Abstract
Chrysanthemum (Chrysanthemum morifolium Ramat.) is a globally important ornamental plant with great economic, cultural, and symbolic value. However, research on chrysanthemum is challenging due to its complex genetic background. Here, we report a near-complete assembly and annotation for C. morifolium comprising 27 pseudochromosomes (8.15 Gb; scaffold N50 of 303.69 Mb). Comparative and evolutionary analyses reveal a whole-genome triplication (WGT) event shared by Chrysanthemum species approximately 6 million years ago (Mya) and the possible lineage-specific polyploidization of C. morifolium approximately 3 Mya. Multilevel evidence suggests that C. morifolium is likely a segmental allopolyploid. Furthermore, a combination of genomics and transcriptomics approaches demonstrate the C. morifolium genome can be used to identify genes underlying key ornamental traits. Phylogenetic analysis of CmCCD4a traces the flower colour breeding history of cultivated chrysanthemum. Genomic resources generated from this study could help to accelerate chrysanthemum genetic improvement.
Similar content being viewed by others
Introduction
Cultivated chrysanthemum (Chrysanthemum morifolium Ramat., Chinese name “Ju Hua”) is a perennial herbaceous ornamental plant that belongs to Asteraceae (also known as Compositae), the largest family of dicotyledons, with more than 1600 genera and 25,000 species1. In addition to being known for its outstanding beauty, cultivated chrysanthemum also holds a key evolutionary position in flowering plants as a representative of Asteraceae. Chrysanthemum originated in China and has been cultivated for over three thousand years2,3. From the 8th century AD to 17th century, several chrysanthemum cultivars were successively introduced to Japan and Europe4, further expanding their breeding for ornamental purposes. At present, chrysanthemum is one of the most economically important floral plants worldwide5, due to the striking diversity of its flower shapes and colours, and accounts for a large proportion of flower industry production, applying as cut-flower, garden and potted types. Additionally, chrysanthemum is widely used in the medical, food, and beverage industries because of its nutritional and biologically active components.
The Chrysanthemum genus harbours more than 41 species with varying ploidies, ranging from 2n = 2x to 10x, and natural hybridisation is prevalent in this genus3,6. Cultivated chrysanthemum is a self-incompatible complex hexaploid frequently showing a chromosome number of 547,8. However, whether chrysanthemum should be genetically classified as an autopolyploid or allopolyploid has long been controversial. Abundant studies have suggested that several wild Chrysanthemum species might have contributed to current complex hybrid chrysanthemum cultivars9,10,11. Hence, following a long period of domestication and artificial selection, cultivated chrysanthemum now displays a much higher diversity of ornamental features than other species of the Chrysanthemum genus. However, the evolutionary history of the Chrysanthemum genus is poorly understood, and the actual ancestry of cultivated chrysanthemum remains elusive.
An available high-quality reference genome sequence is key to elucidating the species’ origin and evolutionary history as well as the genetic basis underlying its phenotypic diversity, which can further accelerate future breeding processes12. Despite the economic importance and evolutionary significance of cultivated chrysanthemum, its genome has not yet been deciphered, mainly due to its polyploidy, high repetitiveness, high heterozygosity, and large size. Recently, two scaffold-level Chrysanthemum genomes, i.e., the ones of C. nankingense13 and C. seticuspe14, as well as three chromosome-scale genomes, i.e., the ones of C. seticuspe15, C. makinoi16, and C. lavandulifolium17, have been reported. These valuable resources allow for dissecting the evolutionary history of the Chrysanthemum genus. However, these genomes all come from diploid wild species and therefore lack critical genetic information of chrysanthemum cultivars, making them less optimal for illuminating the complex genetic basis of phenotypic variation in hexaploid cultivated chrysanthemum. Several transcriptome studies have allowed us to preliminarily isolate a number of homologous genes involved in the regulation of ornamental traits in chrysanthemum18,19,20,21,22, but it remains challenging to reveal novel determinants of these traits through forward genetics approaches due to the lack of a reference genome sequence for cultivated chrysanthemum.
Here, we present a chromosome-scale genome assembly of C. morifolium using a haploid line (n = 3x = 27) derived from pot variety ‘Zhongshanzigui’23. We infer that the recent whole-genome triplication (WGT) and smaller-scale duplications contribute to the expansion of the known genes related to flower development in chrysanthemum and identify several candidate genes underlying petal shape. Furthermore, we trace the flower colour evolution history of cultivated chrysanthemum based on the phylogenetic analysis of CCD4a genes. The C. morifolium reference genome provides insights into the evolution of Chrysanthemum genus and is valuable for future genetic and molecular biology studies of chrysanthemum.
Results
Genome sequencing, assembly, and annotation
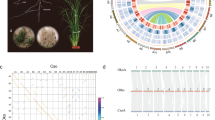
The genome size of the haploid line (Supplementary Fig. 1b, d) was estimated to be approximately 8.47~8.88 Gb by K-mer analysis using 1070.20 Gb clean short reads (Supplementary Fig. 2a–d and Supplementary Tables 1 and 2), which was slightly smaller than the size estimated by flow cytometry (9.02 Gb; Supplementary Fig. 2e). The genome survey also revealed a high repeat content (72.48% ~ 88.85%) in C. morifolium (Supplementary Table 1). We initially corrected and assembled the 1022.3 Gb (120.70×) PacBio continuous long reads (CLR) into contig sequences using Falcon and polished them using Quiver. The consensus sequences were then scaffolded by integration with 907.5 Gb (107.20×) of genome sequences derived from a 10X Genomics library and polished using short reads. Next, we used the AllHiC algorithm24 to improve the C. morifolium genome assembly based on 1002.9 Gb (118.50×) of Hi-C data (Supplementary Table 2 and Supplementary Note 1). This allowed 96.46% of the original assembly sequences to be anchored to 27 pseudochromosomes with lengths ranging from 214.51 to 343.67 Mb (Fig. 1a, Table 1 and Supplementary Tables 3 and 4). The contig and scaffold N50 sizes of the final chromosome-level genome assembly of C. morifolium were 1.87 Mb and 303.69 Mb, respectively (Table 1). The Hi-C interaction heatmap clearly demonstrated that the 27 pseudochromosomes could be clustered into nine homoeologous groups (Fig. 1b). The three pseudochromosomes within a homoeologous group were highly similar in terms of their size, gene number and repeat element content (Supplementary Table 4). Plots of the synteny relationships and Ka/Ks ratios of each syntenic gene pair clearly showed a high degree of conserved synteny, with no substantial overall Ka/Ks ratio difference between any two chromosomes (Fig. 1a).
a Circus plot showing the genomic features of the haploid C. morifolium cv. ‘Zhongshanzigui’. Circular tracks from outside to inside indicate: a the pseudomolecules; b gene density; c density of LTR transposons; d density of Copia transposons; e density of Gypsy transposons; f density of DNA transposons; g GC content; h Ka/Ks of syntenic gene pairs identified between traid1 and either traid2 or traid3 within a homoeologous chromosome group; i Ka/Ks of syntenic gene pairs identified between traid2 and either traid1 or traid3 within a homoeologous chromosome group; j Ka/Ks of syntenic gene pairs identified between traid3 and either traid1 or traid2 within a homoeologous chromosome group; k syntenic genes between different pseudochromosomes. Orange lines indicate syntenic blocks between traid1 and traid2 and traid3, green lines indicate syntenic blocks between traid2 and traid3. b Hi-C heatmap showing the chromosomal interactions. Each homoeologous group contains three pseudomolecules, there are few linkages between different homoeologous groups, indicating high quality chromosome-level scaffolding. c Genome comparison between C. morifolium (Chr1~27) and diploid C. nankingense (Cna1~9). Source data are provided as a Source Data file.
To evaluate the assembled genome quality, we aligned 105,996 expressed sequence tags (ESTs) of C. morifolium downloaded from the US National Center for Biotechnology Information (NCBI) database to the assembled genome and found that 96.43% of the ESTs could be mapped (Supplementary Table 5). BUSCO and Core Eukaryotic Genes Mapping Approach (CEGMA) analyses found that 97.70% and 98.39% of the total eukaryotic conserved genes, respectively, were identified in the chromosome-scale genome of C. morifolium (Supplementary Table 6), which is comparable to results for the C. seticuspe genome15 but higher than the percentages reported for the other released genomes of Asteraceae species (Supplementary Data 1). The annotation of long terminal repeats (LTRs) revealed an LTR Assembly Index (LAI)25 score of 27.99, meeting the gold standard for reference genomes, comparable to the scores of autotetraploid alfalfa (22.30)26 and allohexaploid Echinochloa colona (22.5)27. The high fidelity of the final assembly was also supported by the high mapping rate of 98.81% and the estimated high base accuracy of 57 homozygous single nucleotide polymorphisms (SNPs) per 1 Mb (0.0057%) by using Illumina sequencing data (Supplementary Table 7). The top 10× (approximately 80 Gb) longest PacBio long reads, ranging from 36.32 to 156.36 kb, were extracted and mapped against the assembled genome, and a large majority (82.24%) of these reads could be uniquely mapped to only one chromosome with more than 80% alignment length, indicating that most of the chromosomes were phased correctly. Furthermore, 82.15% of the transcriptomes could be mapped back to the final assembly (Supplementary Table 8), further supporting a high level of genome integrity. Taken together, the above results indicate a high degree of continuity and completeness of the C. morifolium genome.
