Abstract
Purpose
To investigate the clinical features and treatment outcomes of IgG4-related ophthalmic disease (IgG4-ROD) among idiopathic orbital inflammatory disease (IOID) patients.
Methods
The medical records of 165 biopsy-proven IOID patients were retrospectively reviewed. Biopsy specimens were immunostained to detect IgG4 and IgG, and data regarding the clinicopathologic features, treatment outcomes, and recurrence were analyzed.
Results
Among the 165 IOID patients enrolled, 100 (60.6%) were histopathologically IgG4-positive. The IgG4-positive patients had a lower rate of painful swelling or mass (17.0% versus 33.8%, p = 0.013), a longer symptom duration (p = 0.070), and a lower proportion of eyelid hyperemia (39.0% versus 58.5%, p = 0.014) than the IgG4-negative patients. Increased Ki-67 expression (15.02 ± 6.86%, p < 0.001) was observed in the IgG4-positive patients with characteristic pathological manifestations (more lymphocyte infiltration, nodular plasma cell infiltration, and follicular hyperplasia). IgG4-positive group had a higher recurrence rate in the subgroup of patients treated with surgery plus oral glucocorticoids (p = 0.046), and combined radiotherapy group has a higher cumulative proportion with recurrence (p = 0.011).
Conclusion
Over 60% of biopsy-proven IOID were classified as IgG4-ROD with a stronger proliferation potential. Additional radiotherapy after surgical debulking with oral corticosteroids still has a higher relapse rate, and more effective treatments are needed to prevent recurrence.
Similar content being viewed by others
Introduction
IgG4-related disease (IgG4-RD) is a recently recognized tumefactive condition characterized by excessive IgG4-positive plasma cell infiltration [1, 2]. This immune-mediated disease synchronously or metachronously involves various organs, and the preferred nomenclature for orbit and ocular IgG4-RD is IgG4-related ophthalmic disease (IgG4-ROD). Previous studies suggested that IgG4-ROD and idiopathic orbital inflammatory disease (IOID), a non-granulomatous inflammatory process with an unknown etiology and clear heterogeneity, shared overlapping clinical and pathological manifestations [3, 4], indicating that IgG4-ROD might be a component of IOID. However, the precise role of IgG4 in IOID remains to be fully elucidated [5].
IgG4-ROD represents a distinct condition with different clinical and pathological features [6, 7, 8]. However, most reports were small case series involving mainly Asian populations from Japan and Korea as well as Caucasian populations in the West [6], there have been few studies involving Chinese populations. The purpose of this study was to assess the proportion of IgG4-ROD among IOID cases in a large Southern China-based cohort and to explore the clinicopathologic features and treatment outcomes of patients with IOID.
Methods
Patients
This study was a retrospective case series. The medical records of 165 biopsy-proven IOID patients were reviewed from December 2015 to October 2018 at Zhongshan Ophthalmic Center of Sun Yat-sen University. The exclusion criteria were as follows: (1) thyroid-associated ophthalmopathy; (2) granulomatous inflammation or infection; (3) lymphoid hyperplasia or lymphoma; (4) IgG4 and IgG staining was unavailable. Written informed consent was obtained from all patients, and the study was approved by the Institutional Review Board of Zhongshan Ophthalmic Center and carried out according to the tenets of the Declaration of Helsinki.
Clinical, biological, and radiological information
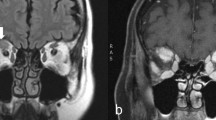
Clinical information, including age at diagnosis, sex, clinical manifestations, disease management, and follow-up data, were retrieved from the medical charts of patients. Whole blood cell counts, serum IgG4 levels, and complement C3 and C4 concentrations were recorded. All participants underwent imaging examination (computerized tomography or magnetic resonance imaging), and the laterality and locations (including the lacrimal gland, orbital fat, extraocular muscle, optic nerve, eyelid, and conjunctiva) as well as the presence of paranasal sinusitis were recorded.
