Abstract
Study design
Systematic review and network meta-analysis.
Objectives
Intermittent catheterization (IC) is considered the standard treatment for neuro-urological patients who are unable to empty their bladders. The present study aimed to conduct a systematic evaluation and network meta-analysis of all available types of intermittent catheters, and determine which one is best suited for clinical use.
Methods
We searched MEDLINE, EMBASE and Cochrane Central Register of Controlled Trials (CENTRAL) databases to identify relevant studies. Only randomized clinical trials (RCTs) were included. Five types of catheters were identified based on the included studies. A Bayesian network meta-analysis was then performed. The surface under the cumulative ranking (SUCRA) curve was used to determine the best catheter for each outcome.
Results
A total of 25 RCTs, involving 1233 participants, were included. The pooled odds ratios of symptomatic UTI were lower for two ready-to-use single-use catheters (gel-lubricated non-coated catheter, OR: 0.30, 95% CI 0.095–0.86; pre-activated hydrophilic-coated catheter, OR: 0.41, 95% CI 0.19–0.83) as compared to single-use non-coated catheter. In terms of patient satisfaction, the SUCRA results showed that the pre-activated hydrophilic-coated catheter may the preferred option (SUCRA = 82.8%). However, there were no significant differences in all outcome measures between traditional single-use non-coated catheters and clean non-coated catheters.
Conclusion
Ready-to-use single-use catheters are associated with lower rates of UTI compared to traditional catheters. Patients may be most satisfied with the pre-activated one. For traditional single-use non-coated catheters and clean non-coated catheters, there is still no convincing evidence as to which is better. Thus, more well-designed trials are needed.
Similar content being viewed by others
Introduction
Intermittent catheterization (IC) is a procedure that involves using a urinary catheter to empty the bladder, which typically is done several times a day. IC is considered as a standard treatment for adults and children with incomplete bladder emptying due to neurological disorders [1]. However, with long-term IC usage, some life-threatening complications may occur. The most frequent complication is urinary tract infection (UTI) [2], which can result in bacteremia/sepsis. Urethral trauma, usually evaluated by haematuria, has also been reported and shown to affect the risk of UTI [3].
Since the first introduction of IC, the material, coating and design of the catheter have undergone continuous improvements to reduce the incidence of UTI, attenuate urinary tract injury and enhance patient satisfaction. IC was originally performed using sterile technique [4] (single-use catheter with sterile gloves and sterile containers). Lapides et al. [5] later proposed a clean reusable catheter, which is more cost-effective without increasing infection rates. Reused IC refers to the reuse of a catheter after washing with soap, boiling and/or soaking in disinfectants and air drying, instead of discarding it. Coated catheters are now available for IC, which are only single-use and designed to improve lubrication and reduce urethral injury. The most widely used one is hydrophilic-coated catheter, with a layer of hydrophilic polymers that absorb water and bind to the catheter surface [6]. The friction of hydrophilic-coated catheter is greatly lower than that of the traditional plastic lubricated catheter [7].
IC can be done by caregivers or by the patients themselves after discharge from the rehabilitation facility. Several new designs, with the concepts of “No touch” or “Ready to use”, have been developed to facilitate the procedure, reducing the risk of infection during the procedure, and improving patient comfort. For non-coated catheters, there are some designs such as a closed, touchless, self-contained catheter system that can be used in a sterile collection bag [8], and the catheters are pre-lubricated without a need to add gel manually [9]. For hydrophilic-coated catheters, the first design required immersion in water for 30 s to activate it before use; the later design is packed in water (pre-activated) and ready for use [10].
Although four meta-analyses [11,12,13,14] have suggested the benefit of hydrophilic-coated catheters over non-hydrophilic ones, the variety of catheters can make data synthesis challenging. The confounding factors may include sterile or clean technique, single-use or re-use, pre-lubricated, pre-activated and other design features.
Another comparison, single-use versus re-use catheters, is still controversial. Two cost-effectiveness studies [11, 15] demonstrated the economic advantage of re-use catheters, and the results suggest that re-use catheters may be more acceptable for long-term use to individuals in developing countries. A Cochran systematic review [13] in 2014 concluded that single-use catheters have no clear advantages over re-use catheters, and this potential endorsement of re-use catheters may have a strong impact on healthcare providers’ decision-making. However, this Cochran review was withdrawn [16], since an independent appraisal [17] had found significant problems with data extraction and analysis.