Additionally, a chromosome-level assembly of diploid C. nankingense (2n = 2x = 18), a potential progenitor of cultivated chrysanthemum, was generated for comparative studies using a similar hybrid assembly strategy (Supplementary Table 9 and Supplementary Note 2). The genome assembly anchored 2.87 Gb of the contigs to 9 pseudochromosomes, representing 93.16% of the estimated 3.09 Gb C. nankingense genome (Supplementary Table 10). The high contig N50 length of 5.98 Mb and scaffold N50 length of 353.78 Mb indicate a superior genome assembly compared to the previously published scaffold-level C. nankingense genome13. Intergenomic comparison analysis revealed, as expected, a distinct 3-to-1 syntenic relationship between C. morifolium and C. nankingense. Only a few inversion or translocation regions were identified (Fig. 1c).
Based on de novo and homology-based predictions and transcriptome data (Supplementary Note 3), we predicted a total of 138,749 protein-coding genes in the C. morifolium genome, which is considerably greater than the number annotated for other Asteraceae plants, ranging from 28,310 genes for globe artichoke to 74,259 for C. seticuspe. The average lengths of total gene regions, coding sequences (CDSs), exon sequences, and intron sequences were 3912, 1090, 242 and 806 bp, respectively (Supplementary Data 2). On average, each predicted gene contained 4.50 exons, and the gene density throughout the genome was approximately one gene per 58.74 kb, with 134,450 genes (96.90%) present on chromosomally anchored contigs. Genes were distributed unevenly, being more abundant towards the ends of the chromosomal arms as expected (Fig. 1a). Approximately 99.3% of the predicted protein-coding genes could be functionally annotated via searches in the NR (90.97%), Swiss-Prot (75.58%), GO (90.78%), KEGG (70.28%), InterPro (98.75%) and Pfam (72.60%) databases (Supplementary Table 11). In addition, we identified 50,848 noncoding RNA (ncRNA) genes encoding 2868 ribosomal RNAs (rRNAs), 4102 transfer RNAs (tRNAs), 2280 microRNAs (miRNAs), and 41,598 small nuclear RNAs (snRNAs) (Supplementary Table 12). Using RNA-seq data from flower buds and nine organs, including roots, stems, shoot leaves, and flower organs dissected from ray florets and disc florets, 103,287 (74.44%) of the identified genes were expressed in at least one tissue, and 57,869 (41.71%) were expressed in all organs analysed (Supplementary Table 13).
Comparative genomics and evolutionary analysis
We inferred the phylogenetic position and divergence times among cultivated chrysanthemum and 14 other plant species (Supplementary Note 4), including 7 asterids I (C. nankingense, C. seticuspe, sunflower, lettuce, globe artichoke, Artemisia annua and carrot), 2 asterids II (tomato and coffee), 1 rosids (grape), 1 Ranunculales (columbine), 2 monocots (rice and maize) and 1 basal angiosperm (Amborella). We constructed a consensus set containing 94,552 genes to represent the monoploid genome of C. morifolium. A total of 507,449 genes were clustered into 48,644 orthologous gene families (orthogroups), including 6193 gene families shared by all 15 species (Supplementary Fig. 3 and Supplementary Data 3). By comparing seven Asteraceae species, we identified 10,234 gene families shared by all members and 3543 families containing 9638 genes that appeared unique to C. morifolium (Supplementary Fig. 4). In addition, gene family evolution analysis revealed 1684 gene families showing possible expansion in C. morifolium, whereas 1926 gene families showed contraction (Fig. 2a). Functional enrichment analysis demonstrated that these expanded gene families were mainly involved in terpene biosynthetic processes, auxin metabolic processes, regulation of hormone levels, regulation of anthocyanin biosynthetic processes, and regulation of double-strand break repair via homologous recombination (Supplementary Fig. 5 and Supplementary Data 4).
a Phylogenetic tree showing the evolutionary relationship of cultivated chrysanthemum and 14 other flowing plants. Expansion (green) and contraction (red) of gene family numbers are shown on the right. The red circles on some nodes represent fossil calibrations points, obtaining from the TIMETREE website (http://www.timetree.org/)112. Predicted divergence times (Mya, million years ago) are labelled in blue at other intermediate nodes as estimated by Maximum Likelihood (PAML), and the numbers in parentheses are 95% confidence intervals of the divergence time between different clades. The WGD and WGT events are marked with coloured dots. b Ks distribution of syntenic paralogues found within C. morifolium and five other Asteraceae sequenced species. The polyploidization events are referenced on the peaks. c Comparison of repetitive sequences among C. morifolium and C. nankingense, A. annua, H. annuus, C. cardunculus. d Insertion time distributions of intact LTRs in different Asteraceae species. Source data are provided as a Source Data file.
As expected, a phylogenetic tree (Fig. 2a) constructed using 491 single-copy gene families agreed with previous studies13,28,29,30. Molecular dating suggested that Asteraceae diverged from carrot (Apiaceae) at approximately 92.4 Mya. Within Anthemideae, cultivated chrysanthemum diverged from the most recent common ancestor of C. nankingense and C. seticuspe ~3.7 Mya, following the divergence of Artemisia annua approximately 6.5 Mya. Anthemideae species are more related to sunflower (Heliantheae, diverged ~34.8 Mya) than to lettuce and globe artichoke (Cichorieae, diverged ~39.3 Mya).
It is widely recognised that whole-genome duplication (WGD) or WGT events have a profound effect on shaping plant genome evolution and speciation31. We first estimated the synonymous substitution rate (Ks) values of collinear paralogous gene pairs within monoploid C. morifolium, C. nankingense and C. seticuspe, sunflower, and globe artichoke to identify WGD/WGT events (Fig. 2b). These results, together with previous reports13,29,32, demonstrated that C. morifolium has a complex palaeopolyploid history. In addition to the ancient WGT-γ (approximately 122~164 Mya) shared by all core eudicots, WGT-1 (~57 Mya) shared by asterids II, and WGD-2 (~38 Mya) in sunflower, we identified a recent lineage-specific WGD event shared by all three Chrysanthemum species that occurred approximately 6 Mya, showing signature Ks peaks at approximately 0.1, in agreement with a previous study13. Moreover, genomic dot plot analysis clearly illustrated a 3:1 syntenic relationship within C. morifolium monoploid pseudochromosomes and a 3:1 syntenic relationship between C. morifolium and globe artichoke (Supplementary Fig. 6a, b), which experienced only the Asteraceae-shared WGT-1 event33. Similar relationships were detected within C. nankingense and between C. nankingense and globe artichoke (Supplementary Fig. 6c, d). These results provide additional evidence that the inferred recent polyploidization event in the common ancestor of Chrysanthemum species was more likely to be a WGT event than a WGD event (herein termed WGT-2). We also calculated the Ks values between paralogous genes within each of the nine homoeologous groups (Fig. 2b) and found another signature peak at approximately 0.05 (~3 Mya) (Fig. 2b), suggesting that apart from the lineage-specific WGT-2 event shared by all three Chrysanthemum species, C. morifolium underwent a very recent polyploidization event. This may be one potential reason for the observed highly conserved triplicate structure without dramatic chromosomal rearrangements in the C. morifolium genome (Fig. 1c).
Gene duplication has been considered a major force for evolution34. In the C. morifolium genome, 136,137 duplicated genes were divided into five categories, i.e., whole-genome duplicates (WGD, 61.5%), dispersed duplicates (DSD, 43.1%), transposed duplicates (TRD, 17.0%), proximal duplicates (PD, 6.9%) and tandem duplicates (TD) (Supplementary Fig. 7 and Supplementary Note 4). Higher Ka/Ks ratios were found for TRD and PD gene pairs (Supplementary Fig. 8a), suggesting an ongoing, continuous process of transposed and proximal duplications and more relaxed selective pressure for duplicates generated via these two modes, while duplications resulting from all five modes showed similar Ks values (Supplementary Fig. 8b). Notably, duplications via all five modes were associated with divergent enriched GO terms. For example, ‘WGD genes’ showed significant enrichment of GO terms involved in various developmental processes, regulation of transcription and signal transduction; the PD gene-enriched categories were implicated in recognition of pollen and sesquiterpene biosynthetic processes, while the TD genes were assigned to monoterpenoid biosynthetic and terpenoid metabolic processes as well as stress responses (Supplementary Fig. 9). Terpene synthases (TPSs) are the key enzymes responsible for the biosynthesis of terpenoid compounds in green plants. We found that the genomes of Anthemideae species contained many more TPSs than those of other Asteraceae plants, specifically for the TPS-a and TPS-b subfamilies (Supplementary Figs. 10 and 11), which are predominantly involved in the synthesis of sesquiterpenes and monoterpenes, respectively35. Several tandem-duplicated TPS-a and TPS-b genes were identified. In particular, 13 TPS-a copies and 10 TPS-b copies were densely distributed on chromosomes 27 and 19, respectively (Supplementary Fig. 10).
Repetitive sequences were identified using a combination of ab initio and homology-based approaches. In total, 83.38% of the assembled sequences were annotated as repetitive elements (TEs), including 79.59% retrotransposons, 8.63% DNA transposons and 0.92% simple repeats and satellites (Supplementary Table 14). The proportion of TEs identified in C. morifolium is, to our knowledge, the highest among the sequenced Asteraceae species, ranging from 58.4% in globe artichoke to 74.7% in sunflower. LTR retrotransposons were found to constitute 72.96% of the C. morifolium genome, with the Copia (40.40%) and Gypsy (24.79%) subfamilies accounting for the largest percentages. The Copia element density increased from the ends of chromosomes towards their centres, yet there was a relatively even distribution of Gypsy elements along the chromosomes (Fig. 1a). Notably, there were large differences in Copia and Gypsy proportions in Asteraceae. In contrast to the situation of more abundant Copia elements in the C. morifolium (1.63), C. nankingense (1.74), C. seticuspe (1.28)15, C. makinoi (3.66)16 and C. lavandulifolium (1.3)17 genomes, the A. annua (0.86)28, sunflower (0.26) 29, and newly released Mikania micrantha (0.37)32 genomes harboured more Gypsy elements (Fig. 2c). These findings suggest that the amplification of LTR/Copia elements was a major driving force for the inflation of the Chrysanthemum genomes. The mechanisms of these distinct behaviours of Copia and Gypsy elements remain to be elucidated. We found very recent bursts (<0.5 Mya) of LTRs in C. morifolium, C. nankingense and sunflower after the three species diverged, which was slightly later than the LTR bursts identified in A. annua (~ 1 Mya) and globe artichoke (~ 2 Mya). Moreover, the bursts of Copia and Gypsy retrotransposon insertions in C. morifolium occurred close in time, and there was no predominant difference in predicted time of Copia/Gypsy bursts among chromosomes (Supplementary Fig. 12). Collectively, these results suggest that the C. morifolium genome is characterised by active transposition, and the accumulation of TE insertions is the main reason for the expansion of the cultivated chrysanthemum genome.
