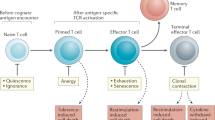
Abstract
Successful adaptive immunity against a broad range of pathogens depends on the diversity of randomly generated T-lymphocyte and B-lymphocyte receptors. A subset of these receptors will be self-reactive and must be regulated to prevent autoimmunity. The process of immunologic tolerance addresses this problem by either purging autoreactive receptors from the system or tuning down their reactivity sufficiently to prevent disease. Immature lymphocytes generate a novel receptor during development in the thymus or bone marrow. Engagement of self antigens by these nascent receptors leads to their purging, either by the apoptotic death of the lymphocyte or by the initiation of receptor editing, a process in which the autoreactive receptor is replaced. If the lymphocytes mature further, the activation threshold of autoreactive cells can be tuned by the co-expression of inhibitory receptors or negative signaling molecules, allowing the persistence of the receptor without an increased risk of autoimmunity. T-cell and B-cell receptors that escape these checkpoints can still be regulated in the peripheral immune system by both purging and tuning mechanisms. A separate set of mechanisms, mediated by various regulatory cells, also operates to tune peripheral receptors in a cell-extrinsic fashion. The combined action of these processes ensures that the organism does not suffer autoimmune pathology, even if autoreactive receptors are generated and maintained in the immune system.
Key Points
-
Immunologic tolerance operates to prevent randomly generated self-reactive T-cell and B-cell receptors from triggering autoimmunity
-
Purging and tuning of the autoreactive receptors are the two strategies used to maintain immunologic tolerance
-
Purging is accomplished by the induction of apoptosis in the lymphocyte expressing the autoreactive receptor or by the process of receptor editing, in which the autoreactive receptor is replaced with a newly rearranged one
-
Tuning can be cell-intrinsic (adaptation) or cell-extrinsic (regulatory) and allows the self-reactive receptor to persist in the periphery without initiating autoimmunity
-
Adaptation is achieved by expression of inhibitory receptors or negative-regulatory signaling molecules in the lymphocyte, which increases its threshold for activation by the autoreactive receptor
-
Cell-extrinsic tuning is mediated by regulatory cells, including CD4+ regulatory T cells, natural killer T cells, and antigen-presenting cells, that might prevent activation of the self-reactive lymphocyte or modulate its differentiation process
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Zerrahn J et al. (1997) The MHC reactivity of the T cell repertoire prior to positive and negative selection. Cell 88: 627–636
Wardemann H et al. (2003) Predominant autoantibody production by early human B cell precursors. Science 301: 1374–1377
Gotter J and Kyewski B (2004) Regulating self-tolerance by deregulating gene expression. Curr Opin Immunol 16: 741–745
Vogel A et al. (2002) The genetic background of autoimmune polyendocrinopathy–candidiasis–ectodermal dystrophy and its autoimmune disease components. J Mol Med 80: 201–211
Vafiadis P et al. (1997) Insulin expression in human thymus is modulated by INS VNTR alleles at the IDDM2 locus. Nat Genet 15: 289–292
Hartley SB et al. (1993) Elimination of self-reactive B lymphocytes proceeds in two stages: arrested development and cell death. Cell 72: 325–335
Ait-Azzouzene D et al. (2005) An immunoglobulin Cκ-reactive single chain antibody fusion protein induces tolerance through receptor editing in a normal polyclonal immune system. J Exp Med 201: 817–828
Nemazee D and Hogquist KA (2003) Antigen receptor selection by editing or downregulation of V(D)J recombination. Curr Opin Immunol 15: 182–189
Chandok MR and Farber DL (2004) Signaling control of memory T cell generation and function. Semin Immunol 16: 285–293
Baxter AG and Hodgkin PD (2002) Activation rules: the two-signal theories of immune activation. Nat Rev Immunol 2: 439–446
Vinuesa CG et al. (2005) A RING-type ubiquitin ligase family member required to repress follicular helper T cells and autoimmunity. Nature 435: 452–458
Lafferty KJ and Cunningham AJ (1975) A new analysis of allogeneic interactions. Aust J Exp Biol Med Sci 53: 27–42
Matzinger P (2002) The danger model: a renewed sense of self. Science 296: 301–305
Janeway CA Jr and Medzhitov R (2002) Innate immune recognition. Ann Rev Immunol 20: 197–216
Grossman Z and Paul WE (2001) Autoreactivity, dynamic tuning and selectivity. Curr Opin Immunol 13: 687–698