Therefore, we undertook a network meta-analysis of all available types of catheters to identify the one that is most suitable for IC. This kind of analysis can reduce the confounding effects in pair-wise meta-analysis due to the variety of catheters, as well as integrate both direct and indirect comparisons.
Methods
Search strategy
This systematic review is reported based on the Preferred Reporting Items for Systematic reviews and Meta-analyses Statement (PRISMA) [18]. The PRISMA checklist is shown in supplementary information. MEDLINE, EMBASE and the Cochrane Central Register of Controlled Trials databases were searched to identify relevant articles published up to June 2020. The following search terms were used: IC, urethral catheterization, self-catheterization, clinical trials as the topic and random. The detailed search strategy is presented in Supplementary Table S1. Relevant references from the 2014 Cochrane review were also identified. Screening of titles and abstracts, as well as full-text reading if necessary, were done by two authors independently. Disagreements were resolved by a third reviewer. The study protocol was registered at PROSPERO (No. CRD42018114852).
Selection criteria
The inclusion criteria were as follows: (i) randomized controlled trials or randomized cross-over trials; (ii) participants who used some form of IC; (iii) comparison of different types of intermittent catheters; and (iv) outcomes were symptomatic UTI, asymptomatic bacteriuria, haematuria and/or patient satisfaction. Observational studies, editorials, commentaries and review articles were excluded. Based on the experimental groups in the included studies, we defined five types of catheters of interest for analysis, as follows: (i) single-use non-coated catheter (SNC)—a single-use uncoated catheter. After removing it from sterile packaging, the user needs to add some lubricant before insertion; (ii) gel-lubricated single-use non-coated catheter (GSNC)—a single-use uncoated catheter with pre-packaged lubricant, which is ready to use; (iii) single-use hydrophilic-coated catheter (SHC)—a single-use catheter with a hydrophilic polymer coating. The user needs to add sterile water for about 30 s to active the hydrophilic polymer coating; (iv) clean reused non-coated catheter (CNC)—a reusable uncoated catheter, which is usually washed with soapy water and air-dried after each use, but the user needs to add some lubricant before use each time; and (v) pre-activated single-use hydrophilic-coated catheter (PSHC)—a single-use catheter with a hydrophilic polymer coating which has been activated, and is ready-to-use. Only studies with at least two of the above-mentioned catheter types were included.
Data extraction
Data extraction was performed by two authors independently. Disagreements were resolved by a third reviewer when necessary. The following information was extracted from each qualified article: author list, publication year, study population, study design, study location, subject age range, sample size, study duration, catheter types, the number of participants who dropped out, and clinical outcomes.
Outcome assessment
The outcomes were symptomatic UTI, asymptomatic bacteriuria, urinary injury measured by haematuria, and patient satisfaction. The definitions of these outcomes were as follows: (i) symptomatic UTI—a positive urine culture of ≥ 103 cfu/mL with at least 1 symptom compatible with UTI, including fever, cloudy/malodorous urine, suprapubic/flank pain, worsening incontinence, malaise and/or pain associated with catheterization [19, 20]. Since some trials had discrepant definitions of symptomatic UTI, we also considered the definitions used in the trials, in a sensitivity analysis; (ii) asymptomatic bacteriuria—the presence of at least 1 bacterial species (≥105 cfu/mL) in a single catheter urine specimen without any symptoms compatible with UTI; (iii) haematuria—the presence of red blood cells in the urine, urethral bleeding, or gross and microscopic haematuria; and (iv) patient satisfaction—the subjective general opinion or preferences. All trials reporting satisfaction included a binary item of overall satisfaction, and the non-binary items were disregarded.
Data quality assessment
Two authors independently evaluated the quality of the studies using the Cochrane Risk of Bias tool covering selection bias, performance bias, detection bias, attrition bias, reporting bias and other potential sources of bias. The studies were classified into three grades: “high”, “unclear” and “low” risk of bias.
Statistical analysis
A network meta-analysis was performed to compare all five types of IC catheters using a Bayesian approach [21], producing four networks with one outcome each. All analyses were performed in R 3.4.4 [22] using the ‘GeMtc’ package. The intervention efficacy or clinical impact are presented as odds ratios (ORs) and 95% confidence intervals (CIs). The ranking probabilities and the surface under the cumulative ranking curves (SUCRA) [23] are utilized to evaluate the relative ranking of different types of catheters for each outcome. Study heterogeneity was evaluated using the Higgins–Thompson I2 method [24]. To evaluate inconsistency among the included trials in the networks, the Z test [25] and node splitting model [26] were used to quantify the degree of difference between direct and indirect comparisons. The level of statistical significance was set at P < 0.05.
