Abstract
Communication difficulties are a core feature of Phelan-McDermid syndrome (PMS). However, a specific speech and language phenotype has not been delineated, preventing prognostic counselling and development of targeted therapies. We examined speech, language, social and functional communication abilities in 21 individuals with PMS (with SHANK3 involvement), using standardised assessments. Mean age was 9.7 years (SD 4.1) and 57% were female. Deletion size ranged from 41 kb to 8.3 Mb. Nine participants (45%) were non-verbal. Four (19%) had greater verbal ability, speaking in at least 4–5 word sentences, but with speech sound errors. Standard scores for receptive and expressive language were low (typically >3 SD below the mean). Language age equivalency was 13–16 months on average (range 2–53 months). There was a significant association between deletion size and the ability to use phrases. Participants with smaller deletion sizes were more likely to be able to use phrases (odds ratio: 0.36, 95% CI: 0.14–0.95, p = 0.040). Adaptive behaviour (life skills) was low in all areas (>2 SD below mean). Scores in communication were markedly lower than for daily living (p = 0.008) and socialisation (p < 0.001). A common linguistic profile was characterised by severe impairment across receptive, expressive and social language domains. Yet data indicated greater communicative intent than appeared to be capitalised by current therapies. Early implementation of augmentative (e.g. computer-assisted) modes of communication, alongside promotion of oral language, is essential to harness this intent, accelerate language development and reduce frustration. Future trials should examine the added benefit of targeted speech motor interventions in those with greater verbal capacity.
Similar content being viewed by others
Introduction
Phelan-McDermid syndrome (PMS) or 22q13 deletion syndrome is typically caused by heterozygous loss of function of SHANK3, through deletion or sequence variant, located at the terminus of the long arm of chromosome 22 [1,2,3]. SHANK3 encodes a scaffolding protein involved in the postsynaptic density of excitatory synapses and is expressed in brain regions important for cognition and language [3, 4]. Most deletions occur de novo, yet 20% of cases are inherited from a parent who carries a balanced chromosome rearrangement involving chromosome 22 [5]. PMS is estimated to occur in around 0.2–0.4% of individuals with neurodevelopmental disorders [1, 6].
PMS characteristics include intellectual disability (ID), autism spectrum disorder (ASD) and language disorder [2, 5, 7, 8]. In two studies of individuals with PMS from the USA (n = 32 and n = 17), all children had ID (~77% in the moderate to severe range), and between 73% and 84% had ASD [7, 9]. ASD characteristics of impaired social communication, poor eye contact, and repetitive and self-stimulatory behaviours [10] are common. ASD diagnosis can be challenging in PMS given the context of ID and severe communication impairment [11].
Gastrointestinal symptoms, epilepsy, mild dysmorphia (e.g. long eyelashes, prominent ears, bulbous nose, pointed chin, fleshy hands and dysplastic toenails), hypotonia and increased pain tolerance [2, 7, 10, 12, 13] are also seen in PMS. Developmental regression is reported in 30–65% of individuals and can impact language, motor and/or behavioural domains [7, 9, 14].
Language difficulties are pervasive and typically severe in PMS. Expressive language is often more impaired than receptive language [1, 7,8,9, 11, 15]. Two large studies have identified an association between deletion size and absence of oral language [6, 16]. One study (n = 84) identified a second locus on 22q13 that was associated with the absence of speech [6]. In this study, participants with breakpoints proximal to 46 Mb were at greater risk for absence of speech compared with those who had smaller deletions [6]. Another study (n = 70) found participants with absent speech had deletion breakpoints between 43.17 and 46.73 Mb and a median deletion size of 7 Mb [16]. Few studies (including those mentioned) have used standardised language tools to assess language. Therefore, a more refined linguistic phenotype beyond this level of present/absent speech has not been delineated such as specific speech (e.g. dyspraxia or dysarthria) or language deficits (e.g. across social communication, functional communication and expressive versus receptive domains) [17, 18].
Here we provide a precise speech and language phenotype in individuals with PMS using standardised clinical tools. We also explore the association between deletion size and receptive/expressive language phenotype. By providing detailed assessments of speech and language we examine whether there is an increased probability of specific linguistic behaviours associated with PMS compared with individuals without the syndrome [19].
Subjects and methods
Participants
Participants were recruited through the Victorian Clinical Genetics Services and Phelan McDermid Syndrome Foundation of Australia via social media. Inclusion criteria were (i) a genetic diagnosis of PMS with SHANK3 involvement, (ii) adequate English language proficiency of caregivers to complete questionnaires, and (iii) between 2 and 19 years of age. Twenty-six eligible children were identified and 21 participated. One family declined participation, one was unable to be contacted and three were unable to schedule appointments due to personal reasons. All participants were Australian. The 22q13 deletions and phenotypic data were submitted to the Decipher database (https://decipher.sanger.ac.uk/).
