Abstract
Active positioning of the nucleus is integral to division, migration and differentiation of mammalian cells1. Fibroblasts polarizing for migration orient their centrosomes by actin-dependent nuclear movement2. This nuclear movement depends on nesprin-2 giant (N2G), a large, actin-binding outer nuclear membrane component of transmembrane actin-associated (TAN) lines that couple nuclei to moving actin cables3. Here, we identify the diaphanous formin FHOD1 as an interaction partner of N2G. Silencing FHOD1 expression or expression of fragments containing binding sites for N2G or FHOD1 disrupted nuclear movement and centrosome orientation in polarizing fibroblasts. Unexpectedly, silencing of FHOD1 expression did not affect the formation or rearward flow of dorsal actin cables required for nuclear positioning. Rather, N2G–FHOD1 interaction provided a second connection to actin cables essential for TAN line formation and thus nuclear movement. These results reveal a unique function for a formin in coupling an organelle to actin filaments for translocation, and suggest that TAN lines require multi-point attachments to actin cables to resist the large forces necessary to move the nucleus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
06 June 2014
In the version of this Letter originally published online, the fourth name in the author list was written incorrectly; it should have read 'G. W. Gant Luxton'. This has been corrected in all versions of the Letter.
References
Gundersen, G. G. & Worman, H. J. Nuclear positioning. Cell 152, 1376–1389 (2013).
Gomes, E. R., Jani, S. & Gundersen, G. G. Nuclear movement regulated by Cdc42, MRCK, myosin, and actin flow establishes MTOC polarization in migrating cells. Cell 121, 451–463 (2005).
Luxton, G. W., Gomes, E. R., Folker, E. S., Vintinner, E. & Gundersen, G. G. Linear arrays of nuclear envelope proteins harness retrograde actin flow for nuclear movement. Science 329, 956–959 (2010).
Chesarone, M. A., DuPage, A. G. & Goode, B. L. Unleashing formins to remodel the actin and microtubule cytoskeletons. Nat. Rev. Mol. Cell Biol. 11, 62–74 (2010).
Bartolini, F. & Gundersen, G. G. Formins and microtubules. Biochim. Biophys. Acta 1803, 164–173 (2010).
Schonichen, A. et al. FHOD1 is a combined actin filament capping and bundling factor that selectively associates with actin arcs and stress fibers. J. Cell Sci. 126, 1891–1901 (2013).
Gasteier, J. E. et al. Activation of the Rac-binding partner FHOD1 induces actin stress fibers via a ROCK-dependent mechanism. J. Biol. Chem. 278, 38902–38912 (2003).
Takeya, R. & Sumimoto, H. Fhos, a mammalian formin, directly binds to F-actin via a region N-terminal to the FH1 domain and forms a homotypic complex via the FH2 domain to promote actin fiber formation. J. Cell Sci. 116, 4567–4575 (2003).
Alvarez, D. E. & Agaisse, H. The formin FHOD1 and the small GTPase Rac1 promote vaccinia virus actin-based motility. J. Cell Biol. 202, 1075–1090 (2013).
Truong, D. et al. Formin-mediated actin polymerization promotes Salmonella invasion. Cell. Microbiol. 15, 2051–2063 (2013).
Iskratsch, T. et al. FHOD1 is needed for directed forces and adhesion maturation during cell spreading and migration. Dev. Cell 27, 545–559 (2013).
Schulte, A. et al. The human formin FHOD1 contains a bipartite structure of FH3 and GTPase-binding domains required for activation. Structure 16, 1313–1323 (2008).
Koka, S. et al. The formin-homology-domain-containing protein FHOD1 enhances cell migration. J. Cell Sci. 116, 1745–1755 (2003).
Simpson, J. G. & Roberts, R. G. Patterns of evolutionary conservation in the nesprin genes highlight probable functionally important protein domains and isoforms. Biochem. Soc. Trans. 36, 1359–1367 (2008).
Autore, F. et al. Large-scale modelling of the divergent spectrin repeats in nesprins: giant modular proteins. PLoS ONE 8, e63633 (2013).
Schmoranzer, J. et al. Par3 and dynein associate to regulate local microtubule dynamics and centrosome orientation during migration. Curr. Biol. 19, 1065–1074 (2009).
Palazzo, A. F., Cook, T. A., Alberts, A. S. & Gundersen, G. G. mDia mediates Rho-regulated formation and orientation of stable microtubules. Nat. Cell Biol. 3, 723–729 (2001).
