Key Points
-
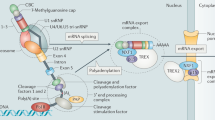
Dicer is a type III cytoplasmic endoribonuclease that is involved in the maturation of several classes of small non-coding RNAs, such as microRNAs.
-
Dicer is involved in the maturation of precursor microRNAs, it loads the derived small RNAs into effector Argonaute proteins, and functions as a scaffold for several interactions in the RNA interference mechanisms.
-
Genetic perturbation of Dicer results in defects affecting functions ranging from embryogenesis, differentiation and homeostasis, to cancer.
-
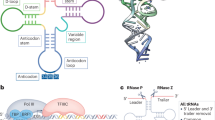
Dicer adopts an overall 'L' shape. The PAZ and Platform domains bind double-stranded RNA (dsRNA) substrate termini, the RNase III domains each cleave along the dsRNA strands, a spacer linker measures the small RNA end products, and an RNA helicase regulates its substrate recognition and activity.
-
Changes in the expression of DICER1 have been associated with various cancers. In general, reduced expression is observed in many cancers, but the data are inconsistent.
-
Germline loss-of-function mutations in DICER1 are associated with a pleiotropic tumour susceptibility syndrome, characterized by pleuropulmonary blastoma, cystic nephroma and other rare entities. Notably, the non-neoplastic condition multinodular goiter seems to feature prominently.
-
In contrast to the loss-of-function mutations, highly specific missense mutations are found as 'second hits' in the tumours that are associated with germline mutations. These missense mutations are almost entirely limited to the RNase IIIb domain of DICER1.
-
Loss-of-heterozygosity of DICER1 is rare and only seems to be frequently associated with a germline DICER1 mutation in the uncommon brain tumour pineoblastoma.
-
Rare phenotypes, such as pituitary blastoma, may be particularly indicative of biallelic mutations in DICER1.
Abstract
Dicer is central to microRNA-mediated silencing and several other RNA interference phenomena that are profoundly embedded in cancer gene networks. Most recently, both germline and somatic mutations in DICER1 have been identified in diverse types of cancer. Although some of the mutations clearly reduce the dosage of this key enzyme, others dictate surprisingly specific changes in select classes of small RNAs. This Review reflects on the molecular properties of the Dicer enzymes in small RNA silencing pathways, and rationalizes the newly discovered mutations on the basis of the activities and functions of its determinants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
10 September 2014
The copyright permission line in Figure 1c was corrected to include the copyright credit text requested by the publisher.
References
Bernstein, E., Caudy, A. A., Hammond, S. M. & Hannon, G. J. Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature 409, 363–366 (2001). This paper reports the discovery of Dicer in mammalian cells. This paper was co-published with another paper that described the discovery of Dicer in several animal models, including C. elegans and Drosophila melanogaster (not cited here).
Grishok, A. et al. Genes and mechanisms related to RNA interference regulate expression of the small temporal RNAs that control C. elegans developmental timing. Cell 106, 23–34 (2001).
Hutvágner, G. et al. A cellular function for the RNA-interference enzyme Dicer in the maturation of the let-7 small temporal RNA. Science 293, 834–838 (2001).
Ketting, R. F. et al. Dicer functions in RNA interference and in synthesis of small RNA involved in developmental timing in C. elegans. Genes Dev. 15, 2654–2659 (2001).
Knight, S. W. & Bass, B. L. A role for the RNase III enzyme DCR-1 in RNA interference and germ line development in Caenorhabditis elegans. Science 293, 2269–2271 (2001).
Zamore, P. D., Tuschl, T., Sharp, P. A. & Bartel, D. P. RNAi: double-stranded RNA directs the ATP-dependent cleavage of mRNA at 21 to 23 nucleotide intervals. Cell 101, 25–33 (2000).