As the definitions of symptomatic UTI varied between various studies, we conducted a sensitivity analysis assessing only studies with clear, detailed definitions and those which only met the standard provided by the Infectious Diseases Society of America (IDSA) 2009 consensus statement [19] and the American Academy of Pediatrics (AAP) guidelines for UTI [20].
Results
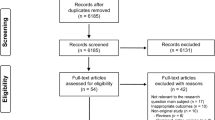
The literature search identified 2914 records, and 40 studies were selected for a full review after excluding duplicate publications and screening title and abstract. After full-text screening, 25 studies [9, 10, 27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49] were included. The PRISMA flow diagram is shown in Fig. 1.
Characteristics of the included studies
Table 1 lists the main characteristics of the studies. Fourteen were randomized controlled trials, and 11 were randomized cross-over trials. Among the 1233 participants, most were diagnosed with neurogenic bladder, while a few had other causes of urinary retention, such as prostatic hyperplasia. Six trials [30, 33, 44, 46, 48, 49] focused on children, and one [47] included individuals with myelomeningocele and involved a small number of children. In two [30, 33] of these seven trials, the children did self-IC, while in the remaining five [44, 46,47,48,49] they did it either themselves or IC was done by a trained caregiver. The remaining 18 trials included only adult subjects. In these 18 trials, IC was performed by the patients themselves [9, 10, 28, 32, 35, 39, 40, 42, 43, 45], by caregivers [29, 31, 34, 37, 38], or by both [27, 36, 41]. Males made up 75.9% of the total study sample. There were 16 trials [9, 27, 29,30,31,32,33, 36, 38,39,40,41, 45, 47,48,49] assessing the incidence of symptomatic UTI, and 10 [27, 28, 30, 32,33,34,35, 38, 45, 49] assessing the incidence of asymptomatic bacteriuria. For haematuria, 8 trials [9, 30, 32, 35,36,37, 40, 41] were included; while for patient satisfaction, 9 trials [10, 32, 36, 37, 40, 42,43,44, 46] were included. The specific data of each comparison arm is shown in Supplementary Table S2. Cochrane tool risk of bias details are listed in Supplementary Fig. S1, which reveals a moderate risk of bias across trials.
Symptomatic UTI
Sixteen trials which reported the incidence of symptomatic UTI, comparing SNC, GSNC, SHC, CNC and PSHC (916 total participants) were included. When SNC was used as the reference, only GSNC (OR 0.30, 95% CI 0.10–0.86) and PSHC (OR 0.41, 95% CI 0.19–0.83) were associated with a statistically significant lower incidence of symptomatic UTI (Fig. 2A). According to the SUCRA values (Fig. 2B), GSNC (SUCRA = 86.7%) was the preferred option to decrease the risk of symptomatic UTI, followed by PSHC (SUCRA = 73.8%), SHC (SUCRA = 57.2%) and CNC (SUCRA = 22.1%). Assessment of heterogeneity (Supplementary Fig. S2) revealed I2 = 29.9% for the comparison of GSNC versus SNC and I2 = 63.1% for CNC versus GSNC; while the remaining comparisons showed no heterogeneity (I2 = 0%). Overall, we found no significant inconsistency (Z = 3.99, p = 0.41) in the analysis.
Sensitivity analysis of symptomatic UTI
The specific definition of symptomatic UTI described in each trial is given in Supplementary Table S3. We conducted a sensitivity analysis for symptomatic UTI assessing only studies with clear, detailed definitions and those that only met the standard of the IDSA 2009 consensus statement and AAP guidelines. Four trials [30, 36, 41, 47] were excluded, and PSHC was not included in this analysis due to the exclusion of two trials [36, 41]. The results of SUCRA-based analysis (Supplementary Fig. S3) showed a similar trend as in the initial analysis. Notably, the probability of GSNC being the preferred choice was 91.0% and that of SHC being the second preferred choice was 67.6%. The estimated risk for the comparison of GSNC versus SNC remained statistically significant (OR 0.29, 95% CI 0.09–0.86).
