Abstract
Speech and language impairments are central features of CDK13-related disorder. While pathogenic CDK13 variants have been associated with childhood apraxia of speech (CAS), a systematic characterisation of communication has not been conducted. Here we examined speech, language, non-verbal communication skills, social behaviour and health and development in 41 individuals with CDK13-related disorder from 10 countries (male = 22, median-age 7 years 1 month, range 1–25 years; 33 novel). Most participants used augmentative and alternative communication (AAC) in early childhood (24/41). CAS was common (14/22). Performance varied widely across intellectual ability, social behaviour and expressive language skills, with participants ranging from within average through to the severely impaired range. Receptive language was significantly stronger than expressive language ability. Social motivation was a relative strength. In terms of a broader health phenotype, a quarter had one or more of: renal, urogenital, musculoskeletal, and cardiac malformations, vision impairment, ear infections and/or sleep disturbance. All had gross and fine motor impairments (41/41). Other conditions included mild-moderate intellectual disability (16/22) and autism (7/41). No genotype-phenotype correlations were found. Recognition of CAS, a rare speech disorder, is required to ensure appropriately targeted therapy. The high prevalence of speech and language impairment underscores the importance of tailored speech therapy, particularly early access to AAC supports.
Similar content being viewed by others
Introduction
CDK13 is part of the family of over 30 cyclin-dependent kinases (CDKs) expressed in humans [1]. CDK13 is involved in transcription and posttranscriptional processing [2] and plays a critical role in embryonic development [3]. CDK13 expression is detectable in the heart, brain and craniofacial area [3].
Pathogenic CDK13 variants cause CDK13-related disorder [4]. The literature reports over 60 individuals with this condition, with most published pathogenic CDK13 variants being missense and occurring de novo [4,5,6,7,8,9,10,11,12,13,14]. Physical features of CDK13-related disorder include recognisable upslanting palpebral fissures, epicanthal folds, a broad nasal bridge, thin upper lip, small mouth, posteriorly rotated ears, peg-shaped teeth, and curly hair [4,5,6,7,8, 14]. Other physical phenotypes include congenital cardiac, renal and skeletal abnormalities, hypotonia, feeding difficulties and a high-arched palate [5,6,7,8, 13].
The neurodevelopmental profile includes average intellectual ability through to moderate intellectual disability (ID), autism spectrum disorder (hereafter autism), attention-deficit/hyperactivity disorder (ADHD), epilepsy and sleep disturbances [14]. Speech and language are reported as among the most commonly impacted areas of neurodevelopment in CDK13-related disorder [5, 6, 8, 10, 13, 14]. Yet whilst communication difficulties are ubiquitously reported, there has been no specificity to the clinical diagnoses, with very general terms such as ‘speech and language delay’ being used. Nor have studies used standardised measures/assessment protocols. A pathogenic variant in CDK13 was recently associated with a rare and severe speech disorder, childhood apraxia of speech (CAS), in a gene discovery cohort of children ascertained for CAS [13]. However, there has been no systematic reverse phenotyping evaluation of speech or language deficits in a cohort of individuals with pathogenic CDK13 variants to date, to confirm this association with CAS. Further, the absence of a comprehensive speech and language evaluation in this population limits prognostic counselling and the provision of targeted intervention. Here, we systematically characterise speech and language abilities, and examine possible genotype-phenotype correlations, in children with CDK13-related disorder using standardised outcome measures.
Methods
Participants
Inclusion criteria were a molecularly confirmed pathogenic diagnosis of CDK13-related disorder. Exclusion criteria were the existence of other pathogenic variants in other genes associated with neurodevelopmental disorders. Participants were recruited internationally via an online CDK13 support group or via their treating clinical geneticist from French, Dutch, German, English and Spanish speaking backgrounds. The Royal Children’s Hospital Human Research Ethics Committee provided ethical approval (HREC 37353 A). Caregivers provided written informed consent for their children to participate, even in the case of the young adults in the study.
Health and development
Families completed caregiver questionnaires concerning individuals’ health and developmental history (Supplementary Table 1). Caregiver questionnaires were completed in the participants’ language: English, French, Dutch, German, and Spanish. Results were confirmed with a case history via telehealth and provision of additional reports (e.g., cognitive assessments, electroencephalogram results, and autism diagnostic reports), a successful method employed previously [15,16,17,18]. All English-speaking, verbal participants (i.e., those who used primarily spoken words to communicate) also completed a telehealth assessment with a university-trained speech pathologist.
Adaptive behaviour and motor skills
The Vineland Adaptive Behaviour Scales (VABS II/III) caregiver version, was completed online for English-, Spanish- (third edition) and French-speaking (second edition) participants [19, 20]. The VABS II/III provides standardised scores for communication, socialisation, self-care, activities of daily living, motor skills and an overall adaptive behaviour score (that does not include the motor skills subtest).
Language and social communication
The VABS II/III communication domain, with receptive, expressive and written skill subdomains, assessed language in English-, French- and Spanish-speaking individuals. English- and Dutch-speaking caregivers of participants younger than 2 years-old completed the Communication and Symbolic Behaviour Scales Developmental Profile (CSBS-DP) standardised questionnaire [21]. Children 4–16 years old completed the Children’s Communication Checklist – Second Edition (CCC-2) [22]. The CSBS-DP and CCC-2 assess speech, receptive and expressive language, non-verbal communication and social communication abilities. These tools were not available in other languages.