Elbashir, S. M., Lendeckel, W. & Tuschl, T. RNA interference is mediated by 21- and 22-nucleotide RNAs. Genes Dev. 15, 188–200 (2001).
Maniataki, E. & Mourelatos, Z. A human, ATP-independent, RISC assembly machine fueled by pre-miRNA. Genes Dev. 19, 2979–2990 (2005).
Pham, J. W. & Sontheimer, E. J. Molecular requirements for RNA-induced silencing complex assembly in the Drosophila RNA interference pathway. J. Biol. Chem. 280, 39278–39283 (2005).
Lee, Y. S. et al. Distinct roles for Drosophila Dicer-1 and Dicer-2 in the siRNA/miRNA silencing pathways. Cell 117, 69–81 (2004).
Duchaine, T. F. et al. Functional proteomics reveals the biochemical niche of C. elegans DCR-1 in multiple small-RNA-mediated pathways. Cell 124, 343–354 (2006).
Ha, M. & Kim, V. N. Regulation of microRNA biogenesis. Nature Rev. Mol. Cell Biol. 15, 509–524 (2014).
Gregory, R. I. et al. The microprocessor complex mediates the genesis of microRNAs. Nature 432, 235–240 (2004).
Han, J. et al. The Drosha-DGCR8 complex in primary microRNA processing. Genes Dev. 18, 3016–3027 (2004).
Landthaler, M., Yalcin, A. & Tuschl, T. The human DiGeorge syndrome critical region gene 8 and Its D. melanogaster homolog are required for miRNA biogenesis. Curr. Biol. 14, 2162–2167 (2004).
Lee, Y. et al. The nuclear RNase III Drosha initiates microRNA processing. Nature 425, 415–419 (2003).
Yi, R., Qin, Y., Macara, I. G. & Cullen, B. R. Exportin-5 mediates the nuclear export of pre-microRNAs and short hairpin RNAs. Genes Dev. 17, 3011–3016 (2003).
Bohnsack, M. T., Czaplinski, K. & Gorlich, D. Exportin 5 is a RanGTP-dependent dsRNA-binding protein that mediates nuclear export of pre-miRNAs. RNA 10, 185–191 (2004).
Lund, E., Guttinger, S., Calado, A., Dahlberg, J. E. & Kutay, U. Nuclear export of microRNA precursors. Science 303, 95–98 (2004).
Gregory, R. I., Chendrimada, T. P., Cooch, N. & Shiekhattar, R. Human RISC couples microRNA biogenesis and posttranscriptional gene silencing. Cell 123, 631–640 (2005).
Wilson, R. C. & Doudna, J. A. Molecular mechanisms of RNA interference. Annu. Rev. Biophys. 42, 217–239 (2013).
Fabian, M. R. & Sonenberg, N. The mechanics of miRNA-mediated gene silencing: a look under the hood of miRISC. Nature Struct. Mol. Biol. 19, 586–593 (2012).
Flamand, M. & Duchaine, T. F. Snapshot: endogenous RNAi pathways. Cell 150, 442–442 e1 (2012); erratum 150, 867 (2012).
Okamura, K. & Lai, E. C. Endogenous small interfering RNAs in animals. Nature Rev. Mol. Cell Biol. 9, 673–678 (2008).
Reinhart, B. J. & Bartel, D. P. Small RNAs correspond to centromere heterochromatic repeats. Science 297, 1831 (2002).
Murchison, E. P., Partridge, J. F., Tam, O. H., Cheloufi, S. & Hannon, G. J. Characterization of Dicer-deficient murine embryonic stem cells. Proc. Natl Acad. Sci. USA 102, 12135–12140 (2005).
Fukagawa, T. et al. Dicer is essential for formation of the heterochromatin structure in vertebrate cells. Nature Cell Biol. 6, 784–791 (2004).
White, S. A. & Allshire, R. C. Loss of Dicer fowls up centromeres. Nature Cell Biol. 6, 696–697 (2004).
