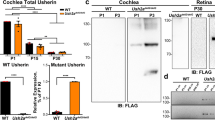
Abstract
Mouse chromosome 10 harbors several loci associated with hearing loss, including waltzer (v), modifier-of deaf waddler (mdfw) and Age-related hearing loss1 (Ahl). The human region that is orthologous to the mouse 'waltzer' region is located at 10q21–q22 and contains the human deafness loci DFNB12 and USH1D (refs. 2,3). Numerous mutations at the waltzer locus have been documented causing erratic circling and hearing loss4,5,6,7. Here we report the identification of a new gene mutated in v. The 10.5-kb Cdh23 cDNA encodes a very large, single-pass transmembrane protein, that we have called otocadherin. It has an extracellular domain that contains 27 repeats; these show significant homology to the cadherin ectodomain. In v6J, a GT transversion creates a premature stop codon. In vAlb, a CT exchange generates an ectopic donor splice site, effecting deletion of 119 nucleotides of exonic sequence. In v2J, a GA transition abolishes the donor splice site, leading to aberrant splice forms. All three alleles are predicted to cause loss of function. We demonstrate Cdh23 expression in the neurosensory epithelium and show that during early hair-cell differentiation, stereocilia organization is disrupted in v2J homozygotes. Our data indicate that otocadherin is a critical component of hair bundle formation. Mutations in human CDH23 cause Usher syndrome type 1D and thus, establish waltzer as the mouse model for USH1D.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
MGD. Mouse Genome Informatics Project (The Jackson Laboratory, Bar Harbor, Maine, 2000).
Chaib, H. et al. Mapping of DFNB12, a gene for a non-syndromal autosomal recessive deafness, to chromosome 10q21–22. Hum. Mol. Genet. 5, 1061–1064 (1996).
Wayne, S. et al. Localization of the Usher syndrome type ID gene (Ush1D) to chromosome 10. Hum. Mol. Genet. 5, 1689– 1692 (1996).
Deol, S.M. A gene for uncomplicated deafness in the mouse. J. Embryol. Exp. Morphol. 4, 190–195 ( 1956).
Deol, M.S. The anatomy and development of the mutants pirouette, shaker-1 and waltzer in the mouse. Proc. Roy. Soc. 145, 206– 213 (1956).
Bryda, E.C., Ling, H. & Flaherty, L. A high-resolution genetic map around waltzer on mouse chromosome 10 and identification of a new allele of waltzer. Mamm. Genome 8, 1–4 ( 1997).
Yonezawa, S. et al. Chromosomal localization of a gene responsible for vestibulocochlear defects of BUS/Idr mice: identification as an allele of waltzer. Hear. Res. 134, 116–122 (1999).
Gumbiner, B.M. Cell adhesion: the molecular basis of tissue architecture and morphogenesis . Cell 84, 345–357 (1996).
Takeichi, M. Morphogenetic roles of classic cadherins. Curr. Opin. Cell Biol. 7, 619–627 ( 1995).
Yagi, T. & Takeichi, M. Cadherin superfamily genes: functions, genomic organization, and neurologic diversity. Genes Dev. 14, 1169–1180 (2000).
Yap, A.S., Brieher, W.M. & Gumbiner, B.M. Molecular and functional analysis of cadherin-based adherens junctions. Annu. Rev. Cell Dev. Biol. 13, 119–146 (1997).
Suzuki, S.T. Protocadherins and diversity of the cadherin superfamily. J. Cell Sci. 109, 2609–2611 ( 1996).
Ozawa, M., Engel, J. & Kemler, R. Single amino acid substitutions in one Ca2+ binding site of uvomorulin abolish the adhesive function. Cell 63, 1033–1038 ( 1990).
Nollet, F., Kools, P. & van Roy, F. Phylogenetic analysis of the cadherin superfamily allows identification of six major subfamilies besides several solitary members. J. Mol. Biol. 299, 551–572 (2000).
Noben-Trauth, K., Zheng, Q.Y., Johnson, K.R. & Nishina, P.M. mdfw: a deafness susceptibility locus that interacts with deaf waddler (dfw). Genomics 44, 266– 272 (1997).
Street, V.A., McKee-Johnson, J.W., Fonseca, R.C., Tempel, B.L. & Noben-Trauth, K. Mutations in a plasma membrane Ca2+-ATPase gene cause deafness in deafwaddler mice. Nature Genet. 19, 390–394 (1998).
