Abstract
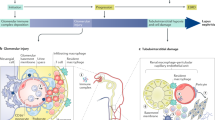
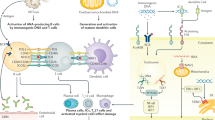
Increased longevity of patients with systemic lupus erythematosus (SLE) leads to chronic organ damage accrual, which reduces the possibility of further survival improvement in patients with the disease. Observations from lupus centres worldwide revealed that the prevalence of damage occurring in the cardiovascular system in patients with SLE has increased over the past four decades. The results of a meta-analysis involving over 70 observational studies demonstrated that lupus-related organ damage involving the neuropsychiatric and renal systems also remains a major factor that limits survival improvement in patients with this disease. While efforts to halt acute lupus-related injury are continuing, through early diagnosis and effective use of immunosuppressive agents, a concomitant strategy to improve survival of patients with SLE would be early detection and timely treatment of lupus-related organ damage with meticulous monitoring. This Review discusses the pattern and trend of organ damage in patients with SLE worldwide, the potential serological and genetic mechanisms of organ damage, and the advances in research on potential tools for early detection of lupus-related organ damage, such as functional brain imaging techniques, measurement of endothelial function, identification of biomarkers from body fluids, and development of risk calculation models.
Key Points
-
Overall survival of patients with systemic lupus erythematosus (SLE) has improved substantially over the past 5 decades, but has slowed down since the 1980s, possibly owing to organ damage
-
Although the most common causes of death in patients with SLE are infection and cardiovascular disease, organ damage predicting survival mainly involves renal, cardiovascular, neuropsychiatric and respiratory systems
-
Multiple factors contributing to organ damage in SLE lead to reduction in overall and organ survival, physical function, productivity and health-related quality of life, eventually leading to psychosocial and economic burden
-
Functional MRI of the brain, endothelium-dependent flow-mediated dilatation, identification of urinary biomarkers and FRAX® assessment are potential tools to detect early neuropsychiatric, cardiovascular and renal damage, and osteoporotic fracture, respectively
-
Further evaluation, standardisation and validation of detection tools are required before they can be widely used
-
Current data suggest that a combination (panel) of biomarkers and biomarker signatures will improve accurate and powerful detection of organ involvement in SLE
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Mak, A., Cheung, M. W., Chiew, H. J., Liu, Y. & Ho, R. C. Global trend of survival and damage of systemic lupus erythematosus: meta-analysis and meta-regression of observational studies from the 1950s to 2000s. Semin. Arthritis Rheum. 41, 830–839 (2012).
Lau, C. S. & Mak, A. The socioeconomic burden of SLE. Nat. Rev. Rheumatol. 5, 400–404 (2009).
Rahman, P., Gladman, D. D., Urowitz, M. B., Hallett, D. & Tam, L, S. Early damage as measured by the SLICC/ACR damage index is a predictor of mortality in systemic lupus erythematosus. Lupus 10, 93–96 (2001).
Chambers, S. A., Allen, E., Rahman, A. & Isenberg, D. Damage and mortality in a group of British patients with systemic lupus erythematosus followed up for over 10 years. Rheumatology (Oxford) 48, 673–675 (2009).
Mok, C. C., Ho, C. T., Wong, R. W. & Lau, C. S. Damage accrual in southern Chinese patients with systemic lupus erythematosus. J. Rheumatol. 30, 1513–1519 (2003).
Danila, M. I. et al. Renal damage is the most important predictor of mortality within the damage index: data from LUMINA LXIV, a multiethnic US cohort. Rheumatology (Oxford) 48, 542–545 (2009).
Guarize, J., Appenzeller, S. & Costallat, L. T. Skin damage occurs early in systemic lupus erythematosus and independently of disease duration in Brazilian patients. Rheumatol. Int. 27, 483–487 (2007).
Wang, Y. et al. Autoantibodies closely relate to the elevation level of in vivo hydrogen peroxide and tissue damage in systemic lupus erythematosus. DNA Cell Biol. 25, 563–570 (2006).
Yung, S. & Chan, T. M. Anti-DNA antibodies in the pathogenesis of lupus nephritis—the emerging mechanisms. Autoimmun. Rev. 7, 317–321 (2008).
