Abstract
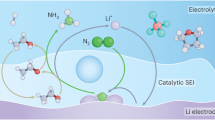
The electrochemical conversion of ammonia to dinitrogen in a direct ammonia fuel cell (DAFC) is a necessary technology for the realization of a nitrogen economy. Previous efforts to catalyse this reaction with molecular complexes required the addition of exogenous oxidizing reagents or application of potentials greater than the thermodynamic potential for the oxygen reduction reaction—the cathodic process of a DAFC. We report a stable metal–metal bonded diruthenium complex that spontaneously produces dinitrogen from ammonia under ambient conditions. The resulting reduced diruthenium material can be reoxidized with oxygen for subsequent reactions with ammonia, demonstrating its ability to spontaneously promote both half-reactions necessary for a DAFC. The diruthenium complex also acts as a redox mediator for the electrocatalytic oxidation of ammonia to dinitrogen at potentials as low as −255 mV versus Fc0/+ and operates below the oxygen reduction reaction potential in alkaline conditions, thus achieving a thermodynamic viability relevant for the future development of DAFCs.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 1945089 (7), 1945090 (2), 1945091 (4), 1945092 (3) and 2006690 (5). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. Source data for Figs. 3 and 4 and Extended Data Fig. 6 are provided with the article. Source data for Extended Data Fig. 1 can be found at https://figshare.com/articles/dataset/Berry_ED_Fig1_mnova/15060798. All other data supporting the findings of this study are available within the article and its Supplementary Information, or from the corresponding author upon reasonable request.
References
Elishav, O., Lewin, D. R., Shter, G. E. & Grader, G. S. The nitrogen economy: economic feasibility analysis of nitrogen-based fuels as energy carriers. Appl. Energy 185, 183–188 (2017).
Adli, N. M., Zhang, H., Mukherjee, S. & Wu, G. Review—ammonia oxidation electrocatalysis for hydrogen generation and fuel cells. J. Electrochem. Soc. 165, J3130–J3147 (2018).
Lan, R., Irvine, J. T. S. & Tao, S. Ammonia and related chemicals as potential indirect hydrogen storage materials. Int. J. Hydrogen Energy 37, 1482–1494 (2012).
Chen, J. G. et al. Beyond fossil fuel-driven nitrogen transformations. Science 360, eaar6611 (2018).
Zhao, Y. et al. An efficient direct ammonia fuel cell for affordable carbon-neutral transportation. Joule 3, 2472–2484 (2019).
Giddey, S., Badwal, S. P. S. & Kulkarni, A. Review of electrochemical ammonia production technologies and materials. Int. J. Hydrogen Energy 38, 14576–14594 (2013).
Hansson, J., Brynolf, S., Fridell, E. & Lehtveer, M. The potential role of ammonia as marine fuel—based on energy systems modeling and multi-criteria decision analysis. Sustainability 12, 3265 (2020).
Valera-Medina, A., Xiao, H., Owen-Jones, M., David, W. I. F. & Bowen, P. J. Ammonia for power. Prog. Energy Combust. Sci. 69, 63–102 (2018).
Wang, D. B. et al. Energy-efficient nitrogen reduction to ammonia at low overpotential in aqueous electrolyte under ambient conditions. ChemSusChem 11, 3416–3422 (2018).
Imamura, K. & Kubota, J. Electrochemical membrane cell for NH3 synthesis from N2 and H2O by electrolysis at 200 to 250 °C using a Ru catalyst, hydrogen-permeable Pd membrane and phosphate-based electrolyte. Sustain. Energy Fuels 2, 1278–1286 (2018).
Guo, X., Zhu, Y. & Ma, T. Lowering reaction temperature: electrochemical ammonia synthesis by coupling various electrolytes and catalysts. J. Energy Chem. 26, 1107–1116 (2017).
Siddharth, K., Chan, Y., Wang, L. & Shao, M. Ammonia electro-oxidation reaction: recent development in mechanistic understanding and electrocatalyst design. Curr. Opin. Electrochem. 9, 151–157 (2018).
Afif, A. et al. Ammonia-fed fuel cells: a comprehensive review. Renew. Sustain. Energy Rev. 60, 822–835 (2016).
