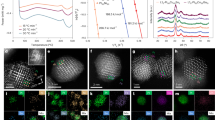
Abstract
Stable catalysts are essential to address energy and environmental challenges, especially for applications in harsh environments (for example, high temperature, oxidizing atmosphere and steam). In such conditions, supported metal catalysts deactivate due to sintering—a process where initially small nanoparticles grow into larger ones with reduced active surface area—but strategies to stabilize them can lead to decreased performance. Here we report stable catalysts prepared through the encapsulation of platinum nanoparticles inside an alumina framework, which was formed by depositing an alumina precursor within a separately prepared porous organic framework impregnated with platinum nanoparticles. These catalysts do not sinter at 800 °C in the presence of oxygen and steam, conditions in which conventional catalysts sinter to a large extent, while showing similar reaction rates. Extending this approach to Pd–Pt bimetallic catalysts led to the small particle size being maintained at temperatures as high as 1,100 °C in air and 10% steam. This strategy can be broadly applied to other metal and metal oxides for applications where sintering is a major cause of material deactivation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Additional data that support the findings of this study are reported in the Supplementary Information. Any additional data are available from the corresponding author upon request. Source data are provided with this paper.
References
Kunwar, D. et al. Investigating anomalous growth of platinum particles during accelerated aging of diesel oxidation catalysts. Appl. Catal. B 266, 118598 (2020).
Yang, A.-C. et al. Revealing the structure of a catalytic combustion active-site ensemble combining uniform nanocrystal catalysts and theory insights. Proc. Natl Acad. Sci. USA 117, 14721–14729 (2020).
Ogel, E. et al. Impact of preparation method and hydrothermal aging on particle size distribution of Pt/γ-Al2O3 and its performance in CO and NO oxidation. J. Phys. Chem. C. 123, 5433–5446 (2019).
Leistner, K., Gonzalez Braga, C., Kumar, A., Kamasamudram, K. & Olsson, L. Volatilisation and subsequent deposition of platinum oxides from diesel oxidation catalysts. Appl. Catal. B 241, 338–350 (2019).
Joo, S. H. et al. Thermally stable Pt/mesoporous silica core–shell nanocatalysts for high-temperature reactions. Nat. Mater. 8, 126–131 (2009).
Lee, I., Zhang, Q., Ge, J., Yin, Y. & Zaera, F. Encapsulation of supported Pt nanoparticles with mesoporous silica for increased catalyst stability. Nano Res. 4, 115–123 (2011).
Moliner, M. et al. Reversible transformation of Pt nanoparticles into single atoms inside high-silica chabazite zeolite. J. Am. Chem. Soc. 138, 15743–15750 (2016).
Zhang, J. et al. Sinter-resistant metal nanoparticle catalysts achieved by immobilization within zeolite crystals via seed-directed growth. Nat. Catal. 1, 540–546 (2018).
Cheng, N. et al. Extremely stable platinum nanoparticles encapsulated in a zirconia nanocage by area-selective atomic layer deposition for the oxygen reduction reaction. Adv. Mater. 27, 277–281 (2015).
Liu, J., Ji, Q., Imai, T., Ariga, K. & Abe, H. Sintering-resistant nanoparticles in wide-mouthed compartments for sustained catalytic performance. Sci. Rep. 7, 41773 (2017).
Yang, X. et al. Taming the stability of Pd active phases through a compartmentalizing strategy toward nanostructured catalyst supports. Nat. Commun. 10, 1611 (2019).
Jeong, H. et al. Highly durable metal ensemble catalysts with full dispersion for automotive applications beyond single-atom catalysts. Nat. Catal. 3, 368–375 (2020).
Li, W.-Z. et al. Stable platinum nanoparticles on specific MgAl2O4 spinel facets at high temperatures in oxidizing atmospheres. Nat. Commun. 4, 2481 (2013).
Jones, J. et al. Thermally stable single-atom platinum-on-ceria catalysts via atom trapping. Science 353, 150–154 (2016).
Datye, A. K. & Votsmeier, M. Opportunities and challenges in the development of advanced materials for emission control catalysts. Nat. Mater. 20, 1049–1059 (2020).
Wynblatt, P. & Gjostein, N. A. Particle growth in model supported metal catalysts—I. Theory. Acta Metall. 24, 1165–1174 (1976).
Plessow, P. N. & Abild-Pedersen, F. Sintering of Pt nanoparticles via volatile PtO2: simulation and comparison with experiments. ACS Catal. 6, 7098–7108 (2016).
Goodman, E. D. et al. Uniform Pt/Pd bimetallic nanocrystals demonstrate platinum effect on palladium methane combustion activity and stability. ACS Catal. 7, 4372–4380 (2017).
