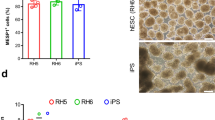
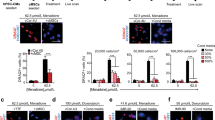
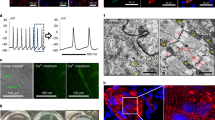
Abstract
A major hurdle in cardiac cell therapy is the lack of a bona fide autologous stem-cell type that can be expanded long-term and has authentic cardiovascular differentiation potential. Here we report that a proliferative cell population with robust cardiovascular differentiation potential can be generated from mouse or human fibroblasts via a combination of six small molecules. These chemically induced cardiovascular progenitor cells (ciCPCs) self-renew long-term in fully chemically defined and xeno-free conditions, with faithful preservation of the CPC phenotype and of cardiovascular differentiation capacity in vitro and in vivo. Transplantation of ciCPCs into infarcted mouse hearts improved animal survival and cardiac function up to 13 weeks post-infarction. Mechanistically, activated fibroblasts revert to a plastic state permissive to cardiogenic signals, enabling their reprogramming into ciCPCs. Expanded autologous cardiovascular cells may find uses in drug discovery, disease modelling and cardiac cell therapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. Source data for the figures are provided with this paper. The RNA-sequencing data are available from the NCBI GEO database, via the accession number GSE159081. Source data are provided with this paper.
References
Laugwitz, K. L. et al. Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature 433, 647–653 (2005).
van Berlo, J. H. et al. c-kit+ cells minimally contribute cardiomyocytes to the heart. Nature 509, 337–341 (2014).
Vagnozzi, R. J. et al. Genetic lineage tracing of Sca-1(+) cells reveals endothelial but not myogenic contribution to the murine heart. Circulation 138, 2931–2939 (2018).
Neidig, L. E. et al. Evidence for minimal cardiogenic potential of stem cell antigen 1-positive cells in the adult mouse heart. Circulation 138, 2960–2962 (2018).
Chien, K. R. et al. Regenerating the field of cardiovascular cell therapy. Nat. Biotechnol. 37, 232–237 (2019).
Li, Y. et al. Genetic lineage tracing of nonmyocyte population by dual recombinases. Circulation 138, 793–805 (2018).
Vagnozzi, R. J. et al. An acute immune response underlies the benefit of cardiac stem cell therapy. Nature 577, 405–409 (2020).
Zhang, Y. et al. Expandable cardiovascular progenitor cells reprogrammed from fibroblasts. Cell Stem Cell 18, 368–381 (2016).
Lalit, P. A. et al. Lineage reprogramming of fibroblasts into proliferative induced cardiac progenitor cells by defined factors. Cell Stem Cell 18, 354–367 (2016).
Li, X., Xu, J. & Deng, H. K. Small molecule-induced cellular fate reprogramming: promising road leading to Rome. Curr. Opin. Genet. Dev. 52, 29–35 (2018).
De, D., Halder, D., Shin, I. & Kim, K. K. Small molecule-induced cellular conversion. Chem. Soc. Rev. 46, 6241–6254 (2017).
Cao, N. et al. Conversion of human fibroblasts into functional cardiomyocytes by small molecules. Science 352, 1216–1220 (2016).
Qian, L. et al. In vivo reprogramming of murine cardiac fibroblasts into induced cardiomyocytes. Nature 485, 593–598 (2012).
Zhang, M. L. et al. Pharmacological reprogramming of fibroblasts into neural stem cells by signaling-directed transcriptional activation. Cell Stem Cell 18, 653–667 (2016).
Cao, S. T. et al. Chemical reprogramming of mouse embryonic and adult fibroblast into endoderm lineage. J. Biol. Chem. 292, 19122–19132 (2017).
Chen, G. et al. Chemically defined conditions for human iPSC derivation and culture. Nat. Methods 8, 424–429 (2011).
Bao, X. et al. Long-term self-renewing human epicardial cells generated from pluripotent stem cells under defined xeno-free conditions. Nat. Biomed. Eng. 1, 0003 (2016).
Yasuda, S. Y. et al. Chemically defined and growth-factor-free culture system for the expansion and derivation of human pluripotent stem cells. Nat. Biomed. Eng. 2, 173–182 (2018).
Wamstad, J. A. et al. Dynamic and coordinated epigenetic regulation of developmental transitions in the cardiac lineage. Cell 151, 206–220 (2012).