The Social Responsiveness Scale-2 (SRS-2) was completed by English- and Dutch-speaking families [23]. The SRS-2 caregiver questionnaire is standardised across three versions from pre-school- (>2 years) and school-aged children, to adulthood. The SRS-2 measures social behaviour based on the Diagnostic and Statistical Manual of Mental Disorders Fifth Edition (DSM-V) autism diagnostic criteria, and examines domains of [23]: social awareness, social cognition, social communication, social motivation, restricted interests, and repetitive behaviour [24].
Alternative communication methods and therapy
Minimally verbal children were defined as having less than 50 spoken words [17, 25] and were assessed using the Inventory of Potential Communicative Acts (IPCA) caregiver questionnaire [26]. This assessment investigates communication behaviours used by individuals of all ages, such as; facial expression, body movement, gesture, and augmentative and alternative communication (AAC) (e.g., sign language, communication devices) across a range of functions including greeting, protesting, and commenting. Caregiver surveys provided information on current therapy goals and AAC systems.
Speech
Verbal children were assessed using standardised tools which examined performance across speech domains of: articulation, phonology, stuttering, dysarthria and CAS.
Articulation disorder (distorted production of a speech sound, e.g., a lisp), phonological delay (where a child is persisting in the use of speech sound error patterns made by >10% of younger children, e.g., fronting of fricatives or velars; gliding, etc) and phonological disorder (atypical speech sound error patterns, defined as errors made by <10% of children in the general population, e.g., initial consonant deletion, backing of sounds) were diagnosed using the Phonology and Inconsistency subtests of the Diagnostic Evaluation of Articulation and Phonology (DEAP) [27] and confirmed during a five minute conversational speech sample. Stuttering was measured with a Likert scale rating from 0 (no stuttering) to 9 (severe stuttering) based on the conversational sample [28]. Ratings for CAS were made using the American Speech and Hearing Association CAS Technical Report Protocol consensus features [29]. CAS diagnoses are made based on three criteria: (i) inconsistent speech production; (ii) disrupted and prolonged co-articulatory transitions; and (iii) prosodic errors. Features of these three criteria were operationally defined and rated using a checklist, previously applied in other populations with genetic disorder [15,16,17,18, 30, 31]. Dysarthria was rated in the presence of neuromuscular tone disruption to one or more of the sub-systems for speech (e.g., phonatory, articulatory) as well as the presence of specific dysarthric features (e.g., hypernasality), as rated using the Mayo Clinic Dysarthria Classification System [32]. Dysarthria and CAS ratings were made based on these operationalised criteria using single word responses to the DEAP Phonology and Inconsistency subtests, the conversational speech sample, and diadochokinetic speech tasks (e.g., ‘pataka’) [27, 30]. Clinician and caregiver reports documented speech diagnoses for non-English speaking individuals, who were not able to be directly assessed.
The Intelligibility in Context Scale (ICS) [33] was completed by caregivers to assess how well a participant is understood (intelligibility) based on their speech in the past month, with different communication partners (e.g., friends, family members) on a scale of 1 (never understood) to 5 (always understood) (Supplementary Fig. 1).
Feeding and nonspeech oral motor skills
English-speaking children aged 6 months to 7 years completed the Child Oral and Motor Proficiency Scale (ChOMPS) [34]. This assessment separates eating and drinking skills into: complex movement patterns (e.g., licking food from lips), basic movements (e.g., sitting), oral motor coordination (e.g., moving jaw to chew), and fundamental oral skills (e.g., closing lips). Caregivers of children who drooled completed the Drooling Impact Scale [35], whereby the frequency and impact of drooling was rated 1 (none) through 10 (all the time). The structure and function of the oral articulators was assessed [36] to support interpretation of the speech and feeding results.
Statistical analyses
Non-parametric statistical analyses were conducted due to the data not being normatively distributed. A Wilcoxon Signed Rank tests compared individual differences between VABS II/III receptive and expressive language scores, CCC-2 domains, and SRS-2 domains. To explore genotype-phenotype associations, a Mann Whitney test compared VABS II/III adaptive behaviour and communication scores between groups with different variants. A Kruskal-Wallis test compared VABS II/III domains. Ages, VABS II/III, CCC-2, CSBS-DP, SRS-2, and ICS data were reported using descriptive statistics detailing central tendency (mean, median) and variability (SD).
Results
Participants
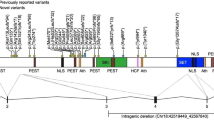
Forty-one participants were recruited, ranging in age from 1 year 6 months to 18 years 9 months (Median = 7 years 1 month; Male = 22) (Table 1). Participants were from the United States (19), France (5), Australia (4), United Kingdom (3), Canada (3), the Netherlands (2), Belgium (1), Spain (1), Switzerland (1) and Qatar (1). Thirty-three participants were novel and eight were previously published [IDs 18, 23 [4], ID 31 [5], ID 7 [5], IDs 5, 9, 33 [14], ID 27 [13, 14]].