Origin of cultivated chrysanthemum
Although the origin of cultivated chrysanthemum has attracted considerable attention, the wild progenitors that contributed to cultivated chrysanthemum remain uncertain. Previous phylogenetic studies aimed at identifying these progenitor species, often based on traditional morphological and cytological taxonomy or a limited number of molecular markers, have produced inconsistent results3. By taking advantage of the C. morifolium genome, we resequenced 12 Chinese wild Chrysanthemum species that are assumed to be the most likely ancestral species of cultivated chrysanthemum, with an average coverage depth of approximately 8.5× for each accession (Supplementary Fig. 13, Supplementary Data 5). The phylogenetic tree (Fig. 3a) generated with 11,755 SNPs clearly illustrated that C. rhombifolium, which bears white ray florets, and tetraploid C. indicum from Nanjing, might be more closely related to cultivated chrysanthemum than other species, consistent with the findings that C. rhombifolium and C. indicum (Nanjing) shared the largest numbers of fragments specific with C. morifolium (Fig. 3b). In addition, identity score (IS) analysis clearly illustrated that C. indicum (Nanjing) (0.866), C. rhombifolium (0.853), C. indicum (Tianzhushan) (0.847), C. indicum (Hubei) (0.844), C. dichrum (0.841) and C. potentilloides (0.833), which formed an independent clade, also exhibited higher genomic similarity to cultivated chrysanthemum than the other wild Chrysanthemum species (Fig. 3c and Supplementary Data 6). Notably, the C. indicum accessions with different geographic regions and ploidy levels were grouped into three main clades and exhibited even more genetic diversity than was observed between species (Fig. 3a), which has also been found in other studies3. We speculate that interspecific hybridisation within the Chrysanthemum genus as well as C. indicum habitat diversity might have contributed to this genetic variation.
a Maximum-likelihood phylogenetic tree of Chrysanthemum plants using sunflower (H. annuus) as the outgroup. Numbers above the branches indicate the bootstrap values. The morphology and cytological characterisations of C. morifolium cv. ‘Zhongshanzigui’ and 12 re-sequenced diploid or tetraploid wild Chrysanthemum species are shown on the right. b UpSet plot showing the number of 100 kb non-overlapping sliding windows with coverage depth larger than 4×. c The identical score (IS) values distribution of 12 wild Chrysanthemum species. The higher IS values indicate a closer relationship with C. morifolium. The central line for each box plot is the median, the top and bottom edges correspond to the first and third quartiles, and the whiskers represent the 1.5 inter-quartile range extending from the edges of the box. The grey points are outliers. The average IS value for each species is indicated as orange dots and marked on the top of box plot. Data were analyzed using one-way ANOVA followed by a two-tailed Tukey’s honestly significant difference (HSD) multiple comparison test (n = 70,524), and different lowercase letters indicate a statistically significant difference at P < 0.01. d Smudgeplots analysis based on kmers for C. morifolium. Source data are provided as a Source Data file.
In a further attempt to track introgression events, we calculated the average IS values within each 100 kb sliding window across the C. morifolium genome. As a result, massive small hotspots with higher IS values and a few large hotspots derived from most of these wild Chrysanthemum species were observed in cultivated chrysanthemum (Supplementary Fig. 14), suggesting that extensive and multiple genomic introgression might have contributed to the formation of cultivated chrysanthemum as well as the complex reticulate evolution history of Chrysanthemum species.
C. nankingense has long been suspected to be a diploid progenitor of cultivated chrysanthemum13. The two genomes assembled in this study enable us to study gene loss and retention following triploidization of C. morifolium. Approximately 74.44% of all genes in C. nankingense were syntenic with at least one homologous copy in C. morifolium, while 55.10% of genes had three homologous copies retained in C. morifolium (Supplementary Fig. 15). However, we could not identify a dominant chromosome set in the C. morifolium genome that could be clearly attributed to C. nankingense, unlike the situation in allopolyploid strawberry36 and broomcorn millet37, suggesting that C. nankingense might not be a direct ancestral donor of cultivated chrysanthemum. This finding was supported by the SNP-based phylogenetic tree (Fig. 3a) and IS analyses (Fig. 3c). Taking advantage of the assembly of C. seticuspe genome15, we performed a similar gene retention analysis and found that C. seticuspe was less closely related to C. morifolium than C. nankingense (Supplementary Fig. 16 and Supplementary Data 7).
It remains unclear whether cultivated chrysanthemum has an autopolyploid or allopolyploid origin. Distinguishing between different forms of polyploidy is not always straightforward. Generally, chromosome behaviour, fertility, segregation ratios, and morphology, coupled with genetic data, are principal criteria for distinguishing between autopolyploids and allopolyploids38. Previous cytological studies revealed that hexaploid cultivated chrysanthemum shows diploid-like meiosis in which bivalents dominate, but univalents and multivalents (mainly quadrivalents) are rarely observed39,40,41. However, the disomic or polysomic inheritance of chrysanthemum represents extreme cases within a continuum42,43. Our karyotype analysis conducted during pollen mother cell (PMC) meiosis showed that 18 chromosomes formed nine bivalents, while nine chromosomes remained univalent (9II + 9I) in the sequenced haploid, and the colchicine-induced doubled haploid behaved like an allopolyploid and almost exclusively formed bivalents (27 II) (Supplementary Fig. 1f and Supplementary Note 5). Phylogenetic analysis of each of the homoeologous groups based on the unique SNPs showed that the three homoeologous chromosomes within the same group were all separated from each other except for Chr17 and Chr18 (Supplementary Fig. 17), which is consistent with the possibility of an allopolyploid origin for C. morifolium.
Nevertheless, we found a pattern of uniform coverage of different wild species in comparison with the C. morifolium genome (Supplementary Fig. 13), which differs from what has been observed for allotetraploid tobacco44 and cotton45. This incongruence might occur because the species within the Chrysanthemum genus are closely related; the extant diploid or tetraploid wild Chrysanthemum species probably have also undergone extensive reticulate evolution with high heterozygosity; and the putative primitive ancestor of cultivated chrysanthemum may have gone extinct, as described by Ma et al.46. To verify that the results were not due to the selected potential progenitors, we also used a 13-mers clustering approach (Supplementary Note 5) based on the distribution of TEs, which did not require access to or even knowledge of living representatives of the progenitor lineages47. Although each set of the three homoeologous chromosomes was clearly clustered based on the identified 4719 chromosome-specific 13-mers, we found obvious differences in 13-mer counts among all homoeologous chromosomes except for Chr7-Chr8-Chr9 (Supplementary Fig. 18), suggesting a non-strict autopolyploid origin of C. morifolium. Moreover, the results indicated that the 17-mers with a depth >120x accounted for a larger portion (Supplementary Fig. 2a) compared with autopolyploid alfalfa48; a significant difference with respect to the Ks distributions for syntenic orthologs between C. morifolium and C. nankingense was observed among four out of the nine homoeologous groups (Supplementary Fig. 19); slightly different gene retention patterns within each homoeologous group (Supplementary Figs. 15 and 16) further supported the possibility that C. morifolium might not be a strict autopolyploid. Intriguingly, a Smudgeplot analysis49 implied that the genome structure of C. morifolium might be “AAB” or “AB” (Fig. 3d). Comparison between the homoeologous chromosomes in a triad by fluorescence in situ hybridisation (FISH) assays, also showed two homoeologous chromosomes often to be generally more similar compared to a third one but also presented differences (Supplementary Fig. 20 and Supplementary Data 8), supporting an “AA’B” relationship of homoeologous chromosomes in C. morifolium.
According to these findings together with evidence from previous reports3,6,46,50,51 as well as comparisons with other typical allopolyploids and autopolyploids, we conclude that C. morifolium cv. ‘Zhongshanzigui’ is likely to be a specific segmental allopolyploid.
Homoeolog expression bias in C. morifolium
To explore the transcriptional behaviour of C. morifolium, we compared genome-wide transcription levels of the annotated genes in different organs (Supplementary Note 6). Similar expression patterns were found for each homoeologous group across organs, except that the expression levels of genes located in the middle of chromosome 13 were significantly lower than those on chromosomes 14 and 15 (Supplementary Figs. 21 and 22). We focused on 11,438 genes showing a 1:1:1 correspondence in syntenic blocks across the three homoeologous chromosomes, referred to as triads. The global analysis combining data across all nine organs showed that 21.65% of syntenic triads presented no statistically significant differences in expression, being assigned to the balanced category, ranging from 19.20% in the Chr1-Chr2-Chr3 chromosome set to 23.50% in the Chr22-Chr23-Chr24 chromosome set (Supplementary Fig. 23); the proportion of syntenic triads with single-homoeolog dominance (41.73%) was significantly higher than that with single-homoeolog suppression (36.61%) (Supplementary Fig. 24), among which Chr8 dominance accounted for the largest proportion (24.7%). GO analysis indicated that the balanced triads were enriched in different biological processes among the nine homoeologous groups associated with various life processes. In particular, ‘protein stabilisation’, ‘reproductive structure development’, ‘regulation of multicellular organismal development’, ‘regulation of transferase activity’, ‘leaf development’, ‘shoot system development’, etc., were enriched in two homoeologous groups, while auxin transport (GO:0060918) was found in three homoeologous groups (Supplementary Data 9). Furthermore, we found a weak correlation between the synteny gene orientation and differential gene expression (Supplementary Note 6, Supplementary Tables 15 and 16), suggesting that the homoeolog expression bias in C. morifolium might not be caused by the change in gene orientation. These observations potentially represent the first steps towards neo- or sub-functionalization of C. morifolium genes.
