Abstract
The ATR checkpoint kinase coordinates cellular responses to DNA replication stress. Budding yeast contain three activators of Mec1 (the ATR orthologue); however, only TOPBP1 is known to activate ATR in vertebrates. We identified ETAA1 as a replication stress response protein in two proteomic screens. ETAA1-deficient cells accumulate double-strand breaks, sister chromatid exchanges, and other hallmarks of genome instability. They are also hypersensitive to replication stress and have increased frequencies of replication fork collapse. ETAA1 contains two RPA-interaction motifs that localize ETAA1 to stalled replication forks. It also interacts with several DNA damage response proteins including the BLM/TOP3α/RMI1/RMI2 and ATR/ATRIP complexes. It binds ATR/ATRIP directly using a motif with sequence similarity to the TOPBP1 ATR-activation domain; and like TOPBP1, ETAA1 acts as a direct ATR activator. ETAA1 functions in parallel to the TOPBP1/RAD9/HUS1/RAD1 pathway to regulate ATR and maintain genome stability. Thus, vertebrate cells contain at least two ATR-activating proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Ciccia, A. & Elledge, S. J. The DNA damage response: making it safe to play with knives. Mol. Cell 40, 179–204 (2010).
Zeman, M. K. & Cimprich, K. A. Causes and consequences of replication stress. Nat. Cell Biol. 16, 2–9 (2014).
Fanning, E., Klimovich, V. & Nager, A. R. A dynamic model for replication protein A (RPA) function in DNA processing pathways. Nucleic Acids Res. 34, 4126–4137 (2006).
Zou, L. & Elledge, S. J. Sensing DNA damage through ATRIP recognition of RPA-ssDNA complexes. Science 300, 1542–1548 (2003).
Ball, H. L., Myers, J. S. & Cortez, D. ATRIP binding to replication protein A-single-stranded DNA promotes ATR-ATRIP localization but is dispensable for Chk1 phosphorylation. 16, 2372–2381 (2005).
Cortez, D. et al. ATR and ATRIP: partners in checkpoint signaling. Science 294, 1713–1716 (2001).
Kumagai, A., Lee, J., Yoo, H. Y. & Dunphy, W. G. TopBP1 activates the ATR-ATRIP complex. Cell 124, 943–955 (2006).
Lindsey-Boltz, L. A., Kemp, M. G., Capp, C. & Sancar, A. RHINO forms a stoichiometric complex with the 9-1-1 checkpoint clamp and mediates ATR-Chk1 signaling. Cell Cycle 14, 99–108 (2015).
Cotta-Ramusino, C. et al. A DNA damage response screen identifies RHINO, a 9-1-1 and TopBP1 interacting protein required for ATR signaling. Science 332, 1313–1317 (2011).
Duursma, A. M., Driscoll, R., Elias, J. E. & Cimprich, K. A. A role for the MRN complex in ATR activation via TOPBP1 recruitment. Mol. Cell 50, 116–122 (2013).
Mordes, D. A., Nam, E. A. & Cortez, D. Dpb11 activates the Mec1-Ddc2 complex. Proc. Natl Acad. Sci. USA 105, 18730–18734 (2008).
Navadgi-Patil, V. M. & Burgers, P. M. The unstructured C-terminal tail of the 9-1-1 clamp subunit Ddc1 activates Mec1/ATR via two distinct mechanisms. Mol. Cell 36, 743–753 (2009).
Navadgi-Patil, V. M. & Burgers, P. M. Yeast DNA replication protein Dpb11 activates the Mec1/ATR checkpoint kinase. J. Biol. Chem. 283, 35853–35859 (2008).
Kumar, S. & Burgers, P. M. Lagging strand maturation factor Dna2 is a component of the replication checkpoint initiation machinery. Genes Dev. 27, 313–321 (2013).
Brosh, R. M. et al. Replication protein A physically interacts with the Bloom’s syndrome protein and stimulates its helicase activity. J. Biol. Chem. 275, 23500–23508 (2000).
