Abstract
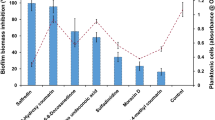
The increased resistance/tolerance of Candida infections to antimicrobial treatment can be attributed to biofilm-associated cells. A way to overcome this situation is to re-purpose non-anti-fungal drugs that could be active against fungi. We have explored the potential of a small library of eighteen non-antifungal drugs used in different human diseases. Candida albicans was cultured in the presence and absence of different concentrations of these drugs. Subsequently, inhibition of growth, germ tube formation, adhesion, and biofilm development were studied. Out of eighteen drug molecules, six showed a reduction in planktonic and biofilm growth in a dose-dependent manner and three drugs inhibited germ tube formation. This study shows the potential of non-antifungal drugs for the development of new anti-Candida agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hasan F, Xess I, Wang X, Jain N, Fries BC. Biofilm formation in clinical Candida isolates and its association with virulence. Microbes Infect. 2009;11:753–61.
Fidel P Jr. Candida-host interactions in HIV disease: relationships in oropharyngeal candidiasis. Adv Dent Res. 2006;19:80–4.
Kuhn DM, Ghannoum MA. Candida biofilms: antifungal resistance and emerging therapeutic options. Curr Opin investigational Drugs. 2004;5:186–97.
Kowalski SL, Kemp CK. Evolving threat of candida resistance. Home Healthc now. 2018;36:332–3.
Krishnasamy L, Krishnakumar S, Kumaramanickavel G, Saikumar C. Molecular mechanisms of antifungal drug resistance in candida species. J Clin Diagn Res. 2018;12:DE01–6.
Swoboda S, Lichtenstern C, Ober MC, Taylor LA, Störzinger D, Michel A, et al. Implementation of practice guidelines for antifungal therapy in a surgical intensive care unit and its impact on use and costs. Chemotherapy. 2009;55:418–24.
Nosanchuk JD. Current status and future of antifungal therapy for systemic mycoses. Recent Pat Antiinfective Drug Discov. 2006;1:75–84.
Robbins N, Caplan T, Cowen LE. Molecular evolution of antifungal drug resistance. Annu Rev Microbiol. 2017;71:753–75.
Odds FC. Antifungal agents: their diversity and increasing sophistication. Mycologist. 2003;17:51–5.
Cannon RD, Lamping E, Holmes AR, Niimi K, Baret PV, Keniya MV, et al. Efflux-mediated antifungal drug resistance. Clin Microbiol Rev. 2009;22:291–321.
Revie NM, Iyer KR, Robbins N, Cowen LE. Antifungal drug resistance: evolution, mechanisms and impact. Curr Opin Microbiol. 2018;45:70–6.
Sanglard D. Finding the needle in a haystack: mapping antifungal drug resistance in fungal pathogen by genomic approaches. PLoS Pathog. 2019;15:e1007478.
Ingham CJ, Boonstra S, Levels S, De Lange M, Meis JF, Schneeberger PM. Rapid susceptibility testing and microcolony analysis of Candida spp. cultured and imaged on porous aluminum oxide. PLoS ONE. 2012;7:e33818.
Spitzer M, Griffiths E, Blakely KM, Wildenhain J, Ejim L, Rossi L, et al. Cross‐species discovery of syncretic drug combinations that potentiate the antifungal fluconazole. Mol Syst Biol. 2011;7:499.
Ji C, Liu N, Tu J, Li Z, Han G, Li J, et al. Drug repurposing of haloperidol: discovery of new benzocyclane derivatives as potent antifungal agents against cryptococcosis and candidiasis. ACS Infect Dis. 2019;6:768–86.
Wall G, Chaturvedi AK, Wormley FL, Wiederhold NP, Patterson HP, Patterson TF, et al. Screening a repurposing library for inhibitors of multidrug-resistant Candida auris identifies ebselen as a repositionable candidate for antifungal drug development. Antimicrobial Agents Chemother. 2018;62:e01084-18.
Judd WR, Martin CA. Antifungal activity of nontraditional antifungal agents. Curr Fungal Infect Rep. 2009;3:86–95.
Richter SS, Galask RP, Messer SA, Hollis RJ, Diekema DJ, Pfaller MA. Antifungal susceptibilities of Candida species causing vulvovaginitis and epidemiology of recurrent cases. J Clin Microbiol. 2005;43:2155–62.
Kathwate GH, Shinde RB, Karuppayil SM. Antiepileptic drugs inhibit growth, dimorphism, and biofilm mode of growth in human pathogen Candida albicans. Assay Drug Dev Technol. 2015;13:307–12.