Most participants had missense variants (n = 37) (Fig. 1). Seventeen had the same missense variant (n = 17, c.2525 A > G, p.Asn842Ser). Six other participants shared a further missense variant (c.2149 G > A, p.Gly717Arg) and a further fifteen participants had other missense variants. Of the four participants who did not have missense variants, three had truncating variants (IDs 39, 40, 41) and one had a splice site variant (ID 38). Thirty-nine were confirmed de novo and two were of unknown inheritance (IDs 14, 18). The average age at genetic diagnosis was 6 years and 4 months.
Health and development
Medical conditions
Cardiac malformations (15/41) and heart surgeries were common (9/14) (Table 1). The most frequent cardiac malformation was atrial septal defect (9/14). Renal and urogenital abnormalities were present in almost half the cohort (19/41) (Supplementary Table 2).
Most participants (34/41) had undergone brain magnetic resonance imaging (MRI) or computerised tomography (CT) scans and almost half had findings (15/34) (Table 1) including hypoplasia of the corpus callosum (6/14) and Chiari malformation (2/14). Three participants had epilepsy (IDs 6, 19, 31) with all taking anticonvulsant medication for seizure management. Insomnia symptoms were evident (23/41), including frequent waking (12/23), early waking (8/23), difficulty falling asleep (9/23) and little sleep (2/23) or a combination of these issues.
Musculoskeletal problems were apparent (15/31), although findings were heterogeneous. Hypotonia was common in infancy (11/41). Small stature and difficulties gaining weight were frequent (11/40).
Infant feeding difficulties (34/41) were treated with nasogastric (10/34) and gastrostomy tubes (5/34) (Supplementary Table 2). Participants 29 and 33 had tracheomalacia in infancy and participant 31 also had a tracheostomy tube in situ at the time of assessment, at age >13 years. Complex dentition was observed (24/41) (Supplementary Table 2).
Ear infections (16/41) were common. One participant had mild, conductive hearing loss (ID 34, 25-39dBHL). A subset of participants had procedures for tympanostomy tubes (7/40), tonsillectomies (7/41), and adenoidectomies (8/41). More than half the group had vision impairment (24/41), with myopia (12/24) and strabismus (11/24) being the predominant diagnosis. Shared facial features were also evident (Supplementary Table 2).
Development
Most participants learned to sit and walk after the expected milestones of 7 (35/41) and 15 months (33/41), respectively (Tables 2, 3 for milestones). Twenty-six participants had delayed acquisition of first words (>15 months) and four had not yet said their first words (aged between 2- to 12-years). Similarly, only eight participants made short sentences at the expected age (2–3 years), and 19 participants (aged up to 15 years old) were not yet combining words. The remaining 15 participants began combining words between 5 to 7 years of age.
Neurodevelopmental conditions
Co-occurring neurodevelopmental conditions were common (Table 2) (25/41). Of the 22 participants with psychometric cognitive assessment data available, most had a moderate ID (12/22, 35-55 FSIQ) and some had a mild ID (4/22, 55-70 FSIQ). Six participants scored in either the very low (4/22, 70-85 FSIQ) or average ranges (2/22, >85 FSIQ). Of the remaining 19 participants without psychometric based cognitive assessments, 16 had paediatrician-reported developmental delays. Intellectual abilities are often not assessed until a child begins school and half of the participants without cognitive assessment had not yet started school (9/19). DSM-V [24] diagnoses reported by caregivers and confirmed by health professional reports included developmental coordination disorder, (8/25), autism (7/25) and ADHD (5/25). One individual had neurobehavioural disorder associated with prenatal alcohol exposure. Other formal diagnoses included sensory processing disorder (13/25), and auditory processing disorder (ID 25). Almost a fifth (7/41) of participants had an anxiety disorder, and participant 3 was also diagnosed with depression.
Education
Twenty-three participants were school aged or older at the time of assessment. Two children were home-schooled, 13 attended special schools and eight attended mainstream settings.
Most participants had accessed speech therapy (39/41). Many accessed physiotherapy (36/41) and occupational therapy (36/41) for gross and fine motor impairments.
Adaptive behaviour and motor skills
A range of profiles was noted in the range of adaptive behaviour composite scores (mean = 61.86). No single domain of daily living, socialisation or communication, was significantly different to any other (p = 0.26).
VABS II/III scores from participants with the same variant (c. 2525 A > G, p.Asn842Ser, n = 15), were compared to the rest of the cohort (n = 22) (Fig. 2). There was no statistically significant difference between participants with this variant and the rest of the cohort on the adaptive behaviour composite score (p = 0.39, p > 0.05) or their communication score (p = 0.36, p > 0.05). However, when descriptively assessed via a boxplot (Fig. 2), the participants with the same variant tended to be more similar to one another on their adaptive behaviour composite score (SD = 15.97) than the rest of the group (SD = 23.39).
The Vineland Adaptive Behaviour Scales Second and Third Edition (mean = 100, SD = 15) scores <70 are low/severe, 71–85 moderately low/moderate, >85 average/within normal limits (minimum score = 20, maximum score = 140). Domain scores for c.2525 A > G, p.Asn842Ser missense variants (indicated by the first box plot in each domain): communication (mean = 63.53), daily living (mean = 55.93), socialisation (mean = 61.20), motor skills (mean = 67.20). Domain scores for all other variants (indicated by the second box plot in each domain): communication (mean = 64.05), daily living (mean = 58.38), socialisation (mean = 68.33), motor skills (mean = 57.95). There were no significant differences between the two groups for the four Vineland domains. Outliers = •, median = centre line, mean = x.