Riscoe, A. R. et al. Transition state and product diffusion control by polymer–nanocrystal hybrid catalysts. Nat. Catal. 2, 852–863 (2019).
Lu, A.-H. & Schüth, F. Nanocasting: a versatile strategy for creating nanostructured porous materials. Adv. Mater. 18, 1793–1805 (2006).
Wynblatt, P. Particle growth in model supported metal catalysts—II. Comparison of experiment with theory. Acta Metall. 24, 1175–1182 (1976).
Jentys, A. Estimation of mean size and shape of small metal particles by EXAFS. Phys. Chem. Chem. Phys. 1, 4059–4063 (1999).
Mitchell, S., Qin, R., Zheng, N. & Pérez-Ramírez, J. Nanoscale engineering of catalytic materials for sustainable technologies. Nat. Nanotechnol. 16, 129–139 (2021).
Zhao, M. et al. Metal–organic frameworks as selectivity regulators for hydrogenation reactions. Nature 539, 76–80 (2016).
Mao, X. et al. Epitaxial and strong support interactions between Pt and LaFeO3 films stabilize Pt dispersion. J. Am. Chem. Soc. 142, 10373–10382 (2020).
Cargnello, M. et al. Control of metal nanocrystal size reveals metal-support interface role for ceria catalysts. Science 341, 771–773 (2013).
Lu, J. et al. Coking- and sintering-resistant palladium catalysts achieved through atomic layer deposition. Science 335, 1205–1208 (2012).
Kwak, J. H. et al. Coordinatively unsaturated Al3+ centers as binding sites for active catalyst phases of platinum on γ-Al2O3. Science 325, 1670–1673 (2009).
Kim, N., Bassiri, R., Fejer, M. M. & Stebbins, J. F. The structure of ion beam sputtered amorphous alumina films and effects of Zn doping: high-resolution 27Al NMR. J. Non-Cryst. Solids 405, 1–6 (2014).
Campbell, C. T. & Mao, Z. Chemical potential of metal atoms in supported nanoparticles: dependence upon particle size and support. ACS Catal. 7, 8460–8466 (2017).
Goodman, E. D. et al. Catalyst deactivation via decomposition into single atoms and the role of metal loading. Nat. Catal. 2, 748–755 (2019).
Malis, T., Cheng, S. C. & Egerton, R. F. EELS log-ratio technique for specimen-thickness measurement in the TEM. J. Electron Microsc. Tech. 8, 193–200 (1988).
Rappé, K. G. et al. Aftertreatment protocols for catalyst characterization and performance evaluation: low-temperature oxidation, storage, three-way, and NH3-SCR catalyst test protocols. Emiss. Control Sci. Technol. 5, 183–214 (2019).
Cargnello, M. et al. Efficient removal of organic ligands from supported nanocrystals by fast thermal annealing enables catalytic studies on well-defined active phases. J. Am. Chem. Soc. 137, 6906–6911 (2015).
Shinotsuka, H., Tanuma, S., Powell, C. J. & Penn, D. R. Calculations of electron inelastic mean free paths. X. Data for 41 elemental solids over the 50 eV to 200 keV range with the relativistic full Penn algorithm. Surf. Interface Anal. 47, 871–888 (2015).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Digne, M., Sautet, P., Raybaud, P., Euzen, P. & Toulhoat, H. Use of DFT to achieve a rational understanding of acid–basic properties of γ-alumina surfaces. J. Catal. 226, 54–68 (2004).
Schäfer, H. & Tebben, A. Gleichgewichtsmessungen im System Platin—Sauerstoff Gasförmiges Platindioxyd. Z. Anorg. Allg. Chem. 304, 317–321 (1960).
Acknowledgements
We gratefully acknowledge financial support from the US Department of Energy, Office of Science, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division, Catalysis Science Program to the SUNCAT Center for Interface Science and Catalysis. Additional support was provided by the Stanford Precourt Institute for Energy through a seed grant, and by the US Department of Energy, Office of Science, Office of Basic Energy Sciences, under contract no. DE-SC0022197 to M.C. A.A. acknowledges support in the form of an American Association University Women International Fellowship and a Siebel Scholar Foundation Fellowship. M.L.S. acknowledges support in the form of a TomKat fellowship at Stanford University. Part of this work was performed at the Stanford Nano Shared Facilities, supported by the National Science Foundation under award ECCS-2026822. Part of this work was performed at the SSRL of the SLAC National Accelerator Laboratory, and use of the SSRL was supported by the US Department of Energy, Office of Science, Office of Basic Energy Science under contract no. DE-AC02-76SF00515, and by CO-ACCESS, supported by the US Department of Energy, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division. Work at the Molecular Foundry was supported by the Office of Science, Office of Basic Energy Sciences of the US Department of Energy under contract no. DE-AC02-05CH11231. P.H. and P.N.P. acknowledge support by the state of Baden-Württemberg through bwHPC (bwunicluster and JUSTUS, RV bw17D011), and funding by the Deutsche Forschungsgemeinschaft (German Research Foundation) under project ID 434253773 and SFB 1441 project ID 426888090. The NMR study was supported by NSF EAR-1521055 to J.F.S. We thank J. Hong (SLAC National Accelerator Laboratory) for help with the XAS data collection, and G. Stewart (SLAC National Accelerator Laboratory) for the Pt@Al2O3 schematic model.