Ellis, N. A. et al. The Bloom’s syndrome gene product is homologous to RecQ helicases. Cell 83, 655–666 (1995).
Chaganti, R. S., Schonberg, S. & German, J. A manyfold increase in sister chromatid exchanges in Bloom’s syndrome lymphocytes. Proc. Natl Acad. Sci. USA 71, 4508–4512 (1974).
Croteau, D. L., Popuri, V., Opresko, P. L. & Bohr, V. A. Human RecQ helicases in DNA repair, recombination, and replication. Annu. Rev. Biochem. 83, 519–552 (2014).
Plank, J. L., Wu, J. & Hsieh, T.-S. Topoisomerase IIIalpha and Bloom’s helicase can resolve a mobile double Holliday junction substrate through convergent branch migration. Proc. Natl Acad. Sci. USA 103, 11118–11123 (2006).
Raynard, S., Bussen, W. & Sung, P. A double Holliday junction dissolvasome comprising BLM, topoisomerase IIIalpha, and BLAP75. J. Biol. Chem. 281, 13861–13864 (2006).
Singh, T. R. et al. BLAP18/RMI2, a novel OB-fold-containing protein, is an essential component of the Bloom helicase-double Holliday junction dissolvasome. Genes Dev. 22, 2856–2868 (2008).
Wu, L. & Hickson, I. D. The Bloom’s syndrome helicase suppresses crossing over during homologous recombination. Nature 426, 870–874 (2003).
Wu, L. et al. BLAP75/RMI1 promotes the BLM-dependent dissolution of homologous recombination intermediates. Proc. Natl Acad. Sci. USA 103, 4068–4073 (2006).
Xu, D., Guo, R. & Sobeck, A. RMI, a new OB-fold complex essential for Bloom syndrome protein to maintain genome stability. Genes Dev. 22, 2843–2855 (2008).
Blackford, A. N. et al. TopBP1 interacts with BLM to maintain genome stability but is dispensable for preventing BLM degradation. Mol. Cell 57, 1133–1141 (2015).
Wang, J., Chen, J. & Gong, Z. TopBP1 controls BLM protein level to maintain genome stability. Mol. Cell 52, 667–678 (2013).
Borowski, A. et al. Structure and function of ETAA16: a novel cell surface antigen in Ewing’s tumours. Cancer Immunol. Immunother. 55, 363–374 (2006).
Matsuoka, S. et al. ATM and ATR substrate analysis reveals extensive protein networks responsive to DNA damage. Science 316, 1160–1166 (2007).
Childs, E. J. et al. Common variation at 2p13.3, 3q29, 7p13 and 17q25.1 associated with susceptibility to pancreatic cancer. Nat. Genet. 47, 911–916 (2015).
Wu, C. et al. Genome-wide association study identifies five loci associated with susceptibility to pancreatic cancer in Chinese populations. Nat. Genet. 44, 62–66 (2012).
Dungrawala, H. et al. The replication checkpoint prevents two types of fork collapse without regulating replisome stability. Mol. Cell 59, 998–1010 (2015).
Bansbach, C. E., Bétous, R., Lovejoy, C. A., Glick, G. G. & Cortez, D. The annealing helicase SMARCAL1 maintains genome integrity at stalled replication forks. Genes Dev. 23, 2405–2414 (2009).
Mer, G. et al. Structural basis for the recognition of DNA repair proteins UNG2, XPA, and RAD52 by replication factor RPA. Cell 103, 449–456 (2000).
Feldkamp, M. D., Mason, A. C., Eichman, B. F. & Chazin, W. J. Structural analysis of replication protein a recruitment of the DNA damage response protein SMARCAL1. Biochemistry 53, 3052–3061 (2014).
Xu, X. et al. The basic cleft of RPA70N binds multiple checkpoint proteins, including RAD9, to regulate ATR signaling. Mol. Cell. Biol. 28, 7345–7353 (2008).
