Abstract
Demonstration of pathological prion protein accumulation in the central nervous system is required to establish the diagnosis of transmissible subacute encephalopathies. In humans, this is frequently achieved using prion protein immunohistochemistry in paraffin-embedded tissue, a technique that requires multiple epitope retrieval and denaturing pretreatments. In addition to being time-consuming, this procedure induces tissue alterations that preclude accurate morphological examination. The aim of this study was to simplify prion protein immunohistochemistry procedure in human tissue, together with increased sensitivity and specificity. We screened a panel of 50 monoclonal antibodies produced using various immunogens (human and ovine recombinant prion protein, prion protein peptides, denatured scrapie-associated fibrils from 263K-infected Syrian hamsters) and directed against different epitopes along the human prion protein sequence. A panel of different forms of genetic, infectious and sporadic transmissible subacute encephalopathies was assessed. The monoclonal 12F10 antibody provided a high specificity and fast immunodiagnosis with very limited denaturing pretreatments. A standardized and reliable fast immunostaining procedure was established using an automated diagnostic system (Nexes, Ventana Medical Systems) and allowed prion protein detection in the central nervous system and in tonsil biopsies. It was evaluated in a series of 300 patients with a suspected diagnosis of transmissible subacute encephalopathies and showed high sensitivity and specificity.
Similar content being viewed by others
Main
The accumulation of a specific protease-resistant isoform (PrPsc) of the host-encoded prion protein (PrPc) characterizes human transmissible spongiform encephalopathies. Identifying PrPsc in the central nervous tissue of suspected patients is a critical step for the diagnosis of definite human transmissible subacute encephalopathies.1, 2 This is frequently achieved using PrP immunohistochemistry on formalin-fixed tissues.
Retrieval of antigens and denaturing pretreatments is required for detecting PrPsc by immunohistochemical procedures in sections of formalin-fixed and paraffin-embedded tissues. Formic acid3 and enzymatic digestion with proteinase K4 or pepsin5 have been used at first. Denaturing treatments using guanidine thiocyanate6 and heating by hydrolytic autoclaving7 or microwave irradiation8 and hydrated autoclaving9 have also been developed. Various combinations of these methods have been successfully tested.10, 11 Two multicentric, prospective studies have assessed several antibodies, including the commercially available and widely used 3F412 antibody in combination with enhancement procedures. Regardless of the antibody in use, they have led to the same reliable standardized pretreatment protocol for PrPsc immunohistochemistry: hydrated autoclaving (121°C for 10 min), followed by formic acid (96% for 5 min) and guanidine thiocyanate (4 M, 4°C for 2 h).13 However, the most sensitive procedure needs an additional antigen-retrieving treatment (ie, proteinase K digestion),14 and these complex pretreatments are time-consuming and induce tissue alterations that preclude fine morphological analysis.
Using a large panel of anti-PrP monoclonal antibodies produced with various immunogens, we have developed a fast, reliable and automated procedure for PrPsc immunohistochemistry in human samples, which enables (1) a high sensitivity and specificity for the diagnosis of human transmissible subacute encephalopathies and (2) a fine preservation of morphological information thanks to a simplified antigen-retrieving procedure.
Materials and methods
Antibody Screening Using a Four-Step Strategy
Step 1: Screening of 50 antibodies on four patients using a combination of four pretreatments
Our strategy was to study a panel of 50 monoclonal antibodies on four patients using a combination of four pretreatments. Four dilutions of primary antibodies (1/100, 1/500, 1/1000, 1/2000) were tested. The staining was compared to that obtained using the 3F4 antibody (Figure 1, Tables 1 and 2).
Step 2: Reducing the pretreatments in the six selected antibodies
The highly efficient antibodies identified in step 1 were used in different simplified pretreatment procedures.
Step 3: Automation of the procedure using the six selected antibodies on 26 patients
These antibodies were checked for their efficiency at 37°C using an automated diagnostic system for immunohistochemistry (Nexes, Ventana Medical Systems, Illkirch, France) on 26 patients. The results were compared to those obtained with 3F4 antibody using the manual and four pretreatment procedures. The highest sensitivity was obtained with the 12F10 antibody.
Step 4: Validation of the automated procedure using an additional series of 25 sporadic Creutzfeldt–Jakob disease patients
Together with the 26 patients used in step 3, sections from a total of 51 patients were used to compare the 12F10 automated procedure to the manual procedure, with the reference 3F4 antibody.