Asymptomatic bacteriuria
For asymptomatic bacteriuria, the network analysis comprised nine trials comparing SNC, GSNC, SHC and CNC (378 total participants). Base on the SUCRA results (Fig. 2D), GSNC (SUCRA = 93.7%) was the preferred choice to prevent asymptomatic bacteriuria, and SHC was likely (SUCRA = 52.0%) to be the second choice. No significant differences were observed for GSNC, SHC and CNC (OR 0.096, 95% CI 0.004–1.2; OR 0.64, 95% CI 0.19–2.3; and OR 0.78, 95% CI 0.37–1.7, respectively), when compared to SNC (Fig. 2C). In addition, there was no evidence of significant heterogeneity and no inconsistency between the studies (Supplementary Fig. S4).
Haematuria
Eight trials comparing SNC, GSNC, SHC, CNC and PSHC (643 total participants) with regards to haematuria or urethral bleeding were included. Although no significant difference in haematuria incidence was observed for the four catheters, the SUCRA results indicated that the hydrophilic-coated catheter may be the preferred option to decrease the risk of haematuria (OR: 0.19, 95% CI 0.01–1.5; SUCRA 87.7%; Fig. 3A, B). No significant inconsistency (Z = 1.57, p = 0.67) was observed between the studies. Assessment of heterogeneity gave I2 = 72.0% for the comparison of PSHC with SNC and I2 = 57.3% for PSHC versus GSNC; the remaining comparisons were not significant (Supplementary Fig. S5).
Patient satisfaction
Nine trials that reported patient satisfaction were included in the analysis (849 total participants). In six [10, 32, 37, 40, 42, 43] of these trials, the satisfaction was reported by the patients themselves; while in the other three [36, 44, 46], part of the evaluation was done by the patient and part by the care provider. SUCRA analysis indicated that in terms of overall patient satisfaction the catheters were ranked as follows: PSHC, CNC, SHC, GSNC and SNC (Fig. 3D). None of the catheters exhibited significant advantages compared with SNC (Fig. 3C). Assessment of heterogeneity (Supplementary Fig. S6) demonstrated no significant heterogeneity for the comparison of SHC versus SNC and GSNC; the remaining comparisons showed a slight but significant heterogeneity. There was no significant inconsistency (Z = 3.46, p = 0.33) observed between the studies.
Discussion
The types of catheters used in IC vary across the globe, including single-use or re-use, coated or uncoated, pre-lubricated, pre-activated and others. Previous pair-wise meta-analyses [12,13,14] have suggested the advantage of using hydrophilic-coated catheters for IC. However, these studies often clustered two different catheters (SNC and CNC) in the category of uncoated catheters when uncoated was compared to hydrophilic-coated catheters, and clustered two different hydrophilic-coated catheters (PSHC and SHC) in the same category. In the 2014 Cochrane review [13], when comparing single-use and re-use catheters, the hydrophilic-coated catheters, gel-lubricated catheters and traditional single-use catheters were classified in the same group (single-use), leading to possibly misleading conclusions. Therefore, the present study attempted to evaluate all available types of catheters as separate entities by performing a network meta-analysis. More importantly, the sample size (1233 total participants) of this meta-analysis and the number of included studies were greater than those of the previous meta-analyses [11,12,13,14]. It is hoped that the results will guide patients and healthcare providers when making treatment decisions.
Some findings are important based on pooled analyses and SUCRA results. First, GSNC and PSHC were associated with a significantly decreased risk of symptomatic UTI compared to SNC. The SUCRA results indicated the GSNC is the preferable option for UTI prevention. The results from asymptomatic bacteriuria gave a similar rank order, but did not reach statistical significance, probably because all patients on IC are at risk of asymptomatic bacteriuria. Asymptomatic bacteriuria has less clinical significance than UTI and requires no treatment. It was noteworthy that when subdividing the hydrophilic-coated catheters into PSHC and SHC, only PSHC shows a statistically significant reduction in UTI rate. These data can be explained by the fact that both GSNC and PSHC are ready-to-use and no-touch techniques, whereas the users need to add sterile water to the traditional hydrophilic-coated catheter (SHC) for about 30 s to active the hydrophilic coating, or manually add some lubricants and disinfectants to SNC before use. These operations may confer some risks. Goessaert et al. [50] reported that even in the hospital setting, nurses and nursing students might make more sterilization errors and spent more time performing the traditional method (sterile with a catheterization-set), than with the no-touch method. Moreover, as a non-hospital procedure, IC is widely performed by patients themselves in real-life circumstances. Many patients with spinal cord injury have decreased manual dexterity or are juveniles. The ready-to-use and no-touch technique that requires less preparation and has less chance of making sterilization errors may be especially useful for those patients.
