Abstract
Neurological soft signs (NSS) are common in patients with schizophrenia. However, the neural substrates of NSS remain poorly understood. Using legacy PubMed, we performed a systematic review and included studies that assessed NSS and obtained neuroimaging data in patients with a schizophrenia spectrum disorder published up to June 2020. We systematically reviewed 35 relevant articles. Studies consistently implicate the basal ganglia and cerebellum as structural substrates of NSS and suggest that somatomotor and somatosensory regions as well as areas involved in visual processing and spatial orientation may underlie NSS in psychosis spectrum disorders. Additionally, dysfunction of frontoparietal and cerebellar networks has been implicated in the pathophysiology of NSS. The current literature outlines several structural and functional brain signatures that are relevant for NSS in schizophrenia spectrum disorder. The majority of studies assessed gray matter structure, but only a few studies leveraged other imaging methods such as diffusion weighted imaging, or molecular imaging. Due to this, it remains unclear if white matter integrity deficits or neurometabolic alterations contribute to NSS in the illness. While a substantial portion of the literature has been conducted in patients in the early illness stages, mitigating confounds of illness chronicity, few studies have been conducted in antipsychotic medication-naïve patients, which is a clear limitation. Furthermore, only little is known about the temporal evolution of NSS and associated brain signatures. Future studies addressing these pivotal gaps in our mechanistic understanding of NSS will be important.
Similar content being viewed by others
Introduction
Neurological soft signs (NSSs) are minor abnormalities in motor coordination, sensory integration, sequencing of complex motor movements, and the disinhibition of those movements1. “Soft” signs are conceptualized as subclinical, non-localized signs of neurological abnormalities, whereas “hard” signs typically refer to impairments in basic sensory, motor, and reflex behaviors which can usually be directly mapped to a specific brain region2. This dichotomy largely exists because of the difficulty in attributing those soft signs to specific brain abnormalities; however, as research and technology advance, that narrative is shifting.
NSSs occur in the majority of patients with schizophrenia2. They are present more frequently than in healthy subjects and have been suggested to represent a core feature of the illness3,4; meta-analyses find that up to 73% of schizophrenia patients perform outside the range of healthy participants on aggregate NSS measures1. Interestingly, NSSs are associated with psychosis proneness within the general population5, and motor abnormalities in adolescents at risk for psychotic disorders may predict subsequent conversion to psychosis6,7,8. NSSs are significantly more common in people with schizophrenia than in their first-degree relatives, and are more common in first-degree relatives than in healthy controls9. While some studies report that NSS scores are diagnostically nonspecific across psychosis spectrum disorders10,11 others found differences in NSS severity between patients with schizophrenia and bipolar disorder12,13.
Studies measuring NSS suggest an abnormal developmental trajectory of NSS in schizophrenia patients14,15. In healthy controls, the relationship between NSS and age tends to be a U-shaped pattern, whereas in schizophrenia patients, the relationship is parallel, with a flat but overall elevated pattern14.
The presence of NSSs has been found to be independent of demographic variables and most medication variables2, supporting the idea that NSSs are related to the pathophysiology and pointing towards a neurodevelopmental component in the pathophysiology2. However, others found NSSs to vary over the clinical course of the disorder16. It appears that NSSs may decrease with remission of psychotic symptoms, especially in patients with a remitting chronic course, but not to levels typically seen in healthy controls16 suggesting potential clinical utility for these measures in monitoring of disease progression.
Currently, NSSs are not generally assessed in clinical practice8 and there is not a universally accepted assessment tool for NSS. Different existing instruments include various items and factor levels17,18,19,20,21,22. The most common instruments include the Neurological Evaluation Scale (NES) which includes 28 items that fall into one of three functional areas of interest (integrative sensory dysfunction, motor incoordination, and impaired sequencing of complex motor acts) or an “Other” domain17; the Heidelberg Scale with 17 items which fall into one of five factors (motor coordination, integrative functions, complex motor tasks, right/left and spatial orientation, and hard signs)22; and the Cambridge Neurological Inventory (CNI) with eight categories (hard neurological signs, motor coordination, sensory integration, primitive reflexes, tardive dyskinesia, catatonic signs, parkinsonism, and failure to suppress inappropriate response)18. The different scales and individual item scores are not directly comparable, but subscales, especially with respect to motor sequencing, motor coordination and sensory integration, are contrastable23.
In 2014, an activation likelihood estimation (ALE) meta-analysis of six structural and fifteen functional neuroimaging studies reported that that NSS patients with schizophrenia and related psychotic disorders were associated with structural and functional abnormalities of brain regions of the cerebello-thalamo-prefrontal network24. Notably, the majority of studies included in this analysis used a go/no-go task as a proxy for motor disinhibition rather than using direct assessments that capture NSS across domains. Building on these efforts, we performed a systematic review of the neuroimaging studies that comprehensively characterized NSS with instruments designed to assess NSS across domains, with the overarching goal to synthesize the neuroimaging literature of neural substrates of NSS in patients with a schizophrenia spectrum disorder.
Results
Study identification
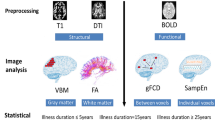
Figure 1 describes outcomes at each level of our study identification process. Of the 35 relevant articles we have included 29 structural MRI studies, 4 functional MRI, one study with both structural and functional MRI, and one molecular imaging study in this systematic review.
