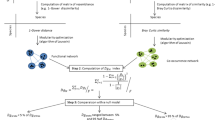
Abstract
Inferring biotic interactions from species co-occurrence patterns has long intrigued ecologists. Yet recent research revealed that co-occurrences may not reliably represent pairwise biotic interactions. We propose that examining network-level co-occurrence patterns can provide valuable insights into community structure and assembly. Analysing ten bipartite networks of empirically sampled biotic interactions and associated species spatial distribution, we find that approximately 20% of co-occurrences correspond to actual interactions. Moreover, the degree distribution shifts from exponential in co-occurrence networks to power laws in networks of biotic interactions. This shift results from a strong interplay between species’ biotic (their interacting partners) and abiotic (their environmental requirements) niches, and is accurately predicted by considering co-occurrence frequencies. Our work offers a mechanistic understanding of the assembly of ecological communities and suggests simple ways to infer fundamental biotic interaction network characteristics from co-occurrence data.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the study results can be found at https://doi.org/10.5281/zenodo.8402455.
Code availability
The code used to analyse the data and generate the results can be found at https://doi.org/10.5281/zenodo.8402455.
References
Gotelli, N. J. & McCabe, D. J. Species co‐occurrence: a meta‐analysis of J.M. Diamond’s assembly rules model. Ecology 83, 2091–2096 (2002).
Araújo, M. B., Rozenfeld, A., Rahbek, C. & Marquet, P. A. Using species co‐occurrence networks to assess the impacts of climate change. Ecography 34, 897–908 (2011).
Connor, E. F., Collins, M. D. & Simberloff, D. The checkered history of checkerboard distributions. Ecology 94, 2403–2414 (2013).
Diamond, J., Pimm, S. L. & Sanderson, J. G. The checkered history of checkerboard distributions: comment. Ecology 96, 3386–3388 (2015).
Hutchinson, G. E. Concluding remarks. Cold Spring Harb. Symp. Quant. Biol. 22, 415–427 (1957).
MacArthur, R. H. Geographical Ecology: Patterns in the Distribution of Species (Harper & Row, 1972).
Peterson, A. T. et al. in Ecological Niches and Geographic Distributions (MPB-49) Ch. 3 (eds Levin, S. A. & Horn, H. S.) (Princeton Univ. Press, 2011).
Boulangeat, I., Gravel, D. & Thuiller, W. Accounting for dispersal and biotic interactions to disentangle the drivers of species distributions and their abundances. Ecol. Lett. 15, 584–593 (2012).
Forbes, S. A. On the Local Distribution of Certain Illinois Fishes: An Essay in Statistical Ecology (Illinois State Laboratory of Natural History, 1907).
Pielou, D. P. & Pielou, E. C. The detection of different degrees of coexistence. J. Theor. Biol. 16, 427–437 (1967).
Pielou, D. P. & Pielou, E. C. Association among species of infrequent occurrence: the insect and spider fauna of Polyporus betulinus (Bulliard) fries. J. Theor. Biol. 21, 202–216 (1968).
Diamond, J. in Ecology and Evolution of Communities (eds Cody, M. L. & Diamond, J.) 342–444 (Belknap, 1975).
Cazelles, K., Araújo, M. B., Mouquet, N. & Gravel, D. A theory for species co-occurrence in interaction networks. Theor. Ecol. 9, 39–48 (2016).
Blanchet, F. G., Cazelles, K. & Gravel, D. Co‐occurrence is not evidence of ecological interactions. Ecol. Lett. 23, 1050–1063 (2020).
Freilich, M. A., Wieters, E., Broitman, B. R., Marquet, P. A. & Navarrete, S. A. Species co‐occurrence networks: can they reveal trophic and non‐trophic interactions in ecological communities? Ecology 99, 690–699 (2018).
Borthagaray, A. I., Arim, M. & Marquet, P. A. Inferring species roles in metacommunity structure from species co-occurrence networks. Proc. Biol. Sci. 281, 20141425 (2014).
Montoya, J. M., Pimm, S. L. & Solé, R. V. Ecological networks and their fragility. Nature 442, 259–264 (2006).
Bascompte, J. & Jordano, P. Plant-animal mutualistic networks: the architecture of biodiversity.Annu. Rev. Ecol. Evol. Syst. 38, 567–593 (2007).
Ings, T. C. et al. Ecological networks—beyond food webs. J. Anim. Ecol. 78, 253–269 (2009).
