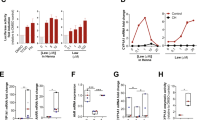
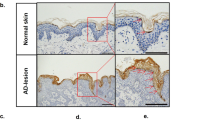
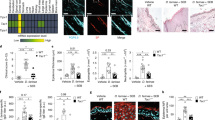
Abstract
Atopic dermatitis is increasing worldwide in correlation with air pollution. Various organic components of pollutants activate the transcription factor AhR (aryl hydrocarbon receptor). Through the use of AhR-CA mice, whose keratinocytes express constitutively active AhR and that develop atopic-dermatitis-like phenotypes, we identified Artn as a keratinocyte-specific AhR target gene whose product (the neurotrophic factor artemin) was responsible for epidermal hyper-innervation that led to hypersensitivity to pruritus. The activation of AhR via air pollutants induced expression of artemin, alloknesis, epidermal hyper-innervation and inflammation. AhR activation and ARTN expression were positively correlated in the epidermis of patients with atopic dermatitis. Thus, AhR in keratinocytes senses environmental stimuli and elicits an atopic-dermatitis pathology. We propose a mechanism of air-pollution-induced atopic dermatitis via activation of AhR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bieber, T. Atopic dermatitis. N. Engl. J. Med. 358, 1483–1494 (2008).
Lewis-Jones, S. Quality of life and childhood atopic dermatitis: The misery of living with childhood eczema. Int. J. Clin. Pract. 60, 984–992 (2006).
Spergel, J.M. & Paller, A.S. Atopic dermatitis and the atopic march. J. Allergy Clin. Immunol. 112, S118–S127 (2003).
Morgenstern, V. et al. Atopic diseases, allergic sensitization, and exposure to traffic-related air pollution in children. Am. J. Respir. Crit. Care Med. 177, 1331–1337 (2008).
Jedrychowski, W. et al. Effects of prenatal and perinatal exposure to fine air pollutants and maternal fish consumption on the occurrence of infantile eczema. Int. Arch. Allergy Immunol. 155, 275–281 (2011).
Song, S. et al. Acute health effects of urban fine and ultrafine particles on children with atopic dermatitis. Environ. Res. 111, 394–399 (2011).
Kim, J. et al. Symptoms of atopic dermatitis are influenced by outdoor air pollution. J. Allergy Clin. Immunol. 132, 495–499 (2013).
Li, R. et al. Ultrafine particles from diesel vehicle emissions at different driving cycles induce differential vascular pro-inflammatory responses: implication of chemical components and NF-κB signaling. Part. Fibre Toxicol. 7, 6 (2010).
Totlandsdal, A.I. et al. Differential effects of the particle core and organic extract of diesel exhaust particles. Toxicol. Lett. 208, 262–268 (2012).
Sanders, C.L., Skinner, C. & Gelman, R.A. Percutaneous absorption of [7.10–14C]benzo[a]pyrene and [7,12–14C]dimethylbenz[a]anthracene in mice. Environ. Res. 33, 353–360 (1984).
Kao, J., Patterson, F.K. & Hall, J. Skin penetration and metabolism of topically applied chemicals in six mammalian species, including man: an in vitro study with benzo[a]pyrene and testosterone. Toxicol. Appl. Pharmacol. 81, 502–516 (1985).
Chu, I., Dick, D., Bronaugh, R. & Tryphonas, L. Skin reservoir formation and bioavailability of dermally administered chemicals in hairless guinea pigs. Food Chem. Toxicol. 34, 267–276 (1996).
Fujii-Kuriyama, Y. & Mimura, J. Molecular mechanisms of AhR functions in the regulation of cytochrome P450 genes. Biochem. Biophys. Res. Commun. 338, 311–317 (2005).
Tauchi, M. et al. Constitutive expression of aryl hydrocarbon receptor in keratinocytes causes inflammatory skin lesions. Mol. Cell. Biol. 25, 9360–9368 (2005).
Van Den Bogaard, E.H. et al. Coal tar induces AHR-dependent skin barrier repair in atopic dermatitis. J. Clin. Invest. 123, 917–927 (2013).
Murota, H. et al. Artemin causes hypersensitivity to warm sensation, mimicking warmth-provoked pruritus in atopic dermatitis. J. Allergy Clin. Immunol. 130, 671–682.e4 (2012).