Hu, Q. et al. DICER- and AGO3-dependent generation of retinoic acid-induced DR2 Alu RNAs regulates human stem cell proliferation. Nature Struct. Mol. Biol. 19, 1168–1175 (2012).
Wang, H. W. et al. Structural insights into RNA processing by the human RISC-loading complex. Nature Struct. Mol. Biol. 16, 1148–1153 (2009).
Lau, P. W. et al. The molecular architecture of human Dicer. Nature Struct. Mol. Biol. 19, 436–440 (2012).
Macrae, I. J. et al. Structural basis for double-stranded RNA processing by Dicer. Science 311, 195–198 (2006). This is a key paper describing how Dicer recognizes its substrates and 'measures' the size of small RNA products. It describes the first multi-domain crystal of Dicer, including the PAZ and RNase III domains.
Tian, Y. et al. A phosphate-binding pocket within the Platform-PAZ-connector helix cassette of human dicer. Mol. Cell 53, 606–616 (2014).
Zhang, H., Kolb, F. A., Jaskiewicz, L., Westhof, E. & Filipowicz, W. Single processing center models for human Dicer & bacterial R.Nase III. Cell 118, 57–68 (2004). This insightful paper established the role of the conserved intramolecular RNase III domain dimer of the catalytic core of Dicer.
Welker, N. C. et al. Dicer's helicase domain discriminates dsRNA termini to promote an altered reaction mode. Mol. Cell 41, 589–599 (2011).
Soifer, H. S. et al. A role for the Dicer helicase domain in the processing of thermodynamically unstable hairpin RNAs. Nucleic Acids Res. 36, 6511–6522 (2008).
Ma, E., MacRae, I. J., Kirsch, J. F. & Doudna, J. A. Autoinhibition of human dicer by its internal helicase domain. J. Mol. Biol. 380, 237–243 (2008).
Pavelec, D. M., Lachowiec, J., Duchaine, T. F., Smith, H. E. & Kennedy, S. Requirement for the ERI/DICER complex in endogenous RNA interference and sperm development in Caenorhabditis elegans. Genetics 183, 1283–1295 (2009).
Park, J. E. et al. Dicer recognizes the 5′ end of RNA for efficient and accurate processing. Nature 475, 201–205 (2011).
Haase, A. D. et al. TRBP, a regulator of cellular PKR and HIV-1 virus expression, interacts with Dicer and functions in RNA silencing. EMBO Rep. 6, 961–967 (2005).
Noland, C. L. & Doudna, J. A. Multiple sensors ensure guide strand selection in human RNAi pathways. RNA 19, 639–648 (2013).
Lee, H. Y., Zhou, K., Smith, A. M., Noland, C. L. & Doudna, J. A. Differential roles of human Dicer-binding proteins TRBP and PACT in small RNA processing. Nucleic Acids Res. 41, 6568–6576 (2013).
Karube, Y. et al. Reduced expression of Dicer associated with poor prognosis in lung cancer patients. Cancer Sci. 96, 111–115 (2005).
Merritt, W. M. et al. Dicer, Drosha, and outcomes in patients with ovarian cancer. N. Engl. J. Med. 359, 2641–2650 (2008).
Pampalakis, G., Diamandis, E. P., Katsaros, D. & Sotiropoulou, G. Down-regulation of dicer expression in ovarian cancer tissues. Clin. Biochem. 43, 324–327 (2010).
Sand, M. et al. Expression levels of the microRNA processing enzymes Drosha and dicer in epithelial skin cancer. Cancer Invest. 28, 649–653 (2010).
Dedes, K. J. et al. Down-regulation of the miRNA master regulators Drosha and Dicer is associated with specific subgroups of breast cancer. Eur. J. Cancer 47, 138–150 (2011).
Chiosea, S. et al. Up-regulation of dicer, a component of the MicroRNA machinery, in prostate adenocarcinoma. Am. J. Pathol. 169, 1812–1820 (2006).