Measures
Caregivers completed a developmental and medical history form and interview [17, 20, 21]. All participants completed the assessment protocol, with the exception of one participant (P21) whose relevant data was extracted from medical reports. See Table 1 for a description of the assessment tools and their scoring. Regression in skills was measured retrospectively by parent report based on the Autism Diagnostic Interview-Revised (ADI-R; [22]). Speech pathologists with PhDs and >20 years of clinical experience (AB, AH, BC) administered the assessments in the home (n = 13) or at a research institute (n = 7).
Statistical analyses
To identify relative strengths, the Wilcoxon signed-ranks test for non-parametric data was used to compare language scores (receptive versus expressive) and other developmental domains (e.g. socialisation versus daily living skills). A non-parametric test was used due to potential violations of the normality assumption. Bonferroni correction was applied with p < 0.01 required to reach statistical significance. Simple logistic regression was used to explore the association between deletion size (Megabases, Mb) and expressive language outcome (presence of phrases/no phrases). Linear regression was used to explore the association between deletion size (Mb) and receptive and expressive domain raw scores on the Vineland Adaptive Behavior Scales- second edtition (VABS-II) and receptive and expressive language raw scores on the Preschool Language Scales - 5th edition (PLS-5) Visualisations of the residuals indicated potential issues of heteroscedasticity, and therefore the dependent variable was log-transformed before fitting the linear model used for analysis. The Wilcoxon rank sum (Mann–Whitney) test for non-parametric data was used to assess the difference in mean deletion size for those participants who did and did not use phrases. All analyses were conducted twice: first, including all participants in the sample (21 participants) and second, excluding participants with additional genetic diagnoses and mosaic presentation (16 participants). The overall results were very similar when excluding the aforementioned participants (i.e. the change in estimate of interest was very small relative to the associated standard error) and therefore we retained all 21 participants in the analyses to increase statistical power. Analyses were conducted using Stata Version 15 [23].
Ethics approval was obtained from the Royal Children’s Hospital, Melbourne, Human Research Ethics Committee (#37353 A).
Results
Participant characteristics
Table 2 describes the participant characteristics. Participants (12 female) were aged between 2 and 18 years (mean = 9.64, SD = 4.12). In three participants (P11, P15 and P18) the 22q13 deletion was associated with a ring chromosome 22, in one participant (P3) the 22q13 deletion was mosaic, being present in 40% of cells, and in one participant (P21) the 22q13 deletion was inherited from the mother, also diagnosed with PMS. The three participants with ring chromosome 22 were minimally verbal, however, they did not present with a notably different phenotype compared with the other participants with PMS. Four participants had additional genetic findings including 1q21.1 duplication, neurofibromatosis type 2 (associated with ring chromosome 22 [24]), Ehlers-Danlos syndrome and premutation carrier for fragile X. Ten participants (48%) had existing clinical diagnoses of ASD (typically from a paediatrician and multidisciplinary, skilled team as per health system processes in Australia) and 19 (100%) over the age of four had some degree of ID, ranging from moderate to severe across the group. Two participants (P1, P6) were too young for formal cognitive testing and ID diagnoses, but had developmental delays. Parents reported that 12 participants (57%) had regressed in areas including language and motor skills before the age of three. Only a small proportion (<25%) had seizures, vision impairment or hearing impairment. One (P18) had a confirmed permanent sensorineural hearing impairment associated with neurofibromatosis type 2 and wore hearing aids. Three had vision impairment: one with severe myopia, alternating exotropia, no binocular vision and optic nerve hyperplasia (P19); one with cortical vision impairment (P18); and one with hyperopia (P7). All were able to eat orally and 15 (71%) had feeding difficulties such as cramming, choking and eating a restricted range of textures. All 21 participants had impaired development of at least some fine and gross motor milestones.