Devine, W. P., Wythe, J. D., George, M., Koshiba-Takeuchi, K. & Bruneau, B. G. Early patterning and specification of cardiac progenitors in gastrulating mesoderm. Elife 3, e03848 (2014).
Moretti, A. et al. Multipotent embryonic isl1+ progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 127, 1151–1165 (2006).
Johnston, P. V. et al. Engraftment, differentiation, and functional benefits of autologous cardiosphere-derived cells in porcine ischemic cardiomyopathy. Circulation 120, 1075–1083 (2009).
Terrovitis, J. V., Smith, R. R. & Marban, E. Assessment and optimization of cell engraftment after transplantation into the heart. Circ. Res. 106, 479–494 (2010).
Lutgens, E. et al. Chronic myocardial infarction in the mouse: cardiac structural and functional changes. Cardiovasc Res 41, 586–593 (1999).
Li, R. H. et al. A mesenchymal-to-epithelial transition initiates and is required for the nuclear reprogramming of mouse fibroblasts. Cell Stem Cell 7, 51–63 (2010).
Zhao, Y. et al. A XEN-like state bridges somatic cells to pluripotency during chemical reprogramming. Cell 163, 1678–1691 (2015).
Han, X. et al. A molecular roadmap for induced multi-lineage trans-differentiation of fibroblasts by chemical combinations. Cell Res. 27, 386–401 (2017).
Thiery, J. P., Acloque, H., Huang, R. Y. & Nieto, M. A. Epithelial-mesenchymal transitions in development and disease. Cell 139, 871–890 (2009).
McCabe, M. T. et al. EZH2 inhibition as a therapeutic strategy for lymphoma with EZH2-activating mutations. Nature 492, 108–112 (2012).
Balasubramanyam, K., Swaminathan, V., Ranganathan, A. & Kundu, T. K. Small molecule modulators of histone acetyltransferase p300. J. Biol. Chem. 278, 19134–19140 (2003).
Gaspar-Maia, A., Alajem, A., Meshorer, E. & Ramalho-Santos, M. Open chromatin in pluripotency and reprogramming. Nat. Rev. Mol. Cell Biol. 12, 36–47 (2011).
Zhou, Y. et al. Bmi1 is a key epigenetic barrier to direct cardiac reprogramming. Cell Stem Cell 18, 382–395 (2016).
Guo, S. et al. Nonstochastic reprogramming from a privileged somatic cell state. Cell 156, 649–662 (2014).
Wang, Y. F. et al. Conversion of human gastric epithelial cells to multipotent endodermal progenitors using defined small molecules. Cell Stem Cell 19, 449–461 (2016).
Wang, B. et al. D609 protects retinal pigmented epithelium as a potential therapy for age-related macular degeneration. Signal Transduct. Target Ther. 5, 20 (2020).
Kattman, S. J. et al. Stage-specific optimization of activin/nodal and BMP signaling promotes cardiac differentiation of mouse and human pluripotent stem cell lines. Cell Stem Cell 8, 228–240 (2011).
Blin, G. et al. A purified population of multipotent cardiovascular progenitors derived from primate pluripotent stem cells engrafts in postmyocardial infarcted nonhuman primates. J. Clin. Invest. 120, 1125–1139 (2010).
Onder, T. T. et al. Chromatin-modifying enzymes as modulators of reprogramming. Nature 483, 598–602 (2012).
Li, X. et al. Direct reprogramming of fibroblasts via a chemically induced XEN-like state. Cell Stem Cell 21, 264–273.e7 (2017).
Kunath, T. et al. Imprinted X-inactivation in extra-embryonic endoderm cell lines from mouse blastocysts. Development 132, 1649–1661 (2005).
Moerkamp, A. T. et al. Extraembryonic endoderm cells as a model of endoderm development. Dev. Growth Differ. 55, 301–308 (2013).
Birket, M. J. et al. Expansion and patterning of cardiovascular progenitors derived from human pluripotent stem cells. Nat. Biotechnol. 33, 970–979 (2015).
Chen, I. Y. & Wu, J. C. Finding expandable induced cardiovascular progenitor cells. Circ. Res. 119, 16–20 (2016).
Masuda, S. et al. Expandable progenitors from induced pluripotent stem cells. Nat. Rev. Cardiol. 13, 574 (2016).
Yamakawa, H. & Fukuda, K. Comment on: expandable cardiovascular progenitor cells reprogrammed from fibroblasts. Stem Cell Investig. 3, 89 (2016).
Kannappan, R. et al. Functionally competent DNA damage-free induced pluripotent stem cell-derived cardiomyocytes for myocardial repair. Circulation 140, 520–522 (2019).