Study characteristics
Sixteen of the structural studies, all of the functional studies, and the study featuring both methods compared patients to healthy controls, while thirteen structural and the one SPECT study only included patients. Of the twenty-one studies comparing patients to healthy controls, fourteen reported higher NSS scores in patients than in healthy controls. Patients’ illness stages in schizophrenia were chronic (n = 14), first episode (n = 15), and recent onset (n = 5); the SPECT study did not specify the patients’ illness stage.
Structural MRI findings
A total of 30 structural imaging studies were included in the systematic review (Table 1a, b). The majority of studies quantified structural indices using T1 weighted imaging, one study used diffusion imaging, and one study used magnetization transfer imaging. Seventeen studies compared structural differences between patients and healthy controls (Table 1a); thirteen investigated patients only (Table 1b).
Associations between global NSS severity and structural imaging measures
To the best of our knowledge, the first neuroimaging study examining biological correlates of soft signs was conducted in 1985, using computed tomography to assess the ventricular-brain ratio. While an increase of ventricle size was evident in patients compared to controls, this was not associated with overall NSS severity25. In a later MRI study, ventricle volumes and lateral ventricular enlargement were found to be associated with NSS severity on at least one item of sensory integration, motor coordination, and sequencing of complex motor acts26, suggesting ventricular volume abnormalities (likely reflecting brain volume loss) may be relevant for NSS severity globally.
Cortical gray matter thickness, surface, gyrification, and sulcation. No differences in cortical thickness or surface area were reported in first episode patients with low versus high NSS burden27. While global cortical sulcus indices were lower in first episode patients with soft signs compared to those without, sulcation indices did not correlate with NSS severity28. In contrast, total NSS scores were found to be associated with gyrification in the orbitofrontal, superior temporal, superior parietal, and supramarginal cortices in recent onset schizophrenia patients29. In chronic patients, reduced cortical thickness in the middle frontal, inferior temporal, superior parietal, postcentral and supramarginal cortex was found to be associated with greater global NSS severity30.
Cortical gray matter volumes or densities. The largest of these studies was conducted in 102 inpatients diagnosed with a first psychotic episode. Global NSS severity was associated with decreased gray matter density in the precentral, postcentral, inferior parietal, and inferior occipital gyri31. The finding was a partial replication of an earlier study in first episode patients that reported an association between lower precentral and postcentral cortical gray matter density and NSS severity. Here, negative correlations were also observed with the lingual gyrus and insula32. Another study in first episode patients reported a relationship between global NSS severity and the dorsolateral prefrontal cortex volumes33. In a longitudinal study monitoring the temporal evolution of soft signs in first episode patients, Kong and colleagues report that those with persistent NSSs after one year of follow up showed more pronounced gray matter volume reductions in the frontal lobe, suggesting that persistence of NSSs may be a clinical marker of gray matter disease progression in first episode patients34.
Subcortical and cerebellar volumes or densities. Global soft sign severity in first episode psychosis patients was found to be associated with volume reductions in the putamen35, thalamus32, brain stem36, and cerebellum37,38. Similar brain signatures were also reported in recent onset schizophrenia patients where associations between NSSs and brain stem volumes36,39, as well as cerebellum volumes11,32,34,37,38,39,40 were detected. Consistent with this, Venkatasubramanian and colleagues reported an association between global soft sign severity and lower gray matter density in the caudate and cerebellum in antipsychotic medication-naïve patients with chronic schizophrenia40.
White matter integrity. Considerably fewer studies have assessed putative associations between white matter measures and soft signs. One study investigated the corpus callosum in chronic schizophrenia patients and found that total NSS scores were associated with the width of the genu of the corpus callosum, but did not find associations with any morphological features of the other six corpus callosum subregions41. In medicated first episode psychosis patients, higher NSS scores were associated with lower white matter density in the inferior frontal gyrus and the cerebellum32. In contrast, a large study conducted in first episode patients failed to reveal associations between white matter volumes and total NSS scores, or differences in white matter volumes between patients with a low or high NSS symptom burden35. This lack of associations between total NSS scores and white matter volumes was later replicated in a study focused on cerebello-thalamo-prefrontal structures in first episode patients33. Another study focused on the cerebellum reported a negative correlation between soft sign severity and white matter volume of the midbrain and cerebellum in recent onset schizophrenia patients39. The only magnetization transfer contrast study examining white matter integrity found a decrease in the temporal magnetization transfer ratio in patients compared to controls, suggestive of myelin or axonal disruption, but did not report an association with NSS total scores42.
Sensory integration and structural imaging measures
Cortical gray matter thickness, surface, gyrification, and sulcation. Studies examining associations of cortical thickness and/or cortical surface area in context of sensory integration deficits found that lower cortical thickness in the pre- and post-central gyrus was associated with greater symptom severity in first episode patients27. In the same patients, the cortical surface area was greater in the lateral occipital cortex in those who had a higher sensory integration scores compared to those with lower scores27. Gyrification in the lateral occipital cortex was associated with sensory integration scores in recent onset schizophrenia patients29. Interestingly, no associations between cortical thickness and sensory integration scores were reported in a small study of patients with chronic schizophrenia30.