Genetic basis of flower evolution and development
Flower shapes have been under intensive selection during cultivated chrysanthemum improvement and continue to be a primary target of breeding programs. Cultivated chrysanthemum has a more diverse capitulum morphology than other Asteraceae species, which is largely determined by the colourful and multiform ray floret petals. According to the degree of corolla tube merging, florets can be divided into flat, spoon-like, and tubular ray florets (Supplementary Fig. 25c), but the genetic basis of floret diversity is not yet clear. Previous studies have shown that the identity of floral organs is regulated by MADS-box genes, as part of the well-characterised ABCE model52. Here, we identified all MADS-box (Supplementary Fig. 26 and Supplementary Data 10) and ABCE genes (Supplementary Fig. 25a) in the C. morifolium genome. The results showed that, as expected from its polyploidy nature, the C. morifolium genome possessed more MADS-box genes than other Asteraceae plants and A. thaliana, especially for the SEP and SVP clades. Synteny analysis, Ks analyses of the orthologous gene pairs and duplication analyses revealed that both the recent WGT-2 as well as smaller-scale duplications contributed to the expansion of the MADS-box family members (Supplementary Fig. 6a, b), resulting in at least three very similar copies of each ABCE model gene in the C. morifolium genome. Compared with unduplicated orthologues, duplicates can undergo relaxed functional constraint, allowing them to diverge in both their gene sequence and expression patterns53 and to undergo subfunctionalization or neofunctionalization. As shown in Supplementary Fig. 25a, the PISTILLATA (PI) paralogues indeed showed different expression patterns. One copy (evm.model.scaffold_4253.83) presented a higher expression level in vegetative organs, while the other members were highly expressed in the reproductive organs, implying that their functions might have undergone subfunctionalization. In contrast, the expression levels of the six paralogues of AGAMOUS (AG) in the disc florets of 9 varieties were significantly higher than those of the corresponding genes in the ray florets, suggesting the functional conservation of AG in determining flower organ identity. Gene expression analysis also indicated that the MADS-box B class genes PI and DEFICIENS (DEF)/APETALA3 (AP3) might play important roles in petal and stamen development.
TCP family CYCLOIDEA2 (CYC2) clade genes regulate floral symmetry, inflorescence architecture and reproductive organ development54,55. Previous studies demonstrated that CYC2 clade genes regulate the differentiation of ray and disc florets in Asteraceae species, such as sunflower, groundsel (Senecio vulgaris) and gerbera54,56,57,58. We identified 25 CYC2-like genes in C. morifolium (Supplementary Fig. 25b), which is a much greater number than the seven and eight members found in C. nankingense and sunflower, respectively. Phylogenetic analysis showed that the CYC2a-like genes of chrysanthemum have undergone several duplication events; genes from one subclade were highly expressed in reproductive organs, while genes from another subclade were highly expressed in vegetative organs, in accordance with the other CYC2 clade genes (Supplementary Fig. 25b). Previous studies showed that the constitutive expression of CmCYC2c in C. lavandulifolium results in increases in both the number of ray florets and the length of the ray floret petal ligule18,59. However, there was no correlation between their expression and the degree of petal fusion, suggesting that petal fusion is controlled not only by CYC2s but also by other genes in C. morifolium.
To further determine the molecular genetic basis of petal shape variation, we performed a whole-genome bulked segregate analysis sequencing (BSA-seq) protocol in an F1 population derived from a flat-type cultivar and a tubular-type cultivar (Fig. 4a, Supplementary Fig. 27 and Supplementary Note 7). Seventy-two consistent genomic regions associated with the degree of petal fusion were detected using ΔSNP-index and ED algorithms (Supplementary Data 11). In particular, the MYB-class gene RAD6 (evm.model.scaffold_2712.53), acting downstream of CYC/DICHOTOMA, antagonistically to DIVARICATA (DIV) in the control of floral symmetry60,61, was found in qPT5-5 (Fig. 4b). The QTL was consistently detected across BSA-seq methods, located in the interval of 201.00-202.10 Mb on chromosome 5, with ΔSNP index and ED peaks of 0.91 and 1.65, respectively, for SNPs and 0.76 and 1.15 for InDels (Supplementary Fig. 28). CmRAD6 was highly expressed in flat-type cultivars, and it was also a hub gene in the turquoise module that negatively correlated with the degree of petal fusion (r = −0.92; P = 5.0E-04) (Fig. 4c, d and Supplementary Fig. 29). Additionally, several candidates for flower development, such as CYC2s, AGL6, MADS33 (AGL12), IBL1, PI, were identified via BSA-seq and weighted correlation network analysis (WGCNA) (Fig. 4d and Supplementary Fig. 28). Several auxin-responsive genes were also identified (Supplementary Data 12), suggesting that auxin might play a crucial role in petal development, consistent with our previous findings21. These results provide insights into the genetic architecture of petal shape in cultivated chrysanthemum, and the reference genome assembled herein offers major resources for accelerating future QTL fine mapping and breeding programs.
a Variation in corolla tube merged degree between parents and offsprings. Bar = 1 cm. b The QTLs controlling petal type on chromosome 5 detected by SNP-index (top) and Euclidean distance (bottom) algorithms, using SNPs. The top 1% threshold is indicated by the horizontal blue dotted line. One significant peak designated as qPT5-5 with 32 genes is shaded and partial enlarged on the right. The green and blue lines represent the gene located on the sense strand and antisense strand, respectively. CmRAD6 (evm.model.scaffold_2712.53) is indicated by a red triangle. c Expression of the 32 genes in the ray (R) and discoid (D) florets of three flat petal type cultivars (R/D1~3), three tubular petal type cultivars (R/D4~6) and three spoon petal type cultivars (R/D7~9). Green star, pink triangle, and purple diamond, respectively, represent the gene is differentially expressed in groups of flat vs tubular, flat vs spoon, tubular vs spoon. d Sub-network for the genes in turquoise module. The heatmap of the hub gene is same as that in c panel. e Phylogenetic tree of CCD4a genes from representative non-yellow wild and cultivated chrysanthemums showing the potential breeding process on flower colour. The orange triangle is merged by all the traditional Japanese chrysanthemums and one traditional Chinese chrysanthemum cultivar. The pink triangle presents a mixture of traditional Chinese chrysanthemums and pot chrysanthemums. The purple and blue triangles represent the group of traditional Chinese chrysanthemums and cut chrysanthemums, respectively. See Supplementary Fig. 31 for the full version of this tree.
Phylogenetic analysis of CmCCD4a revealed the flower colour breeding history of chrysanthemum
The flower colour diversity of cultivated chrysanthemum is mainly due to its colourful ray floret petals, which is one of the main ornamental traits of these plants. According to literature dating from the Tang Dynasty (618-907), cultivated chrysanthemum had only yellow ray floret petals, while various colours of ray floret petals in cultivated chrysanthemum were recorded in literature from the Song Dynasty (960-1279)62. One important question is how the petal colour of cultivated chrysanthemum changed from only yellow to a variety of flower colours. It has been shown that carotenoid cleavage dioxygenase (CmCCD4a) regulates the degradation of yellow pigments (carotenoids) in the petals of chrysanthemum ray florets63 (Supplementary Note 8 and Supplementary Fig. 30c, e). We found that CCD4a was missing from the C. morifolium genome. Furthermore, the original chrysanthemum cultivar ‘Zhongshanzigui’ has purple ray floret petals (Supplementary Fig. 1a). However, the sequenced haploid material showed yellow or slightly orange-ray floret petals (Fig. 3a), and the flower colour could not be recovered by artificial whole-genome doubling (Supplementary Fig. 1b, c). Therefore, we hypothesise that the CCD4a gene of wild Chrysanthemum species was introduced into cultivated chrysanthemum through artificial hybridisation but in low copy numbers, making it easy to lose after hybridisation, resulting in the return to yellow ray floret petals.
To verify this hypothesis and determine how CCD4a was introduced into cultivated chrysanthemum, we sought to amplify the CCD4a genes of 40 cultivated chrysanthemum varieties and 14 related wild species of Chrysanthemum genera. The results showed that there is no CCD4a gene in the genomes of cultivated varieties and wild relatives with yellow or orange ray floret petals. Finally, we cloned 78 CCD4a sequences from 23 cultivated chrysanthemum varieties and nine wild relatives of Chrysanthemum genera. Phylogenetic analysis showed that CCD4a was introduced into different chrysanthemum cultivar groups via multiple independent hybridisation events (Fig. 4e and Supplementary Fig. 31). The CCD4a genes of traditional Chinese chrysanthemums and pot chrysanthemums might have been introduced from C. vestitum, while the CCD4a genes of cut chrysanthemums might have been introduced from C. zawadskii (Fig. 4e). This implies that the breeding process of cut chrysanthemums has involved different artificial hybridisation events compared with those of traditional chrysanthemums and pot chrysanthemums. Interestingly, the CCD4a of traditional Japanese chrysanthemum clustered into an independent clade (Supplementary Fig. 31), which suggests that the flower colour trait of chrysanthemum might have arisen from an independent breeding process in Japan. Overall, multiple introductions of the CCD4a gene by artificial hybridisation have promoted carotenoid degradation in the ray floret petals of the original chrysanthemum, contributing to the flower colour diversity of modern cultivated chrysanthemums. The finding also supports the breeding and transmission history of chrysanthemum.