Author information
Authors and Affiliations
Contributions
A.A. and M.C. conceived the research. A.A. synthesized materials, conducted the physisorption and chemisorption experiments, TGA, XPS, DRIFTS, XAS characterization and much of the catalytic activity tests, and analysed the STEM microscopy and tomography data. M.L.S. contributed with sample preparation, catalytic activity characterization and aging tests. C.Z., A.-C.Y., E.D.G., J.C. and K.C.B. performed microscopy characterization studies. J.S.L.-P. conducted the XRF and PXRD measurements. A.S.H. and S.R.B. assisted with the XAS data collection and analysis. P.H. and P.N.P. performed DFT calculations and sintering simulations. P.E. assisted with tomography reconstruction. J.F.S. performed NMR studies and analysed the data. M.C. supervised the whole research project. A.A. and M.C. wrote the manuscript with revision and participation from all authors.
Corresponding author
Ethics declarations
Competing interests
Stanford University has submitted a provisional patent application entitled ‘Novel approach to make active and stable low-temperature hydrocarbon combustion catalysts’ on which A.A. and M.C. are listed as inventors (US provisional patent number 63/210,723, filed in 2022). The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Abhaya Datye and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Characterization of Pt@Al2O3.
a, TGA upon heating the sample in air to 700 °C. b, N2 physisorption isotherm. c, Pore size distribution.
Extended Data Fig. 2 Characterization of Pt/Al2O3.
a, Transmission electron microscopy image. b, NP size distribution. c, d, XPS of (c) Pt 4d3/2 (332.2 eV) and Pt 4d5/2 (315.2 eV); (d) Al 2p (74.7 eV) and Pt 4f5/2 (71.5 eV) overlapping signals. e, XRD of Pt/Al2O3 calcined at 600 °C. Highlighted peaks are due to metallic Pt. f, TEM: upon calcination the average Pt NP size increases from 3.8 nm to 23.3 nm.
Extended Data Fig. 3 Propene combustion light off curves of Pt@Al2O3.
a, Fresh Pt@Al2O3 and metal-free Al2O3 support. Reaction mixture: 0.15 vol.% C3H6, 3 vol.% O2, and 5 vol.% steam (balance Ar). b, As-prepared Pt@Al2O3 and the aged catalyst after consecutive three cycles (aging performed at 800 °C for 2 h in the reaction mixture). 30 mg of the catalyst was mixed with 160 mg of SiC diluent. Catalyst pretreatment: 300 °C, 30 min 5 vol.% O2 (40 ml/min), 10 min Ar (40 ml/min), 30 min 5 vol.% H2 (40 ml/min). Reaction mixture: 0.15 vol.% C3H6, 3 vol.% O2, 5 vol.% H2O, balance Ar (50 ml/min total flow rate).
Extended Data Fig. 4 Catalytic activity testing and characterization of Pt/!-Al2O3.
a, Light-off curve measurements of fresh and aged Pt/!-Al2O3. Reaction conditions: 0.15 vol. % C3H6, 3 vol. % O2, and 5 vol. % steam, balance Ar (40 ml/min total flow). Aging performed in the reaction mixture at 800 °C for 2 h. b–e, Representative TEM images and particle size distributions of the fresh (b, c) and aged catalysts (d, e).
Extended Data Fig. 5 Characterization of Pt samples.
a, XRD patterns for the samples and the bare support. ^ and * represent diffractions peaks due to γ-Al2O3 and quartz diluent, respectively. Highlighted region at 40° indicates overlapping γ-Al2O3 and Pt(111) diffraction peaks. b, XPS characterization of Pt@Al2O3-800. c, Magnitude and imaginary part of the Fourier Transform. d, EXAFS of the Pt L3-edge data and best-fits characterizing the encapsulated and supported Pt catalysts before and after aging at 800 °C. Black lines indicate the fits. e, f, Pt L3-edge XANES: (e) Pt@Al2O3; (f) Pt/Al2O3.