Cortical gray matter volumes or densities. In first episode patients, higher sensory integration scores were found to be correlated with reduced gray matter density in the superior, medial, and inferior frontal gyri31, as well as reduced gray matter volume in the insula, inferior frontal, middle temporal, and lingual gyrus35, but others reported no relationships between sensory integration scores and gray matter volumes33.
Subcortical volumes or densities. In first episode patients, thalamus35,43, pallidum, and putamen volume reductions were associated with greater sensory integration scores35. Using a slightly different approach compared to the traditional subscales for the NSS, Keshavan and colleagues used a factor analysis method to examine structural imaging correlates of soft signs in a large group of antipsychotic medication-naïve first episode psychosis patients. The factor that was comprised of complex tasks that were cognitively demanding and involved sensory processing showed negative correlations with caudate and cerebellum volumes11.
White matter integrity. First episode psychosis patients with high sensory integration scores had greater left internal capsule volumes compared to those with low sensory integration scores35. In chronic patients, the width of the splenium of the corpus callosum was found to be greater in those with low sensory integration scores compared to those with high sensory integration scores41.
Motor coordination and structural imaging measures
Cortical gray matter thickness, surface, gyrification, and sulcation. Studies examining associations of cortical thickness and/or cortical surface area in the context of motor coordination subscale scores found that first episode patients with higher scores showed reduced cortical thickness in the inferior parietal, superior temporal, and fusiform gyrus, as well as reduced surface area in the superior frontal and orbitofrontal gyrus27. In recent onset schizophrenia patients, higher motor coordination scores were associated with reduced cortical thickness in the precentral gyrus29, and in chronic patients, this association was seen in the inferior frontal and postcentral gyri30. Gyrification in the inferior temporal and supramarginal cortices were associated with motor coordination scores in recent onset schizophrenia patients29.
Cortical gray matter volumes or densities. In first episode patients, greater motor coordination scores were associated with decreased gray matter density in the precentral gyrus and inferior parietal lobe31.
Subcortical volumes or densities. In first episode patients, thalamus volumes were positively correlated with motor coordination scores33. In contrast, motor coordination abnormalities were found to be associated with volume reductions in the caudate11, putamen35, brain stem36, cerebellar volumes11, cerebellar gray matter31, and cerebellar white matter33.
White matter integrity. In first episode patients, one group found an inverse correlation between motor coordination scores and corpus callosum volumes32, whereas others found that patients with high motor coordination scores had greater left internal capsule volumes compared to those with low motor coordination scores35.
Sequencing of complex motor tasks and structural imaging measures
Cortical gray matter thickness, surface, gyrification and sulcation. No relationships between sequencing of complex motor task scores and cortical thickness or surface area were reported27. On the other hand, reduced gyrification in the right temporal pole29 and supramarginal and fusiform cortices29 were found to be associated with complex motor task scores in recent onset schizophrenia patients.
Cortical gray matter volumes or densities. In first episode patients, decreased gray matter densities in the superior and middle frontal gyri31,40, precentral gyrus31, insula31, and posterior cingulate cortex40 were found to be associated with greater sequencing of complex motor tasks scores. However, others report no such associations between cortical gray matter volumes and motor sequencing scores35.
Subcortical volumes or densities. In terms of subcortical and cerebellar regions, scores on the complex motor task subscale were associated with volume reductions in the putamen,40,44 midbrain40, brain stem36, and cerebellum38,40.
White matter integrity. One study investigated the corpus callosum in chronic schizophrenia patients and found that scores on the sequencing of complex motor acts scale were positively correlated with the genu width and genu area, and negatively correlated with the corpus callosum rostral body area41. A tractography study of the corticospinal tract and superior cerebellar peduncle, which had both tracts involved in motor sequencing, found that patients with sequencing abnormalities showed reduced fractional anisotropy in the superior cerebellar peduncle compared to patients without sequencing abnormalities and healthy controls. In contrast, only patients without sequencing abnormalities had reduced corticospinal tract fractional anisotropy and increased radial diffusivity. Both radial and mean diffusivity in the superior cerebellar peduncle were correlated with severity of sequencing abnormality scores in the entire group, suggesting that white matter integrity is involved in NSSs45.
Functional MRI findings
Of the five functional studies included here, four utilized task fMRI, and one was a combined resting state fMRI and structural MRI study (Table 2). All studies were conducted in patients who were on antipsychotic medications at the time of the assessment, and all but one focused on patients in the chronic disease stage.
The four studies that assessed brain function during task performance all used motor tasks. Both Chan et al.46 and Zemankova et al.47 reported aberrant task activation in frontoparietal regions. In first episode patients, reduced task activation and functional connectivity were reported, and patients failed to increase frontal activation with increasing task difficulty46. In contrast, in chronic patients an increase in frontoparietal task activation was noted; this overactivation was interpreted as evidence of a compensatory strategy for patients to achieve adequate motor performance. Complementary frontoparietal network functional connectivity analysis further suggested that an interhemispheric cortical inhibition deficit which was most pronounced in patients who had movement sequencing difficulties47. However, others did not find evidence of aberrant task activation in patients with chronic schizophrenia, regardless of whether patients did or did not display sequencing difficulties44.