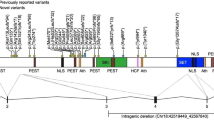
With one exception (P2, who had an additional genetic deletion), the six participants with the smallest 22q13.3 deletions were verbal and able to combine words into phrases. An additional participant (P14) with a deletion size of 3.3 Mb also spoke in phrases. One participant (P20) with a large deletion size of 8 Mb had a vocabulary of around 18 words and some stock phrases (e.g. ‘don’t touch’, ‘Elmo’s shoe’, ‘no mum’). This child also used around 13 key word signs. No discernible factors explained why this participant had stronger expressive language than expected given her deletion size. The remaining participants were minimally verbal (<10 verbal words) or non-verbal. Those participants who could combine words had a mean deletion size of 0.867 Mb and those who were minimally verbal or non-verbal had a mean deletion size of 6.6 Mb. Figure 1 provides the details of the locations and sizes of the 22q13.3 deletions.
Adaptive behaviour
Of the 20 participants with adaptive behaviour data, all scored below 70 (>2 SD below the mean) in the overall composite (mean = 48.6, SD = 10.6), indicating a low adaptive level across the sample (see Fig. 2). In relation to sub-domains, scores were lowest for communication (mean = 46.1, SD = 9.6), followed by daily living (mean = 50.3, SD = 11.8) and socialisation (mean = 52.9, SD = 11.9). The highest scoring domain was motor skills (mean = 61.0, SD = 6.6; >2 SD below mean), yet only five participants completed this composite as standard scores for this domain are only available up to 6 years, 11 months. The median communication composite was significantly lower than those for socialisation and daily living (z = −3.421, p = 0.0006; z = −2.675, p = 0.0075, respectively). There was no difference between social and daily living composite scores (z = 1.121, p = 0.2623).
Language
Most participants had severe language impairment, with standard scores on the PLS-5 (n = 6) greater than 3 SD below the mean in both receptive and expressive language domains. The PLS-5 has a mean standard score of 100, SD = 15. Performance was consistently low across auditory comprehension (a proxy for receptive language, mean = 50.0, SD = 0.0), expressive communication (mean = 52.2, SD = 3.7) and total language (mean = 50.2, SD = 0.4). Receptive vocabulary performance on the Peabody Picture Vocabulary Test- fourth edition (PPVT-4) was also severely impaired in the participants able to complete this test (P12, score 56, <3 SD below mean; P13, score 20, >3 SD below mean).
Figure 3 shows the age-equivalents in months for receptive and expressive language on the PLS-5 and the receptive and expressive sub-domains of the communication domain on the VABS-II. At the mean chronological age of 9.3 years, the mean age-equivalent for receptive language was 14.2 months (SD = 11.8; range 3–53) and for expressive language, 13.9 months (SD = 9.4; range 3–36) on the PLS-5. On the VABS-II, receptive language was a mean age-equivalent of 16.6 months (SD = 14.0; range 3–55) and expressive language was 14.2 months (SD = 12.0; range 2–42). There was no significant difference between receptive and expressive language median age-equivalents on the PLS-5 (z = −0.162, p = 0.9712). However, there was a significant difference between receptive and expressive language on the VABS-II (z = −3.006, p = 0.002), with receptive language being stronger.
Results from the Communication and Symbolic Behavior Scales Developmental Profile™ (CSBS-DP) (Table 3) showed the highest proportion of participants demonstrated strengths in the symbolic composite (8/20; 40%), followed by the speech composite (5/20; 25%), indicating these participants had abilities greater than the average 13–14 month-old child in those areas. No participants showed strengths in the social composite, indicating relative weakness and social communication impairment in 20/20 participants (100%).
The percentage of participants able to complete all sections of the PLS-5, as well as the age level that each task is expected to be acquired by in typically developing children is shown in the Supplementary Materials (Appendix 1 and 2). In auditory comprehension (Appendix 1), all participants were able to complete all tasks in the 0;0–0;2 age level. Thirteen participants (68.4%) responded to their name, 15/19 (78.9%) responded to the word ‘no’ and 17/19 (89.5%) were able to understand at least one specific word or phrase without gesture. The most advanced age level reached was 6;6–6;11, with one participant (P12; 5.3%) being able to complete tasks at this level. Two participants (P14 and P21) did not complete the PLS-5 and qualitative information was collected on their language abilities. P14 could follow routine instructions of 1–2 parts and identify basic body parts, everyday objects, verbs and negatives and some primary colours. P21 was able to follow routine 1–2 step directions and identify everyday objects and functions of objects. Receptive language for these two participants was estimated to be around a 3–4 year old level.
In expressive communication (see Supplementary Materials, Appendix 2), all participants achieved tasks up to the 0;3–0;5 age level. Twelve participants (63.2%) were able to babble two syllables, 10/19 (52.6%) were able to use at least one word, 6/19 (31.6%) were able to use at least five words, 3/19 (15.8%) combined two words and 2/19 (10.5%) produced sentences of four to five words. The highest age-equivalent reached was 4;0–4;5. Two participants (P14 and P21) did not complete the expressive domain of the PLS-5. Based on language samples collected during assessments, both were able to use sentences of up to 5–6 words in length. Morpho-syntax errors were present for both participants. Expressive language was estimated to be around a 3 year old level for both participants.