Alagramam, K.N. The mouse Ames waltzer hearing-loss mutant is caused by mutation of Pcdh15 , a new protocadherin gene. Nature Genet. 27, 99–102 (2000).
Yamoah, E.N. et al. Plasma membrane Ca2+-ATPase extrudes Ca2+ from hair cell stereocilia. J. Neurosci. 18, 610–624 (1998).
Johnson, K.R., Erway, L.C., Cook, S.A., Willott, J.F. & Zheng, Q.Y. A major gene affecting age-related hearing loss in C57BL/6J mice. Hear. Res. 114, 83– 92 (1997).
Bolz, H. et al. Mutation of CDH23, encoding a new member of the cadherin gene family, causes Usher syndrome type 1D. Nature Genet. 27, 105–112 (2000).
Liu, X., Udovichenko, I.P., Brown, S.D., Steel, K.P. & Williams, D.S. Myosin VIIa participates in opsin transport through the photoreceptor cilium. J. Neurosci. 19, 6267–6274 (1999).
Schultz, J., Copley, R.R., Doerks, T., Ponting, C.P. & Bork, P. SMART: a web-based tool for the study of genetically mobile domains. Nucleic Acids Res 28, 231 –234 (2000).
Rost, B. PHD: predicting one-dimensional protein structure by profile-based neural networks. Methods Enzymol. 266, 525– 539 (1996).
Nielsen, H., Engelbrecht, J., Brunak, S. & von Heijne, G. Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Protein Eng. 10, 1–6 (1997).
Flaherty, L., Messer, A., Russell, L.B. & Rinchik, E.M. Chlorambucil-induced mutations in mice recovered in homozygotes. Proc. Natl. Acad. Sci. USA 89, 2859– 2863 (1992).
Noben-Trauth, K., Naggert, J.K., North, M.A. & Nishina, P.M. A candidate gene for the mouse mutation tubby. Nature 380, 534–538 (1996).
Lanford, P.J. et al. Notch signalling pathway mediates hair cell development in mammalian cochlea. Nature Genet. 21, 289 –292 (1999).
Hunter-Duvar, I.M. A technique for preparation of cochlear specimens for assessment with the scanning electron microscope. Acta Otolaryngol. Suppl. 351, 3–23 (1978).
Self, T. et al. Shaker-1 mutations reveal roles for myosin VIIA in both development and function of cochlear hair cells. Development 125 , 557–566 (1998).
Acknowledgements
We thank M. Legare and W. Frankel for the v6J allele; N. Noben-Trauth for discussions; K. Johnson for access to unpublished data; W. Pavan, D. Drayna and H. Arnheiter for valuable comments on the manuscript; P. Lanford and M. Kelly for help with the in situ hybridization; B. O'Dell for technical assistance; and the staff of our animal facilities. This work was supported by NIH grant DC03771 (E.C.B.), by EC (contract QLG2-CT-1999-00988), MRC and Defeating Deafness (K.P.S.) and the NIDCD Intramural program DZ 00036-04 (K.N.T.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Figure A
Mouse otocadherin protein; predicted amino acid sequence of mouse otocadherin derived from the C57BL/6J strain. The signal sequence is shown in purple; each ectodomain is highlighted in blue; the predicted transmembrane domain is shown in green and the tyrosine phosphorylation signal is shown in pink. (GIF 239 kb)
Rights and permissions
About this article
Cite this article
Palma, F., Holme, R., Bryda, E. et al. Mutations in Cdh23, encoding a new type of cadherin, cause stereocilia disorganization in waltzer, the mouse model for Usher syndrome type 1D. Nat Genet 27, 103–107 (2001). https://doi.org/10.1038/83660
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/83660
This article is cited by
-
Comparison of vestibular function in hereditary hearing loss patients with GJB2, CDH23, and SLC26A4 variants
Scientific Reports (2024)
-
Mechanisms in cochlear hair cell mechano-electrical transduction for acquisition of sound frequency and intensity
Cellular and Molecular Life Sciences (2021)
-
Spontaneous mutations of the Zpld1 gene in mice cause semicircular canal dysfunction but do not impair gravity receptor or hearing functions
Scientific Reports (2019)
-
Novel compound heterozygous CDH23 variants in a patient with Usher syndrome type I
Human Genome Variation (2019)
-
Genome-wide association meta-analysis identifies five novel loci for age-related hearing impairment
Scientific Reports (2019)