Nonetheless, movement sequencing abnormalities in patients in this study were found to be linked to abnormal cortico-cerebellar functional connectivity. In a region of interest analysis examining activation of the sensorimotor cortex and supplemental motor cortex, Schröder et al. found reduced task activation in both regions in patients with chronic schizophrenia. The extent of task activation between the sensorimotor cortex and the supplemental motor cortex was correlated, suggesting a functional coupling of these brain regions. The authors concluded that dysfunction in these brain regions may contribute to NSS severity48.
The only relevant study assessing brain function at rest did so in a multimodal approach, also considering gray matter structural integrity in the analysis49. Interestingly, NSS motor scores were found to be associated with a joint structural/functional group-discriminating component encompassing frontoparietal and frontocerebellar networks in patients.
Taken together, aberrant function in motor, frontoparietal and cerebellar networks may be associated with NSS in patients with a schizophrenia spectrum disorder.
Molecular imaging findings
One molecular imaging study using SPECT was included in the systematic review. This report examined dopamine D2 receptor binding in a group of 23 antipsychotic medication-naïve schizophrenia patients. Importantly, they found that NSSs are already present in patients who have never been medicated, and that these may decrease in severity after antipsychotic treatment. However, no direct relationships between NSS severity and baseline D2 receptor binding or antipsychotic medication related change in D2 binding were detected50.
Discussion
Here, we performed a systematic review of the neuroimaging literature of NSS in patients with a schizophrenia spectrum disorder with the goal to synthesize the existing literature. We included studies which have a neuroimaging component and a standardized NSS measure. Studies consistently implicate the basal ganglia and cerebellum as structural substrates of NSS and suggest that structural alterations in somatomotor and somatosensory regions, as well as areas involved in visual processing and spatial orientation, may underlie NSS in psychosis spectrum disorders. Additionally, dysfunction of frontoparietal and cerebellar networks has been implicated in the pathophysiology of NSS in several studies. In contrast, the limited published literature on white matter volume deficits and dopamine D2 receptor dysfunction suggest that these markers may not play a primary role in the development of NSS, though those findings clearly warrant replication.
Our results are consistent with findings from a previous meta-analysis24 that included six structural and fifteen functional neuroimaging studies (the ALE meta-analysis included functional studies that used a go/no-go task in lieu of measurements designed to quantitatively assess NSSs across domains). Despite the difference in inclusion criteria, they identified a number of overlapping brain regions, including frontal regions, basal ganglia, and the cerebellum, that were associated with soft signs. Our findings not only confirm prior meta-analytic data, but they also expand efforts for synthesis of the literature by attempting to dissect the neurobiological correlates of NSS with respect to individual domains of soft signs. Interestingly, studies reported associations between NSS and basal ganglia structural integrity across domains. Only a few findings regarding involvement of a specific brain region in an individual domain of NSSs are replicated at this point. Sensory integration scores were found to be associated with lateral occipital cortex integrity in two studies27,29 and the association of motor coordination scores and precentral as well as inferior parietal gray matter integrity was reported more than once31,32. The lack of replication in structural correlates of NSS in specific domains may be attributable to a number of factors, including the small number of studies that investigated specific neurobiological signatures of NSS within a specific domain, the different instruments used to assess NSS, the heterogeneity in sample characteristics, and the variety of image analysis approaches, to name just a few. Similarly, the limited number of functional imaging studies conducted prevents us from making definitive conclusions on the functional circuitry of NSS.
In conclusion, a number of scientific questions around the central theme of neurobiological signatures of soft signs in psychosis spectrum disorders remain unanswered. For example, the literature is not consistent as to whether soft signs change across time in patients with psychosis spectrum disorders51,52,53. A meta-analysis suggests that the temporal evolution of soft signs may be a function of the illness course16, and different brain structures may differentially contribute to soft signs at different disease stages54, but the underlying pathophysiology remains unclear. To our knowledge, only two longitudinal studies examining NSSs also included an imaging component. One included 20 first episode schizophrenia patients who were followed over one year. While a significant decrease in NSS scores was found in the overall group, the subgroup without reductions in NSS showed pronounced gray matter reductions in the frontal lobe, cingulate gyrus, and cerebellum, suggesting that persistent NSS may be associated with progressive brain changes34. Another study assessed NSS severity in antipsychotic-naïve patients at baseline and after a variable length of follow-up, with imaging done at baseline. Here, smaller left dorsolateral prefrontal lobe volume at baseline predicted greater negative symptoms and poorer functional outcome on follow-up, but there was no significant change in NSS from baseline to follow-up55. Clearly, well-powered, longitudinal studies are needed to comprehensively characterize the neurobiological correlates of temporal changes in NSS, especially given its potential for biomarker development.
Similarly, the extent to which antipsychotic medications confound imaging findings with respect to soft signs has not been determined. The majority of studies investigated patients who were medicated at the time of the assessment, which can influence measurements on two levels. First, antipsychotic medications could affect the extent to which soft signs are behaviorally expressed56,57. NSS severity may improve with treatment and over time58. On the other hand, a study in children and adolescents at ultra-high risk (UHR) for psychosis found that antipsychotic treatment does not affect severity of NSS59, and another found no relationship between current antipsychotic dosage and NSS severity60. Second, medications do affect brain structure, function, and neurochemistry61,62,63,64,65,66,67,68,69,70,71. In this context, pharmacological challenge studies could help determine to what extent antipsychotic medications play a role in the phenotypic and neurobiological expression of NSSs.