Previous studies have mentioned that the definitions of symptomatic UTI are not consistent across trials. Two previous meta-analyses [13, 14] used the definition taken from an outdated consensus statement [51], while the IDSA 2009 consensus statement [19] had already provided the latest definition of UTI. Considering the variation in the definitions, we conducted a sensitivity analysis focusing on trials with clear, detailed definitions, and those that approximately met the IDSA standard. The core definition of symptomatic UTI was significant bacteriuria plus the presence of symptoms or signs. The main differences between the two definitions are as follows: the National Institute on Disability and Rehabilitation Research (NIDRR) defined the significant bacteriuria of UTI as ≥102 cfu/ml; while the IDSA standard defined the significant bacteriuria of UTI as ≥103 cfu/ml. One trial [41] has applied the former definition, which may have led to a risk of overestimating the incidence of UTI. This study was excluded in the sensitivity analysis. The incidence of symptomatic UTI in this trial (82%) was higher than that in the remaining trials. Another three trials [30, 36, 47] were excluded due to the lack of a clear and precise description of the inclusion criteria and the definition of symptomatic UTI, especially the diagnostic standard of bacterial colonies in urine samples. Given the exclusion of the De Ridder [36] and Cardenas [41] studies, the PSHC arm was excluded from the sensitivity analysis. The sensitivity analysis revealed a similar rank order as the initial analysis, and the gel-lubricated catheter was still significantly superior to other catheters in terms of avoiding symptomatic UTI.
Regarding haematuria, the SUCRA results demonstrated the superiority of SHC compared to GSNC and other uncoated catheters. This may be attributed to the fact that hydrophilic catheters have a smooth and slippery surface with lower friction force and higher osmolality [7]. However, Stensballe et al. [37] measured the withdrawal friction and the incidence of haematuria for the hydrophilic catheter and the gel-lubricated catheter and reported that there is no direct relationship between friction force and haematuria, which may partially explain our insignificant results. Haematuria is an acute form of urethral trauma, and trauma to the urethral mucosa can lead to long-term complications such as urethral stricture, which was rarely reported in most trials. The reason may be that a great number of patients might only have mucosal irritation symptoms, which indicate a very slight urethral trauma that cannot be detected by laboratory tests.
Some subjective indicators should be paid more attention than objective ones, such as the overall patient satisfaction and other related parameters. Patient satisfaction or preference is affected by comfort, ease of handling, convenience, cost and insurance reimbursement. The SUCRA analysis revealed that with respect to patient satisfaction PSHC and CNC were the top two. The advantage of PSHC is that it has a slippery surface and is ready-to-use, which offer comfort and ease of operation at the same time. Traditional single-use hydrophilic catheters can make some individuals feel that ‘lubricant [water in the packaging] would leave a mess on the bathroom floor’, and make them prefer to use multi-use catheters [44]. The evaluation criteria of satisfaction varied greatly; however, we noted that all trials reporting satisfaction include a binary measure of overall satisfaction. The remaining non-binary items could not be used for network analysis at present. It is hoped that there will be a unified scale to measure IC satisfaction in the future, which is convenient for accurate evidence synthesis.
Another important issue nowadays is whether the SNC or the multi-use non-coated catheter is preferred. Our meta-analysis showed that if hydrophilic-coated and gel-lubricated catheters were left out of comparison, there were no significant differences for the four outcomes between the SNC and CNC. The 2014 Cochrane review [13] reached a similar conclusion and stated that no benefit was proven for single-use catheters compared to re-use catheters. However, such a statement strongly encourages healthcare insurers to carefully consider the cheaper re-use catheters, thus leading to great potential risks. There still is no general consensus whether the healthcare insurers should cover the cost of single-use catheters.
A cost-effectiveness analysis, which represents a key to interpret the clinical and financial trade-offs, was not included in the current meta-analysis. Two previous cost-effective studies [11, 15] obtained different conclusions based on two probabilistic Markov models with various groups and health states. Bermingham et al. [11] reported that clean re-use catheters were more cost-effective than single-use catheters, whereas Clark et al. [15] supported the use of SHCs, especially when long-term UTI complications were included in the analysis. However, these two studies were both performed based on the National Health Service drug tariff in the UK, and data from developing countries are still missing. More rigorous trials with cost-effectiveness analyses are needed for adequate decision-making, especially in developing countries.