Social communication and characteristics of ASD
Of the 17 participants who completed the Social Communication Questionnaire (SCQ), the mean score was 18.94 (SD = 5.13; range 12–28). In total, 11 participants (65%) had scores above the cut off of 15 indicating a possible profile of ASD on this tool.
Communication forms and functions
The Inventory of Potential Communicative Acts (IPCA) interview assessed verbal and non-verbal communicative acts for all but one participant (n = 20). Appendix 3 (see Supplementary Materials) provides the types of communicative forms for each function. All but one participant (95%) were able to use some social conventions, draw attention to self and reject/protest. Eighteen (90%) were able to request an object and 16 (80%) were able to request an action and make a choice. Fourteen (70%) could make a comment and 11 (55%) could answer in response to questions or requests. Only six participants (30%) could request information and five (25%) could reliably imitate words and/or gestures. All those able to request information and imitate were verbal. Around 25% of participants used words as their primary means of communication and 75% used non-verbal means (e.g. pulling, screaming, laughing). The type of communicative form varied according to the function. For example, when requesting actions, most participants used physical forms of communication such as pulling, reaching and gesture. For social conventions, more social forms were used (e.g. smiling, eye gaze).
Speech
Speech profiles were variable across the six verbal participants (Table 4). P12 and P14 had severely delayed and disordered speech with inconsistent errors, a low percentage of correct vowels and phonemes (<65% phonemes correct for both) and characteristics of childhood apraxia of speech [17]. P13 presented with delayed phonological processes and generally intelligible speech. P21 had mostly intelligible speech with imprecise vowels and consonants. Two participants (P4 and P8) were not able to complete formal speech assessment but demonstrated severely delayed and disordered speech in their spontaneous language.
Oromotor structure and function
Deficits in oromotor function were observed in 4/7 (57.1%) participants assessed (P9, P12, P13 and P15) including difficulties with tongue (n = 4) and lip (n = 2) movements. Those participants who completed formal assessment (P12 and P13) demonstrated tonal and coordination deficits with lips as well as difficulties with tongue elevation, pitch/loudness and coordinated speech movements. Of those participants who were unable to complete formal assessment, n = 14 had an open mouth posture at rest and five demonstrated poor saliva control. Structural abnormalities were observed in 2/7 (28.6%) participants, both participants had a high palate and one also had a severe overbite.
Association between deletion size and language phenotype
A significant association was found between deletion size (Mb) and the participant’s ability to use phrases. Participants with smaller deletion sizes were more likely to be able to use phrases (odds ratio: 0.36, 95% CI: 0.14 to 0.95, p = 0.040). When the deletion size increased by 1 Mb, the odds of an individual being able to use phrases decreased by 64%, on average. There was a strong association between deletion size and VABS-II expressive language raw scores (co-efficient: −3.48, 95% CI: −7.14 to 0.18, p = 0.061): as deletion size increased the mean VABS-II scores decreased. However, there was no significant association between VABS-II receptive language raw scores and deletion size (co-efficient: −1.26, 95% CI: −2.8 to 0.28, p = 0.103). The findings were similar for language outcomes on the PLS-5. There was a strong association between deletion size and expressive language raw scores (co-efficient: −1.57, 95% CI: −3.2 to 0.08, p = 0.061): as deletion size increased, expressive language scores decreased. Yet, there was no significant association between receptive language raw scores and deletion size (co-efficient: −1.70, 95% CI: −3.83 to 0.2, p = 0.109). Appendix 4 (see Supplementary Materials) shows the relationship between deletion size and scores on receptive and expressive language tools. Participants who used words and phrases had a mean deletion size of 0.87 Mb (median = 0.47, range 0.041–3.26 Mb), and those who were minimally verbal had a mean of 6.6 Mb (median = 3.32, range 0.01–8.30 Mb). There was a significant difference (z = −2.647, p = 0.0081) in median deletion size for those who used phrases and those who did not (Fig. 4).
Discussion
Communication impairment is a central feature of PMS; yet, to date, linguistic examination has been limited to binary present/absent ratings of language in large samples, or a restricted battery of tests on small case series. Here we provided a comprehensive clinical examination of speech, language, social and functional communication abilities.