Muralidhar, B. et al. Global microRNA profiles in cervical squamous cell carcinoma depend on Drosha expression levels. J. Pathol. 212, 368–377 (2007).
Sugito, N. et al. RNASEN regulates cell proliferation and affects survival in esophageal cancer patients. Clin. Cancer Res. 12, 7322–7328 (2006).
Chiosea, S. et al. Overexpression of Dicer in precursor lesions of lung adenocarcinoma. Cancer Res. 67, 2345–2350 (2007).
Jakymiw, A. et al. Overexpression of dicer as a result of reduced let-7 MicroRNA levels contributes to increased cell proliferation of oral cancer cells. Genes Chromosomes Cancer 49, 549–559 (2010).
Martin, M. G., Payton, J. E. & Link, D. C. Dicer and outcomes in patients with acute myeloid leukemia (AML). Leuk. Res. 33, e127 (2009).
Melo, S. A. et al. A TARBP2 mutation in human cancer impairs microRNA processing and DICER1 function. Nature Genet. 41, 365–370 (2009).
Melo, S. A. et al. A genetic defect in exportin-5 traps precursor microRNAs in the nucleus of cancer cells. Cancer Cell 18, 303–315 (2010).
Hill, D. A. et al. DICER1 mutations in familial pleuropulmonary blastoma. Science 325, 965 (2009). This is the definitive paper establishing heterozygous germline DICER1 mutations as the molecular defect underlying familial disease involving PPB and related conditions. It was the culmination of years of kindred research by the International Pleuropulmonary Blastoma Registry (see Further information).
Priest, J. R. et al. Pleuropulmonary blastoma: a marker for familial disease. J. Pediatr. 128, 220–224 (1996).
Cross, S. F. et al. Familial pleuropulmonary blastoma in Australia. Pediatr. Blood Cancer 55, 1417–1419 (2010).
Priest, J. R., Williams, G. M., Hill, D. A., Dehner, L. P. & Jaffe, A. Pulmonary cysts in early childhood and the risk of malignancy. Pediatr. Pulmonol. 44, 14–30 (2009).
Delahunt, B. et al. Familial cystic nephroma and pleuropulmonary blastoma. Cancer 71, 1338–1342 (1993).
Boman, F. et al. Familial association of pleuropulmonary blastoma with cystic nephroma and other renal tumors: a report from the International Pleuropulmonary Blastoma Registry. J. Pediatr. 149, 850–854 (2006).
Priest, J. R., Williams, G. M., Mize, W. A., Dehner, L. P. & McDermott, M. B. Nasal chondromesenchymal hamartoma in children with pleuropulmonary blastoma—a report from the International Pleuropulmonary Blastoma Registry registry. Int. J. Pediatr. Otorhinolaryngol. 74, 1240–1244 (2010).
Priest, J. R. et al. Ciliary body medulloepithelioma: four cases associated with pleuropulmonary blastoma—a report from the International Pleuropulmonary Blastoma Registry. Br. J. Ophthalmol. 95, 1001–1005 (2011).
Slade, I. et al. DICER1 syndrome: clarifying the diagnosis, clinical features and management implications of a pleiotropic tumour predisposition syndrome. J. Med. Genet. 48, 273–278 (2011). This paper represents the first broad search for DICER1 mutations in DNA from 823 childhood cancer patients and from 781 cancer cell lines. It confirmed and extended the original report and the large number of specimens evaluated helped to define which tumours are not typically associated with DICER1 mutations.
Foulkes, W. D. et al. Extending the phenotypes associated with DICER1 mutations. Hum. Mutat. 32, 1381–1384 (2011).
de Kock, L. et al. Exploring the association between DICER1 mutations and differentiated thyroid carcinoma. J. Clin. Endocrinol. Metab. 99, E1072–E1077 (2014).