Nossal GJ (1996) Clonal anergy of B cells: a flexible, reversible, and quantitative concept. J Exp Med 183: 1953–1956
Healy JI and Goodnow CC (1998) Positive versus negative signaling by lymphocyte antigen receptors. Annu Rev Immunol 16: 645–670
Cannons JL and Schwartzberg PL (2004) Fine-tuning lymphocyte regulation: what's new with tyrosine kinases and phosphatases? Curr Opin Immunol 16: 296–303
Healy JI et al. (1997) Different nuclear signals are activated by the B cell receptor during positive versus negative signaling. Immunity 6: 419–428
Schwartz RH (2003) T cell anergy. Annu Rev Immunol 21: 305–334
Heissmeyer V et al. Calcineurin imposes T cell unresponsiveness through targeted proteolysis of signaling proteins. Nat Immunol 5: 255–265
Tanchot C et al. Adaptive tolerance of CD4+ T cells in vivo: multiple thresholds in response to a constant level of antigen presentation. J Immunol 167: 2030–2039
Singh NJ and Schwartz RH (2003) The strength of persistent antigenic stimulation modulates adaptive tolerance in peripheral CD4+ T cells. J Exp Med 198: 1107–1117
Leibson PJ (2004) The regulation of lymphocyte activation by inhibitory receptors. Curr Opin Immunol 16: 328–336
Raman C (2002) CD5, an important regulator of lymphocyte selection and immune tolerance. Immunol Res 26: 255–263
Ueda H et al. (2003) Association of the T-cell regulatory gene CTLA4 with susceptibility to autoimmune disease. Nature 423: 506–511
Kronenberg M and Rudensky A (2005) Regulation of immunity by self-reactive T cells. Nature 435: 598–604
Fontenot JD et al. (2005) Regulatory T cell lineage specification by the forkhead transcription factor foxp3. Immunity 22: 329–341
Picca CC and Caton AJ (2005) The role of self-peptides in the development of CD4+ CD25+ regulatory T cells. Curr Opin Immunol 17: 131–136
Tanchot C et al. (2004) Immune regulation by self-reactive T cells is antigen specific. J Immunol 172: 4285–4291
Schwartz RH (2005) Natural regulatory T cells and self-tolerance. Nat Immunol 6: 327–330
Gambineri E et al. (2003) Immune dysregulation, polyendocrinopathy, enteropathy, and X-linked inheritance (IPEX), a syndrome of systemic autoimmunity caused by mutations of FOXP3, a critical regulator of T-cell homeostasis. Curr Opin Rheumatol 15: 430–435
Taniguchi M et al. (2003) The regulatory role of Vα14 NKT cells in innate and acquired immune response. Annu Rev Immunol 21: 483–513
Smits HH et al. (2005) Different faces of regulatory DCs in homeostasis and immunity. Trends Immunol 26: 123–129
Lund FE et al. (2005) Regulatory roles for cytokine-producing B cells in infection and autoimmune disease. Curr Dir Autoimmun 8: 25–54
Leadbetter EA et al. (2002) Chromatin-IgG complexes activate B cells by dual engagement of IgM and Toll-like receptors. Nature 416: 603–607
Weiner HL (1997) Oral tolerance for the treatment of autoimmune diseases. Annu Rev Med 48: 341–351
Goodnow CC (1996) Balancing immunity and tolerance: deleting and tuning lymphocyte repertoires. Proc Natl Acad Sci USA 93: 2264–2271
McHugh RS and Shevach EM (2002) Cutting edge: depletion of CD4+ CD25+ regulatory T cells is necessary, but not sufficient, for induction of organ-specific autoimmune disease. J Immunol 168: 5979–5983
Anderson CC (2004) Conceptual models in immunity/tolerance: application to transplantation in immunobiology of organ transplantation. In Immunology of Organ Transplant, 171–190 (Eds Wilkes DS and Burlingham WJ) New York: Kluwer Academic/Plenum
Acknowledgements
This work was supported by the Intramural Research Program of the National Institutes of Allergy and Infectious Diseases at the National Institutes of Health, Bethesda, MD, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Singh, N., Schwartz, R. Primer: mechanisms of immunologic tolerance. Nat Rev Rheumatol 2, 44–52 (2006). https://doi.org/10.1038/ncprheum0049
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncprheum0049
This article is cited by
-
Transplant Tolerance: Current Insights and Strategies for Long-Term Survival of Xenografts
Archivum Immunologiae et Therapiae Experimentalis (2018)
-
A stepwise model of polyreactivity of the T cell antigen-receptor (TCR): its impact on the self–nonself discrimination and on related observations (receptor editing, anergy, dual receptor cells)
Cellular and Molecular Life Sciences (2014)
-
β-Arrestin 1 Modulates Functions of Autoimmune T Cells from Primary Biliary Cirrhosis Patients
Journal of Clinical Immunology (2011)
-
Granzyme B cleavage of autoantigens in autoimmunity
Cell Death & Differentiation (2010)
-
Immune privilege or privileged immunity?
Mucosal Immunology (2008)