Nakajima, K., Toda, H., Sakata, K. & Nishibayashi, Y. Ruthenium-catalysed oxidative conversion of ammonia into dinitrogen. Nat. Chem. 11, 702–709 (2019).
Habibzadeh, F., Miller, S. L., Hamann, T. W. & Smith, M. R. 3rd Homogeneous electrocatalytic oxidation of ammonia to N2 under mild conditions. Proc. Natl Acad. Sci. USA 116, 2849–2853 (2019).
Mahdi, R. B., Christine, G., Jeffery A., B. & Timothy H. W. Chemical and electrocatalytic ammonia oxidation by ferrocene. Preprint at https://doi.org/10.26434/chemrxiv.9729635.v1 (2019).
Bhattacharya, P. et al. Catalytic ammonia oxidation to dinitrogen by hydrogen atom abstraction. Angew. Chem. Int. Ed. 58, 11618–11624 (2019).
Zott, M. D., Garrido-Barros, P. & Peters, J. C. Electrocatalytic ammonia oxidation mediated by a polypyridyl iron catalyst. ACS Catal. 9, 10101–10108 (2019).
Dunn, P. L., Johnson, S. I., Kaminsky, W. & Bullock, R. M. Diversion of catalytic C–N bond formation to catalytic oxidation of NH3 through modification of the hydrogen atom abstractor. J. Am. Chem. Soc. 142, 3361–3365 (2020).
Zott, M. D. & Peters, J. C. Enhanced ammonia oxidation catalysis by a low-spin iron complex featuring cis coordination sites. J. Am. Chem. Soc. 143, 7612–7616 (2021).
A. J. Bard, L. R. F. Electrochemical Methods: Fundamentals and Applications 2nd edn (John Wiley, 2000).
Lindley, B. M., Appel, A. M., Krogh-Jespersen, K., Mayer, J. M. & Miller, A. J. M. Evaluating the thermodynamics of electrocatalytic N2 reduction in acetonitrile. ACS Energy Lett. 1, 698–704 (2016).
Pegis, M. L. et al. Standard reduction potentials for oxygen and carbon dioxide couples in acetonitrile and N,N-dimethylformamide. Inorg. Chem. 54, 11883–11888 (2015).
Coetzee, J. F. & Padmanabhan, G. R. Properties of bases in acetonitrile as solvent. IV. Proton acceptor power and homoconjugation of mono- and diamines. J. Am. Chem. Soc. 87, 5005–5010 (1965).
Corcos, A. R., Pap, J. S., Yang, T. & Berry, J. F. A synthetic oxygen atom transfer photocycle from a diruthenium oxyanion complex. J. Am. Chem. Soc. 138, 10032–10040 (2016).
Varela-Álvarez, A. et al. Rh2(ii,iii) Catalysts with chelating carboxylate and carboxamidate supports: electronic structure and nitrene transfer reactivity. J. Am. Chem. Soc. 138, 2327–2341 (2016).
Kornecki, K. P. et al. Direct spectroscopic characterization of a transitory dirhodium donor–acceptor carbene complex. Science 342, 351 (2013).
Berry, J. F. The role of three-center/four-electron bonds in superelectrophilic dirhodium carbene and nitrene catalytic intermediates. Dalton Trans. 41, 700–713 (2012).
Chakravarty, A. R., Cotton, F. A. & Tocher, D. A. New isomeric form of the ‘M2(OC5H3NCl)4’ core: a polar arrangement of the four 6-chloro-2-hydroxypyridinato (chp) ligands in a chlorodiruthenium(ii,iii) complex, Ru2Cl(chp)4. Inorg. Chem. 24, 1263–1267 (1985).
Corcos, A. R., Long, A. K. M., Guzei, I. A. & Berry, J. F. A synthetic cycle for nitrogen atom transfer featuring a diruthenium nitride intermediate. Eur. J. Inorg. Chem. 2013, 3808–3811 (2013).
Corcos, A. R. & Berry, J. F. Anilinopyridinate-supported Ru2x+ (x = 5 or 6) paddlewheel complexes with labile axial ligands. Dalton Trans. 46, 5532–5539 (2017).
Cotton, F. A., Kim, Y. & Yokochi, A. Regioisomerism displayed by the 6-chloro-2-oxopyridinate complexes of Ru24+ and Ru25+. Inorg. Chim. Acta 236, 55–61 (1995).