Dunne, J. A., Williams, R. J. & Martinez, N. D. Food-web structure and network theory: the role of connectance and size. Proc. Natl Acad. Sci. USA 99, 12917–12922 (2002).
McCann, K. S. The diversity-stability debate. Nature 405, 228–233 (2000).
Albert, R., Jeong, H., Barabasi, A. & Barabási, A.-L. Error and attack tolerance of complex networks. Nature 406, 378–382 (2000).
Kaiser‐Bunbury, C. N., Muff, S., Memmott, J., Müller, C. B. & Caflisch, A. The robustness of pollination networks to the loss of species and interactions: a quantitative approach incorporating pollinator behaviour. Ecol. Lett. 13, 442–452 (2010).
Mestre, F., Rozenfeld, A. & Araújo, M. B. Human disturbances affect the topology of food webs. Ecol. Lett. 25, 2476–2488 (2022).
Araújo, M. B. & Rozenfeld, A. The geographic scaling of biotic interactions. Ecography 37, 406–415 (2014).
Galiana, N., Lurgi, M., Montoya, J. M., Araújo, M. B. & Galbraith, E. D. Climate or diet? The importance of biotic interactions in determining species range size. Glob. Ecol. Biogeogr. 32, 1178–1188 (2023).
Gaston, K. J. & Fuller, R. A. The sizes of species’ geographic ranges. J. Appl. Ecol. 46, 1–9 (2009).
Manne, L. L., Brooks, T. M. & Pimm, S. L. Relative risk of extinction of passerine birds on continents and islands. Nature 399, 258–261 (1999).
Staude, I. R. et al. Replacements of small- by large-ranged species scale up to diversity loss in Europe’s temperate forest biome. Nat. Ecol. Evol. 4, 802–808 (2020).
Gravel, D., Poisot, T., Albouy, C., Velez, L. & Mouillot, D. Inferring food web structure from predator–prey body size relationships. Methods Ecol. Evol. 4, 1083–1090 (2013).
Morales-Castilla, I., Matias, M. G., Gravel, D. & Araújo, M. B. Inferring biotic interactions from proxies. Trends Ecol. Evol. 30, 347–356 (2015).
Albouy, C. et al. The marine fish food web is globally connected. Nat. Ecol. Evol. 3, 1153–1161 (2019).
Maiorano, L., Montemaggiori, A., Ficetola, G. F. & O’Connor, L. TETRA‐EU 1.0: a species‐level trophic metaweb of European tetrapods. Glob. Ecol. Biogeogr. 29, 1452–1457 (2020).
Mestre, F. et al. Disentangling food-web environment relationships: a review with guidelines. Basic Appl. Ecol. 61, 102–115 (2022).
Hernández‐Castellano, C. et al. A new native plant in the neighborhood: effects on plant–pollinator networks, pollination, and plant reproductive success. Ecology 101, e03046 (2020).
Reverté, S. et al. Spatial variability in a plant–pollinator community across a continuous habitat: high heterogeneity in the face of apparent uniformity. Ecography 42, 1558–1568 (2019).
Torné‐Noguera, A., Arnan, X., Rodrigo, A. & Bosch, J. Spatial variability of hosts, parasitoids and their interactions across a homogeneous landscape. Ecol. Evol. 10, 3696–3705 (2020).
Vázquez, D. P. & Simberloff, D. Changes in interaction biodiversity induced by an introduced ungulate. Ecol. Lett. 6, 1077–1083 (2003).
Grass, I., Jauker, B., Steffan-Dewenter, I., Tscharntke, T. & Jauker, F. Past and potential future effects of habitat fragmentation on structure and stability of plant–pollinator and host–parasitoid networks. Nat. Ecol. Evol. 2, 1408–1417 (2018).
Osorio, S., Arnan, X., Bassols, E., Vicens, N. & Bosch, J. Local and landscape effects in a host–parasitoid interaction network along a forest–cropland gradient. Ecol. Appl. 25, 1869–1879 (2015).
Kaartinen, R. & Roslin, T. Shrinking by numbers: landscape context affects the species composition but not the quantitative structure of local food webs. J. Anim. Ecol. 80, 622–631 (2011).
Kopelke, J.-P. et al. Food-web structure of willow-galling sawflies and their natural enemies across Europe. Ecology 98, 1730 (2017).