Yung, S., Cheung, K. F., Zhang, Q. & Chan, T. M. Anti-dsDNA antibodies bind to mesangial annexin II in lupus nephritis. J. Am. Soc. Nephrol. 21, 1912–1927 (2010).
Yung, S. et al. Anti-DNA antibody induction of protein kinase C phosphorylation and fibronectin synthesis in human and murine lupus and the effect of mycophenolic acid. Arthritis Rheum. 60, 2071–2082 (2009).
Sun, K. H., Tang, S. J., Wang, Y. S., Lin, W. J. & You, R. I. Autoantibodies to dsDNA cross-react with the arginine-glycine-rich domain of heterogeneous nuclear ribonucleoprotein A2 (hnRNP A2) and promote methylation of hnRNP A2. Rheumatology (Oxford) 42, 154–161 (2003).
Wilson, A. M. et al. Asymmetric dimethylarginine correlates with measures of disease severity, major adverse cardiovascular events and all-cause mortality in patients with peripheral arterial disease. Vasc. Med. 15, 267–274 (2010).
Bultink, I. E., Teerlink, T., Heijst, J. A., Dijkmans, B. A. & Voskuyl, A. E. Raised plasma levels of asymmetric dimethylarginine are associated with cardiovascular events, disease activity, and organ damage in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 64, 1362–1365 (2005).
Kiani, A. N., Mahoney, J. A. & Petri, M. Asymmetric dimethylarginine is a marker of poor prognosis and coronary calcium in systemic lupus erythematosus. J. Rheumatol. 34, 1502–1505 (2007).
Xiao, Z. S. et al. Effect of asymmetric dimethylarginine on osteoblastic differentiation. Kidney Int. 60, 1699–1704 (2001).
Lu, R., Hu, C. P., Wu, X. P., Liao, E. Y. & Li, Y. J. Effect of age on bone mineral density and the serum concentration of endogenous nitric oxide synthase inhibitors in rats. Comp. Med. 52, 224–228 (2002).
Oates, J. C., Shaftman, S. R., Self, S. E. & Gilkeson, G. S. Association of serum nitrate and nitrite levels with longitudinal assessments of disease activity and damage in systemic lupus erythematosus and lupus nephritis. Arthritis Rheum. 58, 263–272 (2008).
Saxena, V. et al. Dual roles of immunoregulatory cytokine TGF-beta in the pathogenesis of autoimmunity-mediated organ damage. J. Immunol. 180, 1903–1912 (2008).
Taniguchi, Y. et al. Role of transforming growth factor-beta 1 in glomerulonephritis. J. Int. Med. Res. 25, 71–80 (1997).
Becker-Merok, A., Eilertsen, G. O. & Nossent, J. C. Levels of transforming growth factor-beta are low in systemic lupus erythematosus patients with active disease. J. Rheumatol. 37, 2039–2045 (2010).
Barreto, M. et al. Low frequency of CD4+CD25+ Treg in SLE patients: a heritable trait associated with CTLA4 and TGFbeta gene variants. BMC Immunol. 10, 5 (2009).
Font, J. et al. Association of mannose-binding lectin gene polymorphisms with antiphospholipid syndrome, cardiovascular disease and chronic damage in patients with systemic lupus erythematosus. Rheumatology (Oxford) 46, 76–80 (2007).
Øhlenschlaeger, T., Garred, P., Madsen, H. O. & Jacobsen, S. Mannose-binding lectin variant alleles and the risk of arterial thrombosis in systemic lupus erythematosus. N. Engl. J. Med. 351, 260–267 (2004).
Calvo-Alén, J. et al. Systemic lupus erythematosus in a multiethnic US cohort: XXXIV. Deficient mannose-binding lectin exon 1 polymorphisms are associated with cerebrovascular but not with other arterial thrombotic events. Arthritis Rheum. 54, 1940–1945 (2006).
Stellato, C. et al. Expression of the chemokine RANTES by a human bronchial epithelial cell line. Modulation by cytokines and glucocorticoids. J. Immunol. 155, 410–418 (1995).