While all participants were delayed across all domains of adaptive functioning, with a substantial gap between adaptive age and chronological age, communication was a particular area of difficulty [8]. Severe language disorder was present in all, with performance at 3 SD below the mean across the group in line with previous reports [9, 11, 15, 25]. Whilst receptive language scores were descriptively higher than expressive scores across several assessments, this difference did not meet statistical significance for direct assessments of language. This is an important clinical finding, highlighting that only formalised standardised tests can provide a true indication of the relativity of expressive versus receptive language performance. There is often a propensity for clinical observation to surmise that expressive language is far worse than understanding in children who are minimally verbal given the striking speech production limitations. Certainly, speech or expressive language production was extremely limited in this group, with almost half of participants being non-verbal, in line with previous studies [16, 26, 27]. However, the standardised results showed that receptive language was just as impaired relative to peers. The striking nature of the language involvement is perhaps best highlighted when considering age-equivalent scores which were between 13 and 16 months, considerably lower than the average chronological age of 9.3 years across the group. All participants here had some degree of cognitive impairment. As for many other conditions, it is difficult to determine the degree to which linguistic deficits are specific, related to, or a consequence of cognitive impairment or other comorbidities [28], including ASD [29,30,31,32].
From a social skills perspective, almost three quarters of our sample met an indication of possible autism, replicating prior research [9]. While only 50% of individuals studied here were formally diagnosed with ASD, some participants may not have received a clinical diagnosis because severe to profound ID can make ASD diagnosis challenging due to overlapping characteristics (e.g. delayed play and joint attention, repetitive play and movements). There is also evidence that while formal diagnostic assessments may show an association between autism and some genetic syndromes, there may be subtle qualitative differences in the way autism characteristics present in syndrome groups [32]. Autism symptoms may also be seen as part of the syndrome without an indication for additional ASD diagnosis. Furthermore, while the SCQ has been adapted for participants who are non-verbal, the tool has reduced validity for those with cognitive ability less than 2 years [33].
All participants (all female) who were verbal and used phrases had speech production difficulties, but there was no clear homogeneity to presentation, with deficits in both linguistic (phonological delay) and motor (apraxia of speech) systems being evident. As there are no other studies to our knowledge examining speech production characteristics in PMS, it is unclear whether our participants reflect the typical population of verbal individuals with PMS. Data are too limited on verbal participants here to shed light on whether there is a continuum of perturbation of motor and linguistic tracts that lead to the minimally verbal presentation seen in half the group. Diffusion MRI analyses examining motor and linguistic tracts may help further dissociate the relative involvement and contribution of these systems to the speech and language abilities of children with PMS [34,35,36,37].
For non-verbal participants, there were relative strengths in play and other pre-linguistic skills on the CSBS-DP. The greatest area of strength was the symbolic composite, comprising skills in understanding, and object use (functional, symbolic and pretend play). From a clinical perspective, this finding suggests a strong communicative substrate to build upon in therapy, that is, individuals do appear to have symbolic communication and communicative intent. Certainly, participants in this study communicated a variety of functions using a range of forms. Almost all participants could request an object, draw attention to themselves, protest/reject and display social conventions. Most could request someone perform an action and make a choice. Only a small subset could make a comment, ask for information or imitate. Imitation was observed to be a difficult skill for participants on two tools (CSBS-DP, IPCA). All but five participants used non-verbal communication as their primary means of expressing different functions, and most relied on gestural cues and routines to understand and use words. Overall, most participants expressed basic needs and wants but struggled to use language for social interaction and had difficulty initiating language interactions. Previous research in this specific area is unavailable for comparison, but our results suggest it may be important to focus on skills of imitation and initiation as a foundation for further communication development.
In terms of genotype–phenotype associations, the odds of using phrases decreased as the deletion size increased. Six participants had deletion sizes >4 Mb, and all but one of these participants (P20; deletion size = 8 Mb) were minimally verbal (i.e. used <10 words). This is consistent with previous studies using similar categorical approaches for language (i.e. non-verbal or use of phrases) [6, 11, 16, 25, 27]. Previous studies have also suggested there may be genes in the regions proximal to SHANK3 that influence speech and language development [6, 16]. While the current study confirmed SHANK3 as being an important gene for the speech and language characteristics of PMS, our sample size was insufficient to explore the possible contribution of other genes in the region.