Brown, T. R., Dolinar, B. S., Hillard, E. A., Clérac, R. & Berry, J. F. Electronic structure of Ru2(ii,ii) oxypyridinates: synthetic, structural, and theoretical insights into axial ligand binding. Inorg. Chem. 54, 8571–8589 (2015).
Besora, M., Vidossich, P., Lledós, A., Ujaque, G. & Maseras, F. Calculation of reaction free energies in solution: a comparison of current approaches. J. Phys. Chem. A 122, 1392–1399 (2018).
Weatherburn, M. W. Phenol–hypochlorite reaction for determination of ammonia. Anal. Chem. 39, 971–974 (1967).
Neese, F. ORCA, an ab initio, density functional and semi-empirical program package, v.4.0.0.2 (Max Planck Institute for Chemical Energy Conversion, 2018).
Neese, F., Wennmohs, F., Hansen, A. & Becker, U. Efficient, approximate and parallel Hartree–Fock and hybrid DFT calculations. A ‘chain-of-spheres’ algorithm for the Hartree–Fock exchange. Chem. Phys. 356, 98–109 (2009).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu. J. Chem. Phys. 132, 154104 (2010).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle–Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297–3305 (2005).
Aravena, D., Neese, F. & Pantazis, D. A. Improved segmented all-electron relativistically contracted basis sets for the lanthanides. J. Chem. Theory Comput. 12, 1148–1156 (2016).
Lenthe, E. V., Baerends, E. J. & Snijders, J. G. Relativistic regular two-component Hamiltonians. J. Chem. Phys. 99, 4597–4610 (1993).
Pantazis, D. A., Chen, X.-Y., Landis, C. R. & Neese, F. All-electron scalar relativistic basis sets for third-row transition metal atoms. J. Chem. Theory Comput. 4, 908–919 (2008).
Bühl, M., Reimann, C., Pantazis, D. A., Bredow, T. & Neese, F. Geometries of third-row transition-metal complexes from density-functional theory. J. Chem. Theory Comput. 4, 1449–1459 (2008).
Pantazis, D. A. & Neese, F. All-electron scalar relativistic basis sets for the lanthanides. J. Chem. Theory Comput. 5, 2229–2238 (2009).
Pantazis, D. A. & Neese, F. All-electron scalar relativistic basis sets for the actinides. J. Chem. Theory Comput. 7, 677–684 (2011).
Pantazis, D. A. & Neese, F. All-electron scalar relativistic basis sets for the 6p elements. Theor. Chem. Acc. 131, 1292 (2012).
York, D. M. & Karplus, M. A smooth solvation potential based on the conductor-like screening model. J. Phys. Chem. A 103, 11060–11079 (1999).
PyMOL molecular graphics system, v.2.5.1 (Schrödinger, 2021).
Acknowledgements
We thank the Department of Energy for funding (DE-SC0021021). We also thank S.-C. Wang in the Hermans group as well as E. Canales and K. Rivera-Dones in the Huber group at UW-Madison for help with the mass spectrometry measurements of labelled nitrogen. We thank M. Aristov and A. Wheaton for assistance with crystallographic data. Crystallographic data were collected on a Cu Kα instrument that was funded by the NSF (CHE-1919350) and on a Mo Kα instrument funded by a generous gift from Paul and Margaret Bender.
Author information
Authors and Affiliations
Contributions
M.J.T. performed all electrochemical and spectroelectrochemical experiments and conducted the computational modelling. C.M.W. performed all synthesis and non-crystallographic characterization of diruthenium complexes, preparation of ammonia solutions, ammonia reactivity experiments and other spectroscopic experiments. T.R.B. performed preliminary synthesis and characterization on diruthenium ammine complexes that directly informed this work. S.V.P. performed crystallographic characterization of the diruthenium compounds. M.J.T., C.M.W. and J.F.B. wrote the manuscript. All authors provided feedback during the manuscript preparation and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
M.J.T., C.M.W., T.R.B., S.V.P. and J.F.B. have submitted a provisional patent application based on the work described here (US patent application number P200013US01).
Additional information
Peer review information Nature Chemistry thanks Dai Oyama and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 1H-NMR spectra monitoring the reaction of 2 with ammonia in CH3CN-d3.