Ye, D. Q. et al. Polymorphisms in the promoter region of RANTES in Han Chinese and their relationship with systemic lupus erythematosus. Arch. Dermatol. Res. 297, 108–113 (2005).
Sung, Y. K. et al. Interleukin-10 gene polymorphisms are associated with the SLICC/ACR Damage Index in systemic lupus erythematosus. Rheumatology (Oxford) 45, 400–404 (2006).
Johansson, M., Arlestig, L., Moller, B., Smedby, T. & Rantapaa-Dahlqvist, S. Oestrogen receptor α gene polymorphisms in systemic lupus erythematosus. Ann. Rheum. Dis. 64, 1611–1617 (2005).
Lundström, E. et al. HLA-DRB1*04/*13 alleles are associated with vascular disease and antiphospholipid antibodies in systemic lupus erythematosus. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2012-201760.
Diamond, B. Antibodies and the brain: lessons from lupus. J. Immunol. 185, 2637–2640 (2010).
Aranow, C., Diamond, B. & Mackay, M. Glutamate receptor biology and its clinical significance in neuropsychiatric systemic lupus erythematosus. Rheum. Dis. Clin. North Am. 36, 187–201, x–xi (2010).
Lapteva, L. et al. Anti-N-methyl-D-aspartate receptor antibodies, cognitive dysfunction, and depression in systemic lupus erythematosus. Arthritis Rheum. 54, 2505–2514 (2006).
Kowal, C. et al. Human lupus autoantibodies against NMDA receptors mediate cognitive impairment. Proc. Natl Acad. Sci. USA 103, 19854–19859 (2006).
Gono, T. et al. NR2-reactive antibody decreases cell viability through augmentation of Ca(2+) influx in systemic lupus erythematosus. Arthritis Rheum. 63, 3952–3959 (2011).
Hanly, J. G. et al. Autoantibodies and neuropsychiatric events at the time of systemic lupus erythematosus diagnosis: results from an international inception cohort study. Arthritis Rheum. 58, 843–853 (2008).
Yoshio, T., Onda, K., Nara, H. & Minota, S. Association of IgG anti-NR2 glutamate receptor antibodies in cerebrospinal fluid with neuropsychiatric systemic lupus erythematosus. Arthritis Rheum. 54, 675–678 (2006).
Arinuma, Y., Yanagida, T. & Hirohata, S. Association of cerebrospinal fluid anti-NR2 glutamate receptor antibodies with diffuse neuropsychiatric systemic lupus erythematosus. Arthritis Rheum. 58, 1130–1135 (2008).
Lu, X. Y. et al. Anti-alpha-internexin autoantibody from neuropsychiatric lupus induce cognitive damage via inhibiting axonal elongation and promote neuron apoptosis. PLoS One 5, e11124 (2010).
Denburg, J. A., Carbotte, R. M. & Denburg, S. D. Neuronal antibodies and cognitive function in systemic lupus erythematosus. Neurology 37, 464–467 (1987).
Hanly, J. G. et al. Cognitive impairment and autoantibodies in systemic lupus erythematosus. Br. J. Rheumatol. 32, 291–296 (1993).
Spezialetti, R., Bluestein, H. G., Peter, J. B. & Alexander, E. L. Neuropsychiatric disease in Sjogren's syndrome: anti-ribosomal P and anti-neuronal antibodies. Am. J. Med. 95, 153–160 (1993).
Kozora, E., Ellison, M. C. & West, S. Reliability and validity of the proposed American College of Rheumatology neuropsychological battery for systemic lupus erythematosus. Arthritis Rheum. 51, 810–818 (2004).
DiFrancesco, M. W. et al. Functional magnetic resonance imaging assessment of cognitive function in childhood-onset systemic lupus erythematosus: a pilot study. Arthritis Rheum. 56, 4151–4163 (2007).
Fitzgibbon, B. M. et al. Functional MRI in NPSLE patients reveals increased parietal and frontal brain activation during a working memory task compared with controls. Rheumatology (Oxford) 47, 50–53 (2008).