This study was limited by a small sample size. Statistical tests may have been underpowered due to sample size and it may be simplistic to assume a linear relationship for the regression analyses. For these reasons, we suggest these findings are exploratory. We note that based on numbers provided by the PMS Foundation of Australia, our sample represented around 18% of the population of children and adults living with PMS. This is a sizeable proportion given we only included children and adolescents in our study. Furthermore, the clinical features of participants in our sample are consistent with other samples of individuals with PMS. For example, ID or developmental delay was present in 100% of participants [7, 9] and regression affected 53% of our sample consistent with other studies reporting between 43% and 50% of participants had history of regression [7, 9, 14]. Two of the verbal participants did not complete the PLS assessment and were not included in the median age-equivalent scores. Both participants used phrases and had stronger language abilities than 80% of the sample. This may have resulted in an underestimate of mean language ability in the cohort.
Even with the best available tools to assess speech and language, assessment of individuals with severe ID and limited verbal ability remains challenging. Available assessment tools, developed on neurotypical peers, are often blunt, resulting in floor effects, and providing little differentiation between participants or across other rare conditions. We attempted to counter these issues by reporting raw scores and age-equivalents rather than standard scores, by including adaptive measures of language that spanned a wide range of ages, and by providing detail about individual participants’ abilities for parents and clinicians wanting this information to guide prognostication. We also included standardised parent report measures to supplement formal standardised assessment and provide meaningful comparisons of data collected by families who know their children better than a clinician conducting a single assessment.
Nonetheless, our detailed phenotyping highlights areas of strength and challenge to inform prognostic counselling of speech and language development and to aid development of targeted therapy programs. Interventions should harness relative strengths of the individual. In the current group, for example utilising strengths in symbolic play and focusing on communicative intent, initiation and imitation could be the starting point for building solid communication foundations. Parents are often concerned that the use of alternate or augmentative devices (e.g. speech generating devices) will reduce verbal ability. Yet, there is increasing evidence that speech generating devices are effective in facilitating communication in minimally verbal children with ASD, e.g. [38]. Therefore, it is reasonable to assume these may also be effective for children with PMS. Emphasis on speech production and oral language should also occur in conjunction with the use of these devices, with social communication being central to any therapy goal. Tailored interventions may assist with the reduction of challenging or maladaptive behaviour, facilitate language development and improve wellbeing and quality of life for affected families.
Change history
15 March 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41431-021-00855-4
References
Phelan K, McDermid HE. The 22q13.3 deletion syndrome (Phelan-McDermid syndrome). Mol Syndromol. 2011;2:186–201.
Phelan MC. Deletion 22q13.3 syndrome. Orphanet J Rare Dis. 2008;3:14.
Durand CM, Betancur C, Boeckers TM, Bockmann J, Chaste P, Fauchereau F, et al. Mutations in the gene encoding the synaptic scaffolding protein SHANK3 are associated with autism spectrum disorders. Nat Genet. 2007;39:25–7.
Boeckers TM, Bockmann J, Kreutz MR, Gundelfinger ED. ProSAP/Shank proteins – a family of higher order organizing molecules of the postsynaptic density with an emerging role in human neurological disease. J Neurochem. 2002;81:903–10.
Kolevzon A, Angarita B, Bush L, Wang AT, Frank Y, Yang A, et al. Phelan-McDermid syndrome: a review of the literature and practice parameters for medical assessment and monitoring. J Neurodev Disord. 2014;6:39.
Tabet A-C, Rolland T, Ducloy M, Lévy J, Buratti J, Mathieu A, et al. A framework to identify modifier genes in patients with Phelan-McDermid syndrome. NPJ Genom Med. 2017;2:32. https://doi.org/10.1038/s41525-017-0035-2.
Soorya L, Kolevzon A, Zweifach J, Lim T, Dobry Y, Schwartz L. et al. Prospective investigation of autism and genotype-phenotype correlations in 22q13 deletion syndrome and SHANK3 deficiency. Mol Autism. 2013;4:18.
Zwanenburg RJ, Ruiter SAJ, van den Heuvel ER, Flapper BCT, Van Ravenswaaij-Arts CMA. Developmental phenotype in Phelan-McDermid (22q13.3 deletion) syndrome: a systematic and prospective study in 34 children. J Neurodev Disord. 2016;8:16.
De Rubeis S, Siper PM, Durkin A, Weissman J, Muratet F, Halpern D. et al. Delineation of the genetic and clinical spectrum of Phelan-McDermid syndrome caused by SHANK3 point mutations. Mol Autism. 2018;9:31.
Manning MA, Cassidy SB, Clericuzio C, Cherry AM, Schwartz S, Hudgins L, et al. Terminal 22q deletion syndrome: a newly recognized cause of speech and language disability in the autism spectrum. Pediatrics 2004;114:451–7.