Histopathological analysis
All samples obtained at the time of diagnosis were blindly analyzed by two pathologists (PZ and LT). The formalin-fixed and paraffin-embedded sections were subjected to haematoxylin–eosin and immunohistochemical staining. The intensities of fibrosis (collagenous or storiform), lymphocytic infiltration, plasmacytic infiltration (nodular or diffuse), eosinophilic infiltration, and follicular hyperplasia were semi-quantified as absent (0), small amount (1), moderate amount (2), and marked amount (3) [3].
Immunohistochemical staining for IgG4 (mouse monoclonal, MRQ-44, Gene Tech, Shanghai, China), IgG (mouse polyclonal, Gene Tech, Shanghai, China), and Ki-67 (mouse monoclonal, UMAB107, ZSGB-Bio, Beijing, China) was performed. For each section, the mean numbers of IgG4 and IgG positive cells in three high-power fields (HPFs) were calculated, and the ratio of IgG4/IgG was recorded.
Diagnostic criteria for IgG4-ROD
A diagnosis of IgG4-ROD was made according to the comprehensive diagnostic criteria [9]. Pathologically, patients were categorized into IgG4-positive and IgG4-negative. Specimens with a ratio of IgG4/IgG > 40% and >10 IgG4-positive plasma cells/HPF were scored as positive, which including definite and probable diagnosis of IgG4-ROD.
Treatment and outcomes
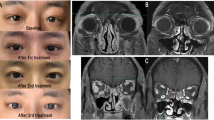
The enrolled patients underwent biopsy or surgical debulking with oral corticosteroids and additional treatment (radiotherapy or radiotherapy plus chemotherapy), if necessary. Notably, patients with diffuse swelling were treated with biopsy, and patients with masses type underwent surgical debulking. 20–24 Gy fractional radiotherapy was used for residual masses or refractory cases.
During the follow-up period (19.49 ± 10.16 months), the participants were examined in the outpatient department every 3–6 months according to their conditions, with telephone follow-up interviews at specific intervals. Treatment response was categorized as complete response (CR) and partial response (PR). CR was defined as complete remission at the last follow-up achieved clinically and radiologically. PR was defined as incomplete clinical remission or more than two treatment courses or therapies required. Recurrence was defined as the re-emergence of symptoms or the enlargement of lesions during the follow-up period.
Statistical analyses
Categorical variables are presented as numbers (percentages) and were evaluated using the chi-square test or Fisher’s exact test. Continuous variables are expressed as the mean ± standard deviation (SD), and a t test or Mann–Whitney U-test was performed unless otherwise indicated. Survival curves for recurrence were using the Kaplan–Meier method and compared between groups using log-rank tests. SPSS version 22.0 (SPSS, Chicago, Illinois, USA) was utilized for statistical analyses, and p < 0.05 was considered significant. As this study was an exploratory work, no priori statistical power calculation has been carried out.
Results
Baseline demographic and laboratory data of patients with IgG4-positive and IgG4-negative
A total of 165 patients with biopsy-proven IOID were ultimately enrolled in the study. Of these, 100 (60.6%) patients were pathologically IgG4-positive. The lacrimal gland was the most commonly affected site and no conjunctival involvement was found in this study. The demographic and laboratory data of the patients are displayed in Table 1. The mean age at diagnosis in the IgG4-positive patients was 43.30 ± 16.50 years, and the male proportion was higher than that in the IgG4-negative patients (60.0% versus 38.5%, p = 0.007). Bilateral involvement was more common in the IgG4-positive patients than in the IgG4-negative patients (49.0% versus 20.0%, p < 0.001). The IgG4-positive patients usually presented with painless swelling or a mass (p = 0.013); eyelid hyperemia was relatively rare (39.0%, p = 0.014), and the symptom duration tended to be longer (median = 12 months, interquartile range (IQR) = 4.00–33.00 months, p = 0.070). An increased proportion of IgG4-positive patients presented paranasal sinusitis (68.0%, p = 0.005), while diplopia was more common in the IgG4-negative patients (29.2%, p = 0.010).