Rio Frio, T. et al. DICER1 mutations in familial multinodular goiter with and without ovarian Sertoli-Leydig cell tumors. JAMA 305, 68–77 (2011).
Schultz, K. A. et al. Ovarian sex cord-stromal tumors, pleuropulmonary blastoma and DICER1 mutations: a report from the International Pleuropulmonary Blastoma Registry. Gynecol. Oncol. 122, 246–250 (2011).
Doros, L. A. et al. DICER1 mutations in childhood cystic nephroma and its relationship to DICER1-renal sarcoma. Mod. Pathol. http://dx.doi.org/10.1038/modpathol.2013.242 (2014).
Bahubeshi, A. et al. Germline DICER1 mutations and familial cystic nephroma. J. Med. Genet. 47, 863–866 (2010).
Doros, L. et al. DICER1 mutations in embryonal rhabdomyosarcomas from children with and without familial PPB-tumor predisposition syndrome. Pediatr. Blood Cancer 59, 558–560 (2012).
de Kock, L. et al. Pituitary blastoma: a pathognomonic feature of germ-line DICER1 mutations. Acta Neuropathol. 128, 111–122 (2014). This paper established germline DICER1 mutations as the major and possibly sole genetic cause of the rare paediatric brain tumour pituitary blastoma.
Sabbaghian, N. et al. Germline DICER1 mutation and associated loss of heterozygosity in a pineoblastoma. J. Med. Genet. 49, 417–419 (2012).
Seki, M. et al. Biallelic DICER1 mutations in sporadic pleuropulmonary blastoma. Cancer Res. 74, 2742–2749 (2014).
Pugh, T. J. et al. Exome sequencing of pleuropulmonary blastoma reveals frequent biallelic loss of TP53 and two hits in DICER1 resulting in retention of 5p-derived miRNA hairpin loop sequences. Oncogene http://dx.doi.org/10.1038/onc.2014.150 (2014).
Schultz, K. A. et al. Judicious DICER1 testing and surveillance imaging facilitates early diagnosis and cure of pleuropulmonary blastoma. Pediatr. Blood Cancer 61, 1695–1697 (2014).
Sabapathy, D. G. et al. Radiographic screening of infants and young children with genetic predisposition for rare malignancies: The case of DICER1 mutations and pleuropulmonary blastoma. Am. J. Roentg. (in the press).
Vogelstein, B. et al. Cancer genome landscapes. Science 339, 1546–1558 (2013).
Wu, M. K. et al. Biallelic DICER1 mutations occur in Wilms tumours. J. Pathol. 230, 154–164 (2013).
Heravi-Moussavi, A. et al. Recurrent somatic DICER1 mutations in nonepithelial ovarian cancers. N. Engl. J. Med. 366, 234–242 (2012). This paper first identified somatic DICER1 mutations affecting metal-ion-binding RNase IIIb residues in DICER1 protein in unselected stromal cell ovarian tumours, particularly in ovarian SLCTs. The authors coined the term 'hotspot' for these crucial, recurrent mutations.
de Kock, L. et al. Germ-line and somatic DICER1 mutations in pineoblastoma. Acta Neuropathol. http://dx.doi.org/10.1007/s00401-014-1318-7 (2014).
Crabtree, M. et al. Refining the relation between 'first hits' and 'second hits' at the APC locus: the 'loose fit' model and evidence for differences in somatic mutation spectra among patients. Oncogene 22, 4257–4265 (2003).
Klein, S. et al. Expanding the phenotype of mutations in DICER1: mosaic missense mutations in the RNase IIIb domain of DICER1 cause GLOW syndrome. J. Med. Genet. 51, 294–302 (2014).
Anglesio, M. et al. Cancer-associated somatic DICER1 hotspot mutations cause defective miRNA processing and reverse strand expression bias to predominantly mature 3p strands through loss of 5p strand cleavage. J. Pathol. 229, 400–409 (2012).