Ponson L, Gomot M, Blanc R, Barthelemy C, Roux S, Munnich A. et al. 22q13 deletion syndrome: communication disorder or autism? Evidence from a specific clinical and neurophysiological phenotype. Translational Psychiatry. 2018;8:146.
Jeffries AR, Curran S, Elmslie F, Sharma A, Wenger S, Hummel M, et al. Molecular and phenotypic characterization of ring chromosome 22. Am J Med Genet Part A. 2005;137A:139–47.
Koolen DA, Reardon W, Rosser EM, Lacombe D, Hurst JA, Law CJ, et al. Molecular characterisation of patients with subtelomeric 22q abnormalities using chromosome specific array-based comparative genomic hybridisation. Eur J Hum Genet. 2005;13:1019–24.
Reierson G, Bernstein J, Froehlich-Santino W, Urban A, Purmann C, Berquist S, et al. Characterizing regression in Phelan McDermid Syndrome (22q13 deletion syndrome). J Psychiatr Res. 2017;91:139–44.
Oberman LM, Boccuto L, Cascio L, Sarasua S, Kaufmann WE. Autism spectrum disorder in Phelan-McDermid syndrome: initial characterization and genotype-phenotype correlations. Orphanet J Rare Dis. 2015;10:105.
Sarasua SM, Dwivedi A, Boccuto L, Chen C-F, Sharp JL, Rollins JD, et al. 22q13.2q13.32 genomic regions associated with severity of speech delay, developmental delay, and physical features in Phelan-McDermid syndrome. Genet Med. 2014;16:318.
Mei C, Fedorenko E, Amor DJ, Boys A, Hoeflin C, Carew P, et al. Deep phenotyping of speech and language skills in individuals with 16p11.2 deletion. Eur J Hum Genet. 2018;26:676–86.
Morgan AT, Haaften LV, van Hulst K, Edley C, Mei C, Tan TY, et al. Early speech development in Koolen de Vries syndrome limited by oral praxis and hypotonia. Eur J Hum Genet. 2018;26:75–84.
Dykens EM. Measuring behavioral phenotypes: provocations from the “new genetics”. Am J Ment Retard. 1995;99:522–32.
St John M, Ponchard C, van Reyk O, Mei C, Pigdon L, Amor DJ, et al. Speech and language in children with Klinefelter syndrome. J Commun Disord. 2019;78:84–96.
Brignell A, St John M, Boys A, Bruce A, Dinale C, Pigdon L, et al. Characterization of speech and language phenotype in children with NRXN1 deletions. Am J Med Genet Part B. 2018;177:700–8.
Rutter M, Le Couteur A, Lord C. Autism diagnostic interview-revised. Los Angeles, CA: Western Psychological Services; 2003.
StataCorp L. Stata statistical software: release 15. College Station, TX: StataCorp LP; 2017.
Ziats CA, Jain L, McLarney B, Vandenboom E, DuPont BR, Rogers C, et al. Neurofibromatosis type 2 in Phelan-McDermid syndrome: institutional experience and review of the literature. Eur J Med Genet. 2020;63:104042.
Webster KT, Raymond GV. 22q13 Deletion syndrome: a report of the language function in two cases. J Med Speech-Lang Pathol. 2004;12:41–6.
Nesslinger NJ, Gorski JL, Kurczynski TW, Shapira SK, Siegel-Bartelt J, Dumanski JP, et al. Clinical, cytogenetic, and molecular characterization of seven patients with deletions of chromosome 22q13.3. Am J Hum Genet. 1994;54:464–72.
Sarasua SM, Dwivedi A, Boccuto L, Rollins JD, Chen C-F, Rogers RC, et al. Association between deletion size and important phenotypes expands the genomic region of interest in Phelan–McDermid syndrome (22q13 deletion syndrome). J Med Genet. 2011;48:761–6.
Liao S-F, Liu J-C, Hsu C-L, Chang M-Y, Chang T-M, Cheng H. Cognitive development in children with language impairment, and correlation between language and intelligence development in kindergarten children with developmental delay. J Child Neurol. 2014;30:42–7.
Norrelgen F, Fernell E, Eriksson M, Hedvall A, Persson C, Sjolin M, et al. Children with autism spectrum disorders who do not develop phrase speech in the preschool years. Autism. 2015;19:934–43.
Levy SE, Giarelli E, Lee L-C, Schieve LA, Kirby RS, Cunniff C, et al. Autism spectrum disorder and co-occurring developmental, psychiatric, and medical conditions among children in multiple populations of the United States. J Dev Behav Pediatr. 2010;31:267–75.