Kabashima, K. New concept of the pathogenesis of atopic dermatitis: Interplay among the barrier, allergy, and pruritus as a trinity. J. Dermatol. Sci. 70, 3–11 (2013).
Barnes, K.C. An update on the genetics of atopic dermatitis: Scratching the surface in 2009. J. Allergy Clin. Immunol. 125, 16–29.e11 (2010).
Tamari, M. & Hirota, T. Genome-wide association studies of atopic dermatitis. J. Dermatol. 41, 213–220 (2014).
Li, M. et al. Retinoid X receptor ablation in adult mouse keratinocytes generates an atopic dermatitis triggered by thymic stromal lymphopoietin. Proc. Natl. Acad. Sci. USA 102, 14795–14800 (2005).
Yoo, J. et al. Spontaneous atopic dermatitis in mice expressing an inducible thymic stromal lymphopoietin transgene specifically in the skin. J. Exp. Med. 202, 541–549 (2005).
Imai, Y. et al. Skin-specific expression of IL-33 activates group 2 innate lymphoid cells and elicits atopic dermatitis-like inflammation in mice. Proc. Natl. Acad. Sci. USA 110, 13921–13926 (2013).
Ricci, G. et al. Long-term follow-up of atopic dermatitis: Retrospective analysis of related risk factors and association with concomitant allergic diseases. J. Am. Acad. Dermatol. 55, 765–771 (2006).
Hamid, Q. & Tulic, M. Immunobiology of asthma. Annu. Rev. Physiol. 71, 489–507 (2009).
Abramovits, W. A clinician's paradigm in the treatment of atopic dermatitis. J. Am. Acad. Dermatol. 53, S70–S77 (2005).
Hashimoto, Y. et al. Scratching of their skin by NC/Nga mice leads to development of dermatitis. Life Sci. 76, 783–794 (2004).
Ikoma, A. et al. Neurophysiology of pruritus: interaction of itch and pain. Arch. Dermatol. 139, 1475–1478 (2003).
Akiyama, T. et al. Mouse model of touch-evoked itch (alloknesis). J. Invest. Dermatol. 132, 1886–1891 (2012).
Wahlgren, C.F., Hagermark, O. & Bergstrom, R. Patients' perception of itch induced by histamine, compound 48/80 and wool fibres in atopic dermatitis. Acta Derm. Venereol. 71, 488–494 (1991).
Tominaga, M. & Takamori, K. Itch and nerve fibers with special reference to atopic dermatitis: therapeutic implications. J. Dermatol. 41, 205–212 (2014).
Bíró, T. et al. TRP channels as novel players in the pathogenesis and therapy of itch. Biochim. Biophys. Acta 1772, 1004–1021 (2007).
Ross, S.E. Pain and itch: insights into the neural circuits of aversive somatosensation in health and disease. Curr. Opin. Neurobiol. 21, 880–887 (2011).
Tominaga, M., Ozawa, S., Ogawa, H. & Takamori, K. A hypothetical mechanism of intraepidermal neurite formation in NC/Nga mice with atopic dermatitis. J. Dermatol. Sci. 46, 199–210 (2007).
Wei, Y.D., Bergander, L., Rannug, U. & Rannug, A. Regulation of CYP1A1 transcription via the metabolism of the tryptophan-derived 6-formylindolo[3,2-b]carbazole. Arch. Biochem. Biophys. 383, 99–107 (2000).
Wincent, E. et al. The suggested physiologic aryl hydrocarbon receptor activator and cytochrome P4501 substrate 6-formylindolo[3,2-b]carbazole is present in humans. J. Biol. Chem. 284, 2690–2696 (2009).
Kim, H.O., Kim, J.H., Chung, B.Y., Choi, M.G. & Park, C.W. Increased expression of the aryl hydrocarbon receptor in patients with chronic inflammatory skin diseases. Exp. Dermatol. 23, 278–281 (2014).
DiMeglio, P. et al. Activation of the aryl hydrocarbon receptor dampens the severity of inflammatory skin conditions. Immunity 40, 989–1001 (2014).
Kerkvliet, N.I. AHR-mediated immunomodulation: The role of altered gene transcription. Biochem. Pharmacol. 77, 746–760 (2009).
