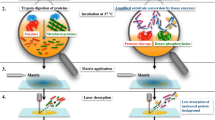
Abstract
Zymography is a technique for studying hydrolytic enzymes on the basis of substrate degradation. It is a powerful, but often misinterpreted, tool yielding information on potential hydrolytic activities, enzyme forms and the locations of active enzymes. In this Review, zymography techniques are compared in terms of advantages, limitations and interpretations. With in gel zymography, enzyme forms are visualized according to their molecular weights. Proteolytic activities are localized in tissue sections with in situ zymography. In vivo zymography can pinpoint proteolytic activity to sites in an intact organism. Future development of novel substrate probes and improvement in detection and imaging methods will increase the applicability of zymography for (reverse) degradomics studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gross, J. & Lapière, C.M. Collagenolytic activity in amphibian tissues: a tissue culture assay. Proc. Natl. Acad. Sci. USA 48, 1014–1022 (1962)This was the first study of collagenolysis in tissue, which resulted in the first description of a matrix metalloproteinase (MMP-1).
Granelli-Piperno, A. & Reich, E. A study of proteases and protease-inhibitor complexes in biological fluids. J. Exp. Med. 148, 223–234 (1978).
Heussen, C. & Dowdle, E.B. Electrophoretic analysis of plasminogen activators in polyacrylamide gels containing sodium dodecyl sulfate and copolymerized substrates. Anal. Biochem. 102, 196–202 (1980).
Hibbs, M.S., Hasty, K.A., Seyer, J.M., Kang, A.H. & Mainardi, C.L. Biochemical and immunological characterization of the secreted forms of human neutrophil gelatinase. J. Biol. Chem. 260, 2493–2500 (1985). One of the first gelatin in gel zymography methods, in which a single gel system was used.
Masure, S., Billiau, A., Van Damme, J. & Opdenakker, G. Human hepatoma cells produce an 85 kDa gelatinase regulated by phorbol 12-myristate 13-acetate. Biochim. Biophys. Acta 1054, 317–325 (1990).
Masure, S., Proost, P., Van Damme, J. & Opdenakker, G. Purification and identification of 91-kDa neutrophil gelatinase. Release by the activating peptide interleukin-8. Eur. J. Biochem. 198, 391–398 (1991).
Houde, M. et al. Differential regulation of gelatinase B and tissue-type plasminogen activator expression in human Bowes melanoma cells. Int. J. Cancer 53, 395–400 (1993).
Paemen, L. et al. The gelatinase inhibitory activity of tetracyclines and chemically modified tetracycline analogues as measured by a novel microtiter assay for inhibitors. Biochem. Pharmacol. 52, 105–111 (1996).
Van den Steen, P.E. et al. Biochemistry and molecular biology of gelatinase B or matrix metalloproteinase-9 (MMP-9). Crit. Rev. Biochem. Mol. Biol. 37, 375–536 (2002).
Galis, Z.S., Sukhova, G.K., Lark, M.W. & Libby, P. Increased expression of matrix metalloproteinases and matrix degrading activity in vulnerable regions of human atherosclerotic plaques. J. Clin. Invest. 94, 2493–2503 (1994).
Galis, Z.S., Sukhova, G.K. & Libby, P. Microscopic localization of active proteases by in situ zymography: detection of matrix metalloproteinase activity in vascular tissue. FASEB J. 9, 974–980 (1995).
Kaijzel, E.L., van der Pluijm, G. & Lowik, C.W. Whole-body optical imaging in animal models to assess cancer development and progression. Clin. Cancer Res. 13, 3490–3497 (2007).
Weissleder, R., Tung, C.H., Mahmood, U. & Bogdanov, A. Jr. In vivo imaging of tumors with protease-activated near-infrared fluorescent probes. Nat. Biotechnol. 17, 375–378 (1999).
Bremer, C., Tung, C.H. & Weissleder, R. In vivo molecular target assessment of matrix metalloproteinase inhibition. Nat. Med. 7, 743–748 (2001).
Crawford, B.D. & Pilgrim, D.B. Ontogeny and regulation of matrix metalloproteinase activity in the zebrafish embryo by in vitro and in vivo zymography. Dev. Biol. 286, 405–414 (2005).
Kleiner, D.E. & Stetler-Stevenson, W.G. Quantitative zymography: detection of picogram quantities of gelatinases. Anal. Biochem. 218, 325–329 (1994).
Kupai, K. et al. Matrix metalloproteinase activity assays: importance of zymography. J. Pharmacol. Toxicol. Methods 61, 205–209 (2010).
Ikeda, M. et al. Inhibition of gelatinolytic activity in tumor tissues by synthetic matrix metalloproteinase inhibitor: application of film in situ zymography. Clin. Cancer Res. 6, 3290–3296 (2000).
