, Sanofi–Genzyme, Vifor, Pharmacosmos, Medice, Pierre-Fabre, the Falk Foundation and grant support from Pharmacosmos and Vifor. The authors declare that none of these conflicts of interest is relevant to the present article. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Graça Porto, Fudi Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Valenti, L., Corradini, E., Adams, L.A. et al. Consensus Statement on the definition and classification of metabolic hyperferritinaemia. Nat Rev Endocrinol 19, 299–310 (2023). https://doi.org/10.1038/s41574-023-00807-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-023-00807-6
This article is cited by
-
Indoleamine 2,3-dioxygenase 1-mediated iron metabolism in macrophages contributes to lipid deposition in nonalcoholic steatohepatitis
Journal of Gastroenterology (2024)
-
Dual nature of ferritin for hematologic, liver functional, and metabolic parameters in older diabetic patients
Scientific Reports (2023)