Language and social communication
At a group level, average receptive language skills (mean = 10.20) were significantly stronger than average expressive language ability (mean = 8.71) (p = 0.03, p < 0.05) on the VABS II/III. Overall communication scores (test standard score mean = 100, SD = 15) indicated generally low communication skills (mean = 63.76), however scores ranged from within average limits to severely impaired (Fig. 2, Table 3).
Six female participants had expressive and/or receptive language skills within the normal range of performance (IDs 5, 26, 27, 36, 38, 40). There were five participants, however, with average social behaviour and moderate to severe language impairment (IDs 7, 13, 14, 22, 29).
CSBS-DP emotion and eye gaze (mean = 9.75), words (mean = 8) and understanding (mean = 7.75) were in the average range (Table 4). High variability in the group was also observed across CSBS-DP subdomains.
Language skills on the CCC-2 (n = 22) and CSBS-DP (n = 4) were low across most subdomains (mean = 10, SD = 3) (Table 4). CCC-2 subdomain scores ranged between −1 to −2 SD of the mean, except for speech, syntax and semantic scores which were greater than −2 SD (Supplementary Fig. 2). Speech was the lowest subdomain and was significantly different to all subdomains except syntax (p < 0.05, Table 4). High variability amongst the group was observed in all subdomains.
SRS-2 T-scores demonstrated a range of social communication abilities, from within normal limits (9/27) to severely impaired (10/27) (normative mean = 60, SD = 10) (Fig. 3, Table 3). Moderate to severe scores indicate a high likelihood of autism (15/27), however only five had confirmed diagnoses of autism and four other individuals had sensory processing disorder. Restricted and repetitive behaviours were moderately impaired (mean = 70.53) and were significantly different from social motivation (p = 0.0003, p < 0.01). All other social communication domains were mildly impaired.
Higher T scores indicate higher autistic traits (mean = 60, SD = 10, range 34–90). ≤59 social behaviour within normal limits, 60–65 mild difficulty, 66–75 moderate difficulty, ≥75 severe difficulty. Social awareness (mean = 65.5), social cognition (mean = 64.3), social communication (mean = 65.03), social motivation (mean = 62.4), restricted interests and repetitive behaviour (mean = 70.53). Individual data points = •, median = centre line, mean = x.
Alternative communication methods and therapy
Caregivers identified speech production (32/41), receptive language skills (23/41), social language skills (20/41), low-technology AAC (e.g., communication boards, 19/41), high-technology AAC (e.g., speech generating device, such as an electronic tablet, 19/41), and Key Word Sign/Makaton (KWS, i.e., using single signs to communicate, 12/41) as beneficial focuses of speech therapy sessions. One participant was receiving specific speech therapy targeting CAS.
More than half of the cohort used AAC (24/41) (Table 3). KWS was commonly the sole AAC system (11/24) and was used by younger children (<3 years old, 4/11) or those with verbal communication who on occasion used single signs while speaking (6/11), with only one older participant (>3 years old) using KWS as their primary communication system. All other AAC users had graphic AAC systems (Table 3). Four minimally verbal participants older than 3 years old, when children typically learn to combine words, did not have an AAC system.
Eleven minimally verbal participants completed the IPCA (Supplementary Table 3). In terms of symbolic communication, almost half the group used speech to greet and farewell others and seek attention (5/11) and used sign to request ‘more’ (6/11). Graphic AAC was mostly used for requesting objects (4/11). Participants also used symbolic gesture (e.g., hugging, pointing) to request objects and seek comfort and answer yes or no. Challenging behaviours were exhibited when participants felt angry (6/11).
Speech
Twenty-two verbal, English-speaking, participants had a standardised speech assessment (Table 3). CAS and phonological delay were the most frequently occurring speech disorders (14/22), with co-occurring diagnoses being common (11/14). Dysarthria (8/22), phonological (10/22) and articulation disorders (8/22, interdental lisp 6/8, lateral lisp 2/8) were also present.
The fourteen participants with CAS had features across all three diagnostic criteria (Fig. 4) [29, 30]. The most prevalent CAS features distinct from dysarthric features were inconsistent production of the same phoneme (consonant or vowel) across different words (92.86%), difficulty sequencing sounds and syllables (85.71%), and increased errors with increased word length and complexity (71.43%). Participant 3 had a history of CAS that had largely resolved. One participant (ID 36) had a mild stutter [28]. Four of the eight participants with dysarthria also had CAS. Dysarthric features were seen across all speech dimensions (Fig. 4). Only five participants had a speech disorder diagnosis in isolation (CAS 1/5, dysarthria 2/5, articulation disorder 1/5, phonological delay 1/5). Two participants did not have a speech disorder.
Figure 4a. Specific speech features in 8 individuals with dysarthria across speech dimensions (prosody, articulation, resonance, pitch, volume, quality) rated by the Mayo Clinic Dysarthria Classification System (Duffy, 2005). Figure 4b. Specific speech features in 13 individuals with childhood apraxia of speech (CAS) rated by the ASHA CAS Technical Report protocol’s diagnostic criteria (2007), operationalised by Mei et al (2008). DDK diadochokinetic speech task (e.g., say ‘pataka’).