Wilkesman, J. & Kurz, L. Protease analysis by zymography: a review on techniques and patents. Recent Pat. Biotechnol. 3, 175–184 (2009).
Vandooren, J. et al. Gelatin degradation assay reveals MMP-9 inhibitors and function of O-glycosylated domain. World J. Biol. Chem. 2, 14–24 (2011).
McKerrow, J.H., Pino-Heiss, S., Lindquist, R. & Werb, Z. Purification and characterization of an elastinolytic proteinase secreted by cercariae of Schistosoma mansoni. J. Biol. Chem. 260, 3703–3707 (1985).
Baragi, V.M. et al. A versatile assay for gelatinases using succinylated gelatin. Matrix Biol. 19, 267–273 (2000).
Lê, J. et al. Quantitative zymography of matrix metalloproteinases by measuring hydroxyproline: application to gelatinases A and B. Electrophoresis 20, 2824–2829 (1999).
Liabakk, N.B., Talbot, I., Smith, R.A., Wilkinson, K. & Balkwill, F. Matrix metalloprotease 2 (MMP-2) and matrix metalloprotease 9 (MMP-9) type IV collagenases in colorectal cancer. Cancer Res. 56, 190–196 (1996).
Curino, A. et al. Detection of plasminogen activators in oral cancer by laser capture microdissection combined with zymography. Oral Oncol. 40, 1026–1032 (2004).
Margulies, I.M. et al. Urinary type IV collagenase: elevated levels are associated with bladder transitional cell carcinoma. Cancer Epidemiol. Biomarkers Prev. 1, 467–474 (1992).
Li, L. et al. Inhibitory effects of GL-V9 on the invasion of human breast carcinoma cells by downregulating the expression and activity of matrix metalloproteinase-2/9. Eur. J. Pharm. Sci. 43, 393–399 (2011).
Wang, Z. et al. Interleukin-lβ induces migration of rat arterial smooth muscle cells through a mechanism involving increased matrix metalloproteinase-2 activity. J. Surg. Res. 169, 328–336 (2011).
Tamura, Y. et al. Profibrinolytic effect of Enzamin, an extract of metabolic products from Bacillus subtilis AK and Lactobacillus. J. Thromb. Thrombolysis 32, 195–200 (2011).
Fontana, V. et al. Consistent alterations of circulating matrix metalloproteinases levels in untreated hypertensives and in spontaneously hypertensive rats: a relevant pharmacological target. Basic Clin. Pharmacol. Toxicol. 109, 130–137 (2011).
Knier, B. et al. Effect of the plasminogen-plasmin system on hypertensive renal and cardiac damage. J. Hypertens. 29, 1602–1612 (2011).
Todorova, L., Bjermer, L., Westergren-Thorsson, G. & Miller-Larsson, A. TGFβ-induced matrix production by bronchial fibroblasts in asthma: budesonide and formoterol effects. Respir. Med. 105, 1296–1307 (2011).
Huang, C.Y. et al. Advanced glycation end products cause collagen II reduction by activating Janus kinase/signal transducer and activator of transcription 3 pathway in porcine chondrocytes. Rheumatology (Oxford) 50, 1379–1389 (2011).
Paemen, L., Olsson, T., Söderström, M., van Damme, J. & Opdenakker, G. Evaluation of gelatinases and IL-6 in the cerebrospinal fluid of patients with optic neuritis, multiple sclerosis and other inflammatory neurological diseases. Eur. J. Neurol. 1, 55–63 (1994).
Rossano, R., Larocca, M. & Riccio, P. 2-D zymographic analysis of Broccoli (Brassica oleracea L. var. Italica) florets proteases: follow up of cysteine protease isotypes in the course of post-harvest senescence. J. Plant Physiol. 168, 1517–1525 (2011).
Geib, S.M., Tien, M. & Hoover, K. Identification of proteins involved in lignocellulose degradation using in gel zymogram analysis combined with mass spectroscopy-based peptide analysis of gut proteins from larval Asian longhorned beetles, Anoplophora glabripennis. Insect Sci. 17, 253–264 (2010).
Velada, I. et al. Expression of genes encoding extracellular matrix macromolecules and metalloproteinases in avian tibial dyschondroplasia. J. Comp. Pathol. 145, 174–186 (2011).
Métayer, S., Dacheux, F., Dacheux, J.L. & Gatti, J.L. Comparison, characterization, and identification of proteases and protease inhibitors in epididymal fluids of domestic mammals. Matrix metalloproteinases are major fluid gelatinases. Biol. Reprod. 66, 1219–1229 (2002).
Kim, S.H., Choi, N.S. & Lee, W.Y. Fibrin zymography: a direct analysis of fibrinolytic enzymes on gels. Anal. Biochem. 263, 115–116 (1998).