Toledo, L. I., Murga, M., Gutierrez-Martinez, P., Soria, R. & Fernandez-Capetillo, O. ATR signaling can drive cells into senescence in the absence of DNA breaks. Genes Dev. 22, 297–302 (2008).
Ball, H. L. et al. Function of a conserved checkpoint recruitment domain in ATRIP proteins. Mol. Cell. Biol. 27, 3367–3377 (2007).
Toledo, L. I. et al. ATR prohibits replication catastrophe by preventing global exhaustion of RPA. Cell 155, 1088–1103 (2013).
Putnam, C. D., Hayes, T. K. & Kolodner, R. D. Post-replication repair suppresses duplication-mediated genome instability. PLoS Genet. 6, e1000933 (2010).
Bussen, W., Raynard, S., Busygina, V., Singh, A. K. & Sung, P. Holliday junction processing activity of the BLM-Topo IIIalpha-BLAP75 complex. J. Biol. Chem. 282, 31484–31492 (2007).
Mordes, D. A., Glick, G. G., Zhao, R. & Cortez, D. TopBP1 activates ATR through ATRIP and a PIKK regulatory domain. Genes Dev. 22, 1478–1489 (2008).
Peter, H. et al. Activation of the ATR kinase by the RPA-binding protein ETAA1. Nat. Cell Biol. http://dx.doi.org/10.1038/ncb3422 (2016).
Delacroix, S., Wagner, J. M., Kobayashi, M., Yamamoto, K.-i. & Karnitz, L. M. The Rad9-Hus1-Rad1 (9-1-1) clamp activates checkpoint signaling via TopBP1. Genes Dev. 21, 1472–1477 (2007).
Kumagai, A. & Dunphy, W. G. Claspin, a novel protein required for the activation of Chk1 during a DNA replication checkpoint response in Xenopus egg extracts. Mol. Cell 6, 839–849 (2000).
German, J. & Alhadeff, B. Analysis of sister-chromatid exchanges Curr. Protoc. Hum. Genet. Chapter 8 Unit 8.6 (2001).
Acknowledgements
The research was supported primarily by R01GM116616 to D.C. with additional support from R01CA102729 and the Vanderbilt-Ingram Cancer Center. NMR experiments were supported by R01GM65484 and P01CA092584 to W.J.C. T.E.B. is supported by training grant T32CA009582-28. We thank R. Guo for performing the initial Flag-RPA1 immunopurifications and W. Hayes McDonald in the Vanderbilt Proteomics Laboratory for performing the mass spectrometry.
Author information
Authors and Affiliations
Contributions
T.E.B., J.W.L., G.K., C.C., H.D., G.G.G. and D.C. performed most of the experiments. M.D.F. and R.P. performed the NMR experiments with supervision from W.J.C. T.E.B. and D.C. conceived of the project and wrote the manuscript. D.C. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 RPA recruits ETAA1 to damaged replication forks.
(a) Examples of GFP-Flag-ETAA1 localization in cells treated with HU or cisplatin for 3 h. (b) Schematic of ETAA1 fragments tested for their ability to bind RPA. (c–e) Nuclear extracts were prepared from HEK293T cells after transfection with the indicated GFP-Flag-NLS-ETAA1 expression vectors. Flag immunoprecipitates were separated by SDS-PAGE and examined by immunoblotting. Mock, mock-transfected. Cells were treated with 100 nM CPT for 3 h prior to lysis where indicated. The interaction between ETAA1 and RPA was not changed substantially in cells treated with CPT. Representative blots from one of two independent experiments are shown. (f) Quantitation of cells containing ETAA1 foci after transfection with the indicated ETAA1 expression vectors. Mean and SEM from n = 3 experiments is shown. (g) U2OS cells were transfected with an ETAA1 expression vector missing the C-terminal RPA32 interaction motif (ETAA1Δ32) in combination with non-targeting or RPA siRNA and imaged for ETAA1 and RPA localization after a challenge with 100 mM CPT. (h) ETAA1Δ cells were transduced with lentivirus to express empty vector (GFP-Flag), GFP-Flag-ETAA1 (WT), or GFP-Flag-ETAA1 with point mutations (residues 606-611 mutated from DVDDDL to NAAIRS) in the RPA70N motif and deletion of the ETAA1-RPA32C (deletion of residues 885–926) interaction motif (ETAA1ΔRPA). Cells were treated with 100 nM CPT for 3 h. RPA, and Flag-GPF-ETAA1 were visualized by immunofluorescence. Representative images from one of two independent experiments in g and h are shown. Scale bars are 5 μm. Unprocessed original scans of blots in c, d and e are shown in Supplementary Fig. 8, and source data for f is in Supplementary Table 1.