For non-English speaking participants who were not able to be assessed over telehealth (n = 19), 10 caregivers reported clinically diagnosed speech disorders and nine individuals were minimally verbal. Of the 10 with clinically diagnosed speech disorders, articulation disorder was most common (9/10), followed by CAS (6/10), phonological delay (5/10) and disorder (5/10), and dysarthria (4/10).
Intelligibility ranged from 1 (never understood) to 5 (always understood) across a variety of communication partners (n = 41) (Supplementary Fig. 1). Participants were most intelligible to their caregivers (mean = 3.98, sometimes to usually understood), and least intelligible to strangers (mean = 2.65, rarely to sometimes understood).
Feeding and nonspeech oral motor skills
The ChOMPS (n = 17) indicated that feeding difficulties were almost universal (16/17) in children younger than 7 years old (Supplementary Fig. 3). Most participants (15/17) had highly concerning feeding skills (<5th percentile). Only one participant had feeding skills within normal limits. Complex movement patterns (e.g., licking food off the top lip) were descriptively most challenging, while basic movement patterns were a strength (e.g., bringing a bottle to mouth). Drooling prevalence ranged from never drooling to frequent drooling, with drooling generally delayed and resolving only by the late primary school years.
Oral motor skills were impaired in all participants able to complete testing (21/21, Table 3). Greatest difficulty was seen in moving the tongue vertically and horizontally (14/21 and 13/21, respectively), as well as in rounding the lips (10/21), and coordinating two or more non-speech movements (13/21, e.g., bite then lick lips).
Discussion
Here we provide the most comprehensive characterisation of speech and language in CDK13-related disorder. With the addition of 33 novel cases to the existing 60 cases in the literature, we also provide a description of over a third of all published cases of CDK13-related disorder to date.
Speech production was substantially more impaired when compared to other communication domains, such as social communication. Speech disorder was the most prevalent phenotypic feature, where CAS was dominant (63.6%) and considerably more prevalent than general population diagnostic frequencies (0.1%) [37, 38]. CAS frequently co-occurred with other speech disorders. Despite the frequency of CAS, only one participant was receiving a CAS-specific intervention. This lack of recognition of CAS may be hindering opportunities for more targeted therapy with negative implications for longer-term outcomes. Expressive syntax (the arrangement of words to form sentences), can also be impacted by severe speech disorder [39]. Development of speech was generally protracted, e.g., not combining words until after 15 months of age, with most participants using AAC to support their communication needs while verbal speech developed. Some participants remained minimally verbal or had severely impaired speech intelligibility, requiring AAC aides into adolescence. Comprehensive AAC supports are required so that individuals can meet all their communication needs where verbal speech is inadequate.
Historically, the non-specific terms speech/language and delay/disorder have been used interchangeably to describe the features observed in CDK13-related disorder. Our systematic characterisation of specific speech and language diagnoses is critical for the provision of tailored interventions. Our findings suggest access to AAC in the early years, with ongoing support for AAC into adolescence if needed, is of paramount importance to optimise communication outcomes. Typically developing children are immersed in their language system from birth and say their first words around 12 months of age. A child with CDK13-related disorder should be exposed to both verbal language and AAC before their first birthday to allow for optimal learning opportunities with a trained speech pathologist [40]. AAC is not used as a replacement for verbal development, but rather it is known to support verbal development [41] and particularly support growth of expressive vocabulary and grammar [42]. AAC should continue to be implemented if the child cannot be understood by different individuals across communication settings (e.g., school, home, with friends), so as to meet all of their communication needs. Consequently, AAC systems that can execute a range of communication functions should be considered. Further, a combination of AAC systems can be used, known as a multi-modal approach, such as KWS and a high-tech graphic AAC [43].
The most common speech disorder in this group, CAS, disrupts motor planning and programming for speech. In line with this speech motor involvement, fine and gross motor impairment were also widespread. The frequency of co-occurring fine, gross and speech motor disorders implicates an underlying mechanism of disordered movement planning abilities in CDK13-related disorder. Evidence for the motor involvement in CDK13-related disorder is consistent with neurobiological evidence showing high CDK13 expression in the cerebellum; which is responsible for precision of speech sounds and physical movement amongst other skills [44,45,46]. Further, almost half of those with MRI findings in our cohort had hypoplasia of the corpus callosum. Callosal aberrations have also been implicated in speech disorder [47]. Further evidence is required to better understand the neurobiological bases of CDK13-related disorder and their association to speech disorder.
Six female participants had average expressive and/or receptive language skills. These participants demonstrate that speech and language disorders may dissociate, given that all of this group had speech disorders in the presence of intact language abilities. Participants with moderate to severe social behavioural impairment all had similarly impaired language skills. Yet there were also participants with average social behaviour and impaired language ability. Hence, impaired social behaviour was always associated with impaired language skills, but not vice versa. It is important to acknowledge that speech and language impairment can be present in the absence of ID, with linguistic behaviours having their own biologically driven neurological pathways [48].
Receptive language was a relative strength when compared to expressive language ability. In genetic conditions with a high prevalence of CAS systematically characterised to date, stronger receptive language skills compared to expressive language skills have not been seen across cohorts [15,16,17,18]. This suggests that receptive language may be a strength for individuals with CDK13-related disorder, at least relative to other genetic conditions involving CAS that are understood at this time [15,16,17,18]. A limitation of our study was that assessment tools and access to trained clinicians for examining speech and language in individuals from non-English speaking backgrounds were more limited than those available for English-speaking individuals.