Discussion
Polyploid genome assembly and analysis are still a great technical challenge due to high sequence similarities and homoeologous exchanges. To overcome these difficulties, we selected a specific haploid cultivated chrysanthemum as the material for sequencing, which greatly reduced complexity in assembly, annotation, and downstream analysis. PacBio long read sequencing allows resolving and assembling regions that are rich in repeats and of high genomic similarity. The generated C. morifolium genome has the contig N50 size of 1.87 Mb, scaffold N50 size of 303.69 Mb, and LAI of 27.99. It provides a valuable resource for evolutionary and comparative genomics studies. Our data suggest that the previously reported recent WGD event in C. nankingense13, estimated at approximately 6 Mya, was more likely to be a WGT event (herein referred to as WGT-2) and was shared by all Chrysanthemum species (Fig. 2b and Supplementary Fig. 6). In addition, a very recent independent triploidization event (~3 Mya) was revealed in cultivated chrysanthemum (Fig. 2b). Therefore, C. morifolium underwent a total of at least three rounds of WGTs after the WGT‐γ, plus a recent burst in LTRs (<0.5 Mya; Fig. 2d), resulting in a huge, complex genome architecture.
Polyploidy has long been recognised as a major force in plant evolution and speciation64. To date, the genomes of seven hexaploid plants have been sequenced27,65,66, and all are recognised as being of allopolyploid origin. A previous study revealed that pot chrysanthemums are genetically more closely related to the wild progenitor Chrysanthemum species than to either cut chrysanthemums or traditional chrysanthemums67. Thus, the ‘Zhongshanzigui’ genome sheds light on the origin of cultivated chrysanthemum. Based on our current findings together with previous reports46,68,69, we infer that the multiple identified introgressions, hybridisations, and polyploidization events have led to evolutionary reticulation in the C. morifolium polyploid complex and that C. nankingense, C. lavandulifolium and C. seticuspe, previously thought to be the progenitors of cultivated chrysanthemum, may not be the direct ancestral donors. The phylogenetic and IS analyses (Fig. 3) demonstrated that C. rhombifolium, which is narrowly distributed in Wushan (Chongqing, China), presented a closer relationship with cultivated chrysanthemum. Questions that remain to be further explored include the possible involvement of C. rhombifolium in the formation of cultivated chrysanthemum and when and how the hybridisation mixture expanded its range. The multiple lines of evidence provided here combined with previous studies40,51,70,71 imply that C. morifolium cv. ‘Zhongshanzigui’ is likely to be a specific segmental allopolyploid (as defined by Stebbins et al.72.) and reveal its ongoing divergent evolution. Unfortunately, we failed to assign the 27 pseudochromosomes to subgenomes, and our conjecture of the “AA’B” genomic composition of C. morifolium remains to be investigated. Interestingly, we found that only ~22% of chrysanthemum homoeologs show balanced expression, which is considerably lower than the percentage in allopolyploid wheat (72.5%)73 and Brassica juncea (83.8%)74. This may be explained by the rapid evolution of cultivated chrysanthemum and its specific asexual reproduction.
Cultivated chrysanthemum possesses greater morphological diversity than other Chrysanthemum species, especially with respect to flower shape and colour. In this study, we inferred that the time period extending back 6 Mya was an important period in the speciation of Chrysanthemum and the tachytelic evolution of cultivated chrysanthemums. The expansion of the MADS-box gene family and CYC2 genes because of the recent WGT-2 and triploidization events might be responsible for the diversity of flower morphology. Based on the reference genome, we were able to preliminarily reveal the variation underlying genetic diversity of cultivated chrysanthemum (Supplementary Data 13) and identify several candidate genes involved in petal development, which will allow further fine mapping and functional verification in chrysanthemum. Moreover, our phylogenetic study of CCD4a provides a perspective on the independent breeding history of each cultivated type, even among different cultivars. In addition, we reconstructed the anthocyanin and flavonol biosynthesis pathways for flower colouration in chrysanthemum (Supplementary Note 8 and Supplementary Fig. 30d).
In conclusion, the genomic resources we present here can help to mine hub genes governing important traits in chrysanthemum and contribute to the study of Chrysanthemum evolution. Additionally, the reference genome will facilitate the application of molecular marker-assisted breeding and genome editing to cultivated chrysanthemum. The allopolyploid genome of C. morifolium may help to guide assembly other large complex polyploid genomes with high heterozygosity and uncertain origins.
Methods
Plant materials and genome sequencing
We sequenced the entire genome of a previously characterized haploid (n = 3x = 27) line of potted and ground-type C. morifolium cv. ‘Zhongshanzigui’ (2n = 6x = 54), derived from the in vitro culture of nonfertilized ovules23 (Supplementary Fig. 1). Genomic DNA was extracted from young leaf tissue using a DNAsecure Plant Kit (TIANGEN, Beijing, China) for Illumina short read sequencing, PacBio long read sequencing, 10X Genomics and Hi-C library construction and sequencing, as described in Supplementary Note 1. Total RNA was extracted from various organs using an RNA extraction kit (Huayueyang, Beijing, China) for sequencing (RNAseq) to support genome annotation. All plant materials used in this study were collected from the Chrysanthemum Germplasm Resource Preserving Centre (Nanjing Agricultural University, Nanjing, Jiangsu, China).
Chromosomal assembly and validation
The de novo assembly of the long reads obtained from the PacBio SMRT Sequencer was performed using FALCON75 with the following parameters: length_cutoff_pr = 11,000, overlap_filtering_setting = --max_diff 500 --max_cov 500. The initial assembly was polished using Quiver76 based on PacBio long reads. Then, 10X Genomics reads were aligned to the assembly by BWA-MEM77 using the default settings. Scaffolding was performed by FragScaff78 with the barcoded sequencing reads. We used Pilon79 to perform error correction based on the Illumina sequences. Subsequently, the Hi-C sequencing reads were used to align to the assembled scaffolds by BWA-MEM4, and these scaffolds were clustered and reordered using ALLHiC80. Scaffolds were fine-tuned, discordant contigs were removed from scaffolds using Juicebox81, and the final chromosome assembly was generated after careful manual inspection. The top 10× longest PacBio reads were mapped to the genome by minimap2 with the parameters: “-k 15 -w 10 -I 9 G” to assess its continuity by calculating the unique mapping (≥80% alignment length) proportion. In addition, we also present a chromosome-scale genome assembly for diploid C. nankingense as described in Supplementary Note 2. Synteny analysis between the genomes of diploid C. nankingense and C. morifolium was performed by MCScan (Python-jcvi).
CEGMA (http://korflab.ucdavis.edu/dataseda/cegma/)82 and BUSCO (http://busco.ezlab.org/)83 with the Embryophyta odb10 database and LTR Assembly Index (LAI)84 were used to assess the completeness and continuity of the genome assembly. Gene completeness was evaluated by mapping ESTs to the assembly using BLAT (http://genome.ucsc.edu/goldenpath/help/blatSpec.html).
Genome annotation
We adapted a combination of protein homology-based, de novo and transcriptome-based predictions to annotate protein-coding genes. The protein sequences from six species (Arabidopsis thaliana, Daucus carota, Helianthus annuus, Lactuca sativa, Solanum lycopersicum and Solanum tuberosum) were aligned to the C. morifolium genome using TBLASTN25, with an E-value cut-off of 1e-5. GeneWise84 was employed to predict the exact gene structure of the corresponding genomic regions in each BLAST hit. For transcriptome-based prediction, RNA-seq data from three tissues were mapped to the C. morifolium genome using TopHat (--splice-mismatches 2 --max-intron-length 500000 --min-intron-length 50) and Cufflinks85 (--max-intron-length 500000 --min-intron-length 50 --max-mle-iterations 5000). In addition, Trinity86 was used to assemble the RNA-seq data (--min_glue 2 --min_kmer_cov 2), followed by the application of PASA software87 to improve the gene structures. The generated gene set was denoted PASA-T-set and was used to train ab initio gene prediction programs. Five ab initio gene prediction programs, namely, Augustus (version 2.5.5)88, GENSCAN (version 1.0)89, GlimmerHMM (version 3.0.1)90, Geneid91, and SNAP92, were used to predict coding regions in the repeat-masked genome with the default parameters. Finally, all the predictions were combined using EVidenceModeller (EVM)93 to produce the nonredundant gene set.
The functional annotation of protein-coding genes was achieved by performing BLASTp searches (E-value ≤ 1e-5)94 against the SwissProt (http://web.expasy.org/docs/swiss-prot_guideline.html), NR (ftp://ftp.ncbi.nih.gov/blast/db/), InterPro (http://www.ebi.ac.uk/interpro/, V32.0), Pfam (http://pfam.xfam.org/, V27.0) and KEGG databases (http://www.kegg.jp/kegg/kegg1.html).
Transposable elements (TEs) in the C. morifolium genome were identified through a combination of de novo and homology-based approaches using several databases and software packages, including RepeatModeler95, LTR_FINDER96, RepeatMasker and RepeatProteinMask97, as described in Supplementary Note 2. The LTR retrotransposons were annotated with a pipeline that uses LTRharvest98, LTR_FINDER96 and LTRdigest99. We extracted the sequences of the long terminal repeats for each LTR, aligned them with MUSCLE100, and then calculated the distance (K) between LTRs with the Kimura two-parameter approach. The LTR insert time was estimated using the formula
where r refers to a general substitution rate of 1.3 × 10−8 per site per year in the Asteraceae family. Additional details are available in Supplementary Note 3.
