Abstract
Background
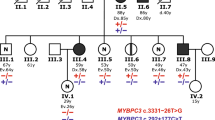
Tetralogy of Fallot (TOF) is a common form of congenital heart disease. The MYH6 gene has important effects on cardiovascular growth and development.
Methods
In 608 subjects, including 315 TOF patients, we investigated the MYH6 gene promoter variants and verified the effect on gene expression by using cellular functional experiments with three cell lines (HEK-293, HL-1, and H9C2 cells) and bioinformatics analysis.
Results
In the MYH6 gene promoter, 12 variants were identified from 608 subjects. Five variants were found only in patients with TOF and two of them (g.3384G>T and g.4518T>C) were novel. Electrophoretic mobility shift assay with three cell lines (HEK-293, HL-1, and H9C2) showed significant changes in the transcription factors bound by the promoter variants compared to the wild-type. Dual luciferase reporter showed that four of the five variants reduced the transcriptional activity of the MYH6 gene promoter (p < 0.05).
Conclusions
This study is the first to test the cellular function of variants in the promoter region of the MYH6 gene in patients with TOF, which provides new insights into the genetic basis of TOF and provides a basis for further study of the mechanism of TOF formation.
Impact
-
DNA from 608 human subjects was sequenced for MYH6 gene promoter region variants with five variants found only in TOF patients and two were novel.
-
EMSA and dual luciferase reporter experiments in three cell lines found these variants pathological.
-
Prediction by JASPAR database indicated that these variants alter the transcription factor binding sites.
-
The study, for the first time, confirmed that there are variants at the MYH6 gene promoter region and these variants alter the cellular function.
-
The variants found in this study suggest the possible pathological role in the formation of TOF.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The datasets generated during and analyzed during the current study are available from https://ngdc.cncb.ac.cn/omix/view/OMIX001938 and the corresponding author upon request.
References
Kakucs, Z., Heidenhoffer, E. & Pop, M. Detection of coronary artery and aortic arch anomalies in patients with tetralogy of Fallot using CT angiography. J. Clin. Med. 11, 5500 (2022).
Apitz, C., Webb, G. D. & Redington, A. N. Tetralogy of Fallot. Lancet 374, 1462–1471 (2009).
Althali, N. J. & Hentges, K. E. Genetic insights into non-syndromic Tetralogy of Fallot. Front. Physiol. 13, 1012665 (2022).
Starr, J. P. Tetralogy of fallot: yesterday and today. World J. Surg. 34, 658–668 (2010).
Faerber, J. A. et al. Identifying risk factors for complicated post-operative course in tetralogy of Fallot using a machine learning approach. Front. Cardiovasc. Med. 8, 685855 (2021).
Jiang, L. et al. Incremental prognostic value of myocardial strain over ventricular volume in patients with repaired tetralogy of Fallot. Eur. Radiol. 33, 1992–2003 (2023).
Gao, Y. et al. Superior prognostic value of right ventricular free wall compared to global longitudinal strain in patients with repaired tetralogy of Fallot. Front. Cardiovasc. Med. 9, 996398 (2022).
Valente, A. M. et al. Contemporary predictors of death and sustained ventricular tachycardia in patients with repaired tetralogy of Fallot enrolled in the INDICATOR cohort. Heart 100, 247–253 (2014).
He, G. W. et al. Corrective surgery alters plasma protein profiling in congenital heart diseases and clinical perspectives. Am. J. Transl. Res. 12, 1319–1337 (2020).
Sohn, S. & Lee, Y. T. Outcome of adults with repaired tetralogy of Fallot. J. Korean Med. Sci. 15, 37–43 (2000).
Khairy, P. et al. Arrhythmia burden in adults with surgically repaired tetralogy of Fallot: a multi-institutional study. Circulation 122, 868–875 (2010).
Tang, C. et al. Sequencing of a Chinese tetralogy of Fallot cohort reveals clustering mutations in myogenic heart progenitors. JCI Insight 7, e152198 (2022).
Reuter, M. S. et al. Clinical genetic risk variants inform a functional protein interaction network for tetralogy of Fallot. Circ. Genom. Precis. Med. 14, e003410 (2021).
Sharma, R. et al. Extra-cardiac diagnoses and postnatal outcomes of fetal tetralogy of fallot. Prenat. Diagn. 42, 260–266 (2022).
Villafañe, J. et al. Hot topics in tetralogy of Fallot. J. Am. Coll. Cardiol. 62, 2155–2166 (2013).
Kan, Z. et al. Identification of circRNA-miRNA-mRNA regulatory network and crucial signaling pathway axis involved in tetralogy of Fallot. Front. Genet 13, 917454 (2022).
Sun, H. et al. Expanding the phenotype associated with SMARCC2 variants: a fetus with tetralogy of Fallot. BMC Med. Genomics 15, 40 (2022).
He, G. W. et al. Identification of novel rare copy number variants associated with sporadic tetralogy of Fallot and clinical implications. Clin. Genet. 102, 391–403 (2022).
Chen, Z. et al. Pathophysiological Pathophysiological role of variants of the promoter region of CITED2 gene in sporadic tetralogy of Fallot patients with cellular function verification. Biomolecules 12, 1644 (2022).
Granados-Riveron, J. T. et al. Alpha-cardiac myosin heavy chain (MYH6) mutations affecting myofibril formation are associated with congenital heart defects. Hum. Mol. Genet. 19, 4007–4016 (2010).
Chen, J. H. et al. Identification of MYH6 as the potential gene for human ischaemic cardiomyopathy. Cell. Mol. Med. 25, 10736–10746 (2021).
