Abstract
Study design
A historical cohort study
Objectives
The aim of the study was to examine the incidence of and predictive factors for VUR in individuals with suprasacral spinal cord injury (SCI).
Setting
Srinagarind Hospital, Khon Kaen University, Thailand
Methods
Medical records were reviewed for all individuals with SCI and neurogenic bladder admitted for urological check-up between 1996 and 2016. The primary outcome was the cumulative incidence of VUR. The statistical tests used included the Nelson–Aalen Estimator and Cox Proportional Hazard Ratio. Harrell’s C concordance statistic was used to evaluate the discrimination ability of the predictive model.
Results
293 participants with SCI (102 tetraplegic and 191 paraplegic) were included. Most participants were male (67%), and the median age was 52 years. The overall incidence of VUR was 7.5 cases per 100 person-years (95% CI, 6.15–9.4). In the multivariate analysis, the predictive factors for VUR were: (1) maximum detrusor pressure at first visit ≥ 75 cm of water (HRadj: 2.4 [95% CI: 1.4–4.1]); (2) indwelling urethral catheterization (IUC) (HRadj: 11.1 [95% CI: 3.9–31.7]) and clean intermittent catheterization (CIC) (HRadj: 6.5 [95% CI: 2.2–18.7]); (3) age ≥ 60 years at onset of SCI (HRadj: 1.7 [95% CI: 1.1–2.8]); and, (4) absence of antimuscarinic medication (HRadj: 3.8 [95% CI: 2.4–6.1]). The predictive model had an overall C-index of 0.78.
Conclusions
The incidence of VUR was high up to 12 years after SCI. High maximum detrusor pressure, IUC, age ≥ 60 years and absence of antimuscarinic medication were predictive factors for VUR.
Similar content being viewed by others
Introduction
Neurogenic bladder is one of the most common problems found after spinal cord injury (SCI). This condition can lead to two types of abnormality in bladder function during the storage and emptying phases: overactive bladder and/or incomplete emptying of the bladder [1]. These problems require bladder management for urinary drainage, such as indwelling urethral catheterization (IUC) or clean intermittent catheterization (CIC) [2]. Individuals with SCI require urological long-term care to maintain their quality of life [3]. Investigations of the urological system include: urinalysis, urine culture, ultrasonography, urodynamic study, and voiding cystourethrography [4]. Despite advances in early urological investigation and medical treatments, affected persons continue to endure urological complications (i.e., vesicoureteric reflux (VUR), renal calculi, hydronephrosis, and urinary tract infection) [2, 5,6,7].
One of the most common complications in neurogenic bladder is VUR which is a condition in which urine flows backward from the bladder to one or both ureters and sometimes to the kidneys. Its prevalence is in the range of 8–24% in individuals with SCI [2, 8,9,10]. It was noted that most people with SCI developed VUR within 4 years after injury [2, 8, 9, 11]. VUR is one of the causes of morbidity and mortality in people with SCI. If VUR is not properly treated, the condition can lead to recurrent urinary tract infection, hydronephrosis, renal failure, and/or death [3, 12].
The vesicoureteral junction (VUJ) is on the border of the upper urinary tract and the lower urinary tract and it protects the upper tract from reflux by active and passive anti-reflux mechanisms [13]. The VUR can be classified into 2 types: the primary type is an anatomical defect that results in incomplete closure of the VUJ; and a secondary type, which is the main cause of VUR in SCI, which is due to elevated bladder pressure either during the filling phase or the voiding phase [14]. Additionally, failure of VUJ can contribute to VUR which may be caused by chronic infections weakening the valve mechanism due to scarring of the VUJ;[15] and bladder trabeculations or diverticula near the ureteral orifice, thus preventing the submucosal ureteral tunnel being passively compressed [14].