Genome evolution
To infer phylogenetic trees, we first generated a consensus gene set to represent the monoploid genome of C. morifolium. Protein sequences from each set of three homoeologous chromosomes were extracted for use as queries against each other, and after c-score filtering (>0.7) by JCVI (https://github.com/tanghaibao/jcvi), MCScanX101 (-a, -e:1e-5, -u:1, -s:5) was used to determine synteny blocks with at least five gene pairs per block. The longest genes among each set of orthologous gene pairs together with the chromosome-unique genes constituted the final C. morifolium consensus gene set.
The protein sequences of C. morifolium, A. annua, A. coerulea, A. trichopoda, C. canephora, C. cardunculus, C. nankingense, C. seticuspe, D. carota, H. annuus, L. sativa, O. sativa, S. lycopersicum, V. vinifera and Z. mays were clustered into paralogues and orthologues using the program OrthoMCL102 with an inflation parameter equal to 1.5. The single-copy gene protein sequences were independently aligned by MUSCLE100 and then concatenated into a superalignment matrix. The phylogenetic tree of the 15 species was constructed by the ML method using RAxML103 with 100 bootstrap replicates. The MCMCTree program implemented in Phylogenetic Analysis by Maximum Likelihood (PAML)104 was applied to infer divergence times based on the phylogenetic tree with the following main parameters: burn-in of 10,000, sample number of 100,000, and sampling frequency of 2. In addition, the analysis of gene family expansion and contraction was performed by the CAFÉ program105.
Synonymous substitutions per synonymous site (Ks) values were calculated for each gene pair in the aligned blocks. The distributions of all Ks values were plotted to infer whole-genome duplication or speciation events that occurred during the evolutionary history. The peak Ks values were converted to divergence times according to the formula
by using a substitution rate of 8.25×10−9 for asterids13. The dot plots between C. cardunculus and C. morifolium as well as the C. nankingense genome were generated with Quota synteny alignment software to visualise palaeopolyploidy events. Additional details are available in Supplementary Note 4.
Gene family identification
The protein sequences of the ABCE floral development genes in Arabidopsis thaliana and reported CYC2 genes in chrysanthemum were used as queries, and the candidates in the C. morifolium reference genome were identified using BLASTp with a threshold of E-value ≤ 1e-5. The aligned hits with at least 50% coverage of seed protein sequences and >50% protein sequence identity were selected as homologues. Then, the domains of these homologues were predicted by PFAM (http://pfam.xfam.org/). Only those genes that had the same protein domain were considered to be homologues. The results of the identification of TPS and MADS-box family members in eight plant species, including C. morifolium, A. annua, C. cardunculus, C. nankingense, C. seticuspe, L. sativa, H. annuus and A. thaliana, are provided in Supplementary Note 3.
The origin of cultivated chrysanthemum
For whole-genome resequencing, 12 Chinese wild Chrysanthemum accessions were chosen, representing all the extant potential diploid and tetraploid ancestors of cultivated chrysanthemum. Based on the filtered SNPs, the phylogenetic relationships were evaluated by constructing a maximum-likelihood tree and calculating identical score (IS) values. Gene retention analysis was performed using the chromosome-scale C. nankingense and C. seticuspe15 genomes as a reference. To further clarify the genome structure of C. morifolium, two kmer-based methods were adopted, and the meiosis behaviours of the sequenced haploid and doubled haploid plants were examined. A FISH analysis was conducted to assess sequence differences among the homoeologous chromosomes. All details are available in Supplementary Note 5.
Mining of quantitative trait loci and candidate genes for flower shape
Based on the reference genome, we were able to identify quantitative trait loci and candidate genes by integrated BSA-seq and WGCNA, as described in Supplementary Note 7. Briefly, for BSA-seq analysis, two extreme DNA pools, flat-bulk (BF) and tubular-bulk (BT), were constructed in an F1 population derived from ‘Hongxiao’ and ‘Q5-12’ by mixing equal amounts of DNA from 20 individuals for each bulk (Supplementary Fig. 25). Four libraries, including two parents and two progeny pools, were sequenced on the DNBseq-T7 platform to obtain 150 bp PE reads with an average coverage of 30×. GATK software106 was used for SNP/InDel variant calling. Two association analysis methods, SNP-index107 and Euclidean distance (ED)107, were used. The top 1% of absolute ΔSNP index and ED75 values were considered to be strongly associated with the corolla tube merged degree (CTMD).
For WGCNA, the ray florets and disc florets of nine representative cultivars were dissected in the early bloom stage for RNA extraction and transcriptome sequencing. Differential gene expression analysis was performed by DESeq108. After filtering low-abundance (FPKM ≤ 1) genes of samples, 74,074 ray-specific genes were used for coexpression network analysis by the WGCNA package109 using a dynamic tree cut algorithm with a minimum module size of 50 genes and a merging threshold of 0.25. The CTMD values were used as phenotypic data to identify petal type-related modules. Coexpression networks were visualised in Cytoscape software110.
Phylogenetic analysis of CCD4a genes
To obtain the CCD4a nucleotide sequences of Chrysanthemum species, the primer set CCD4a-ORF-F (5’-ATGGGCTCTTTTCCCACATCT-3’)/R (5’-ATAATCAAAGCGTTGTTAGGTCATT-3’) was designed based on the CCD4a open reading frame (ORF) sequence. The PCR mixture consisted of 17 μL ddH2O, 25 μL 2× Phanta Max Buffer, 1 μL dNTP Mix, 2 μL of each CCD4a-ORF-F/R (Sangon Biotech, Shanghai, China), 1 μL Phanta Max Super-Fidelity DNA Polymerase (Vazyme Biotech, Nanjing, Jiangsu, China), and 2 μL DNA. The PCR program consisted of 3 min at 95°C, followed by 35 cycles of 15 s at 95°C, 15 s at 55°C, and 120 s at 72°C, followed by a 5 min extension at 72°C. The PCR product was inserted into the pEASY-Blunt Simple Cloning vector (TransGen Biotech, Beijing, China) and then transformed into DH5α for sequencing. MUSCLE software was employed for multisequence alignments100, and a phylogenetic tree was constructed with MEGA X using the ML method111. Internal branching support was estimated using 1000 bootstrap replicates.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The raw sequencing data generated in this study have been deposited in the NCBI under accession PRJNA796762 and PRJNA895586 The chloroplast and mitochondrial genome were also available at GenBank under the accession number OP104251 and OP104742 respectively. The assembled genome sequences and annotations are available at Figshare [https://doi.org/10.6084/m9.figshare.21655364.v2]. The Arabidopsis ABCE and chrysanthemum CYC2 genes were used as query sequences for gene family identification, which are available at Figshare [https://doi.org/10.6084/m9.figshare.21610305]. Source data are provided with this paper.
References
Group, A. P. et al. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot. J. Linn. Soc. 181, 1–20 (2016).
Anderson, N.O. Flower Breeding and Genetics: Issues, Challenges and Opportunities for the 21st Century Ch. 14 (Springer, Dordrecht, 2006).
Chen, J., Wang, C., Zhao, H. & Zhou, J. The Origin of Garden Chrysanthemum (Anhui Science & Technology Publishing House, Hefei, 2012).
Ohmiya, A. Molecular mechanisms underlying the diverse array of petal colors in chrysanthemum flowers. Breed. Sci. 68, 119–127 (2018).
AIPH. International Statistics Flowers and Plants (Oxford, UK, 2020).
Spaargaren, J. & Geest, G.v. Ornamental Crops Ch. 14 (Springer, Dordrecht, 2018).
Dowrick, G. The chromosomes of Chrysanthemum. Heredity 7, 59–72 (1953).
Roxas, N. J., Tashiro, Y., Miyazaki, S., Isshiki, S. & Takeshita, A. Meiosis and pollen fertility in Higo chrysanthemum (Dendranthema×grandiflorum (Ramat.) Kitam. J. Jpn. Soc. Hortic. Sci. 64, 161–168 (1995).
Vogelmann, A. Chrysanthemen (Ulmer Eugen Verlag, Stuttgart, 1969).
Yang, W., Glover, B. J., Rao, G. Y. & Yang, J. Molecular evidence for multiple polyploidization and lineage recombination in the Chrysanthemum indicum polyploid complex (Asteraceae). New Phytol. 171, 875–886 (2006).
Su, J. et al. Current achievements and future prospects in the genetic breeding of chrysanthemum: a review. Hortic. Res. 6, 109 (2019).
Hibrand Saint-Oyant, L. et al. A high-quality genome sequence of Rosa chinensis to elucidate ornamental traits. Nat. Plants 4, 473–484 (2018).
Song, C. et al. The Chrysanthemum nankingense genome provides insights into the evolution and diversification of chrysanthemum flowers and medicinal traits. Mol. Plant 11, 1482–1491 (2018).
Hirakawa, H. et al. De novo whole-genome assembly in Chrysanthemum seticuspe, a model species of Chrysanthemums, and its application to genetic and gene discovery analysis. DNA Res. 26, 195–203 (2019).
Nakano, M. et al. A chromosome-level genome sequence of Chrysanthemum seticuspe, a model species for hexaploid cultivated chrysanthemum. Commun. Biol. 4, 1–11 (2021).
van Lieshout, N. et al. De novo whole-genome assembly of Chrysanthemum makinoi, a key wild chrysanthemum. G3 12, jkab358 (2022).
Wen, X. et al. The Chrysanthemum lavandulifolium genome and the molecular mechanism underlying diverse capitulum types. Hortic. Res. 9, uhab022 (2022).