Left: Portion of spectra focused on the Ar-H signals in the diruthenium complex. Right: Portion of spectra focused on the NH3/NH4+ signal shifting over the course of the experiment. Spectra are collected at 1-hour intervals.
Extended Data Fig. 2 Spectroelectrochemical cell used in controlled current Coulometry experiments.
Left: A photograph of the spectroelectrochemical cell, loaded with a solution of 5 with excess ammonia in the working electrode chamber and a solution of FcPF6 sacrificial oxidant in the counter electrode chamber. Right: A schematic of the spectroelectrochemical cell detailing its assembly.
Extended Data Fig. 3 Electrode and chemical processes occurring during controlled current Coulometry.
Diruthenium species in the [Ru2]4+ oxidation state are electrochemically oxidized to the [Ru2]5+ oxidation state at the surface of the reticulated vitreous carbon (RVC) working electrode (WE). The redox potential for this transformation is −255 mV vs Fc0/+ in CH3CN. Applied potential at WE is adjusted throughout the experiment to maintain a constant oxidizing current of +0.5 mA. Exogenous ammonia in solution spontaneously reacts with [Ru2]5+ species to produce dinitrogen and ammonium while regenerating [Ru2]4+ species. The ferrocenium cation in FcPF6 is electrochemically reduced to neutral ferrocene at the surface of the platinum counter electrode (CE). The working electrode and counter electrode chambers are divided by a fine glass frit allowing for ion exchange.
Extended Data Fig. 6 Titration of 2 with N2H4 in CH3CN monitored by electronic absorption spectroscopy.
The trace of absorbance at 533 nm versus equivalents of N2H4 shows a distinct end to the reaction after the addition 1.25 equivalents.
Supplementary information
Supplementary Information
Supplementary Figs. 1–20, Discussion and Tables 1–18. Supplementary tables providing DFT-optimized coordinates are included in a separate Excel spreadsheet in the additional supplementary files.
Supplementary Data 1
Crystallographic data for compound 2. CCDC reference 1945090.
Supplementary Data 2
Crystallographic data for compound 3. CCDC reference 1945092.
Supplementary Data 3
Crystallographic data for compound 4. CCDC reference 1945091.
Supplementary Data 4
Crystallographic data for compound 5. CCDC reference 2006690.
Supplementary Data 5
Crystallographic data for compound 7. CCDC reference 1945089.
Supplementary Data 7
Statistical source data for Supplementary Fig. 8.
Supplementary Data 8
Statistical source data for Supplementary Fig. 10.
Supplementary Data 9
Statistical source data for Supplementary Fig. 11.
Supplementary Data 10
Statistical source data for Supplementary Fig. 12.
Supplementary Data 11
Statistical source data for Supplementary Fig. 13.
Supplementary Data 12
Statistical source data for Supplementary Fig. 14.
Supplementary Data 13
Statistical source data for Supplementary Fig. 15.
Supplementary Data 14
Statistical source data for Supplementary Fig. 16.
Supplementary Data 15
Statistical source data for Supplementary Fig. 17.
Supplementary Data 16
Statistical source data for Supplementary Fig. 18.
Supplementary Data 17
Statistical source data for Supplementary Fig. 19.
Supplementary Data 18
Statistical source data for Supplementary Fig. 20.
Source data
Source Data Fig. 3
Statistical source data for Fig. 3.
Source Data Fig. 4
Statistical source data for Fig. 4.
Source Data Extended Data Fig. 6
Statistical source data for Extended Data Fig. 6.
Rights and permissions
About this article
Cite this article
Trenerry, M.J., Wallen, C.M., Brown, T.R. et al. Spontaneous N2 formation by a diruthenium complex enables electrocatalytic and aerobic oxidation of ammonia. Nat. Chem. 13, 1221–1227 (2021). https://doi.org/10.1038/s41557-021-00797-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00797-w
This article is cited by
-
Recent Progress in Electrochemical Synthesis and Conversion of Nitrates in Aqueous Electrolyte
Topics in Catalysis (2024)
-
Direct synthesis of hydrazine by efficient electrochemical ruthenium-catalysed ammonia oxidation
Nature Catalysis (2023)
-
Wettability control of defective TiO2 with alkyl acid for highly efficient photocatalytic ammonia synthesis
Nano Research (2023)