Park, S.G. et al. A functional proteomic analysis of secreted fibrinolytic enzymes from Bacillus subtilis 168 using a combined method of two-dimensional gel electrophoresis and zymography. Proteomics 2, 206–211 (2002).
Hasson, S.S., Theakston, R.D. & Harrison, R.A. Antibody zymography: a novel adaptation of zymography to determine the protease-neutralising potential of specific antibodies and snake antivenoms. J. Immunol. Methods 292, 131–139 (2004).
Hughes, A.J. & Herr, A.E. Quantitative enzyme activity determination with zeptomole sensitivity by microfluidic gradient-gel zymography. Anal. Chem. 82, 3803–3811 (2010).
Thimon, V., Belghazi, M., Labas, V., Dacheux, J.L. & Gatti, J.L. One- and two-dimensional SDS-PAGE zymography with quenched fluorogenic substrates provides identification of biological fluid proteases by direct mass spectrometry. Anal. Biochem. 375, 382–384 (2008).
Pan, D., Hill, A.P., Kashou, A., Wilson, K.A. & Tan-Wilson, A. Electrophoretic transfer protein zymography. Anal. Biochem. 411, 277–283 (2011).
Choi, N.S. et al. Mixed-substrate (glycerol tributyrate and fibrin) zymography for simultaneous detection of lipolytic and proteolytic enzymes on a single gel. Electrophoresis 30, 2234–2237 (2009).
Choi, N.S. et al. Multiple-layer substrate zymography for detection of several enzymes in a single sodium dodecyl sulfate gel. Anal. Biochem. 386, 121–122 (2009).
Oliver, G.W., Leferson, J.D., Stetler-Stevenson, W.G. & Kleiner, D.E. Quantitative reverse zymography: analysis of picogram amounts of metalloproteinase inhibitors using gelatinase A and B reverse zymograms. Anal. Biochem. 244, 161–166 (1997).
Yan, S.J. & Blomme, E.A. In situ zymography: a molecular pathology technique to localize endogenous protease activity in tissue sections. Vet. Pathol. 40, 227–236 (2003).
Duran-Vilaregut, J. et al. Role of matrix metalloproteinase-9 (MMP-9) in striatal blood-brain barrier disruption in a 3-nitropropionic acid model of Huntington's disease. Neuropathol. Appl. Neurobiol. 37, 525–537 (2011).
Mungall, B.A. & Pollitt, C.C. In situ zymography: topographical considerations. J. Biochem. Biophys. Methods 47, 169–176 (2001).
Hu, J., Van den Steen, P.E., Sang, Q.X. & Opdenakker, G. Matrix metalloproteinase inhibitors as therapy for inflammatory and vascular diseases. Nat. Rev. Drug Discov. 6, 480–498 (2007).
Van den Steen, P.E. et al. Matrix metalloproteinases, tissue inhibitors of MMPs and TACE in experimental cerebral malaria. Lab. Invest. 86, 873–888 (2006).
Redondo-Muñoz, J. et al. Matrix metalloproteinase-9 promotes chronic lymphocytic leukemia B cell survival through its hemopexin domain. Cancer Cell 17, 160–172 (2010).
Agrawal, S. et al. Dystroglycan is selectively cleaved at the parenchymal basement membrane at sites of leukocyte extravasation in experimental autoimmune encephalomyelitis. J. Exp. Med. 203, 1007–1019 (2006). This paper illustrates the power of combining IGZ, ISZ and IHC.
Louboutin, J.P., Agrawal, L., Reyes, B.A., Van Bockstaele, E.J. & Strayer, D.S. HIV-1 gp120-induced injury to the blood-brain barrier: role of metalloproteinases 2 and 9 and relationship to oxidative stress. J. Neuropathol. Exp. Neurol. 69, 801–816 (2010).
Oh, L.Y. et al. Matrix metalloproteinase-9/gelatinase B is required for process outgrowth by oligodendrocytes. J. Neurosci. 19, 8464–8475 (1999).
Wyatt, R.A. et al. The zebrafish embryo: a powerful model system for investigating matrix remodeling. Zebrafish 6, 347–354 (2009).
Keow, J.Y., Herrmann, K.M. & Crawford, B.D. Differential in vivo zymography: a method for observing matrix metalloproteinase activity in the zebrafish embryo. Matrix Biol. 30, 169–177 (2011).
Scherer, R.L., VanSaun, M.N., McIntyre, J.O. & Matrisian, L.M. Optical imaging of matrix metalloproteinase-7 activity in vivo using a proteolytic nanobeacon. Mol. Imaging 7, 118–131 (2008). An example of how nanoparticles are useful tools for IVZ with the use of near-infrared fluorophores with low tissue absorption.
Saghatelian, A., Jessani, N., Joseph, A., Humphrey, M. & Cravatt, B.F. Activity-based probes for the proteomic profiling of metalloproteases. Proc. Natl. Acad. Sci. USA 101, 10000–10005 (2004).