Liu, H. et al. Whole-transcriptome analysis of differentially expressed genes in the ray florets and disc florets of Chrysanthemum morifolium. BMC Genomics 17, 1–17 (2016).
Wang, J. et al. Transcriptomic and hormone analyses reveal mechanisms underlying petal elongation in Chrysanthemum morifolium ‘Jinba’. Plant Mol. Biol. 93, 593–606 (2017).
Ding, L. et al. Comprehensive characterization of a floral mutant reveals the mechanism of hooked petal morphogenesis in Chrysanthemum morifolium. Plant Biotechnol. J. 17, 2325–2340 (2019).
Ding, L. et al. The core regulatory networks and hub genes regulating flower development in Chrysanthemum morifolium. Plant Mol. Biol. 103, 669–688 (2020).
Dong, W. et al. Transcriptome analysis of the molecular mechanism of Chrysanthemum flower color change under short-day photoperiods. Plant Physiol. Bioch. 146, 315–328 (2020).
Wang, H. et al. Characterization of in vitro haploid and doubled haploid Chrysanthemum morifolium plants via unfertilized ovule culture for phenotypical traits and DNA methylation pattern. Front. Plant Sci. 5, 738 (2014).
Zhang, J. et al. Allele-defined genome of the autopolyploid sugarcane Saccharum spontaneum L. Nat. Genet. 50, 1565–1573 (2018).
Ou, S., Chen, J. & Jiang, N. Assessing genome assembly quality using the LTR Assembly Index (LAI). Nucleic Acids Res. 46, e126–e126 (2018).
Shen, C. et al. The chromosome-level genome sequence of the autotetraploid alfalfa and resequencing of core germplasms provide genomic resources for alfalfa research. Mol. Plant 13, 1250–1261 (2020).
Wu, D. et al. Genomic insights into the evolution of Echinochloa species as weed and orphan crop. Nat. Commun. 13, 1–16 (2022).
Shen, Q. et al. The genome of Artemisia annua provides insight into the evolution of Asteraceae family and artemisinin biosynthesis. Mol. Plant 11, 776–788 (2018).
Badouin, H. et al. The sunflower genome provides insights into oil metabolism, flowering and Asterid evolution. Nature 546, 148–152 (2017).
Yang, F. S. et al. Chromosome-level genome assembly of a parent species of widely cultivated azaleas. Nat. Commun. 11, 1–13 (2020).
Wendel, J. F., Jackson, S. A., Meyers, B. C. & Wing, R. A. Evolution of plant genome architecture. Genome Biol. 17, 1–14 (2016).
Liu, B. et al. Mikania micrantha genome provides insights into the molecular mechanism of rapid growth. Nat. Commun. 11, 1–13 (2020).
Scaglione, D. et al. The genome sequence of the outbreeding globe artichoke constructed de novo incorporating a phase-aware low-pass sequencing strategy of F1 progeny. Sci. Rep. 6, 1–17 (2016).
Panchy, N., Lehti-Shiu, M. & Shiu, S. H. Evolution of gene duplication in plants. Plant Physiol. 171, 2294–2316 (2016).
Chen, F., Tholl, D., Bohlmann, J. & Pichersky, E. The family of terpene synthases in plants: a mid‐size family of genes for specialized metabolism that is highly diversified throughout the kingdom. Plant J. 66, 212–229 (2011).
Edger, P. P. et al. Origin and evolution of the octoploid strawberry genome. Nat. Genet. 51, 541–547 (2019).
Shi, J. et al. Chromosome conformation capture resolved near-complete genome assembly of broomcorn millet. Nat. Commun. 10, 1–9 (2019).
Grant, V. Plant Speciation (Columbia Univ. Press, 1981).
Watanabe, K. Studies on the control of diploid-like meiosis in polyploid taxa of Chrysanthemum. Theor. Appl. Genet. 66, 9–14 (1983).
Chen, F. D., Zhao, H. B., Li, C., Chen, S. M. & Fang, W. M. Advances in cytology and molecular cytogenetics of the genus Chrysanthemum. J. Nanjing Agric. Univ. 31, 118 (2008).
Xu, Y. F., Chen, F. D., Chen, S. M., Teng, N. J. & Li, F. T. Research on self-setting characteristics, meiotic behavior and pollen fertility of five chrysanthemum cultivars. Acta Botanica Boreal-Occidentalia Sin. 29, 469–474 (2009).
Klie, M., Schie, S., Linde, M. & Debener, T. The type of ploidy of chrysanthemum is not black or white: a comparison of a molecular approach to published cytological methods. Front. Plant Sci. 5, 479 (2014).
Nguepjop, J. R. et al. Evidence of genomic exchanges between homeologous chromosomes in a cross of peanut with newly synthetized allotetraploid hybrids. Front. Plant Sci. 7, 1635 (2016).
Edwards, K. et al. A reference genome for Nicotiana tabacum enables map-based cloning of homeologous loci implicated in nitrogen utilization efficiency. BMC Genomics 18, 1–14 (2017).
Huang, G. et al. Genome sequence of Gossypium herbaceum and genome updates of Gossypium arboreum and Gossypium hirsutum provide insights into cotton A-genome evolution. Nat. Genet. 52, 516–524 (2020).
Ma, Y. P. et al. Origins of cultivars of Chrysanthemum—Evidence from the chloroplast genome and nuclear LFY gene. J. Syst. Evol. 58, 925–944 (2020).
Mitros, T. et al. Genome biology of the paleotetraploid perennial biomass crop Miscanthus. Nat. Commun. 11, 1–11 (2020).
Chen, H. et al. Allele-aware chromosome-level genome assembly and efficient transgene-free genome editing for the autotetraploid cultivated alfalfa. Nat. Commun. 11, 1–11 (2020).
Ranallo-Benavidez, T. R., Jaron, K. S. & Schatz, M. C. GenomeScope 2.0 and Smudgeplot for reference-free profiling of polyploid genomes. Nat. Commun. 11, 1–10 (2020).
Liu, P. L., Wan, Q., Guo, Y. P., Yang, J. & Rao, G. Y. Phylogeny of the genus Chrysanthemum L.: evidence from single-copy nuclear gene and chloroplast DNA sequences. PLoS One 7, e48970 (2012).
Dowrick, G. J. & El-Bayoumi, A. The origin of new forms of the garden. Chrysanthemum. Euphytica 15, 32–38 (1966).
Theißen, G., Melzer, R. & Rümpler, F. MADS-domain transcription factors and the floral quartet model of flower development: linking plant development and evolution. Development 143, 3259–3271 (2016).
Kondrashov, F. A., Rogozin, I. B., Wolf, Y. I. & Koonin, E. V. Selection in the evolution of gene duplications. Genome Biol. 3, 1–9 (2002).
Juntheikki‐Palovaara, I. et al. Functional diversification of duplicated CYC2 clade genes in regulation of inflorescence development in Gerbera hybrida (Asteraceae). Plant J. 79, 783–796 (2014).
Elomaa, P., Zhao, Y. & Zhang, T. Flower heads in Asteraceae—recruitment of conserved developmental regulators to control the flower-like inflorescence architecture. Hortic. Res. 5, 36 (2018).
Tähtiharju, S. et al. Evolution and diversification of the CYC/TB1 gene family in Asteraceae—a comparative study in Gerbera (Mutisieae) and sunflower (Heliantheae). Mol. Biol. Evol. 29, 1155–1166 (2012).
Bello, M. A., Cubas, P., Álvarez, I., Sanjuanbenito, G. & Fuertes-Aguilar, J. Evolution and expression patterns of CYC/TB1 genes in Anacyclus: phylogenetic insights for floral symmetry genes in Asteraceae. Front. Plant Sci. 8, 589 (2017).
Shen, C. Z., Chen, J., Zhang, C. J., Rao, G. Y. & Guo, Y. P. Dysfunction of CYC2g is responsible for the evolutionary shift from radiate to disciform flowerheads in the Chrysanthemum group (Asteraceae: Anthemideae). Plant J. 106, 1024–1038 (2021).
Huang, D. et al. Identification and characterization of CYC-like genes in regulation of ray floret development in Chrysanthemum morifolium. Front. Plant Sci. 7, 1633 (2016).
Costa, M. M. R., Fox, S., Hanna, A. I., Baxter, C. & Coen, E. Evolution of regulatory interactions controlling floral asymmetry. Development 132, 5093–5101 (2005).
Raimundo, J. et al. A subcellular tug of war involving three MYB‐like proteins underlies a molecular antagonism in Antirrhinum flower asymmetry. Plant J. 75, 527–538 (2013).
Liu, M. Liu’s Chrysanthemum Manuals (1104).
Ohmiya, A., Kishimoto, S., Aida, R., Yoshioka, S. & Sumitomo, K. Carotenoid cleavage dioxygenase (CmCCD4a) contributes to white color formation in chrysanthemum petals. Plant Physiol. 142, 1193–1201 (2006).
Mason, A. S. & Wendel, J. F. Homoeologous exchanges, segmental allopolyploidy, and polyploid genome evolution. Front. Genet. 11, 1014 (2020).
Sun, Y., Shang, L., Zhu, Q. H., Fan, L. & Guo, L. Twenty years of plant genome sequencing: achievements and challenges. Trends Plant Sci. 27, 391–401 (2021).
Zheng, Y. et al. Allele‐aware chromosome‐scale assembly of the allopolyploid genome of hexaploid Ma Bamboo (Dendrocalamus latiflorus Munro). J. Integr. Plant Biol. 64, 649–670 (2022).
Chong, X. et al. A SNP-enabled assessment of genetic diversity, evolutionary relationships and the identification of candidate genes in chrysanthemum. Genome Biol. Evol. 8, 3661–3671 (2016).