An increased proportion of eosinophils (median=3.45%, IQR = 1.50–7.78%, p < 0.001), significantly elevated serum IgG4 levels (median=3.68 g/L, IQR = 0.74–13.48 g/L, p = 0.002) and decreased C3 levels (1.17 ± 0.22 g/L, p = 0.020) were found in the IgG4-positive group, albeit there was no difference with regard to previous autoimmune diseases or an allergy history (p = 0.362).
Histological features and correlations between IgG4+ plasma cells and clinical variables in IOID patients
To estimate the pathological implications of IgG4 in tissue, we compared the histological characteristics of the IgG4-positive and IgG4-negative patients (Table 2). The IgG4-positive group showed an increased lymphocytic infiltration (87.0%, p < 0.001), nodular plasmacytic infiltration (p < 0.001), follicular hyperplasia (p < 0.001), and Ki-67 expression (15.02 ± 6.86%, p < 0.001). Notably, storiform fibrosis was rare (18.2%) and coexisted with collagen fibrosis. The comparisons of orbital fibrosis type and eosinophilic infiltration did not reach statistical significance. Correlations between IgG4+ plasma cells and clinical parameters are summarized in Table 3. IgG4+ plasma cells were positively correlated with eosinophils (r = 0.226, p = 0.004) and Ki-67 (r = 0.396, p < 0.001), while negatively correlated with serum C3 (r = −0.338, p = 0.029). Figure S1 displayed representative pathologic findings of IgG4-ROD of present study.
Clinical outcomes of IgG4-positive and IgG4-negative IOID patients
During follow-up period (19.49 ± 10.16 months), the treatment outcomes and recurrence of 134 patients were recorded. One hundred and twenty-four patients treated with oral prednisolone were administrated at dose of 0.6–0.8 mg/kg/day for 1st week and then tapered by 10 mg every 3–4 weeks. Additional radiotherapy was given in ten cases, and one patient progressed to require chemotherapy after radiotherapy had failed.
Sixty-five (48.5%) patients reached a CR and thirty-seven (27.6%) patients experienced a relapse. Recurrence occurred in 32 patients in surgery plus oral glucocorticoids group, 5/10 patients in the additional treatment group relapsed. Recurrence was more frequent in patients with IgG4-positive group than in those with IgG4-negative group (32.2% versus 19.1%) (Table 1), especially in the subgroup of patients treated with surgery plus oral glucocorticoids (31.3% versus 14.6%, p = 0.046) (Fig. 1). Furthermore, the cumulative proportion without recurrence was higher in the surgery plus oral glucocorticoids group than additional treatment (radiotherapy) group (p = 0.011) (Fig. 2).
Discussion
Compared with other published articles, this study contained the largest sample size to undergo IgG4 immunohistochemistry. Most of the literature consisted of small case series [7, 10] and it is difficult to generalize their results. In addition, large samples are needed due to the differential diagnoses are still obstacles in the study of IgG4-RD.
This study analysed the prevalence of IgG4-RD in pathologically confirmed IOID patients. Regardless of the pathological or comprehensive diagnostic criteria, more than 60% of IOID patients were classified as IgG4-RD. A higher proportion of IgG4-ROD in IOID patients was observed in Japanese studies from 2008 to 2012 [11,12,13], suggesting that the increasing rate may be caused by an increasing awareness of IgG4-RD among clinicians. Our results (60.6%) were concordant with those from a study from Japan (61.5%) [13] and slightly reduced in Korea (45.8%) [7]. However, our results were higher than those from western countries, France (37.1 and 40.0%) [8, 14] and Australia (23.6%) [10], and slightly higher than the United States (52.4%) [3]. These results suggest that the prevalence of IgG4-ROD in IOID patients may be influenced by ethnicity. IOID is a group of heterogeneous diseases, and the differences in the clinical, imaging, and histology characteristics suggest that IgG4-ROD may have different pathogenesis and pathophysiology mechanisms. IgG4-positive IOID was predominant in males, while other autoimmune diseases were more prevalent in females [15]. Nevertheless, there was no significant gender advantage when compared with IgG4-associated AIP [16, 17]. Consistent with the results of a previous study [8], IgG4-positive was mainly manifested as more diplopia, less eyelid congestion, a trend toward longer symptom duration, painless swelling and bilateral involvement, which was contrary to the literature that suggested more monocular involvement [18, 19]. In addition, this study has suggested that IgG4-positive is dominated by space-occupying effects and is more insidious and causes less “inflammation” than IgG4-negative.