Gurtan, A. M., Lu, V., Bhutkar, A. & Sharp, P. A. In vivo structure-function analysis of human Dicer reveals directional processing of precursor miRNAs. RNA 18, 1116–1122 (2012).
Torrezan, G. T. et al. Recurrent somatic mutation in DROSHA induces microRNA profile changes in Wilms tumour. Nature Commun. 5, 4039 (2014).
Murray, M. J. et al. Serum levels of mature microRNAs in DICER1-mutated pleuropulmonary blastoma. Oncogenesis. 3, e87 (2014).
Aksoy, B. A. et al. Cancer-associated recurrent mutations in RNase III domains of DICER1. bioRxiv β http://dx.doi.org/10.1101/005686 (2014).
Lu, J. et al. MicroRNA expression profiles classify human cancers. Nature 435, 834–838 (2005).
Yu, F. et al. let-7 regulates self renewal and tumorigenicity of breast cancer cells. Cell 131, 1109–1123 (2007).
Kumar, M. S., Lu, J., Mercer, K. L., Golub, T. R. & Jacks, T. Impaired microRNA processing enhances cellular transformation and tumorigenesis. Nature Genet. 39, 673–677 (2007).
Gaur, A. et al. Characterization of microRNA expression levels and their biological correlates in human cancer cell lines. Cancer Res. 67, 2456–2468 (2007).
Kumar, M. S. et al. Dicer1 functions as a haploinsufficient tumor suppressor. Genes Dev. 23, 2700–2704 (2009).
Zhang, B. et al. A dosage-dependent pleiotropic role of Dicer in prostate cancer growth and metastasis. Oncogene 33, 3099–3108 (2014).
Nittner, D. et al. Synthetic lethality between Rb, 53 and Dicer or miR-17-92 in retinal progenitors suppresses retinoblastoma formation. Nature Cell Biol. 14, 958–965 (2012).
Lopez-Serra, P. & Esteller, M. DNA methylation-associated silencing of tumor-suppressor microRNAs in cancer. Oncogene 31, 1609–1622 (2012).
Zhang, L. et al. Dicer ablation impairs prostate stem cell activity and causes prostate atrophy. Stem Cells 28, 1260–1269 (2010).
Iliou, M. S. et al. Impaired DICER1 function promotes stemness and metastasis in colon cancer. Oncogene 33, 4003–4015 (2014).
Jensen, R. D., Norris, H. J. & Fraumeni, J. F. Jr. Familial arrhenoblastoma and thyroid adenoma. Cancer 33, 218–223 (1974).
Rath, S. et al. Multinodular goiter in children: an important pointer to a germline DICER1 mutation. J. Clin. Endocrinol. Metab. 99, 1947–1948 (2014).
Dehner, L. P. Pleuropulmonary blastoma is THE pulmonary blastoma of childhood. Semin. Diagn. Pathol. 11, 144–151 (1994).
Allan, B. T., Day, D. L. & Dehner, L. P. Primary pulmonary rhabdomyosarcoma of the lung in children. Report of two cases presenting with spontaneous pneumothorax. Cancer 59, 1005–1011 (1987).
McDermott, M. B., Ponder, T. B. & Dehner, L. P. Nasal chondromesenchymal hamartoma: an upper respiratory tract analogue of the chest wall mesenchymal hamartoma. Am. J. Surg. Pathol. 22, 425–433 (1998).
Ramasubramanian, A., Correa, Z. M., Augsburger, J. J., Sisk, R. A. & Plager, D. A. Medulloepithelioma in DICER1 syndrome treated with resection. Eye 27, 896–897 (2013).
Priest, J. R. et al. Cerebral metastasis and other central nervous system complications of pleuropulmonary blastoma. Pediatr. Blood Cancer 49, 266–273 (2007).
Scheithauer, B. W. et al. Pituitary blastoma. Acta Neuropathol. 116, 657–666 (2008).