Suprasacral SCI is associated with VUR [16]. This may be because participants with suprasacral SCI have detrusor overactivity which results in high filling pressure and bladder outlet obstruction from detrusor external sphincter dyssynergia (DESD) which results in high voiding pressure, thus contributing to VUR and upper tract deterioration [12]. An SCI between thoracic level 10 and lumbar level 2 which involves the sympathetic nervous system shows a significantly higher risk of VUR than in other spinal cord regions, which may be due to sympathetic injury affecting the VUJ mechanism and bladder compliance [17]. Age at onset of SCI, duration of SCI, types of bladder management especially indwelling urethral catheterization (IUC), duration of IUC of more than 6 months, urodynamics findings of low bladder capacity, low bladder compliance, high urethral pressure, high maximum detrusor pressure, high detrusor leak point pressure, abnormal radiologic lower urinary tract findings, recurrent urinary tract infection and the absence of antimuscarinic usage were found to be associated with VUR in univariate and multivariate analysis [10, 18,19,20,21,22].
If the aim of urologic surveillance is early detection of such urological complications, it may be useful to see the natural history of VUR development (i.e., incidence of VUR). Since follow-up is time and resource limited, it would be best to allocate the resource to the group with the highest risk and/or at a specific time point of highest risk. To the best of our knowledge, most published studies analyzed data from cross-sectional or retrospective studies, and there have been no published studies on the incidence of VUR in individuals with SCI from a cohort study. The purpose of the present study was to describe the incidence of VUR and to identify the predictive factors associated with the development of VUR in individuals with suprasacral SCI.
Methods
Study design and participants
Our historical cohort study was reviewed and approved by the Khon Kaen University Ethics Committee in Human Research (HE591494). We obtained data from the hospital’s OPD records and IPD reviews. The data set contained: sex, age, cause of SCI, neurological level, duration of SCI, duration from SCI to first urological checkup and to outcome, medication, radiologic findings, methods of bladder managements, and urodynamic results.
Methodology
We analyzed the data from all participants with neurogenic bladder after SCI who underwent the urological check-up between January, 1996 and December, 2016 at Srinagarind Hospital, Thailand. The urological check-up data included annual urodynamic studies and vesicoureterography. The outcome of the study was a first episode of VUR identified by vesicoureterography.
Demographics and clinical data were collected on all available participants who had had a urological checkup. Excluded from the study were participants who had myelomeningocele or did not go to early follow-up for a urological check-up (time after SCI and first check-up more than 2 years apart). We were thus able to identify 293 participants.
Urodynamics were determined by standardized procedure. The bladder was filled and the intravesical pressure recorded via a transurethral double-lumened catheter while abdominal pressure was recorded by a rectal indwelling catheter. Sterile saline solution was used to fill the bladder at a rate of 20 cc/min [23]. Bladder compliance was calculated by the detrusor pressure and corresponding bladder volume at cystometric bladder capacity or immediately before the start of detrusor contraction causing significant leakage. Maximum detrusor pressure was defined as the highest detrusor pressure measured during filling cystometry [24].
The primary outcome measure was presence of VUR. For each cohort member, person-years at risk were calculated commencing upon diagnosis of SCI, and finishing on the date of VUR documentation, loss of follow up, death or the end of December 2016 when the data set was frozen.
Terminology
‘Neurogenic bladder’’ was diagnosed according to the definition of adult neurogenic lower urinary tract dysfunction (ANLUTD) which refers to abnormal or difficult function of the bladder or urethra in mature individuals in the context of clinically confirmed relevant neurologic disorder;[25] specifically, in the present study, disorder refers to SCI with AIS classification A–D. ‘Balanced bladder’ describes a state where patients can empty their bladder with a maximum detrusor pressure below 60 cm H20 and with a post-void residual urine less than 100 ml [26] for 3 days. ‘Intermittent catheterization’ describes patients who drain their urine from the bladder with subsequent removal of the catheter either combined without or with voluntary voiding (IC based on residual urine).