This study has some limitations. First, the variety of definitions of clinical outcomes in the included studies might contribute to statistical heterogeneity. The above sensitivity analysis was an attempt to address this issue. Second, most studies had limitations of high dropout rate, which in turn resulted in a low quality of evidence. That is, the clinical outcomes of various IC options could be different for individuals who did not complete the study than for those who had completed the study, which may have resulted in an underestimate of UTI risks. Third, most studies had a short follow-up period, hence, some long-term complications such as urethral stricture and renal failure were not observed and reported. Forth, due to the obvious differences in the operation of diverse catheters, the blinding method was not used in any of the included studies. Fifth, the cleaning technique is another variable that should be considered. Cleaning technique is the procedure of IC with or without a rigorous aseptic technique (sterile gloves, container and asepsis of genitals), and it can be performed with a single-use catheter or a reused catheter. Unfortunately, few trials have evaluated cleaning techniques, which may introduce confounding. Finally, most of the study subjects included were male, and seven trials [29, 30, 32, 35,36,37, 40] enroled only males, which could reduce the significance of the results in guiding IC use by female. Despite these limitations, the present network meta-analysis is the first to integrate all current literature and thus determine the most suitable type of catheter for IC.
Conclusion
Our network meta-analysis indicated that GSNC and PSHC reduce the risk of UTI. The ready-to-use and no-touch technique showed significant advantages compared to traditional techniques. For reasons such as ease of use and comfort, patients were most satisfied with PSHC. In terms of urethral injury measured by haematuria, the hydrophilic-coated catheter seemed to perform best. However, based on the present analysis and previous cost-effective studies, there is still no convincing evidence whether SNC or CNC is better. Further well-designed and high-quality RCTs with a strict definition of outcomes, adequate sample size, follow-up periods and cost-effectiveness analysis, are necessary to obtain more conclusive results.
References
Blok B, Padilla-Fernández B, Pannek J, Castro-Diaz D, Del Popolo G, Groen J, et al. EAU Guidelines on Neuro-Urology 2018. European Association of Urology Guidelines 2018 Edition. presented at the EAU Annual Congress Copenhagen 2018. Arnhem: European Association of Urology Guidelines Office; 2018.
Wyndaele JJ. Complications of intermittent catheterization: their prevention and treatment. Spinal Cord. 2002;40:536–41.
Abrams P, Andersson KE, Birder L, Brubaker L, Cardozo L, Chapple C, et al. Fourth International Consultation on Incontinence Recommendations of the International Scientific Committee: evaluation and treatment of urinary incontinence, pelvic organ prolapse, and fecal incontinence. Neurourol Urodyn. 2010;29:213–40.
Guttmann L, Frankel H. The value of intermittent catheterisation in the early management of traumatic paraplegia and tetraplegia. Paraplegia. 1966;4:63–84.
Lapides J, Diokno AC, Lowe BS, Kalish MD. Followup on unsterile intermittent self-catheterization. J Urol. 1974;111:184–7.
Diokno AC, Mitchell BA, Nash AJ, Kimbrough JA. Patient satisfaction and the LoFric catheter for clean intermittent catheterization. J Urol. 1995;153:349–51.
Waller L, Telander M, Sullivan L. The importance of osmolality in hydrophilic urethral catheters: a crossover study. Spinal Cord. 1997;35:229–33.
Charbonneau-Smith R. No-touch catheterization and infection rates in a select spinal cord injured population. Rehabil Nurs. 1993;18:296–9. 305.
Giannantoni A, Di Stasi SM, Scivoletto G, Virgili G, Dolci S, Porena M. Intermittent catheterization with a prelubricated catheter in spinal cord injured patients: a prospective randomized crossover study. J Urol. 2001;166:130–3.
Pascoe G, Clovis S. Evaluation of two coated catheters in intermittent self-catheterization. Br J Nurs. 2001;10:325–9.
Bermingham SL, Hodgkinson S, Wright S, Hayter E, Spinks J, Pellowe C. Intermittent self catheterisation with hydrophilic, gel reservoir, and non-coated catheters: a systematic review and cost effectiveness analysis. BMJ. 2013;346:e8639.
Li L, Ye W, Ruan H, Yang B, Zhang S, Li L. Impact of hydrophilic catheters on urinary tract infections in people with spinal cord injury: systematic review and meta-analysis of randomized controlled trials. Arch Phys Med Rehabil. 2013;94:782–7.
Prieto J, Murphy CL, Moore KN, Fader M. Intermittent catheterisation for long-term bladder management. Cochrane Database Syst Rev. 2014;9:CD006008.
Rognoni C, Tarricone R. Intermittent catheterisation with hydrophilic and non-hydrophilic urinary catheters: systematic literature review and meta-analyses. BMC Urol. 2017;17:4.