Mak, A., Ren, T., Fu, E. H., Cheak, A. A. & Ho, R. C. A prospective functional MRI study for executive function in patients with systemic lupus erythematosus without neuropsychiatric symptoms. Semin. Arthritis Rheum. 41, 849–858 (2012).
Ren, T,, Ho, R. C. & Mak, A. Dysfunctional cortico-basal ganglia-thalamic circuit and altered hippocampal-amygdala activity on cognitive set-shifting in non-neuropsychiatric, SLE. Arthritis Rheum. 64, 4048–4059 (2012).
Folsom, A. R. et al. Coronary artery calcification compared with carotid intima-media thickness in the prediction of cardiovascular disease incidence: the Multi-Ethnic Study of Atherosclerosis (MESA). Arch. Intern. Med. 168, 1333–1339 (2008).
Simon, A., Chironi, G. & Levenson, J. Performance of subclinical arterial disease detection as a screening test for coronary heart disease. Hypertension 48, 392–396 (2006).
Thompson, T. et al. Progression of carotid intima-media thickness and plaque in women with systemic lupus erythematosus. Arthritis Rheum. 58, 835–842 (2008).
Asanuma, Y. et al. Premature coronary-artery atherosclerosis in systemic lupus erythematosus. N. Engl. J. Med. 349, 2407–2415 (2003).
Rho, Y. H. et al. Novel cardiovascular risk factors in premature coronary atherosclerosis associated with systemic lupus erythematosus. J. Rheumatol. 35, 1789–1794 (2008).
Yiu, K. H. et al. Pattern of arterial calcification in patients with systemic lupus erythematosus. J. Rheumatol. 36, 2212–2217 (2009).
Halcox, J. P. et al. Prognostic value of coronary vascular endothelial dysfunction. Circulation 106, 653–658 (2002).
Furchgott, R. F. & Zawadzki, J. V. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature 288, 373–376 (1980).
Corretti, M. C. et al. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: a report of the International Brachial Artery Reactivity Task Force. J. Am. Coll. Cardiol. 39, 257–265 (2002).
Mak, A., Liu, Y. & Ho, R. C. Endothelium-dependent but not endothelium-independent flow-mediated dilation is significantly reduced in patients with systemic lupus erythematosus without vascular events: a metaanalysis and metaregression. J. Rheumatol. 38, 1296–1303 (2011).
Lima, D. S., Sato, E. I., Lima, V. C., Miranda, F. Jr & Hatta, F. H. Brachial endothelial function is impaired in patients with systemic lupus erythematosus. J. Rheumatol. 29, 292–297 (2002).
El-Magadmi, M. et al. Systemic lupus erythematosus: an independent risk factor for endothelial dysfunction in women. Circulation 110, 399–404 (2004).
Mak, A. et al. Lumbar spine bone mineral density predicts endothelial reactivity in patients with systemic lupus erythematosus. Clin. Exp. Rheumatol. 29, 261–268 (2011).
Asahara, T., Kawamoto, A. & Masuda, H. Concise review: circulating endothelial progenitor cells for vascular medicine. Stem Cells 29, 1650–1655 (2011).
Lau, K. K. et al. Incremental predictive value of vascular assessments combined with the Framingham Risk Score for prediction of coronary events in subjects of low-intermediate risk. Postgrad. Med. J. 84, 153–157 (2008).
Lau, K. K. et al. Burden of carotid atherosclerosis in patients with stroke: relationships with circulating endothelial progenitor cells and hypertension. J. Hum. Hypertens. 21, 445–451 (2007).
Loomans, C. J., De Koning, E. J., Staal, F. J., Rabelink, T. J. & Zonneveld, A. J. Endothelial progenitor cell dysfunction in type 1 diabetes: another consequence of oxidative stress? Antioxid. Redox Signal. 7, 1468–1475 (2005).
Moonen, J. R. et al. Reduced number and impaired function of circulating progenitor cells in patients with systemic lupus erythematosus. Arthritis Res. Ther. 9, R84 (2007).
Grisar, J. et al. Systemic lupus erythematosus patients exhibit functional deficiencies of endothelial progenitor cells. Rheumatology (Oxford) 47, 1476–1483 (2008).