Statistical analysis
All participants were enrolled into the cohort on the date of their first recorded SCI. Follow-up continued until December 31, 2016, unless they had VUR. Participants who were lost to follow-up were censored at the time of their last urological check-up. Person-years at risk (PYAR) were calculated from the date of censoring and of VUR. Nelson–Aalen estimator was used to demonstrate the cumulative risk of VUR. The log-rank test was used to assess differences between the incidence rate across age at index date of SCI, sex, level of spinal cord injury, bladder compliance, cystometric bladder capacity, maximum detrusor pressure at first visit, antimuscarinic medication, the presence of abnormal radiologic findings classified by Ogawa’s classification of bladder deformity [27] and bladder management methods. Univariate and Multivariate Cox Proportional hazard ratios (HRs) and the respective 95% confidence intervals (CIs) were calculated to determine the independent effect of each variable and the overall rates of VUR. The age at index date of SCI, maximum detrusor pressure at first visit, antimuscarinic medication and bladder management methods were included in the adjusted model. Multicollinearity was tested on all variables before adding each into the regression model. In order to find the optimum cut-off point for maximum detrusor pressure, each individual cut-off score was tested to determine sensitivity and specificity for having VUR. The optimum cut-off was determined by the Youden’s index. Discrimination ability of the predictive model was evaluated from Harrell’s C concordance statistics and reported in overall C-index [28]. A C-index of more than 0.9 indicates high accuracy, a value from 0.7 to 0.9 indicates moderate accuracy and a value around 0.5 indicates no predictive ability [29]. All statistical tests were two-sided and the significance level was 0.05. All statistical analyses were done using Stata version 13.0 (Stata Statistical Software: Release 13. College Station, TX: Stata Corp LP).
Results
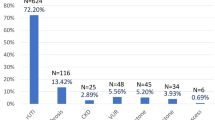
293 participants with SCI were included (Fig. 1): 102 were tetraplegic and 191 were paraplegic. Most of the participants were male (67%), and the median age was 52 years. The median duration of documented SCI to first urological check-up was 6 months. The median follow-up time in this cohort was 2 years and 7 months. The types of bladder management used included: (1) indwelling urethral catheterization (IUC) in 106 (37%) participants, (2) clean intermittent catheterization (CIC) in 108 (38%), and (3) balanced bladder in 70 (25%) (Table 1).
Of participants with SCI, there were 85 VUR events in the analysis cohort. The overall incidence of VUR was 7.5 cases/100 person-years at risk (PYAR) (95% CI, 6.1–9.3) (Table 2). The Nelson–Aalen estimator in Fig. 2 presents the estimates cumulative hazard of VUR in the overall cohort. The median cumulative hazard is 11.6 years.
In the univariate analysis and multivariate analysis, the predictive factors for VUR were (1) maximum detrusor pressure at first visit of more than 75 cm H2O (HRadj: 2.4 [95% CI: 1.4–4.1]); (2) bladder management methods by IUC (HRadj: 11.1 [95% CI: 3.9–31.7]) and clean intermittent catheterization (HRadj: 6.5 [95% CI: 2.2–18.7]); (3) age more than 60 years at the onset of SCI (HRadj: 1.7 [95% CI: 1.1–2.8]); and, (4) absence of antimuscarinic medication (HRadj: 3.7 [95% CI: 2.4–6.1]) (Table 3). The estimates cumulative hazard of VUR for each variable are presented in Fig. 3. The prediction model yielded an overall C-index of 0.78 which indicates moderate accuracy.
Discussion
Our study aimed to determine the incidence of VUR and to determine the predictive factors for VUR in participants after SCI. Most studies reported a prevalence of VUR between 8–24% among individuals with SCI [2, 8,9,10]. after SCI. To our knowledge, ours is the first study to report the incidence of VUR (viz., 7.5 cases per100 PYAR [95% CI, 6.1–9.3]). The incidence of VUR was high up to 12 years
As with previous studies, we found that IUC was a strong predictive factor for VUR [10, 20]. There are a few reasons that could account for this. It is possible that IUC may promote an inflammatory response and infectious process which may lead to changes at the ureteral orifice from infection [30] or changes in bladder connective tissue content [31]. These process together with continual bladder decompression may accelerate bladder wall fibrosis. Larsen et al. suggested that severe, histological, detrusor fibrosis was likely a risk factor in upper urinary tract deterioration [32]. Alternatively, IUC may be related to unknown neurological mechanisms—such as aberrant local reflexes or altered bladder and outlet interaction—that may contribute to VUR [33]. Our study supported Lamid who reported that having an IUC did not prevent reflux formation and did not protect the refluxing kidney from damage [11]. IUC was, moreover, shown to be associated with higher urological complications such as urinary tract infection, bladder calculi, and severe urethral incompetence [32, 34,35,36].