Clark JF, Mealing SJ, Scott DA, Vogel LC, Krassioukov A, Spinelli M, et al. A cost-effectiveness analysis of long-term intermittent catheterisation with hydrophilic and uncoated catheters. Spinal Cord. 2015;54:73–7.
Prieto J, Murphy CL, Moore KN, Fader M. WITHDRAWN: Intermittent catheterisation for long-term bladder management. Cochrane Database Syst Rev. 2017;8:Cd006008.
Christison K, Walter M, Wyndaele JJM, Kennelly M, Kessler TM, Noonan VK, et al. Intermittent catheterization: the devil is in the details. J Neurotrauma. 2018;35:985–9.
Hutton B, Salanti G, Caldwell DM, Chaimani A, Schmid CH, Cameron C, et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med. 2015;162:777–84.
Hooton TM, Bradley SF, Cardenas DD, Colgan R, Geerlings SE, Rice JC, et al. Diagnosis, prevention, and treatment of catheter-associated urinary tract infection in adults: 2009 International Clinical Practice Guidelines from the Infectious Diseases Society of America. Clin Infect Dis. 2010;50:625–63.
Roberts KB. Urinary tract infection: clinical practice guideline for the diagnosis and management of the initial UTI in febrile infants and children 2 to 24 months. Pediatrics. 2011;128:595–610.
van Valkenhoef G, Lu G, de Brock B, Hillege H, Ades AE, Welton NJ. Automating network meta-analysis. Res Synth Methods. 2012;3:285–99.
Neupane B, Richer D, Bonner AJ, Kibret T, Beyene J. Network meta-analysis using R: a review of currently available automated packages. PLoS ONE. 2014;9:e115065.
Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol. 2011;64:163–71.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58.
Lu G, Ades AE. Assessing evidence inconsistency in mixed treatment comparisons. J AM Stat Assoc 2006;101:447–59.
Dias S, Welton NJ, Caldwell DM, Ades AE. Checking consistency in mixed treatment comparison meta-analysis. Stat Med. 2010;29:932–44.
King RB, Carlson CE, Mervine J, Wu Y, Yarkony GM. Clean and sterile intermittent catheterization methods in hospitalized patients with spinal cord injury. Arch Phys Med Rehabil. 1992;73:798–802.
Quigley PA, Riggin OZ. A comparison of open and closed catheterization techniques in rehabilitation patients. Rehabil Nurs. 1993;18:26–9. 33.
Duffy LM, Cleary J, Ahern S, Kuskowski MA, West M, Wheeler L, et al. Clean intermittent catheterization: safe, cost-effective bladder management for male residents of VA nursing homes. J Am Geriatr Soc. 1995;43:865–70.
Sutherland RS, Kogan BA, Baskin LS, Mevorach RA. Clean intermittent catheterization in boys using the LoFric catheter. J Urol. 1996;156:2041–3.
Prieto-Fingerhut T, Banovac K, Lynne CM. A study comparing sterile and nonsterile urethral catheterization in patients with spinal cord injury. Rehabil Nurs. 1997;22:299–302.
Pachler J, Frimodt-Moller C. A comparison of prelubricated hydrophilic and non-hydrophilic polyvinyl chloride catheters for urethral catheterization. BJU Int. 1999;83:767–9.
Schlager TA, Clark M, Anderson S. Effect of a single-use sterile catheter for each void on the frequency of bacteriuria in children with neurogenic bladder on intermittent catheterization for bladder emptying. Pediatrics. 2001;108:E71.
Day RA, Moore KN, Albers MK. A pilot study comparing two methods of intermittent catheterization: limitations and challenges. Urol Nurs. 2003;23:143–7. 58.
Vapnek JM, Maynard FM, Kim J. A prospective randomized trial of the LoFric hydrophilic coated catheter versus conventional plastic catheter for clean intermittent catheterization. J Urol. 2003;169:994–8.
De Ridder DJ, Everaert K, Fernandez LG, Valero JV, Duran AB, Abrisqueta ML, et al. Intermittent catheterisation with hydrophilic-coated catheters (SpeediCath) reduces the risk of clinical urinary tract infection in spinal cord injured patients: a prospective randomised parallel comparative trial. Eur Urol. 2005;48:991–5.
Stensballe J, Looms D, Nielsen PN, Tvede M. Hydrophilic-coated catheters for intermittent catheterisation reduce urethral micro trauma: a prospective, randomised, participant-blinded, crossover study of three different types of catheters. Eur Urol. 2005;48:978–83.