This review must be considered in the context of some limitations. It is important to note that we did not use quantitative meta-analytic techniques, in part because the heterogeneity in data acquisition and analytic techniques was significant, which would make it difficult to make meaningful quantitative comparisons. Most studies had a relatively small sample size, and the majority of studies were conducted in patients who were treated with an antipsychotic medication. While there have been a relatively large number of studies conducted in first-episode psychosis patients, only two were performed in antipsychotic medication-naïve patients, which is a clear limitation. Next, it is important to note that the vast majority of studies assessed gray matter structure, but only few studies leveraged other imaging methods such as diffusion weighted imaging, or molecular imaging. Because of this, it remains unclear if white matter integrity deficits or neurometabolic alterations contribute to NSS in the illness. Another limitation is the use of various soft sign instruments in the reviewed studies, which could have contributed to the limited replications of findings. Developing a gold standard instrument for NSS assessments could be helpful in this regard. Future studies filling these important gaps in the literature will be helpful in advancing our mechanistic understanding of NSS and in assessing the potential of clinical and neurobiological markers of NSS in patients suffering from a psychosis spectrum disorder in the development neuroimaging biomarkers72.
Methods
Eligibility criteria
We included studies that assessed NSSs and obtained neuroimaging data in patients with a schizophrenia spectrum disorder published up to June 2020 (date of last literature search: June 15, 2020). We excluded the following:
-
review articles
-
studies published in languages other than English
-
non-human studies
-
studies that did not include a neuroimaging component
-
studies that did not include a specific instrument designed to measure NSSs
-
studies with fewer than 10 subjects
-
studies expressly including subjects with neurological or genetic diseases, or intellectual disabilities
We did not exclude any studies based on the age of their study sample.
Literature search
Using legacy PubMed, we used the following search term: “(neurological evaluation scale OR soft signs OR NES) AND (schizophrenia OR psychosis OR schizoaffective) AND (neuroimaging OR MRI OR Spectroscopy OR functional OR structural MRI OR white matter OR diffusion OR DTI OR gyrification OR cortical thickness OR neurometabolite OR functional connectivity OR network OR PET OR SPECT)”. Reference lists of those studies, as well as the initial results which were review articles or meta-analyses, were inspected for additional relevant publications.
Study selection
After removing duplicate articles, GDS and NVK screened titles and abstracts to exclude irrelevant articles. Both authors applied eligibility criteria, and a list of eligible full text articles was developed through consensus. Full text articles that were not immediately available through the university library were requested, and all requests were fulfilled.
Data extraction
We extracted the following information from each study: name of first author, year of publication, number of participants per diagnostic category, average age of patients, illness duration and medication status of patients, instrument used to quantify NSSs, magnetic field strength, neuroimaging modality, data analysis type, and reported main study outcomes for NSS and neuroimaging data, and tabulated findings separately for structural and functional neuroimaging studies.
To complement the high-level summary of each study presented in the table, we also provide a narrative description of main results in the Results section. The section for structural MRI findings was thematically arranged as follows: “cortical gray matter thickness, surface, gyrification, and sulcation”, “cortical gray matter volumes or densities”, “subcortical and cerebellar volumes or densities”, and “white matter integrity”, for which findings will be separately discussed with respect to global soft signs, sensory integration, motor coordination, and sequencing of complex motor task scores. These domains were chosen because they have been reliably implicated as subscales in different factor analyses of NSS battery items2,73,74. Because the number of functional and molecular imaging studies were much smaller, such a structure was not warranted, and we provided a narrative review summarizing major findings and highlighting similarities across studies instead.
References
Chan, R. C., Xu, T., Heinrichs, R. W., Yu, Y. & Wang, Y. Neurological soft signs in schizophrenia: a meta-analysis. Schizophr. Bull. 36, 1089–1104 (2010).
Bombin, I., Arango, C. & Buchanan, R. W. Significance and meaning of neurological signs in schizophrenia: two decades later. Schizophr. Bull. 31, 962–977 (2005).
Fountoulakis, K. N., Panagiotidis, P., Gonda, X., Kimiskidis, V. & Nimatoudis, I. Neurological soft signs significantly differentiate schizophrenia patients from healthy controls. Acta Neuropsychiatr. 30, 97–105 (2018).
Fountoulakis, K. N., Panagiotidis, P., Kimiskidis, V., Nimatoudis, I. & Gonda, X. Prevalence and correlates of neurological soft signs in healthy controls without family history of any mental disorder: a neurodevelopmental variation rather than a specific risk factor? Int. J. Dev. Neurosci. 68, 59–65 (2018).
Barkus, E., Stirling, J., Hopkins, R. & Lewis, S. The presence of neurological soft signs along the psychosis proneness continuum. Schizophr. Bull. 32, 573–577 (2006).
Mittal, V. A. et al. Movement abnormalities and the progression of prodromal symptomatology in adolescents at risk for psychotic disorders. J. Abnorm. Psychol. 116, 260–267 (2007).
Mittal, V. A. & Walker, E. F. Movement abnormalities predict conversion to Axis I psychosis among prodromal adolescents. J. Abnorm. Psychol. 116, 796–803 (2007).
