Abstract
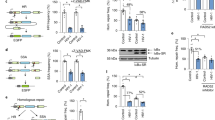
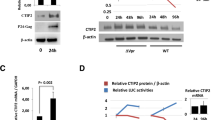
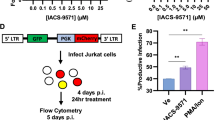
Host factors that silence provirus transcription in CD4+ memory T cells help HIV-1 escape eradication by the host immune system and by antiviral drugs1. These same factors, however, must be overcome for HIV-1 to propagate. Here we show that Vpx and Vpr encoded by diverse primate immunodeficiency viruses activate provirus transcription. Vpx and Vpr are adaptor proteins for the DCAF1-CUL4A/B E3 ubiquitin ligase that degrade SAMHD1 and increase reverse transcription2,3,4. Nonetheless, Vpx and Vpr have effects on reporter gene expression that are not explained by SAMHD1 degradation5,6,7,8. A screen for factors that mimic these effects identified the human silencing hub (HUSH) complex, FAM208A (TASOR/RAP140), MPHOSPH8 (MPP8), PPHLN1 (PERIPHILIN) and MORC29,10,11,12,13. Vpx associated with the HUSH complex and decreased steady-state level of these proteins in a DCAF1/CUL4A/B/proteasome-dependent manner14,15. Replication kinetics of HIV-1 and SIVMAC was accelerated to a similar extent by vpx or FAM208A knockdown. Finally, vpx increased steady-state levels of LINE-1 ORF1p, as previously described for FAM208A disruption11. These results demonstrate that the HUSH complex represses primate immunodeficiency virus transcription, and that, to counteract this restriction, viral Vpx or Vpr proteins degrade the HUSH complex.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper or in the supplementary table. The plasmids described in Supplementary Table 1, along with their complete nucleotide sequences, are available at https://www.addgene.org/Jeremy_Luban/.
References
Siliciano, J. D. et al. Long-term follow-up studies confirm the stability of the latent reservoir for HIV-1 in resting CD4+ T cells. Nat. Med. 9, 727–728 (2003).
Hrecka, K. et al. Vpx relieves inhibition of HIV-1 infection of macrophages mediated by the SAMHD1 protein. Nature 474, 658–661 (2011).
Laguette, N. et al. SAMHD1 is the dendritic- and myeloid-cell-specific HIV-1 restriction factor counteracted by Vpx. Nature 474, 654–657 (2011).
Lim, E. S. et al. The ability of primate lentiviruses to degrade the monocyte restriction factor SAMHD1 preceded the birth of the viral accessory protein Vpx. Cell Host Microbe. 11, 194–204 (2012).
Pertel, T., Reinhard, C. & Luban, J. Vpx rescues HIV-1 transduction of dendritic cells from the antiviral state established by type 1 interferon. Retrovirology 8, 49 (2011).
Reinhard, C., Bottinelli, D., Kim, B. & Luban, J. Vpx rescue of HIV-1 from the antiviral state in mature dendritic cells is independent of the intracellular deoxynucleotide concentration. Retrovirology 11, 12 (2014).
Goh, W. C. et al. HIV-1 Vpr increases viral expression by manipulation of the cell cycle: a mechanism for selection of Vpr in vivo. Nat. Med. 4, 65–71 (1998).
Miller, C. M. et al. Virion-associated Vpr alleviates a postintegration block to HIV-1 infection of dendritic cells. J. Virol. 91, e00051-17 (2017).
Tchasovnikarova, I. A. et al. GENE SILENCING. Epigenetic silencing by the HUSH complex mediates position-effect variegation in human cells. Science 348, 1481–1485 (2015).
Tchasovnikarova, I. A. et al. Hyperactivation of HUSH complex function by Charcot–Marie–Tooth disease mutation in MORC2. Nat. Genet. 49, 1035–1044 (2017).
Liu, N. et al. Selective silencing of euchromatic L1s revealed by genome-wide screens for L1 regulators. Nature 553, 228–232 (2018).
Liu, L. et al. A whole genome screen for HIV restriction factors. Retrovirology 8, 94 (2011).
Blewitt, M. E. et al. An N-ethyl-N-nitrosourea screen for genes involved in variegation in the mouse. Proc. Natl Acad. Sci. USA 102, 7629–7634 (2005).
Le Rouzic, E. et al. HIV1 Vpr arrests the cell cycle by recruiting DCAF1/VprBP, a receptor of the Cul4-DDB1 ubiquitin ligase. Cell Cycle 6, 182–188 (2007).
Srivastava, S. et al. Lentiviral Vpx accessory factor targets VprBP/DCAF1 substrate adaptor for cullin 4 E3 ubiquitin ligase to enable macrophage infection. PLoS Pathog. 4, e1000059 (2008).