Lo, R. & Matthews, J. High-resolution genome-wide mapping of AHR and ARNT binding sites by ChIP-Seq. Toxicol. Sci. 130, 349–361 (2012).
Frericks, M., Meissner, M. & Esser, C. Microarray analysis of the AHR system: tissue-specific flexibility in signal and target genes. Toxicol. Appl. Pharmacol. 220, 320–332 (2007).
Elitt, C.M. et al. Artemin overexpression in skin enhances expression of TRPV1 and TRPA1 in cutaneous sensory neurons and leads to behavioral sensitivity to heat and cold. J. Neurosci. 26, 8578–8587 (2006).
Malin, S.A. et al. Glial cell line-derived neurotrophic factor family members sensitize nociceptors in vitro and produce thermal hyperalgesia in vivo. J. Neurosci. 26, 8588–8599 (2006).
Yoshida, N. et al. Inhibition of TRPA1 channel activity in sensory neurons by the glial cell line-derived neurotrophic factor family member, artemin. Mol. Pain 7, 41 (2011).
Kopec, A.K. et al. Automated dose-response analysis and comparative toxicogenomic evaluation of the hepatic effects elicited by TCDD, TCDF, and PCB126 in C57bl/6 mice. Toxicol. Sci. 118, 286–297 (2010).
Farmahin, R., Crump, D., O'Brien, J.M., Jones, S.P. & Kennedy, S.W. Time-dependent transcriptomic and biochemical responses of 6-formylindolo[3,2-b]carbazole (FICZ) and 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) are explained by AHR activation time. Biochem. Pharmacol. 115, 134–143 (2016).
Wei, Y.D., Helleberg, H., Rannug, U. & Rannug, A. Rapid and transient induction of CYP1A1 gene expression in human cells by the tryptophan photoproduct 6-formylindolo[3,2- b]carbazole. Chem. Biol. Interact. 110, 39–55 (1998).
Kuriyama, S. et al. The Tohoku Medical Megabank Project: design and mission. J. Epidemiol. 26, 493–511 (2016).
Hong, J., Buddenkotte, J., Berger, T.G. & Steinhoff, M. Management of itch in atopic dermatitis. Semin. Cutan. Med. Surg. 30, 71–86 (2011).
Walisser, J.A., Glover, E., Pande, K., Liss, A.L. & Bradfield, C.A. Aryl hydrocarbon receptor-dependent liver development and hepatotoxicity are mediated by different cell types. Proc. Natl. Acad. Sci. USA 102, 17858–17863 (2005).
Tarutani, M. et al. Tissue-specific knockout of the mouse Pig-a gene reveals important roles for GPI-anchored proteins in skin development. Proc. Natl. Acad. Sci. USA 94, 7400–7405 (1997).
Hennings, H. et al. Calcium regulation of growth and differentiation of mouse epidermal cells in culture. Cell 19, 245–254 (1980).
Kuraishi, Y., Nagasawa, T., Hayashi, K. & Satoh, M. Scratching behavior induced by pruritogenic but not algesiogenic agents in mice. Eur. J. Pharmacol. 275, 229–233 (1995).
Hirotsu, Y. et al. Nrf2-MafG heterodimers contribute globally to antioxidant and metabolic networks. Nucleic Acids Res. 40, 10228–10239 (2012).
Fujita, P.A. et al. The UCSC genome browser database: update 2011. Nucleic Acids Res. 39, 876–882 (2011).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Zhang, Y. et al. Model-based analysis of ChIP-Seq (MACS). Genome Biol. 9, R137 (2008).
Quinlan, A.R. & Hall, I.M. BEDTools: A flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Heo, Y., Saxon, A. & Hankinson, O. Effect of diesel exhaust particles and their components on the allergen-specific IgE and IgG1 response in mice. Toxicology 159, 143–158 (2001).
Thornton, P. et al. Artemin-GFRα3 interactions partially contribute to acute inflammatory hypersensitivity. Neurosci. Lett. 545, 23–28 (2013).
Sagai, M., Saito, H., Ichinose, T., Kodama, M. & Mori, Y. Biological effects of diesel exhaust particles. I. In vitro production of superoxide and in vivo toxicity in mouse. Free Radic. Biol. Med. 14, 37–47 (1993).