Ramage G, Walle KV, Wickes BL, López-Ribot JL. Standardized method for in vitro antifungal susceptibility testing of Candida albicansbiofilms. Antimicrobial Agents Chemother. 2001;45:2475–9.
Chandra J, Kuhn DM, Mukherjee PK, Hoyer LL, McCormick T, Ghannoum MA. Biofilm formation by the fungal pathogenCandida albicans: development, architecture, and drug resistance. J J Bacteriol. 2001;183:5385–94.
Chauhan NM, Raut JS, Karuppayil SM. A morphogenetic regulatory role for ethyl alcohol in Candida albicans. Mycoses. 2011;54:e697–e703.
Hudson DA, Sciascia QL, Sanders RJ, Norris GE, Edwards PJ, Sullivan PA, et al. Identification of the dialysable serum inducer of germ-tube formation in Candida albicans. Microbiology. 2004;150:3041–9.
Wood N, Nugent K. Inhibitory effects of chlorpromazine on Candida species. Antimicrobial Agents. Chemotherapy. 1985;27:692–4.
Sharma S, Kaur H, Khuller G. Cell cycle effects of the phenothiazines: trifluoperazine and chlorpromazine in Candida albicans. FEMS Microbiol Lett. 2001;199:185–90.
Cooke CE, Mehra IV. Oral ondansetron for preventing nausea and vomiting. Am J Health-Syst Pharm. 1994;51:762–71.
Ye JH, Ponnudurai R, Schaefer R. Ondansetron: a selective 5‐HT3 receptor antagonist and its applications in CNS‐related disorders. CNS Drug Rev. 2001;7:199–213.
Versele M, Lemaire K, Thevelein JM. Sex and sugar in yeast: two distinct GPCR systems. EMBO Rep. 2001;2:574–9.
Lin C-H, Choi A, Bennett RJ. Defining pheromone-receptor signaling in Candida albicans and related asexual Candida species. Mol Biol Cell. 2011;22:4918–30.
Bölker M. Sex and crime: heterotrimeric G proteins in fungal mating and pathogenesis. Fungal Genet Biol. 1998;25:143–56.
Maidan MM, De Rop L, Serneels J, Exler S, Rupp S, Tournu H, et al. The G protein-coupled receptor Gpr1 and the Gα protein Gpa2 act through the cAMP-protein kinase A pathway to induce morphogenesis in Candida albicans. Mol Biol Cell. 2005;16:1971–86.
Berdicevsky I, Silbermann M. Effect of glucocorticoid hormones on calcium uptake and the morphology of Candida albicans. Cell Biol Int Rep. 1982;6:783–90.
Blasko G. Pharmacology, mechanism of action and clinical significance of a convenient antispasmodic agent: drotaverine. J Am Med Assoc India. 1998;1:63–9.
Jung WH, Stateva LI. The cAMP phosphodiesterase encoded by CaPDE2 is required for hyphal development in Candida albicans. Microbiology. 2003;149:2961–76.
Jung WH, Warn P, Ragni E, Popolo L, Nunn CD, Turner MP, et al. Deletion of PDE2, the gene encoding the high‐affinity cAMP phosphodiesterase, results in changes of the cell wall and membrane in Candida albicans. Yeast. 2005;22:285–94.
Yost CS. A new look at the respiratory stimulant doxapram. CNS Drug Rev. 2006;12:236–49.
Hänel H, Kirsch R, Schmidts HL, Kottmann H. New systematically active antimycotics from the beta‐blocker category: Neue, systemisch wirksame Antimykotika aus der Klasse der β‐Blocker. Mycoses. 1995;38:251–64.
da Silveira Derengowski L, Pereira AL, Andrade AC, Kyaw CM, Silva-Pereira I. Propranolol inhibits Candida albicans adherence and biofilm formation on biotic and abiotic surfaces. Int J Antimicrobial Agents. 2009;34:614–6.
Ma B, Huang H-h, Chen X-y, Sun Y-m, Lin L-h, Zhong D-f. Biotransformation of metoprolol by the fungus Cunninghamella blakesleeana. Acta Pharmacologica Sin. 2007;28:1067–74.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kathwate, G.H., Shinde, R.B. & Mohan Karuppayil, S. Non-antifungal drugs inhibit growth, morphogenesis and biofilm formation in Candida albicans. J Antibiot 74, 346–353 (2021). https://doi.org/10.1038/s41429-020-00403-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41429-020-00403-0