Moderate ID generally corresponded with moderate to severely impaired language skills. However, three participants with very low to average FSIQ had moderate to severely impaired language skills. Consequently, intellectual and language ability are typically congruent, but can be distinct from one another in some individuals, indicating distinct neurobiological pathways underpinning language impairment and intellectual disability.
The incidence of ID was less in this cohort than previously reported [14], with around one quarter of assessed participants having borderline to average FSIQ. However, this cohort may be biased, as caregivers may only self-refer to a speech and language study for children with stronger language and intellectual ability. Additionally, previous literature largely characterises individuals drawn from cohorts of children ascertained for ID, so here we broaden the phenotype of CDK13-related disorder with the inclusion of individuals without ID.
The occurrence of other neurodevelopmental conditions such as autism and ADHD was consistent with previously published cases [14]. However, most participants with moderate to severe social behaviour impairment in this study did not have a clinical diagnosis of autism. For the first time, sensory processing disorder was identified as a commonly occurring feature of CDK13-related disorder, with over one-quarter of the cohort affected. However, sensory processing disorder and autism are difficult to differentially diagnose, especially on a background of intellectual and language impairment, and sensory processing disorder is not considered a DSM-V diagnosis [24, 49]. The range of co-occurring neurodevelopmental conditions highlights the importance of systematic neuropsychological assessment, to provide optimal, individualised support.
With regards to genotype-phenotype correlations, there was little evidence to indicate that genetic variants were closely associated with specific phenotypes. Of the 17 participants who shared the same variant, considerable heterogeneity emerged in intellectual, language, speech, and medical presentations.
The health and medical profile in CDK13-related disorder was expanded here with our addition of 33 novel cases to the literature. Feeding problems had a significant impact in infancy and early childhood. Similarly, renal, urogenital, and musculoskeletal malformations, and vision impairment were more common than cardiac malformations in our cohort who, as noted earlier, may have been a more biased group. Cryptorchidism was present in our cohort (38% of males), having been recently described in individuals with pathogenic CDK13 variants [14].
We are the first to characterise sleep disturbances in CDK13-related disorder, highlighting prevalent insomnia features. Sleep quality and duration can also negatively impact receptive and expressive language skills [50].
Rouxel & colleagues [14] linked anxiety with CDK13-related disorder in 50% of their cohort (all >7 years). Anxiety disorder was also present in our cohort, although less prevalent (17.5%, all >8 years bar one 5-year-old). The median age of Rouxel et al.’s cohort was markedly older (median age = 12 years) than our cohort (median age = 7 years).
In terms of clinical impacts of our work, neuropsychological assessments are recommended to assess cognitive abilities, given the incidence of ID and other neurodevelopmental conditions. Likewise, occupational and physiotherapy are warranted as fine and gross motor impairment was ubiquitous. Critically, our work suggests speech pathology services should be sought to implement AAC in early childhood, and then provide targeted speech and language therapy (e.g., evidence-based CAS therapy) when verbal speech develops.
In conclusion, we characterise speech and language in CDK13-related disorder and identify CAS as a common feature. Until this study, CAS had only been described in one individual (ID 27) in the literature, included in two previous studies [13, 14]. The profile of speech, language and ID, on the background of significant health disorders, emphasises the importance of comprehensive, multidisciplinary assessment and intervention for individuals with CDK13-related disorder.
Data availability
The datasets generated and analysed during this study are not publicly available because participants have not given permission for data to be made public but may be requested from the corresponding author (A.T.M) who could go back to the participants to request data sharing. Genotypic data were submitted to Decipher (https://decipher.sanger.ac.uk/).
Change history
08 December 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41431-023-01515-5
References
Colas P. Cyclin-dependent kinases and rare developmental disorders. Orphanet J Rare Dis. 2020;15:1–14.
Kohoutek J, Blazek D. Cyclin K goes with Cdk12 and Cdk13. Cell Div. 2012;7:1–10.
Nováková M, Hampl M, Vrábel D, Procházka J, Petrezselyová S, Procházková M, et al. Mouse model of congenital heart defects, dysmorphic facial features and intellectual developmental disorders as a result of non-functional CDK13. Front Cell Dev Biol. 2019:155. https://doi.org/10.3389/fcell.2019.00155.
Sifrim A, Hitz M-P, Wilsdon A, Breckpot J, Al Turki SH, Thienpont B, et al. Distinct genetic architectures for syndromic and nonsyndromic congenital heart defects identified by exome sequencing. Nat Genet. 2016;48:1060–5.
Bostwick BL, McLean S, Posey JE, Streff HE, Gripp KW, Blesson A, et al. Phenotypic and molecular characterisation of CDK13-related congenital heart defects, dysmorphic facial features and intellectual developmental disorders. Genome Med. 2017;9:1–9.
van den Akker W, Brummelman I, Martis L, Timmermans R, Pfundt R, Kleefstra T, et al. De novo variants in CDK13 associated with syndromic ID/DD: molecular and clinical delineation of 15 individuals and a further review. Clin Genet. 2018;93:1000–7.