Wincent, E. et al. Inhibition of cytochrome P4501-dependent clearance of the endogenous agonist FICZ as a mechanism for activation of the aryl hydrocarbon receptor. Proc. Natl. Acad. Sci. USA 109, 4479–4484 (2012).
Acknowledgements
We thank K. Kuroda for bioinformatics support; M. Nakagawa, M. Kikuchi, M. Tsuda, S. Inomata, A. Gotoh and A. Uchiyama for technical assistance; the Biomedical Research Core of Tohoku University Graduate School of Medicine for technical support; J. Takeda (Osaka University) for Krt5-Cre mice; N. E. Fusenig (Deutsches Krebsforschungszentrum) for HaCaT keratinocytes; S. Ikawa (Tohoku University) for adenoviral vectors; and M. Sagai (Aomori University of Health and Welfare) for DEPs. Supported by the Japan Society for the Promotion of Science (KAKENHI 25840001 and 15K08257 to E.H.K.; 25461688 to E.O.; 22118002, 24249015 and 26111002 to M.Y.; and AMED-CREST (chronic inflammation) to M.Y.), the Mitsubishi Foundation (M.Y.) and the Takeda Science Foundation (M.Y.).
Author information
Authors and Affiliations
Contributions
T.H., E.H.K. and M.Y. designed the study; T.H. performed the experiments involving mice and cells, analyzed the data and wrote the paper; E.O. and R.O. performed ChIP sampling and experiments with human specimens; E.H.K. supervised the study, performed ChIP analyses, analyzed the data and wrote the paper; T.S., T.F. and S.A were involved in the study design; R.F. organized and K.N. supervised the deep sequencing; T.N. performed bioinformatics analyses; and M.Y. supervised the project and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Allergic inflammation in AhR-CA mice.
(a) Gate strategy for assessing allergic inflammatory cells in the skin of AhR-CA mice. CD4+ T cells were gated on CD45+SSClowFSClowCD3+CD4+ cells. Mast cells were gated on CD45+CD3–B220–SSChiFSChiCD117+FcɛRIα+. ILC2s were gated on CD45+SSClowFSClowLin–(CD3–CD5–B220–CD11b–Gr-1–NK1.1–FcɛRIα–) CD25+IL-33Rα+CD127+KLEG1+ cells. Eosinophils were gated on CD45+CD3–B220–SSChiFSClowCD11b+Siglec-F+ cells. Basophils were gated on CD45+CD3–B220–CD117–CD49b+FcɛRIα+ cells. (b) Number of inflammatory cells detected in the skin of 5 AhR-CA mice and that of 5 WT littermates. Dots represent individual mice. (c) Relative expression of barrier-related genes in the epidermis of 5 AhR-CA mice and 5 WT littermates; box-and-whisker plot (line, median; box, 25th to 75th percentiles; bar, minimum to maximum). (d) Relative expression of Tslp, Il33 and Cyp1a1 in AhR-CA overexpressing keratinocytes; each dot represents an independent sampling, n=3. (e) AhR-CA ChIP-qPCR results from 3 independent samplings. (f, g) Immunostaining of the skin for TSLP (f) and IL-33 (g). Dotted line, the epidermis-dermis boundary; scale bar, 50 µm for f and 100 µm for g; representative images of 3 mice each. (h) Intracellular cytokine staining of CD4+ T cells isolated from regional lymph nodes of AhR-CA mice and WT littermates (5 mice each). (i) Dot-plot presentation of the intracellular cytokine staining observed in h. Dots represent individual mice. Data in b, d, and i; lines represent the mean; unpaired t-test; *p <0.05, **p < 0.01 and ***p<0.001.
Supplementary Figure 2 Influence of skin-barrier destruction and cytokine expression on AhR-CA mouse phenotypes.