Bartolini, F. et al. The formin mDia2 stabilizes microtubules independently of its actin nucleation activity. J. Cell Biol. 181, 523–536 (2008).
Luxton, G. W., Gomes, E. R., Folker, E. S., Worman, H. J. & Gundersen, G. G. TAN lines: a novel nuclear envelope structure involved in nuclear positioning. Nucleus 2, 173–181 (2011).
Ciobanasu, C., Faivre, B. & Le Clainche, C. Integrating actin dynamics, mechanotransduction and integrin activation: the multiple functions of actin binding proteins in focal adhesions. Eur. J. Cell Biol. 92, 339–348 (2013).
Pellegrin, S. & Mellor, H. Actin stress fibres. J. Cell Sci. 120, 3491–3499 (2007).
Starr, D. A. & Fridolfsson, H. N. Interactions between nuclei and the cytoskeleton are mediated by SUN-KASH nuclear-envelope bridges. Annu. Rev. Cell Dev. Biol. 26, 421–444 (2010).
Korobova, F., Ramabhadran, V. & Higgs, H. N. An actin-dependent step in mitochondrial fission mediated by the ER-associated formin INF2. Science 339, 464–467 (2013).
Riedl, J. et al. Lifeact: a versatile marker to visualize F-actin. Nat. Methods 5, 605–607 (2008).
Madrid, R. et al. Oligomerization of the diaphanous-related formin FHOD1 requires a coiled-coil motif critical for its cytoskeletal and transcriptional activities. FEBS Lett. 579, 441–448 (2005).
Cook, T. A., Nagasaki, T. & Gundersen, G. G. Rho guanosine triphosphatase mediates the selective stabilization of microtubules induced by lysophosphatidic acid. J. Cell Biol. 141, 175–185 (1998).
Iyer, K. et al. Utilizing the split-ubiquitin membrane yeast two-hybrid system to identify protein-protein interactions of integral membrane proteins. Sci. STKE 2005, pl3 (2005).
Gasteier, J. E. et al. FHOD1 coordinates actin filament and microtubule alignment to mediate cell elongation. Exp. Cell Res. 306, 192–202 (2005).
Gomes, E. R. & Gundersen, G. G. Real-time centrosome reorientation during fibroblast migration. Methods Enzymol. 406, 579–592 (2006).
Palazzo, A. F. et al. Cdc42, dynein, and dynactin regulate MTOC reorientation independent of Rho-regulated microtubule stabilization. Curr. Biol. 11, 1536–1541 (2001).
Chang, W., Folker, E. S., Worman, H. J. & Gundersen, G. G. Emerin organizes actin flow for nuclear movement and centrosome orientation in migrating fibroblasts. Mol. Biol. Cell. 24, 3869–3880 (2013).
Acknowledgements
We thank D. Discher and M. Geyer for helpful discussion. This work was financially supported in part by the Deutsche Forschungsgemeinschaft (GRK1188 to S.K., grant FA 378/6-2 to O.T.F.) and NIH (grant GM099481 to G.G.G.). O.T.F. is a member of the CellNetworks Cluster of Excellence EXC81.
Author information
Authors and Affiliations
Contributions
G.G.G. and O.T.F. conceived the study, designed experiments and wrote the manuscript. S.K., R.Z., S.A., G.W.L. and I.S. designed and conducted experiments and discussed and interpreted the data together with O.T.F. and G.G.G.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Additional evidence that the interaction between FHOD1s N-terminus and nesprin-2G (N2G) is specific and phylogenetic comparison of the FHOD1 interacting region of N2G.
(a) Schematic of N2G and FHOD1 with boundaries for constructs used in membrane yeast-two hybrid. (b) Legend for yeast two-hybrid indicating the FHOD1 fragments used as a bait for the experiment shown in panel c. (c) Yeast two-hybrid results for the interaction between N2G J, H and I fragments and FHOD1 fragments indicated in panel b. Bar, 5 mm. (d) N2G HI fragment interacts with HA-FHOD WT in cell lysates. GFP-N2G HI was expressed alone or co-expressed with HA-FHOD1 WT in 293T cells and lysates were immunoprecipitated with HA antibody. Western blots were probed with antibodies for HA and GFP. Input shows level of expression of transfected proteins. (e) Phylogenetic comparison between spectrin repeats (SRs) 9–13 of N2G. Red indicates residues conserved between at least four of the five species; pink indicates residues that are conserved in at least three of the species. Consensus residues are shown below for highly conserved positions. Sequences were obtained from the following sources. Human (H. sapiens, NP_878918.2, NCBI), Mouse (M. musculus, NIH3T3 fibroblast cDNA), Chicken (G. gallus, XP_003641488, NCBI), Frog (X. tropicanis, XP_002933763, NCBI), Fish (D. rerio, F1QVC9, Uniport).