Extended Data Fig. 6 Catalytic activity testing and characterization of Pt@Al2O3 and Pt/Al2O3.
a, Arrhenius plots with 95% confidence intervals of Pt@Al2O3 and Pt/Al2O3 for propene combustion collected at conversions less than 5%. The data points are averages for three measurements. b, CO diffuse reflectance infrared spectroscopy. c, Arrhenius plots with 95% confidence intervals of Pt@Al2O3 and Pt/Al2O3 for CO oxidation collected at conversions less than 5%. The data points are averages for three measurements. d, 27Al MAS NMR spectra of fresh and aged Pt@Al2O3 and Pt/Al2O3. Peaks for four-, five-, and six-coordinated Al (AlO4, AlO5, AlO6) are labeled, with approximate percentages of each shown below the spectra. e, Proposed model schematic of Pt@Al2O3.
Extended Data Fig. 7 DFT and sintering models.
a, Side-on views of the atomic models optimized for the interfaces of Pt(100) and Pt(111) with the γ-Al2O3(100) surface. b, Illustration of sintering models and underlying assumptions: Left: No diffusion limitations lead to immediate equilibration within the gas phase and thus a constant background pressure. Right: Significant diffusion limitations through the encapsulating material lead to local equilibration between particle and gas phase for each particle, so that locally the equilibrium pressure is established.
Extended Data Fig. 8 Catalytic activity testing of Pt@Al2O3 and Pd@Al2O3.
a, Propene combustion activity for fresh Pt@Al2O3 and 1,100 °C aged Pt@Al2O3. Reaction mixture: 0.15 vol.% C3H6, 3 vol.% O2, and 5 vol.% steam. Aging performed at 1,100 °C for 5 h in air with 10 vol. % steam (40 ml/min total flow). b, TEM image of the 1,100 °C aged Pt@Al2O3 catalyst. c, d, TEM image and particle size distribution of as-synthesized Pd@Al2O3. e, Propene combustion activity for fresh Pd@Al2O3 and 1,100 °C aged Pd@Al2O3. Reaction mixture: 0.15 vol.% C3H6, 3 vol.% O2, and 5 vol.% steam. Aging performed at 1,100 °C for 5 h in air with 10 vol. % steam (40 ml/min total flow). f, TEM image of the 1,100 °C aged Pd@Al2O3 catalyst.
Extended Data Fig. 9 Catalytic activity and characterization of PdPt@Al2O3.
a, Propene combustion activity of the fresh and 1,100 °C aged metal-free alumina support. Reaction mixture: 0.15 vol.% C3H6, 3 vol.% O2, and 5 vol.% steam. Aging performed at 1,100 °C for 5 h in air with 10 vol. % steam (40 ml/min total flow). b, c, Characterization of the assynthesized and 1,100 °C aged PdPt@Al2O3: b, XPS. c, XRF. d, e, Representative HAADF-STEM image of 1,100 °C aged PdPt@Al2O3 and EDS spectrum showing no Pd or Pt detectable signal. Fe, Co, Cu, Zr and Sn normal signal from the TEM column/holder. f, XRD and g, 27Al NMR of as-synthesized and 1,100 °C aged PdPt@Al2O3.
Extended Data Fig. 10 XAS characterization of PdPt@Al2O3.
a–c, Magnitude and imaginary component of the Fourier Transform and EXAFS at the Pd K-edge (a, b) and Pt L3-edge (c, d) characterizing the fresh PdPt@Al2O3 and PdPt@Al2O3-1100-air samples. Black lines indicate the fits. The range in R was 1.2–3.0 Å (Pd) and 1.7–3.2 Å (Pt); the range in k was 2.7–12.5 Å−1 (Pd) and 3.1–12.0 Å−1 (Pt). e, f, PdPt@Al2O3 XANES: e, Pt L3-edge; f, Pd K-edge.
Supplementary information
Supplementary Information
Sintering simulations and Supplementary Figs. 1–16, Tables 1–9 and references.
Source data
Source Data Fig. 3
Source data file (text format) for DFT structures.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aitbekova, A., Zhou, C., Stone, M.L. et al. Templated encapsulation of platinum-based catalysts promotes high-temperature stability to 1,100 °C. Nat. Mater. 21, 1290–1297 (2022). https://doi.org/10.1038/s41563-022-01376-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-022-01376-1
This article is cited by
-
Synthesis of core@shell catalysts guided by Tammann temperature
Nature Communications (2024)
-
Challenging thermodynamics: combining immiscible elements in a single-phase nano-ceramic
Nature Communications (2024)
-
Hybrid oxide coatings generate stable Cu catalysts for CO2 electroreduction
Nature Materials (2024)
-
Nanoparticle proximity controls selectivity in benzaldehyde hydrogenation
Nature Catalysis (2024)
-
Selective hydrogenation via precise hydrogen bond interactions on catalytic scaffolds
Nature Communications (2023)