(a) OVA uptake by MHC class II+ (MHCII+) antigen-presenting cells at the cortex of regional lymph nodes. Scale bar, 20 µm; 3 mice each. (b) Number of OVA+ and OVA+MHCII+ cells observed in a. (c-e) Relative mRNA expressions of Ephb2 (c), Artn (d), and St3gal3 (e). The expressions of Ephb2 and St3gal3 were evaluated in the RT-qPCR analysis of epidermal samples from neonatal AhR-CA mice compared with their expression in samples from wild-type littermates (5 mice each). Artn expression was examined in the skin from WT and AhR-CA mice with or without nail-clipping (6 mice each). (f) Relative expression of AD-related genes at the rostral back after Artemin-neutralization (4 mice each). (g-i) Appearance (g, Scale bar, 10 mm), number of scratching behaviors in 10 min (h) and alloknesis score (i, lines represent the median; Mann-Whitney U test) of 4 Tslp-KO, 5 AhR-CA, and 6 AhR-CATslp-KO compound mice. Data in b-f, and h, dots represent individual mice; lines represent the mean; unpaired t-test; *p < 0.05 and **p < 0.01.
Supplementary Figure 3 Exposure to air pollutants induces expression of Tslp and Il33 in keratinocytes.
(a) Knockout efficiency determined via PCR of the Ahr gene using isolated epidermal samples of Ahrfl/fl mice and Ahrfl/flKrt5-Cre mice (dots represent individual mice, 5 mice each). (b, c) Relative expressions of Tslp, Il33 and Cyp1a1 in mouse primary keratinocytes exposed to different concentrations of benzo[a]pyrene (B[a]P; 3 independent samplings; b) and in 1 µM B[a]P-exposed mouse primary keratinocytes with or without AhR knockdown (3 independent samplings, c). (d) Relative expression of CYP1A1 in HaCaT cells exposed to FICZ (5 independent samplings). (e) Relative expression of TSLP, IL33, FLG and CYP1A1 in differentiated or undifferentiated HEKns exposed to DMBA or FICZ (6 independent samplings). Data in a - d, coral lines represent the mean. Data in e, box-and-whisker plot (line, median; box, 25th to 75th percentiles; bar, minimum to maximum). Data in a-e, unpaired t-test. *p < 0.05, **p < 0.01, ***p < 0.001.
Supplementary Figure 4 Evaluation of CYP1A1 expression and hyper-innervation in human skin.
(a) Representative immunostaining for CYP1A1 in the specimens classified as –, + and ++. Images from the same experiments as in Fig. 7g and Supplementary Table 1. –, negative or positive only in basal layer; +, positive in less than the half of the epidermis; ++, positive in more than the half of the epidermis. Immunostaining for TSLP in the same specimens are presented in the lower panels. Scale bars, 100 µm. (b) Immunostaining for neuronal marker PGP9.5 in skin samples of normal subjects, AD patients, and contact dermatitis patients. Dotted line, the epidermis-dermis boundary; scale bar, 20 µm; representative images of 3 individuals each.
Supplementary Figure 5 Epidermal activation of AhR by air pollutants causes AD-like phenotypes through artemin expression and hyper-innervation into the epidermis.
Schema of AhR-mediated development and the exacerbation of AD. Note that AhR is activated by air pollutants, such as traffic-derived particulate matter. Activated AhR induces the expression of TSLP, IL-33 and ARTEMIN in keratinocytes. ARTEMIN induces hyper-innervation of the epidermis by sensory nerves, resulting in alloknesis. Subsequent scratching behaviors lead to barrier damage and increased antigen penetration, resulting in enhanced sensitization that promotes a predisposition to other allergic diseases.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Table 1 (PDF 1884 kb)
Rights and permissions
About this article
Cite this article
Hidaka, T., Ogawa, E., Kobayashi, E. et al. The aryl hydrocarbon receptor AhR links atopic dermatitis and air pollution via induction of the neurotrophic factor artemin. Nat Immunol 18, 64–73 (2017). https://doi.org/10.1038/ni.3614
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3614
This article is cited by
-
Region-specific activation in the accumbens nucleus by itch with modified scratch efficacy in mice – a model-free multivariate analysis
Molecular Brain (2024)
-
Spatial modeling connecting childhood atopic dermatitis prevalence with household exposure to pollutants
Communications Medicine (2024)
-
The Burden of Air Pollution on Skin Health: a Brief Report and Call to Action
Dermatology and Therapy (2024)
-
Atopic dermatitis: an expanding therapeutic pipeline for a complex disease
Nature Reviews Drug Discovery (2022)
-
Experts’ Consensus on the Use of Pimecrolimus in Atopic Dermatitis in China: A TCS-Sparing Practical Approach
Dermatology and Therapy (2022)