Moss J, Howlin P, Oliver C. The assessment and presentation of autism spectrum disorder and associated characteristics in individuals with severe intellectual disability and genetic syndromes. The Oxford handbook of intellectual disability and development. New York: Oxford University Press; 2011. p. 275–302.
Moss J, Howlin P. Autism spectrum disorders in genetic syndromes: implications for diagnosis, intervention and understanding the wider autism spectrum disorder population. J Intellect Disabil Res. 2009;53:852–73.
Rutter M, Bailey A, Lord C. The social communication questionnaire. Los Angeles, CA: Western Psychological Services; 2003.
Morgan AT, Masterton R, Pigdon L, Connelly A, Liegeois FJ. Functional magnetic resonance imaging of chronic dysarthric speech after childhood brain injury: reliance on a left-hemisphere compensatory network. Brain. 2013;136:646–57.
Morgan AT, Su M, Reilly S, Conti-Ramsden G, Connelly A, Liégeois FJ. A brain marker for developmental speech disorders. J Pediatr. 2018;198:234–9.e1.
Liégeois F, Tournier J-D, Pigdon L, Connelly A, Morgan AT. Corticobulbar tract changes as predictors of dysarthria in childhood brain injury. Neurology. 2013;80:926–32.
Liégeois FJ, Turner SJ, Mayes A, Bonthrone AF, Boys A, Smith L, et al. Dorsal language stream anomalies in an inherited speech disorder. Brain. 2019;142:966–77.
Kasari C, Kaiser A, Goods K, Nietfeld J, Mathy P, Landa R, et al. Communication interventions for minimally verbal children with autism: a sequential multiple assignment randomized trial. J Am Acad Child Adolesc Psychiatr. 2014;53:635–46.
Sparrow SS, Cicchetti DV, Balla DA. Vineland adaptive behavior scales. 2nd ed. Minneapolis, MN: American Guidance Service; 2005.
Zimmerman IL, Steinger VG, Pond RE. Preschool language scales. 5th ed. San Antonio, TX: Psychological Corporation; 2011.
Dunn LM, Dunn DM. Peabody picture vocabulary test. 4th ed. Bloomington, MN: Pearson; 2007.
Wetherby AM, Prizant BM. Communication and symbolic behavior scales developmental profile. Baltimore, MD: Paul H. Brookes; 2002.
Mei C, Reilly S, Reddihough D, Mensah F, Pennington L, Morgan A. Language outcomes of children with cerebral palsy aged 5 years and 6 years: A population‐based study. Dev Med Child Neurol. 2016;58:605–11.
Dodd B, Zhu H, Crosbie S, Holm A, Ozanne A. Diagnostic evaluation of articulation and phonology. London, England: Psychological Corporation; 2002.
Robbins J, Klee T. Clinical assessment of oropharyngeal motor development in young children. J Speech Hearing Disord. 1987;52:271–7.
Sigafoos J, Woodyatt G, Keen D, Tait K, Tucker M, Roberts-Pennell D, et al. Identifying potential communicative acts in children with developmental and physical disabilities. Commun Disord Q. 2000;21:77–86.
Acknowledgements
We thank the individuals with PMS and their families for their participation. We also thank Megan Toole for her assistance with recruitment through the Phelan McDermid Syndrome Foundation of Australia.
Funding
Funding was provided by National Health and Medical Research Council (NHMRC) Practitioner Fellowship #1105008 (A.M.); NHMRC Centre of Research Excellence in Speech and Language Neurobiology #1116976 awarded to A.M., D.A.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised due to a typo
Supplementary information
Rights and permissions
About this article
Cite this article
Brignell, A., Gu, C., Holm, A. et al. Speech and language phenotype in Phelan-McDermid (22q13.3) syndrome. Eur J Hum Genet 29, 564–574 (2021). https://doi.org/10.1038/s41431-020-00761-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41431-020-00761-1
This article is cited by
-
Genetic architecture of childhood speech disorder: a review
Molecular Psychiatry (2024)
-
Characterization of speech and language phenotype in the 8p23.1 syndrome
European Child & Adolescent Psychiatry (2024)
-
Sensory processing and adaptive behavior in Phelan-McDermid syndrome: a cross-sectional study
European Journal of Pediatrics (2022)
-
What’s new in EJHG in April
European Journal of Human Genetics (2021)
-
Speech, language and communication phenotyping in rare genetic syndromes: Commentary on Speech and language deficits are central to SETBP1 haploinsufficiency disorder
European Journal of Human Genetics (2021)