Patients and Samples
The panel of 51 patients, which included 46 patients with neuropathologically confirmed transmissible subacute encephalopathies, is described in Table 1. Paraffin-embedded blocks of 10% formalin-fixed brains were sampled from the frontal isocortex and the cerebellar vermis. Serial sections cut at 7 μm on superfrost-plus slides were dried at 56°C for 24 h before immunohistochemical procedures.
Immunohistochemical Procedures
Pretreatments
A combination of four pretreatments was used: hydrated autoclaving in distilled water (25 min), formic acid (5 min, 99%, 24°C), guanidine thiocyanate (2 h, 4 M, 4°C), and proteinase K (8 min, 10 μg/ml, 24°C).
Anti-PrP antibodies
Antibodies were produced in PrP0/0 mice using different immunogens (human and ovine recombinant PrP, PrP peptides, denatured scrapie-associated fibrils from 263K-infected Syrian hamsters), and have been described elsewhere.15, 16 They recognized different epitopes along the PrP sequence (Table 2). Monoclonal antibodies were purified using either caprylic acid precipitation17 or protein A affinity chromatography. Epitope mapping for identifying linear epitopes possibly recognized by monoclonal antibodies was performed as previously described.16 3F4 antibody (ref. 9620) was produced and commercialized by Signet Laboratories (MA, USA).
Manual procedure
After a saturation step using 10% goat serum for 30 min, a 3% H2O2 blockade of endogenous peroxidases and different combinations of pretreatments, the primary antibody was applied overnight at room temperature. A standard streptavidin–biotin peroxidase kit (ref. K5001) with diaminobenzidine substrate (Dako, Trappes, France) was used for single immunolabelling according to the manufacturer's instructions. Sections were counterstained with haematoxylin, dehydrated and mounted.
Automated procedure
A saturation step using 3% BSA for 20 min and hydrated autoclaving pretreatment was applied. The NexES IHC automated diagnostic system for immunohistochemistry and the iVIEW DAB Paraffin kit (ref. 760-091) were used according to the manufacturer's instructions (Ventana Medical Systems). The blocking of endogenous peroxidases, primary antibody incubation (32 min), DAB/copper revelation and section counterstaining with haematoxylin were performed at 37°C working temperature. Slides were dehydrated and mounted.
PrP Patterns
For assessing the pattern of immunolabelling with anti-PrP antibodies on brain samples, we followed criteria derived from those used previously.3, 10, 13, 18, 19, 20 Six types of labelling were distinguished: synaptic labelling (diffuse staining), granular deposits (small scattered immunolabelled spots less than 5 μm wide), focal deposits (large, 5–50 μm wide, non-amyloid rounded positive blots), vacuolar deposits (surrounding vacuoles of spongiform change), morula-type deposits (focal, non-amyloid deposits surrounded by vacuoles of spongiform change) and kuru-type amyloid plaques, 10–50 μm wide (plaques were also identified on Congo red and periodic acid Schiff-stained sections).14, 21 We did not consider other types of labelling as significant, such as that of isolated neurons or astrocytes. PrP immunostaining was considered as positive when at least one typical variety of labelling was observed in the frontal isocortex and/or the cerebellar vermis of the patient. In the study of tonsil biopsies, only patients with five or more secondary lymphoid follicles were considered22, 23 for immunostaining. The presence of intense immunoreactivity within germinal centres was considered as a positive result.
Evaluation of the 12F10 Automated Fast Procedure in a Series of 300 Brains and 47 Tonsil Biopsies from Patients with a Suspected Diagnosis of Human Prion Diseases
Between January 2001 and January 2006, 300 brains and 47 tonsil biopsies of patients with a suspected diagnosis of transmissible subacute encephalopathies were received at the R Escourolle Neuropathology Laboratory. PrP immunohistochemistry using the 12F10 automated fast procedure was performed on samples from frontal isocortex and cerebellar vermis, and tonsil biopsies. The slides were checked by two investigators (JJH and SH), who were unaware of the diagnosis. Interobserver agreement for the diagnosis of transmissible subacute encephalopathies was higher than 0.95. A consensus was reached by joint examination of the remaining samples. The gold standard was the neuropathological examination of various brain areas using standard staining procedures coupled with the western blot demonstration of PrPsc.
Results
Antibodies Screening
Step 1
When the 50 antibodies were screened using the 3F4 reference procedure that includes four pretreatments, we found a strong immunoreactivity with three antibodies that had been produced using human recombinant PrP (4F2, 3B5, 12F10), two produced using denatured scrapie-associated fibrils from 263K-infected Syrian hamsters (SAF34, SAF54) and one produced using ovine recombinant PrP (BAR233) (Table 3). Working concentrations varied between 0.8 and 2 μg/ml.