Theis, J. L. et al. Recessive MYH6 mutations in hypoplastic left heart with reduced ejection fraction. Circ. Cardiovasc. Genet. 8, 564–571 (2015).
Bjornsson, T. et al. A rare missense mutation in MYH6 associates with non-syndromic coarctation of the aorta. Eur. Heart J. 39, 3243–3249 (2018).
Xia, Y. et al. Novel mutation in MYH6 in 2 unrelated Chinese Han families with familial atrial septal defect. Circ. Genom. Precis. Med. 12, e002732 (2019).
Jin, S. C. et al. Contribution of rare inherited and de novo variants in 2,871 congenital heart disease probands. Nat. Genet. 49, 1593–1601 (2017).
Zuo, J. Y. et al. Identification and functional analysis of variants of MYH6 gene promoter in isolated ventricular septal defects. BMC Med. Genomics 15, 213 (2022).
Yang, S. et al. Embelin prevents LMP1-induced TRAIL resistance via inhibition of XIAP in nasopharyngeal carcinoma cells. Oncol. Lett. 11, 4167–4176 (2016).
Castro-Mondragon, J. A. et al. JASPAR 2022: the 9th release of the open-access database of transcription factor binding profiles. Nucleic Acids Res. 50, D165–D173 (2022).
Zuo, J. Y. et al. Genetic analysis of variants of the MYH6 gene promoter in congenital atrial septal defects. Congenital Heart Disease 18, 7–21 (2023).
Benzeno, S. et al. Cyclin-dependent kinase inhibition by the KLF6 tumor suppressor protein through interaction with cyclin D1. Cancer Res. 64, 3885–3891 (2004).
Kumazaki, M. et al. Comparative effects of statins on murine cardiac gene expression profiles in normal mice. Eur. J. Pharmacol. 707, 71–77 (2013).
Schachterle, W. et al. ETS-dependent regulation of a distal Gata4 cardiac enhancer. Dev. Biol. 361, 439–449 (2012).
Anfinson, M. et al. Significance of α-Myosin Heavy Chain (MYH6) variants in hypoplastic left heart syndrome and related cardiovascular diseases. J. Cardiovasc. Dev. Dis. 9, 144 (2022).
Abu-Daya, A. et al. Absence of heartbeat in the Xenopus tropicalis mutation muzak is caused by a nonsense mutation in cardiac myosin myh6. Dev. Biol. 336, 20–29 (2009).
Li, F. et al. A NAC transcriptional factor BrNAC029 is involved in cytokinin-delayed leaf senescence in postharvest Chinese flowering cabbage. Food Chem. 404, 134657 (2023).
Suzuki, T. et al. Vascular implications of the Krüppel-like family of transcription factors. Arterioscler. Thromb. Vasc. Biol. 25, 1135–1141 (2005).
Subramaniam, M. et al. Functional role of KLF10 in multiple disease processes. Biofactors 36, 8–18 (2010).
Oo, J. A. et al. ZNF354C is a transcriptional repressor that inhibits endothelial angiogenic sprouting. Sci. Rep. 10, 19079 (2020).
Zheng, H. et al. Nkx2-3 induces autophagy inhibiting proliferation and migration of vascular smooth muscle cells via AMPK/mTOR signaling pathway. J. Cell. Physiol. 236, 7342–7355 (2021).
Westphal, D. S. et al. Identification of a de novo microdeletion 1q44 in a patient with hypogenesis of the corpus callosum, seizures and microcephaly – a case report. Gene 616, 41–44 (2017).
Hartmann, D. et al. MicroRNA-based therapy of GATA2-deficient vascular disease. Circulation 134, 1973–1990 (2016).
Ma, X. & Li, W. Amisulbrom causes cardiovascular toxicity in zebrafish (Danio rerio). Chemosphere 283, 131236 (2021).
Acknowledgements
We thank the patients and their family members for cooperation. The assistance of surgeons and Nursing staff at the Division of Pediatric Cardiac Surgery, Department of Cardiovascular Surgery is gratefully acknowledged.
Funding
This work was funded by the National Natural Science Foundation of China [82170353]; Tianjin Science and Technology Commission [22ZYQYSY00020];the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences [2020-PT310-007]; TEDA International Cardiovascular Hospital [2021-ZX-002]; and Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-019A).
Author information
Authors and Affiliations
Contributions
G.-W.H. obtained the funding for this project. J.-Y.Z. and H.-X.C. performed experiments. J.-Y.Z., H.-X.C., Z.-G.L. and G.-W.H. analyzed experimental data. J.-Y.Z., H.-X.C., Z.-G.L. and G.-W.H. collected the human blood samples. Q.Y. discussed the findings and participated in the project. J.-Y.Z. and G.-W.H. wrote the manuscript. G.-W.H. supervised the whole project. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study involving human participants was reviewed and approved by the ethics committee of TEDA International Cardiovascular Hospital, China (No. 0715-4, 2021, August 2, 2021).
Consent for publication
Patients signed informed consent regarding publishing their data and photographs.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zuo, JY., Chen, HX., Yang, Q. et al. Tetralogy of Fallot: variants of MYH6 gene promoter and cellular functional analyses. Pediatr Res 96, 338–346 (2024). https://doi.org/10.1038/s41390-023-02955-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02955-x
This article is cited by
-
Genetic insights into Tetralogy of Fallot: Oh MYH(6)
Pediatric Research (2024)
-
Molecular and cellular role of variants of the promoter region of HAND1 gene in sporadic and isolated ventricular septal defect
Molecular and Cellular Biochemistry (2024)