Deng, X. L., Li, X. X., Liu, X. Y., Sun, L. & Liu, R. Comparative study on circulating endothelial progenitor cells in systemic lupus erythematosus patients at active stage. Rheumatol. Int. 30, 1429–1436 (2010).
Hibbert, B. et al. Pre-procedural atorvastatin mobilizes endothelial progenitor cells: clues to the salutary effects of statins on healing of stented human arteries. PLoS One 6, e16413 (2011).
Grisar, J. et al. Endothelial progenitor cells in active rheumatoid arthritis: effects of tumour necrosis factor and glucocorticoid therapy. Ann. Rheum. Dis. 66, 1284–1288 (2007).
Distler, J. H. et al. EULAR Scleroderma Trials and Research group statement and recommendations on endothelial precursor cells. Ann. Rheum. Dis. 68, 163–168 (2009).
Devarajan, P. Neutrophil gelatinase-associated lipocalin (NGAL): a new marker of kidney disease. Scand. J. Clin. Lab. Invest. Suppl. 241, 89–94 (2008).
Pitashny, M. et al. Urinary lipocalin-2 is associated with renal disease activity in human lupus nephritis. Arthritis Rheum. 56, 1894–1903 (2007).
Nielsen, B. S. et al. Induction of NGAL synthesis in epithelial cells of human colorectal neoplasia and inflammatory bowel diseases. Gut 38, 414–420 (1996).
Wu, T. et al. Elevated urinary VCAM-1, P-selectin, soluble TNF receptor-1, and CXC chemokine ligand 16 in multiple murine lupus strains and human lupus nephritis. J. Immunol. 179, 7166–7175 (2007).
Abd-Elkareem, M. I., Al Tamimy, H. M., Khamis, O. A., Abdellatif, S. S. & Hussein, M. R. Increased urinary levels of the leukocyte adhesion molecules ICAM-1 and VCAM-1 in human lupus nephritis with advanced renal histological changes: preliminary findings. Clin. Exp. Nephrol. 14, 548–557 (2010).
Kiani, A. N. et al. Urinary vascular cell adhesion molecule, but not neutrophil gelatinase-associated lipocalin, is associated with lupus nephritis. J. Rheumatol. 39, 1231–1237 (2012).
Ikeda, Y., Fujimoto, T., Ameno, M., Shiiki, H. & Dohi, K. Relationship between lupus nephritis activity and the serum level of soluble VCAM-1. Lupus 7, 347–354 (1998).
Spronk, P. E., Bootsma, H., Huitema, M. G., Limburg, P. C. & Kallenberg, C. G. Levels of soluble VCAM-1, soluble ICAM-1, and soluble E-selectin during disease exacerbations in patients with systemic lupus erythematosus (SLE); a long term prospective study. Clin. Exp. Immunol. 97, 439–444 (1994).
Avihingsanon, Y. et al. Measurement of urinary chemokine and growth factor messenger RNAs: a noninvasive monitoring in lupus nephritis. Kidney Int. 69, 747–753 (2006).
Reyes-Thomas, J., Blanco, I. & Putterman, C. Urinary biomarkers in lupus nephritis. Clin. Rev. Allergy Immunol. 40, 138–150 (2011).
Schwartz, N. et al. Urinary TWEAK and the activity of lupus nephritis. J. Autoimmun. 27, 242–250 (2006).
Varghese, S. A. et al. Urine biomarkers predict the cause of glomerular disease. J. Am. Soc. Nephrol. 18, 913–922 (2007).
Suzuki, M. et al. Initial validation of a novel protein biomarker panel for active pediatric lupus nephritis. Pediatr. Res. 65, 530–536 (2009).
Zhang, X. et al. Biomarkers of lupus nephritis determined by serial urine proteomics. Kidney Int. 74, 799–807 (2008).
Cauley, J. A. et al. Clinical risk factors for fractures in multi-ethnic women: the Women's Health Initiative. J. Bone Miner. Res. 22, 1816–1826 (2007).
van den Bergh, J. P., van Geel, T. A., Lems, W. F. & Geusens, P. P. Assessment of individual fracture risk: FRAX and beyond. Curr. Osteoporos. Rep. 8, 131–137 (2010).