Peralta, V. & Cuesta, M. J. Motor abnormalities: from neurodevelopmental to neurodegenerative through “functional” (neuro)psychiatric disorders. Schizophr. Bull. 43, 956–971 (2017).
Neelam, K., Garg, D. & Marshall, M. A systematic review and meta-analysis of neurological soft signs in relatives of people with schizophrenia. BMC Psychiatry 11, 139 (2011).
Dazzan, P. et al. Neurological abnormalities and cognitive ability in first-episode psychosis. Br. J. Psychiatry 193, 197–202 (2008).
Keshavan, M. S. et al. Diagnostic specificity and neuroanatomical validity of neurological abnormalities in first-episode psychoses. Am. J. Psychiatry 160, 1298–1304 (2003).
Bora, E., Akgul, O., Ceylan, D. & Ozerdem, A. Neurological soft signs in bipolar disorder in comparison to healthy controls and schizophrenia: a meta-analysis. Eur. Neuropsychopharmacol. 28, 1185–1193 (2018).
Owoeye, O. et al. Epidemiological and clinical characterization following a first psychotic episode in major depressive disorder: comparisons with schizophrenia and bipolar I disorder in the Cavan-Monaghan First Episode Psychosis Study (CAMFEPS). Schizophr. Bull. 39, 756–765 (2013).
Chan, R. C. et al. Clinical utility and lifespan profiling of neurological soft signs in schizophrenia spectrum disorders. Schizophr. Bull. 42, 560–570 (2016).
Herold, C. J. et al. Neurological soft signs and psychopathology in chronic schizophrenia: a cross-sectional study in three age groups. Front. Psychiatry 9, 98 (2018).
Bachmann, S., Degen, C., Geider, F. J. & Schroder, J. Neurological soft signs in the clinical course of schizophrenia: results of a meta-analysis. Front. Psychiatry 5, 185 (2014).
Buchanan, R. W. & Heinrichs, D. W. The Neurological Evaluation Scale (NES): a structured instrument for the assessment of neurological signs in schizophrenia. Psychiatry Res. 27, 335–350 (1989).
Chen, E. Y. et al. The Cambridge Neurological Inventory: a clinical instrument for assessment of soft neurological signs in psychiatric patients. Psychiatry Res. 56, 183–204 (1995).
Krebs, M. O., Gut-Fayand, A., Bourdel, M., Dischamp, J. & Olie, J. Validation and factorial structure of a standardized neurological examination assessing neurological soft signs in schizophrenia. Schizophr. Res. 45, 245–260 (2000).
Merriam, A. E., Kay, S. R., Opler, L. A., Kushner, S. F. & van Praag, H. M. Neurological signs and the positive-negative dimension in schizophrenia. Biol. Psychiatry 28, 181–192 (1990).
Rossi, A. et al. Neurological soft signs in schizophrenia. Br. J. Psychiatry 157, 735–739 (1990).
Schroder, J. et al. Neurological soft signs in schizophrenia. Schizophr. Res. 6, 25–30 (1991).
Bachmann, S. & Schroder, J. Neurological soft signs in schizophrenia: an update on the state- versus trait-perspective. Front. Psychiatry 8, 272 (2017).
Zhao, Q. et al. Neurological soft signs are not “soft” in brain structure and functional networks: evidence from ALE meta-analysis. Schizophr. Bull. 40, 626–641 (2014).
Williams, A. O., Reveley, M. A., Kolakowska, T., Ardern, M. & Mandelbrote, B. M. Schizophrenia with good and poor outcome. II: Cerebral ventricular size and its clinical significance. Br. J. Psychiatry. 146, 239–246 (1985).
Bersani, G. et al. Neurological soft signs and cerebral measurements investigated by means of MRI in schizophrenic patients. Neurosci Lett 413, 82–87 (2007).
Ciufolini, S. et al. Cortical thickness correlates of minor neurological signs in patients with first episode psychosis. Schizophr. Res. 200, 104–111 (2018).
Gay, O. et al. Cortex morphology in first-episode psychosis patients with neurological soft signs. Schizophr. Bull. 39, 820–829 (2013). Jul.
Hirjak, D. et al. Local brain gyrification as a marker of neurological soft signs in schizophrenia. Behav. Brain Res. 292, 19–25 (2015). Oct 1.
Kong, L. et al. Association of cortical thickness and neurological soft signs in patients with chronic schizophrenia and healthy controls. Neuropsychobiology 71, 225–233 (2015).
Heuser, M., Thomann, P. A., Essig, M., Bachmann, S. & Schroder, J. Neurological signs and morphological cerebral changes in schizophrenia: An analysis of NSS subscales in patients with first episode psychosis. Psychiatry Res. 192, 69–76 (2011).
Thomann, P. A. et al. Neurological soft signs and brain morphology in first-episode schizophrenia. Psychol. Med. 39, 371–379 (2009).
Mouchet-Mages, S. et al. Correlations of cerebello-thalamo-prefrontal structure and neurological soft signs in patients with first-episode psychosis. Acta Psychiatr. Scand. 123, 451–458 (2011).
Kong, L., Bachmann, S., Thomann, P. A., Essig, M. & Schroder, J. Neurological soft signs and gray matter changes: a longitudinal analysis in first-episode schizophrenia. Schizophr. Res. 134, 27–32 (2012).