Galiana, N. et al. Ecological network complexity scales with area. Nat. Ecol. Evol. 6, 307–314 (2022).
Broido, A. D. & Clauset, A. Scale-free networks are rare. Nat. Commun. 10, 1017 (2019).
Acknowledgements
N.G. received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement BIOFOODWEB (no. 101025471). M.B.A. acknowledges funding from the Spanish Ministry of Science, Innovation and Universities through the PredWeb project (PGC2018–099363-B-I00) and, together with A.R., from the European Union’s Horizon 2020 research and innovation programme under grant agreement AQUACOSM-Plus (no. 871081). J.-F.A. was supported by the Laboratoires d’Excellences (LABEX) TULIP (ANR-10-LABX-41).
Author information
Authors and Affiliations
Contributions
N.G., J.-F.A. and M.B.A. designed the research. N.G. performed the research and analysed the data. J-F.A. and N.G. developed the theoretical framework. N.G. wrote the manuscript with feedback from J.-F.A. and M.B.A., and comments from F.M. and A.R.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Kevin Cazelles and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Comparison of the frequency distributions of resources degree among co-occurrence networks and realised biotic interactions networks.
Red lines represent co-occurrence networks and yellow lines represent realised biotic interactions networks in the 10 datasets investigated. Top row are plant-pollinator networks and bottom row are host-parasite networks. Black dashed lines indicate the more parsimonious functions among all tested (see Methods).
Extended Data Fig. 2 Relationship between the number of potential interactions and the number of realised interactions and model prediction for resource species.
Top row are plant-pollinator networks and bottom row are host-parasite interactions. Each black point represents a species in the empirical system and the green line and points indicate the predicted proportion of realised interactions by the proposed model based on the frequency of co-occurrences. Green lines represent the mean tendency and shaded areas represent 95% confidence intervals.
Extended Data Fig. 3 Relationship between number of potential interactions for resource species and the mean frequency of co-occurrence with their consumers.
Top row are plant-pollinator networks and bottom row are host-parasite interactions. Each black point represents a species in the empirical system. Blue lines represent a gam fit only for visualisation purposes and shaded areas represent 95% confidence intervals.
Extended Data Fig. 4 Relationship between the number of expected interactions based on the frequencies of species co-occurrences and the number of realised interactions for each species.
(a) Shows the relationship from the consumer’s perspective (that is indegree is the number of resources each consumer has) while (b) represents the resources perspective (that is outdegree corresponds to the number of consumers each resource has). Each point represents a species in the empirical system. Black line shows the 1:1 line indicating a perfect relationship between the predicted and the realised number of interactions.
Extended Data Fig. 5 Comparison of the degree distributions of resource species among co-occurrence networks, realised biotic interactions networks and our theoretical predictions accounting for the frequency of interactions.
Red lines represent co-occurrence networks, yellow lines represent realised biotic interactions networks and our theoretical predictions accounting for the frequency of interactions are represented in multicolor (each color represents a replicate) in the 10 datasets investigated. Top row are plant-pollinator networks and bottom row are host-parasite networks.
Extended Data Fig. 6 Comparison of the degree distributions among co-occurrence networks, realised biotic interactions networks and the null model predictions.
Red lines represent co-occurrence networks, yellow lines represent realised biotic interactions networks and the null model predictions are represented in multicolor (each color represents a replicate) for the 10 datasets investigated. The null model prunes the co-occurrence networks using a constant proportion of links to keep across species. Therefore, it results in a random pruning of the co-occurrence network. Top rows correspond to consumers and bottom rows correspond to resource species.
Extended Data Fig. 7 Relationship between the per-site interactions rate (p) and the proportion of realised links (f) across datasets.
Each color represents a different dataset.
Supplementary information
Supplementary Information
Supplementary Text 1. Dataset description. Table 1. Basic network properties of each dataset. Text 2. Mathematical transformations of degree distributions: how to get from an exponential to a power law. Table 2. Values of the per-site interaction rate (P) for each dataset. Table 3. Degree distribution fits for each dataset.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Galiana, N., Arnoldi, JF., Mestre, F. et al. Power laws in species’ biotic interaction networks can be inferred from co-occurrence data. Nat Ecol Evol 8, 209–217 (2024). https://doi.org/10.1038/s41559-023-02254-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-023-02254-y
This article is cited by
-
Isolating interactions from co-occurrences
Nature Ecology & Evolution (2023)