Fraser, L. A. et al. Fracture prediction and calibration of a Canadian FRAX(R.) tool: a population-based report from CaMos. Osteoporos. Int. 22, 829–837 (2011).
Mak, A., Lim, J. Q., Liu, Y., Cheak, A. A. & Ho, R. C. Significantly higher estimated 10-year probability of fracture in lupus patients with bone mineral density comparable to that of healthy individuals. Rheumatol. Int. http://dx.doi.org/10.1007/s00296-012-2389-1.
Mok, C. C. et al. Raloxifene for prevention of glucocorticoid-induced bone loss: a 12-month randomised double-blinded placebo-controlled trial. Ann. Rheum. Dis. 70, 778–784 (2011).
Baker-LePain, J. C., Nakamura, M. C., Shepherd, J. & von Scheven, E. Assessment of bone remodelling in childhood-onset systemic lupus erythematosus. Rheumatology (Oxford) 50, 611–619 (2011).
Abu-Shakra, M., Urowitz, M. B., Gladman, D. D. & Gough, J. Mortality studies in systemic lupus erythematosus. Results from a single center. I. Causes of death. J. Rheumatol. 22, 1259–1264 (1995).
Manger, K. et al. Definition of risk factors for death, end stage renal disease, and thromboembolic events in a monocentric cohort of 338 patients with systemic lupus erythematosus. Ann. Rheum. Dis. 61, 1065–1070 (2002).
Mok, C. C., Mak, A., Chu, W. P., To, C. H. & Wong, S. N. Long-term survival of southern Chinese patients with systemic lupus erythematosus: a prospective study of all age-groups. Medicine (Baltimore) 84, 218–224 (2005).
Cervera, R. et al. Morbidity and mortality in systemic lupus erythematosus during a 10-year period: a comparison of early and late manifestations in a cohort of 1,000 patients. Medicine (Baltimore) 82, 299–308 (2003).
Ruiz-Irastorza, G., Egurbide, M. V., Ugalde, J. & Aguirre, C. High impact of antiphospholipid syndrome on irreversible organ damage and survival of patients with systemic lupus erythematosus. Arch. Intern. Med. 164, 77–82 (2004).
Bernatsky, S. et al. Mortality in systemic lupus erythematosus. Arthritis Rheum. 54, 2550–2557 (2006).
Nossent, J. et al. Current causes of death in systemic lupus erythematosus in Europe, 2000--2004: relation to disease activity and damage accrual. Lupus 16, 309–317 (2007).
Urowitz, M. B., Gladman, D. D., Tom, B. D., Ibanez, D. & Farewell, V. T. Changing patterns in mortality and disease outcomes for patients with systemic lupus erythematosus. J. Rheumatol. 35, 2152–2158 (2008).
Kang, K. Y. et al. The causes of death in Korean patients with systemic lupus erythematosus over 11 years. Lupus 20, 989–997 (2011).
Acknowledgements
We thank Mr Tao Ren, a PhD student at the National University of Singapore, for his editorial assistance and for providing critical comments on the draft of the manuscript.
Author information
Authors and Affiliations
Contributions
A. Mak researched the data for the article. All authors contributed substantially to discussion of content, writing, reviewing and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Mak, A., Isenberg, D. & Lau, CS. Global trends, potential mechanisms and early detection of organ damage in SLE. Nat Rev Rheumatol 9, 301–310 (2013). https://doi.org/10.1038/nrrheum.2012.208
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2012.208
This article is cited by
-
Systemic lupus erythematosus with high disease activity identification based on machine learning
Inflammation Research (2023)
-
Lack of patient education is risk factor of disease flare in patients with systemic lupus erythematosus in China
BMC Health Services Research (2019)
-
Early cerebral volume reductions and their associations with reduced lupus disease activity in patients with newly-diagnosed systemic lupus erythematosus
Scientific Reports (2016)
-
Treat to target in systemic lupus erythematosus: a commentary
Clinical Rheumatology (2016)
-
Functional MRI in SLE—the current state
Nature Reviews Rheumatology (2015)