Keow, J.Y., Pond, E.D., Cisar, J.S., Cravatt, B.F. & Crawford, B.D. Activity-based labeling of matrix metalloproteinases in living vertebrate embryos. PLoS ONE 7, e43434 (2012).
Smith, C. Keeping tabs on fluorescent tags. Nat. Methods 4, 755–761 (2007).
López-Otín, C. & Overall, C.M. Protease degradomics: a new challenge for proteomics. Nat. Rev. Mol. Cell Biol. 3, 509–519 (2002). A critical review of degradomics as a methodology to study the substrate repertoires of single proteases.
Overall, C.M. et al. Protease degradomics: mass spectrometry discovery of protease substrates and the CLIP-CHIP, a dedicated DNA microarray of all human proteases and inhibitors. Biol. Chem. 385, 493–504 (2004).
Schilling, O. & Overall, C.M. Proteome-derived, database-searchable peptide libraries for identifying protease cleavage sites. Nat. Biotechnol. 26, 685–694 (2008).
Dean, R.A. & Overall, C.M. Proteomics discovery of metalloproteinase substrates in the cellular context by iTRAQ labeling reveals a diverse MMP-2 substrate degradome. Mol. Cell Proteomics 6, 611–623 (2007).
Cauwe, B. & Opdenakker, G. Intracellular substrate cleavage: a novel dimension in the biochemistry, biology and pathology of matrix metalloproteinases. Crit. Rev. Biochem. Mol. Biol. 45, 351–423 (2010).
Tsiatsiani, L., Gevaert, K. & Van Breusegem, F. Natural substrates of plant proteases: how can protease degradomics extend our knowledge? Physiol. Plant. 145, 28–40 (2012).
Piccard, H. et al. “Reverse degradomics”, monitoring of proteolytic trimming by multi-CE and confocal detection of fluorescent substrates and reaction products. Electrophoresis 30, 2366–2377 (2009). Definition of reverse degradomics as a substrate-based methodology to study the protease repertoire of single substrates. In fact, substrate zymography is a reverse degradomics method.
Kaberdin, V.R. & McDowall, K.J. Expanding the use of zymography by the chemical linkage of small, defined substrates to the gel matrix. Genome Res. 13, 1961–1965 (2003).
Foltmann, B., Szecsi, P.B. & Tarasova, N.I. Detection of proteases by clotting of casein after gel electrophoresis. Anal. Biochem. 146, 353–360 (1985).
Till, O., Baumann, E. & Linss, W. Zymography with caseogram prints: quantification of pepsinogen. Anal. Biochem. 292, 22–25 (2001).
Kwon, M.A., Kim, H.S., Hahm, D.H. & Song, J.K. Synthesis activity-based zymography for detection of lipases and esterases. Biotechnol. Lett. 33, 741–746 (2011).
Law, B., Hsiao, J.K., Bugge, T.H., Weissleder, R. & Tung, C.H. Optical zymography for specific detection of urokinase plasminogen activator activity in biological samples. Anal. Biochem. 338, 151–158 (2005).
Hattori, S., Fujisaki, H., Kiriyama, T., Yokoyama, T. & Irie, S. Real-time zymography and reverse zymography: a method for detecting activities of matrix metalloproteinases and their inhibitors using FITC-labeled collagen and casein as substrates. Anal. Biochem. 301, 27–34 (2002).
Acknowledgements
The present study was supported by funding from the European Union Seventh Framework Programme (FP7/2007-2013) under grant agreement no. 263307, by “Geconcerteerde OnderzoeksActies” GOA 2012 017 and GOA 2013 014, by the Fund for Scientific Research of Flanders (FWO-Vlaanderen) and by the University of Leuven Research Fund. The authors thank B. Crawford and J. Keow for providing Figure 2c.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1, Supplementary Tables 1–4 and Supplementary Note (PDF 716 kb)
Rights and permissions
About this article
Cite this article
Vandooren, J., Geurts, N., Martens, E. et al. Zymography methods for visualizing hydrolytic enzymes. Nat Methods 10, 211–220 (2013). https://doi.org/10.1038/nmeth.2371
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.2371
This article is cited by
-
Proteases of Acanthamoeba
Parasitology Research (2024)
-
Matrix metalloproteinases in arthritis: towards precision medicine
Nature Reviews Rheumatology (2023)
-
Combretastatin A-4 suppresses the invasive and metastatic behavior of glioma cells and induces apoptosis in them: in-vitro study
Medical Oncology (2023)
-
Purification and characterization of amylases from three freshwater fish species providing new insight application as enzyme molecular markers for zymography
Fish Physiology and Biochemistry (2023)
-
The role of mechanical control of biofilm in the salivary pH after sucrose exposure in children with early childhood caries
Scientific Reports (2021)