Uehara T, Takenouchi T, Kosaki R, Kurosawa K, Mizuno S, Kosaki K. Redefining the phenotypic spectrum of de novo heterozygous CDK13 variants: three patients without cardiac defects. Eur J Med Genet. 2018;61:243–7.
Hamilton MJ, Caswell RC, Canham N, Cole T, Firth HV, Foulds N, et al. Heterozygous mutations affecting the protein kinase domain of CDK13 cause a syndromic form of developmental delay and intellectual disability. J Med Genet. 2018;55:28–38.
Carneiro TN, Krepischi AC, Costa SS, da Silva IT, Vianna-Morgante AM, Valieris R, et al. Utility of trio-based exome sequencing in the elucidation of the genetic basis of isolated syndromic intellectual disability: illustrative cases. Application Clin Genet. 2018;11:93.
Trinh J, Kandaswamy KK, Werber M, Weiss ME, Oprea G, Kishore S, et al. Novel pathogenic variants and multiple molecular diagnoses in neurodevelopmental disorders. J neurodevelopmental Disord. 2019;11:1–6.
Yakubov R, Ayman A, Kremer AK, van den Akker M. One-month-old girl presenting with pseudohypoaldosteronism leading to the diagnosis of CDK13-related disorder: a case report and review of the literature. J Med Case Rep. 2019;13:1–5.
Wang T, Hoekzema K, Vecchio D, Wu H, Sulovari A, Coe BP, et al. Large-scale targeted sequencing identifies risk genes for neurodevelopmental disorders. Nat Commun. 2020;11:1–13.
Hildebrand MS, Jackson VE, Scerri TS, Van Reyk O, Coleman M, Braden RO, et al. Severe childhood speech disorder: gene discovery highlights transcriptional dysregulation. Neurology. 2020;94:e2148–e67.
Rouxel F, Relator R, Kerkhof J, McConkey H, Levy M, Dias P, et al. CDK13-related disorder: Report of a series of 18 previously unpublished individuals and description of an epigenetic signature. Genet Med. 2022;24:1096–107.
Braden RO, Amor DJ, Fisher SE, Mei C, Myers CT, Mefford H, et al. Severe speech impairment is a distinguishing feature of FOXP1‐related disorder. Developmental Med Child Neurol. 2021;63:1417–26.
Morgan A, Braden R, Wong MM, Colin E, Amor D, Liégeois F, et al. Speech and language deficits are central to SETBP1 haploinsufficiency disorder. Eur J Hum Genet. 2021;29:1216–25.
Morison LD, Braden RO, Amor DJ, Brignell A, van Bon BW, Morgan AT. Social motivation a relative strength in DYRK1A syndrome on a background of significant speech and language impairments. Eur J Hum Genet. 2022;30:800–11.
St John M, Amor DJ, Morgan AT. Speech and language development and genotype–phenotype correlation in 49 individuals with KAT6A syndrome. Am J Med Genet Part A. 2022;188:3389–400.
Sparrow SS, Cicchetti DV, Balla DA. Vineland adaptive behavior scales Vineland-II: survey forms manual: Pearson Minneapolis, MN; 2005.
Sparrow SS, Cicchetti DV, Saulnier CA. Vineland-3: Vineland adaptive behavior scales. London: Psychological Corporation; 2016.
Wetherby AM, Prizant BM. Communication and symbolic behavior scales: developmental profile. Baltimore: Paul H Brookes; 2002.
Bishop DV. The Children’s communication checklist. London: Psychological Corporation; 2003.
Constantino JN, Gruber CP. Social responsiveness scale: SRS-2. Torrance, CA: Western psychological services; 2012.
Association AP. Diagnostic and statistical manual of mental disorders: DSM-5. Washington, DC: American Psychiatric Association; 2013.
Brignell A, Chenausky KV, Song H, Zhu J, Suo C, Morgan AT. Communication interventions for autism spectrum disorder in minimally verbal children. Cochrane Database Syst Rev. 2018.
Sigafoos J, Arthur-Kelly M, Butterfield N. Enhancing everyday communication for children with disabilities. Baltimore: Paul H Brookes Publishing; 2006.
Dodd B, Zhu H, Crosbie S, Holm A, Ozanne A. Diagnostic evaluation of articulation and phonology (DEAP). London: Psychology Corporation; 2002.
O’Brian S, Packman A, Onslow M, O’Brian N. Measurement of stuttering in adults. J Speech, Lang, Hearing Res. 2004;47:1081–7.
ASHA. Childhood apraxia of speech Rockville, MD: American speech-language-hearing association; 2007. Available from: https://www2.asha.org/articlesummary.aspx?id=8589947136.
Mei C, Fedorenko E, Amor DJ, Boys A, Hoeflin C, Carew P, et al. Deep phenotyping of speech and language skills in individuals with 16p11. 2 deletion. Eur J Hum Genet. 2018;26:676–86.
Morison LD, Meffert E, Stampfer M, Steiner-Wilke I, Vollmer B, Schulze K, et al. In-depth characterisation of a cohort of individuals with missense and loss-of-function variants disrupting FOXP2. J Med Genet. 2022. https://doi.org/10.1136/jmg-2022-108734.
Duffy JR. Motor speech disorders: substrates, differential diagnosis, and management second edition. St. Louis, Missouri: Elsevier Mosby; 2005.