Gao, L. et al. Large cardiac muscle patches engineered from human induced-pluripotent stem cell-derived cardiac cells improve recovery from myocardial infarction in swine. Circulation 137, 1712–1730 (2018).
Zhu, K. et al. Lack of remuscularization following transplantation of human embryonic stem cell-derived cardiovascular progenitor cells in infarcted nonhuman primates. Circ. Res. 122, 958–969 (2018).
Zhu, W., Zhao, M., Mattapally, S., Chen, S. & Zhang, J. CCND2 overexpression enhances the regenerative potency of human induced pluripotent stem cell-derived cardiomyocytes: remuscularization of injured ventricle. Circ. Res. 122, 88–96 (2018).
Konermann, S. et al. Genome-scale transcriptional activation by an engineered CRISPR-Cas9 complex. Nature 517, 583–588 (2015).
Shalem, O. et al. Genome-scale CRISPR-Cas9 knockout screening in human cells. Science 343, 84–87 (2014).
Wang, H. et al. Small molecules enable cardiac reprogramming of mouse fibroblasts with a single factor, oct4. Cell Rep. 6, 951–960 (2014).
Lin, Y. et al. Heparin promotes cardiac differentiation of human pluripotent stem cells in chemically defined albumin-free medium, enabling consistent manufacture of cardiomyocytes. Stem Cells Transl. Med. 6, 527–538 (2017).
Tohyama, S. et al. Distinct metabolic flow enables large-scale purification of mouse and human pluripotent stem cell-derived cardiomyocytes. Cell Stem Cell 12, 127–137 (2013).
Cao, N. et al. Highly efficient induction and long-term maintenance of multipotent cardiovascular progenitors from human pluripotent stem cells under defined conditions. Cell Res. 23, 1119–1132 (2013).
Ewels, P., Magnusson, M., Lundin, S. & Käller, M. MultiQC: summarize analysis results for multiple tools and samples in a single report. Bioinformatics 32, 3047–3048 (2016).
Wingett, S. W. & Andrews, S. FastQ Screen: A tool for multi-genome mapping and quality control. F1000Res. 7, 1338 (2018).
Anders, S., Pyl, P. T. & Huber, W. HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Li, H. et al. The sequence alignment/map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Acknowledgements
We thank Y. Zhou (University of North Carolina) for technical support and all lab members for helpful discussion. N.C. was funded by the National Key R&D Program of China (2018YFA0109100 and 2018YFA050830), the National Natural Science Foundation of China (92057113, 82061148011 and 31771508), and the Guangdong Innovative and Entrepreneurial Research Team Program (2016ZT06S029). J.W. was funded by the National Key R&D Program of China (2018YFA0109600) and the National Natural Science Foundation of China (81700233). Z.Z. was funded by the National Key R&D Program of China (2019YFA0111500).
Author information
Authors and Affiliations
Contributions
J.W., S.G. and N.C. conceived the project, carried out the experiments and wrote the manuscript. Zihao Chen, W.C. and T.X. performed bioinformatics analysis. F.L., H.X., Z.L., L.W., Zhongyan Chen, D.C., X.C., F.Z., Z.Z. and M.Z. performed data analysis, provided materials or assisted with the experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Ke Cheng, Timothy Kamp and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Main Supplementary Information
Supplementary figures, tables and video captions.
Supplementary Video 1
Beating cluster generated after MEF-derived ciCPCs were cultured in cardiac differentiation conditions for 7 d.
Supplementary Video 2
Beating cluster generated after TTF-derived ciCPCs were cultured in cardiac differentiation conditions for 7 d.
Supplementary Video 3
Beating cluster generated after HFF-derived ciCPCs were cultured in cardiac differentiation conditions for 14 d.
Source data
SD for Fig. 1
Source data.
SD for Fig. 2
Source data.
SD for Fig. 4
Source data.
SD for Fig. 5
Source data.
SD for Fig. 6
Source data.
SD for Fig. 7
Source data.
Rights and permissions
About this article
Cite this article
Wang, J., Gu, S., Liu, F. et al. Reprogramming of fibroblasts into expandable cardiovascular progenitor cells via small molecules in xeno-free conditions. Nat. Biomed. Eng 6, 403–420 (2022). https://doi.org/10.1038/s41551-022-00865-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-022-00865-7
This article is cited by
-
Harnessing stem cell and lineage reprogramming technology to treat cardiac fibrosis
Cell Regeneration (2023)