The proliferation indicator Ki-67 can be used as a prognostic and predictive marker [20]. Ki-67 expression was significantly higher in IgG4-positive patients than in IgG4-negative patients, indicating a stronger proliferation potential in IgG4-ROD, and positively correlated with IgG4+ plasma cells. Lymphoma was detected in patients with prior IgG4-RD diagnoses [21]; two IgG4-positive patients developed large B-cell lymphoma and MALT lymphoma during the follow-up period, as previously mentioned in the meta-analysis [6]. We inferred that IgG4 in tissue increased the risk of lymphoma development in IOID patients.
There are no international treatment guidelines for IgG4-ROD. Systemic glucocorticoids are the first-line agent for IgG4-RD, but recurrence is common [2]. Disease-modifying antirheumatic drugs (methotrexate, mycophenolate mofetil, infliximab, adalimumab, and rituximab (RTX)), radiotherapy and surgery are reported treatment modalities for IgG4-RD [22]. Adjunctive orbital radiotherapy with systemic corticosteroids may help to achieve better disease control, but the precise role of adjunctive orbital radiotherapy on the ocular adnexal IgG4-RD is still unknown [23]. In this study, 27.6% of IOID patients experienced recurrence in the follow-up. Incomplete surgical resection, biopsy with residual mass, or unalleviated clinical manifestation after corticosteroids treatment in patients treated with additional radiotherapy may help to explain why combined radiotherapy group has a higher cumulative proportion with recurrence. Inflammatory masses were basically removed to avoid disease recurrence in the surgical debulking with oral glucocorticoids patients.
Due to limited patient data, the efficacy of orbital radiotherapy is still uncertain and the value of surgical debulking requires future investigation [24]. There are few reports of radiotherapy in patients with IOID, and this is the first report to document the long-term outcomes of radiotherapy in the IOID patients. Radiation therapy has a well-defined role in cases of steroid failure or in patients unable to tolerate steroid therapy. Orbital radiotherapy with systemic corticosteroids achieved stable clinical manifestations in refractory ocular adnexal IgG4-RD [23]. RTX is used for refractive patients [25], but relapses are frequent after B-cell reconstitution. Systematic RTX maintenance retreatment is statistically associated with a longer relapse-free survival [26]. To avoid excessive destructive treatment, we need to find more effective treatments (such as other immunosuppressants) to prevent recurrence.
Our study had the following limitations: (1) this study was retrospective; (2) the postoperative serum IgG4 levels were not detected; (3) most of the patients did not undergo extraorbital systemic evaluation; and (4) RTX was not used for refractory cases. IgG4 is considered to be a less reliable marker for diagnosis and assessing response to treatment. IgG4-secreting plasmablasts/plasma cells was newly proposed to be a potentially useful biomarker have been proposed [27]. Prospective studies and long-term follow-up are required to ascertain serological, clinical, and imaging changes after RTX or other effective treatments, and look for new biomarkers for diagnosis and assess prognosis.