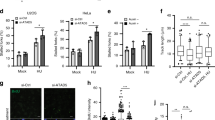
Supplementary Figure 2 ETAA1 knockdown causes hypersensitivity to replication stress.
(a–b) HeLa, H157, and BT549 cells transfected with non-targeting (NT) or ETAA1 siRNAs were left untreated or treated with 100 nM CPT for 3 h. Cells were fixed and RPA foci and γH2AX intensity were quantified by immunofluorescence imaging. The intensity of each nucleus, mean intensity, and number of nuclei imaged is depicted. (c) U2OS cells were transfected with non-targeting or ETAA1 siRNAs. All siRNAs target different regions of the ETAA1 coding sequence or 3′ UTR. siE1, siE3, and siE3 are different from the four ETAA1 siRNAs in the pool. Viability was measured 48 h after challenging the cells with 0.5 μM CPT for 24 h. Data show the mean of three independent experiments for siNT and siETAA1 pool and two for the other siRNAs. [OK?] (d–k) HeLa, HCT116, H157, and BT549 cells were transfected with non-targeting or ETAA1 siRNAs and viability was measured 72 h after challenge with CPT or HU. Data show the mean from three technical replicates and the experiment was performed once. (l) U2OS cells were transfected with non-targeting or ETAA1 siRNAs and exposed to 0, 3, or 5 Gy ionizing radiation. Cell viability was determined by clonogenic assay. (m–o) U2OS cells were transfected with non-targeting or ETAA1 siRNAs and viability was measured 72 h after challenge with cisplatin, olaparib, or BMN673. ATR siRNA was used as a positive control. In l–o the mean viability from from three replicates is shown. One representative experiment is shown and the experiments were completed twice independently. Source data for all panels is in Supplementary Table 1.
Supplementary Figure 3 ETAA1 interacts with multiple DNA damage response proteins.
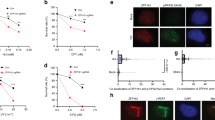
(a) Flag-ETAA1 was immunoprecipitated from nuclear extracts of HEK293T cells, and immunoprecipitated proteins were identified by mass spectrometry. Extracts in the second replicate were treated with benzonase and RNAse prior to the immunopurification. Shown are peptide counts from two experiments including untransfected cell populations (Ctl). (b,c) HEK293T cells were mock transfected (lanes 1 and 3) or transfected with a Flag-GFP-ETAA1 expression vector (lanes 2 and 4) and ETAA1 was immunoprecipitated using Flag antibodies. Co-precipitating proteins were separated by SDS-PAGE and detected by immunoblotting. (d) HEK293T nuclear extracts were fractionated on a Superdex 200 column. Protein fractions were then separated using SDS-PAGE and immunoblotted with the indicated antibodies. To detect ETAA1, it was immunoprecipitated from the fractions prior to immunoblotting. The reason ETAA1 migrates as a doublet in some gel conditions is not known but may represent post-translational modifications. Unprocessed original scans of blots in b,c and d are shown in Supplementary Fig. 8. Blots in b and c are representative from one of two experiments. The experiment in d was completed once.
Supplementary Figure 4 ETAA1 is required for RPA phosphorylation in multiple cell types.