CIC has a higher chance of causing the development of VUR compared to participants with balanced bladder. The ability to void with acceptable residual urine of less than 100 ml in patients with balanced bladder may imply a lesser degree of neurological involvement such as lower degrees of detrusor overactivity and detrusor external sphincter dyssynergia thus such patients would be less likely to develop VUR. Nevertheless, in patients with residual urine and unable to reach a balanced bladder, CIC is a better choice of bladder management as the incidence of VUR and other urological complications is lower than IUC.
Absence of anticholinergic medication, high maximum detrusor pressure (≥75 cm H2O), and leak point pressure (≥40 cm H2O) were risk factors and predictive factors for upper tract deterioration in participants with SCI [21, 22, 37]. Our study showed that the presence of maximum detrusor pressure of ≥75 cmH2O at first urological checkup and absence of antimuscarinic drug usage are predictors for developing VUR in the future. The cut-off point for maximum detrusor pressure found in our study (≥75 cm H2O)—obtained from the ROC curve analysis—was similar to that found by Cetinel et al. [22]. In contrast to previous studies, we did not find abnormal radiographic findings, low cystometric bladder capacity, or low bladder compliance as predictors for developing VUR [10, 22]. We propose that since previous studies were retrospective, the results may indicate an association between the latest urodynamic or radiologic findings and the presence of VUR. Our study used the early data from the first urological checkup, thus in the beginning the low bladder capacity or abnormal radiologic findings might not yet be fully demonstrated.
Age over 60 years was a significant predictive factor for VUR. Weld et al. reported that IUC trended to be used in elderly patients [20]. The significant association of age over 60 years at SCI in the multivariate analysis implies that there may be another factor that increases the chance of developing VUR in older persons that is independent of the methods used for bladder management. This may be due to weakening of the vesicoureteric sphincter in old age, or it may be that antimuscarinic medication used in old age is lower dosage than in younger age groups because of side effects thus making detrusor pressure not well-controlled.
Similar to a previous study, sex and completeness of lesion are not significantly associated with VUR [19]. We did not find any evidence of predominance of lesion level. By comparison, one study reported that lesions between T10-L2 were associated with a higher risk of VUR than other levels perhaps because the sympathetic system supplies the area of trigone such that lesions between T10-L2 would disrupt the sympathetic system [2, 17] Further studies are needed to confirm this.
It should be noted that the current study mainly focused on predictive factors. Although the analyzing methods are the same for the study of risk and predictive factors, predictive factors do not necessarily indicate causation which is different from risk factors [38]. Any association, causal or non-causal, can serve as predictive factors. Additionally, the concepts of confounding, mediation and effect measure modification are not applicable to predictive models and variables that cannot be manipulated can be included [39, 40]. The findings in our study should be interpreted with caution. For example, CIC and age may not serve as risk factors themselves, but as predictive factors [41, 42].
The main limitation of our study was that the design of our study involved a historical cohort. There were missing data and some missed factors that are common risk factors associated with VUR, such as UTIs or the presence of detrusor external sphincter dyssynergia, which may add more accuracy in the predictive model. The rate of occurrence of VUR more than 12 years after SCI should be interpreted with caution because of a low number of individuals at risk mainly due to loss of follow up in the later years. Generalizability of data may be limited due to a single center source and the lost to follow up population. Additionally, loss of follow up population may cause an over- or underestimate of the association between the predictor and the outcome. Notwithstanding, the results of the current study provide a basis for further studies and may guide the development of VUR surveillance concepts.
In conclusion, the incidence of VUR was high up to 12 years after SCI. The occurrence of VUR should be considered, especially among participants with SCI (1) with IUC, (2) with high maximum detrusor pressure (≥75 cm H2O) at first visit, (3) who are older than 60 years and, (4) with absence of antimuscarinic medication.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Lapides J Medical management of neurogenic bladder with oral therapy. In: Campbell M, Harrison J, editors. Urology. Philadelphia USA: WB Saunders; 1997. p. 1343–79.
Foley S, McFarlane J, Shah P. Vesico-ureteric reflux in adult patients with spinal injury. Br J Urol. 1997;79:4.
Persu C, Caun V, Dragomiriteanu I, Geavlete P. Urological management of the patient with traumatic spinal cord injury. J Med Life. 2009;2:296–302.