Supplementary Figure 2 FHOD1 knock down by siRNAs.
Western blot of FHOD1 levels in NIH3T3 fibroblasts after knockdown with four different siRNAs targeting FHOD1. Control siRNA knockdown of GAPDH is shown for comparison. Vinculin is a loading control.
Supplementary Figure 3 FHOD1 knockdown does not affect actin structures induced by serum.
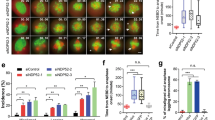
Starved NIH3T3 fibroblasts were stimulated with 20% FCS, fixed at indicated time points and stained with rhodamine phalloidin for F-actin (red) and DAPI for DNA (blue). (a) Fluorescence images of dorsal actin cables over the nucleus. Bar, 10 m. (b) Quantification of the number of actin cables over the nucleus per cell in control siRNA cells at various time points after serum stimulation. (c) Quantification of centrosome orientation in serum-stimulated wound edge NIH3T3 fibroblasts. (d) Comparison of dorsal actin cables over nuclei in siFHOD1 treated cells with oriented and non-oriented centrosomes. Data in b–d are from 3 experiments; n, number of cells analysed per experiment is shown in b–d. Error bars: s.d. ∗∗, P < 0.01; ∗, P < 0.05; ns, not significantly different by two-tailed t-test.
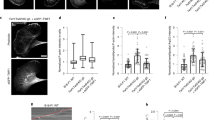
Supplementary Figure 4 Localization of FHOD1 ΔC with endogenous TAN lines and effect of FHOD1 knockdown on endogenous TAN lines.
(a) Immunofluorescence images of GFP-FHOD1 ΔC and endogenous N2G on the dorsal surface of nuclei in LPA-stimulated NIH3T3 fibroblasts. Arrowheads, TAN lines containing N2G and dorsal actin cables and GFP-FHOD1 ΔC (bottom panels). Leading edge of the cell is toward the top. Bar, 10 m. (b) Immunofluorescence images of endogenous N2G (N2G antibody-stained) and F-actin (rhodamine phalloidin) in NIH3T3 fibroblasts treated with the indicated siRNAs. Arrowheads, TAN lines containing N2G co-localized with dorsal actin cables in control siRNA-treated cells. TAN lines are not observed in FHOD1 siRNA-treated cells. Bar, 5 m. (c) Quantification of endogenous TAN lines in control siRNA- and FHOD1 siRNA-treated cells. Data are from 3 experiments; n, number of cells analysed per experiment shown in c. ∗∗, P < 0.01 by Fishers exact test.
Supplementary Figure 5 The I705A mutation in active FHOD1 ΔC disrupts its induction of and localization with thick actin filament bundles.
NIH3T3 cells expressing the indicated GFP-FHOD1 variants were stained for F-actin with rhodamine phalloidin. Note that active FHOD1 ΔC induces the formation of thick F-actin bundles associates with them. Actin bundle formation and actin filament association of FHOD1 is potently disrupted by the I705A mutation. Bar, 20 μm.
Supplementary Figure 6
Full scans.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1683 kb)
Supplementary Table 1
Supplementary Information (XLSX 12 kb)
Rights and permissions
About this article
Cite this article
Kutscheidt, S., Zhu, R., Antoku, S. et al. FHOD1 interaction with nesprin-2G mediates TAN line formation and nuclear movement. Nat Cell Biol 16, 708–715 (2014). https://doi.org/10.1038/ncb2981
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2981
This article is cited by
-
LINC complex protein nesprin-2 has pro-apoptotic activity via Bcl-2 family proteins
Cell Death Discovery (2024)
-
Mena regulates nesprin-2 to control actin–nuclear lamina associations, trans-nuclear membrane signalling and gene expression
Nature Communications (2023)
-
Shielding of actin by the endoplasmic reticulum impacts nuclear positioning
Nature Communications (2022)
-
B16 Melanoma Cancer Cells with Higher Metastatic Potential are More Deformable at a Whole-Cell Level
Cellular and Molecular Bioengineering (2021)
-
Apoptotic stress induces Bax-dependent, caspase-independent redistribution of LINC complex nesprins
Cell Death Discovery (2020)