Ma, Y. P. et al. Origin of Chrysanthemum cultivars—Evidence from nuclear low-copy LFY gene sequences. Biochem. Syst. Ecol. 65, 129–136 (2016).
Qi, S. et al. Natural interploidy hybridization among the key taxa involved in the origin of horticultural chrysanthemums. J. Syst. Evol. 60, 1–10 (2021).
Park, S. K., Arens, P., Esselink, D., Lim, J. H. & Shin, H. K. Analysis of inheritance mode in chrysanthemum using EST-derived SSR markers. Sci. Hortic. 192, 80–88 (2015).
Chen, F. D., Chen, P. D. & Li, H. J. Genome analysis and their phylogenetic relationships of several wild species of Chrysanthemum in China. Acta Hort. Sin. 23, 67–72 (1996).
Stebbins, G. L. Jr. Types of polyploids: their classification and significance. Adv. Genet. 1, 403–429 (1947).
Ramírez-González, R. et al. The transcriptional landscape of polyploid wheat. Science 361, eaar6089 (2018).
Yang, J. et al. The genome sequence of allopolyploid Brassica juncea and analysis of differential homeolog gene expression influencing selection. Nat. Genet. 48, 1225–1232 (2016).
Chin, C. S. et al. Phased diploid genome assembly with single-molecule real-time sequencing. Nat. Methods 13, 1050–1054 (2016).
Chin, C. S. et al. Nonhybrid, finished microbial genome assemblies from long-read SMRT sequencing data. Nat. Methods 10, 563–569 (2013).
Li, H. Toward better understanding of artifacts in variant calling from high-coverage samples. Bioinformatics 30, 2843–2851 (2014).
Adey, A. et al. In vitro, long-range sequence information for de novo genome assembly via transposase contiguity. Genome Res. 24, 2041–2049 (2014).
Walker, B. J. et al. Pilon: an integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS One 9, e112963 (2014).
Zhang, X., Zhang, S., Zhao, Q., Ming, R. & Tang, H. Assembly of allele-aware, chromosomal-scale autopolyploid genomes based on Hi-C data. Nat. Plants 5, 833–845 (2019).
Durand, N. C. et al. Juicebox provides a visualization system for Hi-C contact maps with unlimited zoom. Cell Syst. 3, 99–101 (2016).
Parra, G., Bradnam, K. & Korf, I. CEGMA: a pipeline to accurately annotate core genes in eukaryotic genomes. Bioinformatics 23, 1061–1067 (2007).
Simão, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V. & Zdobnov, E. M. BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212 (2015).
Cook, C. E., Bergman, M. T., Cochrane, G., Apweiler, R. & Birney, E. The European Bioinformatics Institute in 2017: data coordination and integration. Nucleic Acids Res. 46, D21–D29 (2018).
Ghosh, S. & Chan, C. K. Analysis of RNA-seq data using TopHat and Cufflinks. Methods Mol. Biol. 1374, 339–361 (2016).
Haas, B. J. et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat. Protoc. 8, 1494–1512 (2013).
Haas, B. J. et al. Improving the Arabidopsis genome annotation using maximal transcript alignment assemblies. Nat. Protoc. 31, 5654–5666 (2003).
Stanke, M. & Morgenstern, B. AUGUSTUS: a web server for gene prediction in eukaryotes that allows user-defined constraints. Nucleic Acids Res. 33, W465–W467 (2005).
Burge, C. & Karlin, S. Prediction of complete gene structures in human genomic DNA. J. Mol. Biol. 268, 78–94 (1997).
Majoros, W. H., Pertea, M. & Salzberg, S. L. TigrScan and GlimmerHMM: two open source ab initio eukaryotic gene-finders. Bioinformatics 20, 2878–2879 (2004).
Guigo, R. Assembling genes from predicted exons in linear time with dynamic programming. J. Comput. Biol. 5, 681–702 (1998).
Johnson, A. D. et al. SNAP: a web-based tool for identification and annotation of proxy SNPs using HapMap. Bioinformatics 24, 2938–2939 (2008).
Haas, B. J. et al. Automated eukaryotic gene structure annotation using EVidenceModeler and the Program to Assemble Spliced Alignments. Genome Biol. 9, 1–22 (2008).
Altschul, S. F. et al. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 25, 3389–3402 (1997).
Smit, A. F. & Hubley, R. RepeatModeler Open-1.0 (Institute for Systems Biology, 2008).
Xu, Z. & Wang, H. LTR_FINDER: an efficient tool for the prediction of full-length LTR retrotransposons. Nucleic Acids Res. 35, W265–W268 (2007).
Chen, N. Using Repeat Masker to identify repetitive elements in genomic sequences. Curr. Protoc. Bioinforma. 5, 4–10 (2004).
Ellinghaus, D., Kurtz, S. & Willhoeft, U. LTRharvest, an efficient and flexible software for de novo detection of LTR retrotransposons. BMC Bioinforma. 9, 1–14 (2008).
Steinbiss, S., Willhoeft, U., Gremme, G. & Kurtz, S. Fine-grained annotation and classification of de novo predicted LTR retrotransposons. Nucleic Acids Res. 37, 7002–7013 (2009).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Tang, H. et al. Synteny and collinearity in plant genomes. Science 320, 486–488 (2008).
Li, L., Stoeckert, C. J. & Roos, D. S. OrthoMCL: identification of ortholog groups for eukaryotic genomes. Genome Res. 13, 2178–2189 (2003).
Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313 (2014).
Yang, Z. PAML 4: phylogenetic analysis by maximum likelihood. Mol. Biol. Evol. 24, 1586–1591 (2007).
Han, M. V., Thomas, G. W., Lugo-Martinez, J. & Hahn, M. W. Estimating gene gain and loss rates in the presence of error in genome assembly and annotation using CAFE 3. Mol. Biol. Evol. 30, 1987–1997 (2013).
McKenna, A. et al. The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Takagi, H. et al. QTL‐seq: rapid mapping of quantitative trait loci in rice by whole genome resequencing of DNA from two bulked populations. Plant J. 74, 174–183 (2013).
Anders, S. & Huber, W. Differential expression analysis for sequence count data. Genome Biol. 11, R106 (2010).
Langfelder, P. & Horvath, S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinforma. 9, 1–13 (2008).
Shannon, P. et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 13, 2498–2504 (2003).
Kumar, S. et al. MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 35, 1547 (2018).
Kumar, S., Stecher, G., Suleski, M. & Hedges, S. B. TimeTree: a resource for timelines, timetrees, and divergence times. Mol. Biol. Evol. 34, 1812–1819 (2017).
Acknowledgements
This research was funded by the National Natural Science Foundation of China (32230098 to F.D.C., 31930100 to J.F.J., 32030098 to S.M.C., 32172613 to H.B.W., 32172609 to A.P.S.), the Natural Science Fund of Jiangsu Province (BK20190076 to A.P.S., BK20210395 to J.S.S.), China Agriculture Research System (CARS-23-A18 to F.D.C.), the National Key Research and Development Program of China (2021YFD1200205 to J.F.J., 2022YFF1003104 to S.M.C. 2022YFD1200504 to F.Z.), the “JBGS” Project of Seed Industry Revitalisation in Jiangsu Province (JBGS[2021]020 to F.D.C.), the European Union’s Horizon 2020 research and innovation program from European Research Council (833522 to Y.V.dP.) and the Methusalem funding from Ghent University (BOF.MET.2021.0005.01 to Y.V.dP.), and a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institution.
Author information
Authors and Affiliations
Contributions
F.D.C., A.P.S., J.F.J. and S.M.C. conceived and designed the study. H.B.W. prepared the genome sequencing material. A.P.S., Z.R.Z., X.T.Z. and J.S.S. led the bioinformatics analyses. F.D.C., W.M.F., S.M.C., Z.Y.G., J.F.J, F.Z. and S.Z. provided the chrysanthemum materials. H.B.W. and J.H. conducted the FISH analysis. F.Z., L.D., Y.L., D.W.J. and C.W.C. performed the BSA-seq and RNA-seq analyses. L.J.Z., J.L.Z., Z.Y.Y. and D.J.S. cloned the CCD4 genes. J.S.S. developed the figures. A.P.S., J.S.S., Z.R.Z., X.T.Z., Y.V.dP. and F.D.C. interpreted the data and drafted the manuscript. J.S.S., A.P.S., Z.R.Z. and X.T.Z. wrote the manuscript. Y.V.dP., A.P.S., J.S.S., X.T.Z., Z.X.W., L.K.W., B.Q.D., F.C. and F.D.C. revised the manuscript. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Sachiko Isobe, Jianquan Liu, and Amit Rai for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Song, A., Su, J., Wang, H. et al. Analyses of a chromosome-scale genome assembly reveal the origin and evolution of cultivated chrysanthemum. Nat Commun 14, 2021 (2023). https://doi.org/10.1038/s41467-023-37730-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-023-37730-3
This article is cited by
-
CmDOF18 positively regulates salinity tolerance in Chrysanthemum morifolium by activating the oxidoreductase system
BMC Plant Biology (2024)
-
Comparative transcriptome analysis provides molecular insights into heterosis of waterlogging tolerance in Chrysanthemum indicum
BMC Plant Biology (2024)
-
Haplotype-resolved genome assembly provides insights into evolutionary history of the Actinidia arguta tetraploid
Molecular Horticulture (2024)
-
Genomic evidence for rediploidization and adaptive evolution following the whole-genome triplication
Nature Communications (2024)
-
Transcriptomic and metabolomic analyses reveal CmMYB308 as a key regulator in the pink flower color variation of ‘Dante Purple’ chrysanthemum
Plant Cell Reports (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.