Neagu, M. R. et al. Potent inhibition of HIV-1 by TRIM5-cyclophilin fusion proteins engineered from human components. J. Clin. Invest. 119, 3035–3047 (2009).
Re, F., Braaten, D., Franke, E. K. & Luban, J. Human immunodeficiency virus type 1 Vpr arrests the cell cycle in G2 by inhibiting the activation of p34cdc2-cyclin B. J. Virol. 69, 6859–6864 (1995).
Chang, F., Re, F., Sebastian, S., Sazer, S. & Luban, J. HIV-1 Vpr induces defects in mitosis, cytokinesis, nuclear structure, and centrosomes. Mol. Biol. Cell 15, 1793–1801 (2004).
He, J. et al. Human immunodeficiency virus type 1 viral protein R (Vpr) arrests cells in the G2 phase of the cell cycle by inhibiting p34cdc2 activity. J. Virol. 69, 6705–6711 (1995).
McCarthy, K. R. & Johnson, W. E. Plastic proteins and monkey blocks: How lentiviruses evolved to replicate in the presence of primate restriction factors. PLoS Pathog. 10, e1004017 (2014).
Fregoso, O. I. et al. Evolutionary toggling of Vpx/Vpr specificity results in divergent recognition of the restriction factor SAMHD1. PLoS Pathog. 9, e1003496 (2013).
Weinberg, M. S. & Morris, K. V. Transcriptional gene silencing in humans. Nucleic Acids Res. 44, 6505–6517 (2016).
Chéné, I. et al. Suv39H1 and HP1γ are responsible for chromatin-mediated HIV-1 transcriptional silencing and post-integration latency. EMBO J. 26, 424–435 (2007).
Wolf, D. & Goff, S. P. TRIM28 mediates primer binding site-targeted silencing of murine leukemia virus in embryonic cells. Cell 131, 46–57 (2007).
Peterlin, B. M. et al. Hili inhibits HIV replication in activated T cells. J. Virol. 91, e00237-17 (2017).
Wang, G. Z. & Goff, S. P. Transcriptional silencing of moloney murine leukemia virus in human embryonic carcinoma cells. J. Virol. 91, e02075-16 (2017).
Pertel, T. et al. TRIM5 is an innate immune sensor for the retrovirus capsid lattice. Nature 472, 361–365 (2011).
Robbez-Masson, L. et al. The HUSH complex cooperates with TRIM28 to repress young retrotransposons and new genes. Genome Res. 28, 836–845 (2018).
Jordan, A., Bisgrove, D. & Verdin, E. HIV reproducibly establishes a latent infection after acute infection of T cells in vitro. EMBO J. 22, 1868–1877 (2003).
Chen, H.-C., Martinez, J. P., Zorita, E., Meyerhans, A. & Filion, G. J. Position effects influence HIV latency reversal. Nat. Struct. Mol. Biol. 24, 47–54 (2017).
Acknowledgements
The authors wish to dedicate these experiments to the memory of Jan Svoboda (1934–2017), whose demonstration that cells may carry Rous sarcoma virus genetic information in the absence of any infectious virus production provided support to the proviral hypothesis. We thank L. Cavacini for antibodies and N. Landau for SIV plasmids. The following reagents were obtained through the AIDS Reagent Program, Division of AIDS, NIAID, NIH: J-Lat Tat-GFPCells (A1) from Dr. Eric Verdin and SIVmac239 SpX and SIVmac239 SpX ΔVpx from Dr. Ronald C. Desrosiers. This research was supported by National Institutes of Health (USA) grants nos. R01AI111809, RO1AI117839 and DP1DA034990 to J.L.
Author information
Authors and Affiliations
Contributions
L.Y. and J.L. designed the experiments. L.Y. performed the experiments with assistance from M.H.G., K.K., S.L.G., S.M., A.D. and W.E.D. L.Y. and J.L. analysed the experimental data. All authors contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary Figures 1–5 and Supplementary Table 1.
Rights and permissions
About this article
Cite this article
Yurkovetskiy, L., Guney, M.H., Kim, K. et al. Primate immunodeficiency virus proteins Vpx and Vpr counteract transcriptional repression of proviruses by the HUSH complex. Nat Microbiol 3, 1354–1361 (2018). https://doi.org/10.1038/s41564-018-0256-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-018-0256-x
This article is cited by
-
DCAF1 interacts with PARD3 to promote hepatocellular carcinoma progression and metastasis by activating the Akt signaling pathway
Journal of Experimental & Clinical Cancer Research (2024)
-
The HIV-1 gag p6: a promising target for therapeutic intervention
Retrovirology (2024)
-
Keep quiet: the HUSH complex in transcriptional silencing and disease
Nature Structural & Molecular Biology (2024)
-
HIV-2/SIV Vpx antagonises NF-κB activation by targeting p65
Retrovirology (2022)
-
The KT Jeang Retrovirology prize 2022: Florence Margottin-Goguet
Retrovirology (2022)