Harrison SC. Managing the urinary tract in spinal cord injury. Indian J Urol. 2010;26:245–52.
Hall M, Hackler R, Zampieri T, Zampieri J. Renal calculi in spinal cord-injured patient: association with reflux, bladder stones, and foley catheter drainage. Urology. 1989;34:3.
Price M, Kottke F, Olson M. Renal function in patients with spinal cord injury: the eighth year of a ten-year continuing study. Arch Phys Med Rehabil. 1975;56:3.
Cardenas DD, Hooton TM. Urinary tract infection in persons with spinal cord injury. Arch Phys Med Rehabil. 1995;76:272–80.
Thongchim C, Tamnanthong N, Arayawichanont P. Prevalence of vesicoureteric reflux in neurogenic bladder dysfunction patients from spinal cord lesion. J Thai Rehabil Med. 2010;20:52–7.
Ku JH, Choi WJ, Lee KY, Jung TY, Lee JK, Park WH, et al. Complications of the upper urinary tract in patients with spinal cord injury: a long-term follow-up study. Urological Res. 2005;33:435–9. PubMed PMID: WOS:000233869200006
Suksathien R, Ingkasuthi K, Bumrungna S. Factors associated with hydronephrosis and vesicoureteral reflux in spinal cord injured patients. Asean J Rehabil Med. 2019;29:51–7.
Lamid S. Long-term follow-up of spinal cord injury patients with vesicoureteral reflux. Paraplegia. 1988;26:27–34.
Taweel WA, Seyam R. Neurogenic bladder in spinal cord injury patients. Res Rep. Urol. 2015;7:85–99.
Arena S, Iacona R, Impellizzeri P, Russo T, Marseglia L, Gitto E, et al. Physiopathology of vesico-ureteral reflux. Italian J Pediatrics. 2016;42:103.
Wu CQ, Franco I. Management of vesicoureteral reflux in neurogenic bladder. Investigative Clin Urol. 2017;58:554–8.
Peters C, Rushton HG. Vesicoureteral reflux associated renal damage: congenital reflux nephropathy and acquired renal scarring. J Urol. 2010;184:265–73.
Hackler RH, Dalton JJ, Bunts RC. Changing concepts in the preservation of renal function in the paraplegic. J Urol. 1965;94:107–11.
Suzuki T, Ushiyama T. Vesicoureteral reflux in the early stage of spinal cord injury: a retrospective study. Spinal Cord. 2001;39:23–5.
Lee JS, Koo BI, Shin MJ, Chang JH, Kim SY, Ko HY. Differences in urodynamic variables for vesicoureteral reflux depending on the neurogenic bladder type. Ann Rehabil Med. 2014;38:347–52.
Zhang Z, Liao L. Risk factors predicting upper urinary tract deterioration in patients with spinal cord injury: a prospective study. Spinal Cord. 2014;52:468–71.
Weld KJ, Wall BM, Mangold TA, Steere EL, Dmochowski RR. Influences on renal function in chronic spinal cord injured patients. J Urol. 2000;164:1490–3.
Musco S, Padilla-Fernandez B, Del Popolo G, Bonifazi M, Blok BFM, Groen J, et al. Value of urodynamic findings in predicting upper urinary tract damage in neuro-urological patients: a systematic review. Neurourol Urodyn. 2018;37:1522–40.
Cetinel B, Onal B, Can G, Talat Z, Erhan B, Gunduz B. Risk factors predicting upper urinary tract deterioration in patients with spinal cord injury: a retrospective study. Neurourol Urodyn. 2017;36:653–8.
Stöhrer M, Goepel M, Kondo A, Kramer G, Madersbacher H, Millard R, et al. The standardization of terminology in neurogenic lower urinary tract dysfunction: with suggestions for diagnostic procedures. International Continence Society Standardization Committee. Neurourol Urodyn. 1999;18:139–58.
Pannek J, Kennelly M, Kessler T, Linsenmeyer T, Wyndaele J, Biering-Sorensen F. International spinal cord injury urodynamic basic data set (version 2.0). Spinal Cord Ser Cases. 2018;4:98.