McLeod S, Harrison LJ, McCormack J. The intelligibility in context scale: Validity and reliability of a subjective rating measure. J Speech Lang Hear Res. 2012;5-648–56.
Pados BF, Thoyre SM, Park J. Age‐based norm‐reference values for the child oral and motor proficiency scale. Acta Paediatrica. 2018;107:1427–32.
Reid SM, Johnson HM, Reddihough DS. The Drooling Impact Scale: a measure of the impact of drooling in children with developmental disabilities. Developmental Med Child Neurol. 2010;52:e23–e8.
Robbins J, Klee T. Clinical assessment of oropharyngeal motor development in young children. J Speech Hearing Disord. 1987;52:271–7.
Yoss K. Editor developmental apraxia of speech in children: familial patterns and behavioral characteristics. ASHA north central regional conference, Minneapolis; 1975.
Morley ME. The development and disorders of speech in childhood. Austin, TX: Churchill Livingstone; 1972.
Ekelman BL, Aram DM. Syntactic findings in developmental verbal apraxia. J Commun Disord. 1983;16:237–50.
Branson D, Demchak M. The use of augmentative and alternative communication methods with infants and toddlers with disabilities: a research review. Augmentative Alternative Commun. 2009;25:274–86.
Walters C, Sevcik RA, Romski M. Spoken vocabulary outcomes of toddlers with developmental delay after parent-implemented augmented language intervention. Am J Speech-Lang Pathol. 2021;30:1023–37.
Binger C, Light J. The morphology and syntax of individuals who use AAC: research review and implications for effective practice. Augmentative Alternative Commun. 2008;24:123–38.
lacono T, Mirenda P, Beukelman D. Comparison of unimodal and multimodal AAC techniques for children with intellectual disabilities. Augmentative Alternative Commun. 1993;9:83–94.
Atlas THP. CDK13 The Human Protein Atlasn.d. Available from: http://www.proteinatlas.org/ENSG00000065883-CDK13/brain.
Schmahmann JD, Sherman JC. The cerebellar cognitive affective syndrome. Brain: J Neurol. 1998;121:561–79.
Morgan AT, Liégeois F, Liederkerke C, Vogel AP, Hayward R, Harkness W, et al. Role of cerebellum in fine speech control in childhood: persistent dysarthria after surgical treatment for posterior fossa tumour. Brain Lang. 2011;117:69–76.
Luders E, Kurth F, Pigdon L, Conti-Ramsden G, Reilly S, Morgan AT. Atypical callosal morphology in children with speech sound disorder. Neuroscience 2017;367:211–8.
Kaspi A, Hildebrand MS, Jackson VE, Braden R, van Reyk O, Howell T, et al. Genetic aetiologies for childhood speech disorder: novel pathways co-expressed during brain development. Mol Psychiatry. 2022. https://doi.org/10.1038/s41380-022-01764-8.
Thurm A, Farmer C, Salzman E, Lord C, Bishop S. State of the field: differentiating intellectual disability from autism spectrum disorder. Front Psychiatry. 2019:526. https://doi.org/10.3389/fpsyt.2019.00526.
Bonuck K, Battino R, Barresi I, McGrath K. Sleep problem screening of young children by speech-language pathologists: a mixed-methods feasibility study. Autism Developmental Lang Impairments. 2021;6: https://doi.org/10.1177/23969415211035066.
Acknowledgements
Sincere thanks to the children, families and clinicians who took part in this project.
Funding
This work was supported by a National Health and Medical Research Council (NHMRC) Centre of Research Excellence in Speech and Language Neurobiology (CRE-SLANG) #1116976 awarded to A.T.M.; NHMRC Practitioner Fellowship #1105008 awarded to A.T.M., and an NHMRC Investigator grant #1195955 awarded to A.T.M. This work is also supported by the Victorian Government’s Operational Infrastructure Support Program.
Author information
Authors and Affiliations
Contributions
LDM: generated data, analysed data, interpreted data, wrote manuscript. OV: generated data, analysed data, interpreted data, revised manuscript. EF: analysed data, interpreted data, revised manuscript. FR: generated data, analysed data, revised manuscript. LF: generated data, analysed data, revised manuscript. FB: generated data, analysed data, revised manuscript. MV: generated data, analysed data, revised manuscript. MJ: generated data, analysed data, revised manuscript. NLD: generated data, analysed data, revised manuscript. DG: generated data, analysed data, revised manuscript. DJA: generated data, analysed data, interpreted data, revised manuscript. ATM: designed and conceptualised study, directed project, generated data, analysed data, interpreted data, wrote manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Ethics approval was obtained from the Royal Children’s Hospital, Melbourne, Human Research Ethics Committee (HREC 37353A). Adult participants and caregivers of child participants provided informed consent to participate in the study and for results of this study to be published.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Morison, L.D., van Reyk, O., Forbes, E. et al. CDK13-related disorder: a deep characterization of speech and language abilities and addition of 33 novel cases. Eur J Hum Genet 31, 793–804 (2023). https://doi.org/10.1038/s41431-022-01275-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41431-022-01275-8
This article is cited by
-
Speech and language classification in the human phenotype ontology
European Journal of Human Genetics (2024)
-
Unusual genomic variants require unusual analyses
European Journal of Human Genetics (2023)