In conclusion, our data indicated that more than 60% of patients were diagnosed with IgG4-ROD and this is the largest audit of treatment outcomes in Southern China. According to the IgG4 classification in the tissues of IOID patients, patients with IgG4-positive and IgG4-negative had significant differences in clinical pathology, suggesting that there may be distinct pathogenic mechanisms for IgG4-ROD. Our results highlighted the following aspects: (1) IgG4-positive is dominated by space-occupying effects and is more insidious; (2) Ki-67 is positively correlated with IgG4+ plasma cells, indicting IgG4-ROD has a stronger proliferation potential; (3) IgG4-positive group has a higher recurrence rate in the subgroup of patients treated with surgery plus oral glucocorticoids; (4) combined radiotherapy group has a higher cumulative proportion with recurrence, and other treatment is urgently needed to prevent recurrence. Further studies should focus on the pathogenesis of IgG4-ROD, and prospective studies to observe the natural history and long-term prognosis of IOID patients should be conducted.
Summary
What was known before
-
IgG4-related IOID represents a distinct condition with different clinical and pathological features. However, most reports were small case series involving mainly Asian populations from Japan and Korea as well as Caucasian populations in the West.
What this study adds
-
This study is a large sample study of IgG4-related IOID involving Chinese population and depicts the relative differences in clinical features and treatment outcomes.
References
Stone JH, Zen Y, Deshpande V. IgG4-related disease. N Engl J Med. 2012;366(6):539–51.
Kamisawa T, Zen Y, Pillai S, Stone JH. IgG4-related disease. Lancet. 2015;385(9976):1460–71.
Plaza JA, Garrity JA, Dogan A, Ananthamurthy A, Witzig TE, Salomao DR. Orbital inflammation with IgG4-positive plasma cells: manifestation of IgG4 systemic disease. Arch Ophthalmol. 2011;129(4):421–8.
Wah C, Yuen HKL, Chan JKC. Chronic sclerosing dacryoadenitis: part of the spectrum of IgG4-related Sclerosing disease? Am J Surg Pathol. 2007;31(4):643–5.
Mahajan VS, Mattoo H, Deshpande V, Pillai SS, Stone JH. IgG4-related disease. Annu Rev Pathol. 2014;9:315–47.
Nicholas A, Daniel K, Dinesh S. IgG4-related orbital disease: a meta-analysis and review. Acta Ophthalmol. 2013;91(8):694–700.
Sa HS, Lee JH, Woo KI, Kim YD. IgG4-related disease in idiopathic sclerosing orbital inflammation. Br J Ophthalmol. 2015;99(11):1493–7.
Deschamps R, Deschamps L, Depaz R, Coffin-Pichonnet S, Belange G, Jacomet PV, et al. High prevalence of IgG4-related lymphoplasmacytic infiltrative disorder in 25 patients with orbital inflammation: a retrospective case series. Br J Ophthalmol. 2013;97(8):999–1004.
Hisanori U, Kazuichi O, Yasufumi M, Mitsuhiro K, Motohisa Y, Takako S, et al. Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD), 2011. Mod Rheumatol. 2012;22(1):21–30.
Andrew NH, Sladden N, Kearney DJ, Selva D. An analysis of IgG4-related disease (IgG4-RD) among idiopathic orbital inflammations and benign lymphoid hyperplasias using two consensus-based diagnostic criteria for IgG4-RD. Br J Ophthalmol. 2015;99(3):376–81.
Sato Y, Ohshima K, Ichimura K, Sato M, Yamadori I, Tanaka T, et al. Ocular adnexal IgG4-related disease has uniform clinicopathology. Pathol Int. 2008;58(8):465–70.
Kubota T, Moritani S, Katayama M, Terasaki H. Ocular adnexal IgG4-related lymphoplasmacytic infiltrative disorder. Arch Ophthalmol. 2010;128(5):577–84.
Takahira M, Ozawa Y, Kawano M, Zen Y, Hamaoka S, Yamada K, et al. Clinical aspects of IgG4-related orbital inflammation in a case series of ocular adnexal lymphoproliferative disorders. Int J Rheumatol. 2012;2012:635473.
