Abstract
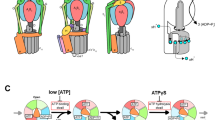
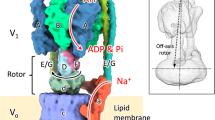
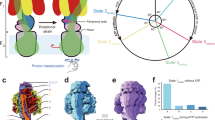
In all organisms, adenosine triphosphate (ATP) provides metabolic energy for driving energy-dependent processes. It is synthesized and/or utilized by enzymes known as F-type and V-type ATPases, which are small rotary motors1,2. Both types consist of a headpiece, F1 or V1, respectively, which is connected by a stalk region to the membrane-bound part, FO or VO. Electron microscopic analysis of negatively stained particles has revealed a peripheral stalk, or stator, between V1 and VO of the V-type (Na+)ATPase of the thermophilic bacterium Clostridium fervidus3,4, like that in F-type ATPases5,6. We have analysed many more particles and now present a more complete structure of the V-type ATPase stator moiety.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Boyer, P. D. FASEB J. 9, 559–561 (1995).
Junge, W., Lill, H. & Engelbrecht, S. Trends Biochem. Sci. 22, 420–423 (1997).
Boekema, E. J., Ubbink-Kok, T., Lolkema, J. S., Brisson, A. & Konings, W. N. Proc. Natl Acad. Sci. USA 94, 14291–14293 (1997).
Boekema, E. J., Ubbink-Kok, T., Lolkema, J. S., Brisson, A. & Konings, W. N. Photosynth. Res. 57, 267–273 (1998).
Wilkens, S. & Capaldi, R. A. Nature 393, 29 (1998).
Böttcher, B., Schwarz, L. & Gräber, P. J. Mol. Biol. 281, 757–762 (1998).
Yasuda, R., Noji, H., Kinosita, K. & Yoshida, M. Cell 93, 1117–1124 (1998).
Abrahams, J. P., Leslie, A. G. W., Lutter, R. & Walker, J. E. Nature 370, 621–628 (1994).
Dschida, W. J. & Bowman, B. J. J. Biol. Chem. 267, 18783–18789 (1992).
Dunn, S. D. & Chandler, J. J. Biol. Chem. 273, 8646–8651 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boekema, E., van Breemen, J., Brisson, A. et al. Connecting stalks in V-type ATPase. Nature 401, 37–38 (1999). https://doi.org/10.1038/43369
Issue Date:
DOI: https://doi.org/10.1038/43369
This article is cited by
-
The dynamic stator stalk of rotary ATPases
Nature Communications (2012)
-
The structure of the peripheral stalk of Thermus thermophilus H+-ATPase/synthase
Nature Structural & Molecular Biology (2010)
-
Structural and functional analysis of the coupling subunit F in solution and topological arrangement of the stalk domains of the methanogenic A1AO ATP synthase
Journal of Bioenergetics and Biomembranes (2006)
-
The ntp operon encoding the Na+ V-ATPase of the thermophile Caloramator fervidus
Archives of Microbiology (2006)
-
Structure and Regulation of the V-ATPases
Journal of Bioenergetics and Biomembranes (2005)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.