Dazzan, P. et al. The structural brain correlates of neurological soft signs in AESOP first-episode psychoses study. Brain 127, 143–153 (2004).
Hirjak, D. et al. Neurological soft signs and brainstem morphology in first-episode schizophrenia. Neuropsychobiology 68, 91–99 (2013).
Bottmer, C. et al. Reduced cerebellar volume and neurological soft signs in first-episode schizophrenia. Psychiatry Res. 140, 239–250 (2005).
Thomann, P. A. et al. Cerebellar substructures and neurological soft signs in first-episode schizophrenia. Psychiatry Res. 173, 83–87 (2009).
Hirjak, D. et al. Neurological soft signs in recent-onset schizophrenia: focus on the cerebellum. Prog. Neuropsychopharmacol. Biol. Psychiatry 60, 18–25 (2015).
Venkatasubramanian, G., Jayakumar, P. N., Gangadhar, B. N. & Keshavan, M. S. Neuroanatomical correlates of neurological soft signs in antipsychotic-naive schizophrenia. Psychiatry Res. 164, 215–222 (2008).
Bersani, G. et al. Neurological Soft Signs and Corpus Callosum morphology in schizophrenia. Neurosci. Lett. 499, 170–174 (2011).
Foong, J. et al. In vivo investigation of white matter pathology in schizophrenia with magnetisation transfer imaging. J. Neurol. Neurosurg. Psychiatry. 68, 70–74 (2000).
Janssen, J. et al. Brain morphology and neurological soft signs in adolescents with first-episode psychosis. Br. J. Psychiatry 195, 227–233 (2009).
Kasparek, T. et al. Cortico-cerebellar functional connectivity and sequencing of movements in schizophrenia. BMC Psychiatry 12, 17 (2012).
Huttlova, J. et al. Abnormalities in myelination of the superior cerebellar peduncle in patients with schizophrenia and deficits in movement sequencing. Cerebellum 13, 415–424 (2014).
Chan, R. C. et al. Prefrontal cortex connectivity dysfunction in performing the Fist-Edge-Palm task in patients with first-episode schizophrenia and non-psychotic first-degree relatives. Neuroimage Clin. 9, 411–417 (2015).
Zemankova, P. et al. Neuronal substrate and effective connectivity of abnormal movement sequencing in schizophrenia. Prog. Neuropsychopharmacol. Biol. Psychiatry 67, 1–9 (2016).
Schroder, J., Wenz, F., Schad, L. R., Baudendistel, K. & Knopp, M. V. Sensorimotor cortex and supplementary motor area changes in schizophrenia. A study with functional magnetic resonance imaging. Br J Psychiatry 167, 197–201 (1995).
Hirjak, D. et al. Patterns of co-altered brain structure and function underlying neurological soft signs in schizophrenia spectrum disorders. Hum Brain Mapp 40, 5029–5041 (2019).
Schroder, J. et al. D2 dopamine receptor up-regulation, treatment response, neurological soft signs, and extrapyramidal side effects in schizophrenia: a follow-up study with 123I-iodobenzamide single photon emission computed tomography in the drug-naive state and after neuroleptic treatment. Biol. Psychiatry 43, 660–665 (1998).
Chen, E. Y. et al. A 3-year prospective study of neurological soft signs in first-episode schizophrenia. Schizophr. Res. 75, 45–54 (2005).
Herold, C. J., Duval, C. Z., & Schroder, J. Neurological soft signs and cognition in the late course of chronic schizophrenia: a longitudinal study. Eur. Arch. Psychiatry Clin. Neurosci. https://doi.org/10.1007/s00406-020-01138-7 (2020)
Mayoral, M. et al. Longitudinal study of neurological soft signs in first-episode early-onset psychosis. J. Child Psychol. Psychiatry 53, 323–331 (2012).
Fritze, S. et al. Differential contributions of brainstem structures to neurological soft signs in first- and multiple-episode schizophrenia spectrum disorders. Schizophr. Res. 210, 101–106 (2019).
Behere, R. V. Dorsolateral prefrontal lobe volume and neurological soft signs as predictors of clinical social and functional outcome in schizophrenia: A longitudinal study. Indian J. Psychiatry 55, 111–116 (2013).
Goldstein, G. et al. The effects of antipsychotic medication on factor and cluster structure of neurologic examination abnormalities in schizophrenia. Schizophr. Res. 75, 55–64 (2005).
Peralta, V., de Jalon, E. G., Campos, M. S. & Cuesta, M. J. Phenomenological differences between spontaneous and drug-related extrapyramidal syndromes in patients with schizophrenia-spectrum disorders. J. Clin. Psychopharmacol. 33, 438–440 (2013).
Emsley, R. et al. Neurological soft signs in first-episode schizophrenia: State- and trait-related relationships to psychopathology, cognition and antipsychotic medication effects. Schizophr. Res. 188, 144–150 (2017).
Pitzianti, M. et al. Antipsychotics do not influence neurological soft signs in children and adolescents at ultra-high risk for psychosis: a pilot study. J Psychiatr Pract 25, 186–191 (2019).
Fritze, S. et al. Neurological soft signs in schizophrenia spectrum disorders are not confounded by current antipsychotic dosage. Eur. Neuropsychopharmacol. 31, 47–57 (2020).