Abad S, Martin A, Heran F, Cucherousset N, Mouriaux F, Heron E, et al. IgG4-related disease in patients with idiopathic orbital inflammation syndrome: data from the French SIOI prospective cohort. Acta Ophthalmol. 2019;97:e648–56.
Yamamoto M, Harada S, Ohara M, Suzuki C, Naishiro Y, Yamamoto H, et al. Clinical and pathological differences between Mikulicz’s disease and Sjogren’s syndrome. Rheumatology 2005;44(2):227–34.
Nishimori I, Tamakoshi A, Otsuki M, Research Committee on Intractable Diseases of the Pancreas, Ministry of Health, Labour, and Welfare of Japan. Prevalence of autoimmune pancreatitis in Japan from a nationwide survey in 2002. J Gastroenterol. 2007;42(18):6–8.
Kanno A, Masamune A, Okazaki K, Kamisawa T, Kawa S, Nishimori I, et al. Nationwide epidemiological survey of autoimmune pancreatitis in Japan in 2011. Pancreas. 2015;44(4):535–9.
Origuchi T, Yano H, Nakamura H, Hirano A, Kawakami A. Three cases of IgG4-related orbital inflammation presented as unilateral pseudotumor and review of the literature. Rheumatol Int. 2013;33(11):2931–6.
Pasquali T, Schoenfield L, Spalding SJ, Singh AD. Orbital inflammation in IgG4-related sclerosing disease. Orbit. 2011;30(5):258–60.
Yerushalmi R, Woods R, Ravdin PM, Hayes MM, Gelmon KA. Ki67 in breast cancer: prognostic and predictive potential. Lancet Oncol. 2010;11(2):174–83.
Igawa T, Hayashi T, Ishiguro K, Maruyama Y, Takeuchi M, Takata K, et al. IgG4-producing lymphoma arising in a patient with IgG4-related disease. Med Mol Morphol. 2016;49(4):243–9.
Poo SX, Tham CSW, Smith C, Lee J, Cairns T, Galliford J, et al. IgG4-related disease in a multi-ethnic community: clinical characteristics and association with malignancy. QJM. 2019;112:763–9.
Lin YH, Yen SH, Tsai CC, Kao SC, Lee FL. Adjunctive orbital radiotherapy for ocular adnexal IgG4-related disease: preliminary experience in patients refractory or intolerant to corticosteroid therapy. Ocul Immunol Inflamm. 2015;23(2):162–7.
Detiger SE, Karim AF, Verdijk RM, van Hagen PM, van Laar JAM, Paridaens D. The treatment outcomes in IgG4-related orbital disease: a systematic review of the literature. Acta Ophthalmol. 2019;97(5):451–9.
Aouidad I, Schneider P, Zmuda M, Gottlieb J, Viguier M. IgG4-related disease with orbital pseudotumors treated with rituximab combined with palpebral surgery. JAMA Dermatol. 2017;153(3):355–6.
Ebbo M, Grados A, Samson M, Groh M, Loundou A, Rigolet A, et al. Long-term efficacy and safety of rituximab in IgG4-related disease: data from a French nationwide study of thirty-three patients. PLoS One. 2017;12(9):e0183844.
Lin W, Zhang P, Chen H, Chen Y, Yang H, Zheng W, et al. Circulating plasmablasts/plasma cells: a potential biomarker for IgG4-related disease. Arthritis Res Ther. 2017;19(1):25.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81700875, 81670887, and 81870689).
Author information
Authors and Affiliations
Contributions
All authors contributed to the collection, analysis, interpretation, and preparation of the data, as well as the review and approval of the manuscript and the final decision to submit the manuscript for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Chen, J., Zhang, P., Ye, H. et al. Clinical features and outcomes of IgG4-related idiopathic orbital inflammatory disease: from a large southern China-based cohort. Eye 35, 1248–1255 (2021). https://doi.org/10.1038/s41433-020-1083-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-020-1083-x
This article is cited by
-
Local recurrence as extraocular muscle lymphoma after 6 years of chronic myositis: a case report
BMC Ophthalmology (2022)