Wildi-Runge, S. et al. New phenotype in the familial DICER1 tumor syndrome: Pituitary blastoma presenting at age 9 months. Endocr. Rev. 32, 1–777 (2011).
Scheithauer, B. W. et al. Pituitary blastoma: a unique embryonal tumor. Pituitary. 15, 365–373 (2012).
Pugh, T. J. et al. Medulloblastoma exome sequencing uncovers subtype-specific somatic mutations. Nature 488, 106–110 (2012).
Shaheen, I. S. et al. Bilateral progressive cystic nephroma in a 9-month-old male infant requiring renal replacement therapy. Pediatr. Nephrol. 25, 1755–1758 (2010).
Pasini, B. et al. Unusual familial aggregation of cervical sarcoma botyroides, thyroid goiter and possibly ovarian Sertoli-Leydig cell tumour: a new syndrome? Hormones 7 (suppl. 2) [online], (2008).
Lallier, M. et al. Pleuropulmonary blastoma: a rare pathology with an even rarer presentation. J. Pediatr. Surg. 34, 1057–1059 (1999).
Nur, S., Badr, R., Sandoval, C., Brudniki, A. & Yeh, A. Syndromic presentation of a pleuropulmonary blastoma associated with congenital cystic adenomatoid malformation. A case report. J. Pediatr. Surg. 42, 1772–1775 (2007).
Lucia-Casadonte, C. et al. An unusual case of pleuropulmonary blastoma in a child with jejunal hamartomas. Case Rep. Pediatr. 2013, 140508 (2013).
Miniati, D. N. et al. Prenatal presentation and outcome of children with pleuropulmonary blastoma. J. Pediatr. Surg. 41, 66–71 (2006).
Hill, D. A. et al. Type I pleuropulmonary blastoma: pathology and biology study of 51 cases from the international pleuropulmonary blastoma registry. Am. J. Surg. Pathol. 32, 282–295 (2008).
Priest, J. R. et al. Pleuropulmonary blastoma: a clinicopathologic study of 50 cases. Cancer 80, 147–161 (1997).
Vujanic, G. M., Kelsey, A., Perlman, E. J., Sandstedt, B. & Beckwith, J. B. Anaplastic sarcoma of the kidney: a clinicopathologic study of 20 cases of a new entity with polyphenotypic features. Am. J. Surg. Pathol. 31, 1459–1468 (2007).
Lashkari, H. P. et al. Treatment of high risk Sertoli-Leydig cell tumors of the ovary using a gonadotropin releasing hormone (GnRH) analog. Pediatr. Blood Cancer 60, E16–E18 (2013).
Acknowledgements
The authors sincerely thank L. de Kock, N. Hamel, A. Sawh and B. Rivera for helping to compile the data shown in Figure 1a and Supplementary information S3 (figure), as well as for their artistic and conceptual contributions to Figure 1a–c. Work carried out in the authors' laboratories described in this Review has been supported by Alex's Lemonade Stand Foundation, the Canadian Cancer Society Research Institute, the Jewish General Hospital Foundation (to W.D.F), the Canadian Institute of Health Research (CIHR) MOP 86577 and the Chercheur-Boursier Salary Award J2 (to T.F.D.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Supplementary information
Supplementary information S1 (table)
Frequency of proven germ-line DICER1 mutations in individuals with various tumors (PDF 141 kb)
Supplementary information S2 (table)
Frequency of presumed somatic DICER1 mutations in various tumors (PDF 162 kb)
Supplementary information S3 (figure)
Germline and somatic mutations in DICER1 (including mutations not confirmed to be somatic in origin) Compare to Fig 1A in main paper, which indicates sites only of mutations proven to be somatic. (PDF 1151 kb)
Glossary
- Small RNAs
-
Includes all classes of small non-coding RNAs, small interfering RNAs (siRNAs), endogenous siRNAs (endo-siRNAs), microRNAs, and PIWI-interacting RNAs (piRNAs).