Moore KN, Burt J, Voaklander DC. Intermittent catheterization in the rehabilitation setting: a comparison of clean and sterile technique. Clin Rehabil. 2006;20:461–8.
Cardenas DD, Hoffman JM. Hydrophilic catheters versus noncoated catheters for reducing the incidence of urinary tract infections: a randomized controlled trial. Arch Phys Med Rehabil. 2009;90:1668–71.
Sarica S, Akkoc Y, Karapolat H, Aktug H. Comparison of the use of conventional, hydrophilic and gel-lubricated catheters with regard to urethral micro trauma, urinary system infection, and patient satisfaction in patients with spinal cord injury: a randomized controlled study. Eur J Phys Rehabil Med. 2010;46:473–9.
Cardenas DD, Moore KN, Dannels-McClure A, Scelza WM, Graves DE, Brooks M, et al. Intermittent catheterization with a hydrophilic-coated catheter delays urinary tract infections in acute spinal cord injury: a prospective, randomized, multicenter trial. PM R. 2011;3:408–17.
Denys P, Prévinaire JG, Aegerter P, de Sèze M, Karsenty G, Amarenco G. Intermittent self-catheterization habits and opinion on aseptic VaPro catheter in French neurogenic bladder population. Spinal Cord. 2012;50:853–8.
Chartier-Kastler E, Amarenco G, Lindbo L, Soljanik I, Andersen HL, Bagi P, et al. A prospective, randomized, crossover, multicenter study comparing quality of life using compact versus standard catheters for intermittent self-catheterization. J Urol. 2013;190:942–7.
Chick HE, Hunter KF, Moore KN. Parent and child experiences using a hydrophilic or reused PVC catheter for intermittent catheterisation. J Clin Nurs. 2013;22:513–20.
Leek H, Stephenson Z, Reus A, Karantanis E, Moore K. Clean intermittent self-catheterisation: a randomised controlled crossover trial of single-use versus multiple re-use of non-coated catheters; is cystitis rate altered? Neurourol Urodyn. 2013;32:759–60.
Kiddoo D, Sawatzky B, Bascu CD, Dharamsi N, Afshar K, Moore KN. Randomized Crossover Trial of Single Use Hydrophilic Coated vs Multiple Use Polyvinylchloride Catheters for Intermittent Catheterization to Determine Incidence of Urinary Infection. J Urol. 2015;194:174–9.
Lucas EJ, Baxter C, Singh C, Mohamed AZ, Li B, Zhang J, et al. Comparison of the microbiological milieu of patients randomized to either hydrophilic or conventional PVC catheters for clean intermittent catheterization. J Pediatr Urol. 2016;12:172.e1–8.
DeFoor W, Reddy P, Reed M, VanderBrink B, Jackson E, Zhang B, et al. Results of a prospective randomized control trial comparing hydrophilic to uncoated catheters in children with neurogenic bladder. J Pediatr Urol. 2017;13:373.e1–e5.
Madero-Morales PA, Robles-Torres JI, Vizcarra-Mata G, Guillén-Lozoya AH, Mendoza-Olazarán S, Garza-González E, et al. Randomized clinical trial using sterile single use and reused polyvinylchloride catheters for intermittent catheterization with a clean technique in spina bifida cases: short-term urinary tract infection outcomes. J Urol. 2019;202:153–8.
Goessaert AS, Antoons S, Van Den Driessche M, Tourchi A, Pieters R, Everaert K. No-touch intermittent catheterization: caregiver point of view on sterility errors, duration, comfort and costs. J Adv Nurs. 2013;69:2000–7.
NIDRR. The prevention and management of urinary tract infections among people with spinal cord injuries. National Institute on Disability and Rehabilitation Research Consensus Statement. J Am Paraplegia Soc. 1992;15:194–204.
Funding
This study is supported by grants from 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYGD18011, ZY2016104 and ZYJC18015).
Author information
Authors and Affiliations
Contributions
Study design: DHY and YTC; data collection: ZYJ and BHL; data analysis: DHY, XJ, and LYX; writing of this paper: DHY and YTC; revision of this paper: DHY, HL, and KJW.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ye, D., Chen, Y., Jian, Z. et al. Catheters for intermittent catheterization: a systematic review and network meta-analysis. Spinal Cord 59, 587–595 (2021). https://doi.org/10.1038/s41393-021-00620-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-021-00620-w