Cadena, E. J., White, D. M., Kraguljac, N. V., Reid, M. A. & Lahti, A. C. Evaluation of fronto-striatal networks during cognitive control in unmedicated patients with schizophrenia and the effect of antipsychotic medication. NPJ Schizophr. 4, 8 (2018).
Kraguljac, N. V. et al. A longitudinal neurite and free water imaging study in patients with a schizophrenia spectrum disorder. Neuropsychopharmacology 44, 1932–1939 (2019).
Kraguljac, N. V. et al. Micro- and macrostructural white matter integrity in never-treated and currently unmedicated patients with schizophrenia and effects of short-term antipsychotic treatment. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 4, 462–471 (2019).
Kraguljac, N. V. & Lahti, A. C. Neuroimaging as a window into the pathophysiological mechanisms of schizophrenia. Front. Psychiatry 12, 613764 (2021).
Kraguljac, N. V. et al. Abnormalities in large scale functional networks in unmedicated patients with schizophrenia and effects of risperidone. Neuroimage Clin. 10, 146–158 (2016).
Lottman, K. K. et al. Risperidone effects on brain dynamic connectivity—a prospective resting-state fMRI Study in schizophrenia. Front Psychiatry 8, 14 (2017).
Morrison, P. D. & Murray, R. M. The antipsychotic landscape: dopamine and beyond. Ther. Adv. Psychopharmacol. 8, 127–135 (2018).
Nelson, E. A. et al. A prospective longitudinal investigation of cortical thickness and gyrification in schizophrenia. Can. J. Psychiatry 65, 381–391 (2020).
Nelson, E. A., White, D. M., Kraguljac, N. V. & Lahti, A. C. Gyrification connectomes in unmedicated patients with schizophrenia and following a short course of antipsychotic drug treatment. Front. Psychiatry 9, 699 (2018).
Reynolds, G. P. Antipsychotic drug mechanisms and neurotransmitter systems in schizophrenia. Acta Psychiatr. Scand. Suppl. 380, 36–40 (1994).
Tomelleri, L. et al. Brain structural changes associated with chronicity and antipsychotic treatment in schizophrenia. Eur. Neuropsychopharmacol. 19, 835–840 (2009).
Kraguljac, N. V. et al. Neuroimaging biomarkers in schizophrenia. Am. J. Psychiatry 178, 509–521 (2021).
Sanders, R. D. et al. Confirmatory factor analysis of the Neurological Evaluation Scale in unmedicated schizophrenia. Psychiatry Res. 133, 65–71 (2005).
Sewell, R. A. et al. Clinical significance of neurological soft signs in schizophrenia: factor analysis of the Neurological Evaluation Scale. Schizophr. Res. 124, 1–12 (2010).
Galderisi, S. et al. ‘Simple schizophrenia’: a controlled MRI and clinical/neuropsychological study. Psychiatry Res. 91, 175–184 (1999).
Hüttlova, J. et al. Abnormalities in myelination of the superior cerebellar peduncle in patients with schizophrenia and deficits in movement sequencing. Cerebellum (London, England) 13, 415–424 (2014).
Kašpárek, T. et al. Movement sequencing abilities and basal ganglia morphology in first-episode schizophrenia. The world journal of biological psychiatry : the official journal of the World Federation of Societies of Biological Psychiatry 10, 752–762 (2009).
Sachdev, P., Rose, N. & Cathcart, S. Schizophrenia with onset after age 50 years. 2: Neurological, neuropsychological and MRI investigation. The British journal of psychiatry: the journal of mental science 175, 416–421 (1999).
Szendi, I. et al. Correlations between clinical symptoms, working memory functions and structural brain abnormalities in men with schizophrenia. Psychiatry research 147, 47–55 (2006).
Gay, O. et al. Cognitive control deficit in patients with first-episode schizophrenia is associated with complex deviations of early brain development. J psychiatry neurosci.: JPN 42, 87–94 (2017).
Hirjak, D. et al. Cortical signature of neurological soft signs in recent onset schizophrenia. Brain topogr. 27, 296–306 (2014).
Hirjak, D. et al. Neurological soft signs and subcortical brain morphology in recent onset schizophrenia. J. psychiatr res 46, 533–539 (2012).
Schröder, J., Wenz, F., Schad, L. R., Baudendistel, K. & Knopp, M. V. Sensorimotor cortex and supplementary motor area changes in schizophrenia. A study with functional magnetic resonance imaging. The British journal of psychiatry : thejournal of mental science 167, 197–201 (1995).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
This work was supported by the National Institute of Mental Health (R01MH102951 and R01MH112800, ACL; R01MH118484 and K23MH106683, NVK) Dr. Kraguljac served as consultant for Neurocrine Biosciences, Inc. All other authors report no conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Samson, G.D., Lahti, A.C. & Kraguljac, N.V. The neural substrates of neurological soft signs in schizophrenia: a systematic review. Schizophr 8, 42 (2022). https://doi.org/10.1038/s41537-022-00245-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41537-022-00245-9
This article is cited by
-
Specific association between retinal neural layer thinning and neurological soft signs in schizophrenia
European Archives of Psychiatry and Clinical Neuroscience (2024)