- MicroRNAs
-
(miRNAs). Small RNAs that are derived from genome-encoded hairpins, acting in trans on mRNA translation and stability. Canonical biogenesis involves Dicer, although alternative pathways have been described.
- Long dsRNA substrates
-
A generic term that is used to describe exogenous Dicer substrates such as virus RNA genomes, or experimentally-introduced double-stranded RNA (dsRNA).
- Small interfering RNAs
-
(siRNAs). Small RNAs that are derived from long double-stranded substrates, usually involve full base-pairing with mRNA targets and a mode of action through Argonaute slicer activity. siRNAs can be genome-encoded (endogenous siRNAs) or from exogenous sources (viruses or experimental use of siRNA).
- Endogenous siRNAs
-
(Endo-siRNAs). Small interfering RNAs (siRNAs) that are derived from endogenous genomic loci. This term is often used to describe small RNAs derived from repetitive and transposable elements. Endo-siRNAs can function in cis (on the locus encoding the trigger itself) or in trans by targeting repeated genomic sequences.
- Pulmonary mesenchyme
-
The respiratory epithelium of the lung is surrounded by mesenchyme, derived from the mesoderm embryonic layer and from which several cell types can arise: smooth muscle, pericytes, lipocytes and fibroblasts. It is from mesenchyme that DICER1-related pleuropulmonary blastoma (PPB) is believed to arise.
- Cystic nephroma
-
These tumours are comprised of liquid-filled cysts, often multi-locular in nature. They do not contain any renal parenchyma and are lined by a simple epithelium that has a so-called 'hobnail' appearance. The stroma usually has a spindle-cell morphology. Although they can overtake functioning renal tissue, malignant transformation (to sarcoma) is rare but has been observed in DICER1 mutation carriers.
- Dysontogenic
-
A process by which embryonic differentiation is perturbed but does not result in neoplasia.
- Pulmonary sequestration
-
Occurs when a section of the developing lung is not connected to the respiratory (bronchial) tree. There are two types — intralobar and extralobar, depending on whether the sequestered lobes are within or external to the main, functioning part of the lung. The blood supply for both types of sequestration is separate to the normal lung. Importantly, 50% of pulmonary sequestrations are accompanied by cystic dysplasias that are distinct from the cysts of pleuropulmonary blastoma (PPB).
- Botryoides
-
This descriptive term is from the Greek, meaning 'taking the form of a bunch of grapes'; they are sarcomatous multi-polyp clusters occurring in hollow organs lined by mucus membrane, such as sinuses, nasopharynx, pulmonary cysts, bladder, cervix, vagina and uterus.
- Phthisis bulbi
-
Phthisis is a Latin word, derived from the Greek phthinein (to waste away). Bulbi refers to the globe of the eye. This term is a general one, applied to any destructive process resulting in a non-functional and usually shrunken eye. When it is present at birth, it is referred to as congenital phthisis bulbi.
Rights and permissions
About this article
Cite this article
Foulkes, W., Priest, J. & Duchaine, T. DICER1: mutations, microRNAs and mechanisms. Nat Rev Cancer 14, 662–672 (2014). https://doi.org/10.1038/nrc3802
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc3802
This article is cited by
-
Poorly differentiated thyroid carcinomas: conceptual controversy and clinical impact
Virchows Archiv (2024)
-
Familial syndromes associated with testicular and paratesticular neoplasms: a comprehensive review
Virchows Archiv (2024)
-
Atrophic changes in thyroid tumors are strong indicators of underlying DICER1 mutations: a bi-institutional genotype–phenotype correlation study
Virchows Archiv (2024)
-
DICER1 Mutations Do Not Always Indicate Dismal Prognosis in Pediatric Poorly Differentiated Thyroid Carcinomas
Endocrine Pathology (2023)
-
An update on, and genetics of refractory adenomas of childhood
Pituitary (2023)