(a) U2OS, HeLa, HCT116, H157, BT549, and A549 cells transfected with non-targeting or ETAA1 siRNAs were left untreated or treated with 100 nM CPT for 8 h. Proteins were separated by SDS-PAGE and detected by immunoblotting. Unprocessed original scans of blots are shown in Supplementary Fig. 8. This experiment with all of the cell lines was completed once although the results in U2OS, HeLa, and HCT116 were confirmed in independent replicates.
Supplementary Figure 5 ETAA1 over-expression causes DNA damage signaling.
(a–h) U2OS cells were transfected with the indicated GFP-Flag-NLS-ETAA1 protein expression vectors, and GFP and γH2AX were visualized by immunofluorescence imaging. (a, b) Cell Profiler was used to quantitate γH2AX intensity in ETAA1 expressing cells. The intensity of each nucleus, mean intensity, and number of nuclei imaged is depicted. Data represent one out of three experiments in a and one out of two experiments in b. (c,d) The percentage of cells with γH2AX or ETAA1 foci were scored manually in blinded samples. Mean and SEM from n = 4 experiments is graphed. (e) Immunoblot of proteins to examine expression levels. (f) Immunoblot of untransfected (lane 1), transiently transfected (lane 3), and stable ETAA1 expressing cell lines after sorting (lane 2). (g,h) GFP-Flag-ETAA1-2-250 or 2-250-W107A intensity versus γH2AX intensity is plotted. Pearson correlation coefficient is shown. Data in e–h are representative of two experiments. Unprocessed original scans of blots in e are shown in Supplementary Fig. 8, and source data for a–d is in Supplementary Table 1.
Supplementary Figure 6 An interaction with RPA is needed for ETAA1 to maintain genome integrity.
(a,b) ETAA1Δ cells were infected with wild-type or RPA binding-deficient ETAA1 lentiviruses. After antibiotic selection, the cell populations were sorted to select for the 10% of cells expressing the lowest amount of ETAA1 protein. (b) Immunoblot shows that the complemented cells overexpress approximately equal amounts of wild-type (WT) and ETAA1ΔRPA proteins. (c) Cell populations were challenged with the indicated concentrations of CPT and cell viability measured after 72 h. The mean viability from three technical replicates from one of two independent experiments is shown. (d) The percentage of cells with micronuclei was scored in the indicated cell populations. Data from two independent complemented ETAA1Δ clones are shown. The mean percentage of cells with micronuclei and total number of nuclei scored from five technical replicates of one of two biological replicates is shown. Unprocessed original scans of blots in b are shown in Supplementary Fig. 8, and source data for c and d are in Supplementary Table 6.
Supplementary Figure 7 ETAA1- and TOPBP1-deficient cells maintain BLM expression.
WT and ETAA1Δ U2OS cells were transfected with non-targeting or two siRNAs targeting TOPBP1. Cell lysates were immunoblotted with the indicated antibodies. Unprocessed original scans of blots are shown in Supplementary Fig. 8. Representative blots from one out of two experiments is shown.
Supplementary information
Supplementary Information
Supplementary Information (PDF 3547 kb)
Supplementary Table 1
Supplementary Information (XLSX 74 kb)
Rights and permissions
About this article
Cite this article
Bass, T., Luzwick, J., Kavanaugh, G. et al. ETAA1 acts at stalled replication forks to maintain genome integrity. Nat Cell Biol 18, 1185–1195 (2016). https://doi.org/10.1038/ncb3415
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3415
This article is cited by
-
Replication protein-A, RPA, plays a pivotal role in the maintenance of recombination checkpoint in yeast meiosis
Scientific Reports (2024)
-
ZNF827 is a single-stranded DNA binding protein that regulates the ATR-CHK1 DNA damage response pathway
Nature Communications (2024)
-
ATR kinase supports normal proliferation in the early S phase by preventing replication resource exhaustion
Nature Communications (2023)
-
WASp modulates RPA function on single-stranded DNA in response to replication stress and DNA damage
Nature Communications (2022)
-
Therapeutic targeting of ATR in alveolar rhabdomyosarcoma
Nature Communications (2022)