Gajewski JB, Schurch B, Hamid R, Averbeck M, Sakakibara R, Agro EF, et al. An International Continence Society (ICS) report on the terminology for adult neurogenic lower urinary tract dysfunction (ANLUTD). Neurourol Urodyn. 2018;37:1152–61.
Wyndaele JJ, De, Taeye N. Early intermittent self-catheterisation after spinal cord injury. Paraplegia. 1990;28:76–80.
Sekido N, Igawa Y, Kakizaki H, Kitta T, Sengoku A, Takahashi S, et al. Clinical guidelines for the diagnosis and treatment of lower urinary tract dysfunction in patients with spinal cord injury. Int J Urol. 2020;27:276–88.
Pencina MJ, D’Agostino RB. Overall C as a measure of discrimination in survival analysis: model specific population value and confidence interval estimation. Stat Med. 2004;23:2109–23.
Chen L, Wang H, Zeng H, Zhang Y, Ma X. Evaluation of CT-based radiomics signature and nomogram as prognostic markers in patients with laryngeal squamous cell carcinoma. Cancer imaging. 2020;20:28.
BORS E, COMARR AE. Vesico-ureteral reflux in paraplegic patients. J Urol. 1952;68:691–8.
Landau EH, Jayanthi VR, Churchill BM, Shapiro E, Gilmour RF, Khoury AE, et al. Loss of elasticity in dysfunctional bladders: urodynamic and histochemical correlation. J Urol. 1994;152:702–5.
Larsen LD, Chamberlin DA, Khonsari F, Ahlering TE. Retrospective analysis of urologic complications in male patients with spinal cord injury managed with and without indwelling urinary catheters. Urology. 1997;50:418–22.
Weld KJ, Graney MJ, Dmochowski RR. Differences in bladder compliance with time and associations of bladder management with compliance in spinal cord injured patients. J Urol. 2000;163:1228–33.
Singh R, Rohilla RK, Sangwan K, Siwach R, Magu NK, Sangwan SS. Bladder management methods and urological complications in spinal cord injury patients. Indian J Orthop. 2011;45:141–7.
Bennett CJ, Young MN, Adkins RH, Diaz F. Comparison of bladder management complication outcomes in female spinal cord injury patients. J Urol. 1995;153:1458–60.
Jacobs SC, Kaufman JM. Complications of permanent bladder catheter drainage in spinal cord injury patients. J Urol. 1978;119:740–1.
Dewire DM, Owens RS, Anderson GA, Gottlieb MS, Lepor H. A comparison of the urological complications associated with long-term management of quadriplegics with and without chronic indwelling urinary catheters. J Urol. 1992;147:1069–71.
Chao R, Clowers D, Mayo ME. Fate of upper urinary tracts in patients with indwelling catheters after spinal cord injury. Urology. 1993;42:259–62.
McGuire EJ, Woodside JR, Borden TA, Weiss RM. Prognostic value of urodynamic testing in myelodysplastic patients. J Urol. 1981;126:205–9.
Rothman KJ, Greenland S. Causation and causal inference in epidemiology. Am J Public Health. 2005;95:S144–S50.
Herbert RD. Cohort studies of aetiology and prognosis: they’re different. J Physiother. 2014;60:241–4.
Schooling CM, Jones HE. Clarifying questions about “risk factors”: predictors versus explanation. Emerg Themes Epidemiol. 2018;15:10.
Acknowledgements
We would like to thank (a) Mrs Kaewjai Tepsuthumarat at the Clinical Epidemiology Unit for guidance with the biostatistical analysis, and (b) Mr. Bryan Roderick Hamman under the aegis of the Publication Clinic KKU, Thailand, for assistance with the English-language presentation of the manuscript.
Author information
Authors and Affiliations
Contributions
PS designed the research question, collected the data, drafted the paper and wrote the final version of the paper. JS designed the research question, collected the data, analyzed the data and commented on the final version of the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of Ethics
This study was approved by the Ethics Review Committee of Khon Kaen University, Thailand (HE591494). We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of the research.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sirasaporn, P., Saengsuwan, J. Incidence and predictive factors for developing vesicoureteric reflux in individuals with suprasarcral spinal cord injury: a historical cohort study. Spinal Cord 59, 753–760 (2021). https://doi.org/10.1038/s41393-020-00573-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-020-00573-6