Step 2
When only one pretreatment (hydrated autoclaving) was used, a strong immunoreactivity was preserved for the six antibodies selected in step 1 (Table 3). As expected, when the number of pretreatments was reduced, the staining obtained using 3F4 antibody dramatically decreased.
Step 3
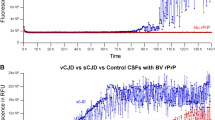
The 12F10, 3B5, 4F2, SAF34 and SAF54 antibodies used in automated procedure provided a strong immunoreactivity, regardless of the pattern of PrP deposits. The 3F4 reference antibody did not work efficiently in the Nexes automated system (71.4% sensitivity) (Table 4, Figure 2). Only the fatal familial insomnia (FFI) patient yielded negative results. There were two false-negative patients using 4F2 and SAF54 antibodies (90.5% sensitivity) and three false-negative patients using 3B5 and SAF34 antibodies (85.7% sensitivity). BAR233 showed the lowest sensitivity (47.6%). The 3B5, SAF34, 4F2 and SAF54 antibodies showed background on the neuropile and positive staining on the senile plaques of the three Alzheimer disease (AD) patients tested (Figure 2). When used in the automated system, 12F10 was the most efficient antibody: sensitivity (95.5%) and deposit patterns were similar to those of manual procedure of the 3F4 reference antibody (Table 4, Figures 2 and 3).
Immunostaining obtained with reference antibody 3F4 using manual procedure compared to 3F4, BAR233, 12F10, SAF34, SAf54, 4F2 and 3B5 antibodies using automated procedure. AD, Alzheimer disease patient; a.p., automated procedure; Cv, cerebellar vermis; FC, frontal isocortex; GSS, Gerstmann–Sträussler–Scheinker syndrome with the P102L mutation; MM1, methionine homozygosity at codon 129 of PRNP and a PrPsc type 1; m.p., manual procedure; MV2, methionine/valine heterozygosity at codon 129 and a PrPsc type 2A; NND, patient dead from a non-neurological disease; sCJD, sporadic Creutzfeldt–Jakob disease. Scale bar=100 μm.
Immunostaining obtained with reference 3F4 antibody using a manual procedure compared to 12F10 antibody using automated procedure. AD, Alzheimer disease; Cv, cerebellar vermis; fCJD E200K, familial Creutzfeldt–Jakob disease with the E200K mutation; FC, frontal isocortex; iCJD, iatrogenic Creutzfeldt–Jakob disease after extractive growth hormone treatment; MV1, methionine/valine heterozygosity at codon 129 and a PrPsc type 1; NND, patient dead from a non-neurological disease; sCJD, sporadic Creutzfeldt–Jakob disease. Scale bar=100 μm.
Step 4
We compared the results of 3F4 manual procedure to the 12F10 automated one using 51 patients (Table 5, Figure 3). The sensitivity of the 12F10 procedure was slightly higher than that of the 3F4 (95.7 vs 91.3%). In this step, we observed the characteristic patterns of the various studied transmissible subacute encephalopathies forms, regardless of the immunostaining procedure (Figure 3).
Evaluation of the 12F10 Automated Fast Procedure in a Series of 300 Patients with a Suspected Diagnosis of Human Transmissible Subacute Encephalopathies
To further validate the use of 12F10 antibody in an automated procedure for the diagnosis of human transmissible subacute encephalopathies, we tested tissue sections from 300 brains of patients with suspected transmissible subacute encephalopathies. Neuropathological examination and western blot demonstration of proteinase K-resistant PrP confirmed the diagnosis of transmissible subacute encephalopathies in 169 patients out of the 300 suspected patients. Among these 169 patients, 161 were found positive in the frontal isocortex (n=142), the cerebellar vermis (n=159) or in both samples (n=140). Only eight patients were negative in the two examined areas (95.3% sensitivity) (Table 6). The 12F10 automated procedure was efficient for the detection of PrPsc deposits in all forms of studied transmissible subacute encephalopathies: sporadic Creutzfeldt–Jakob disease (sCJD) (n=147), iatrogenic CJD (iCJD) after extractive growth hormone treatment (n=5), variant CJD (vCJD) (n=3), and familial CJD (fCJD) (n=14) with various mutations (n=10). Only one out of the 131 patients with non-prion diseases was immunopositive (99.2% specificity).
Evaluation of the 12F10 Automated Fast Procedure in a Series of 47 Tonsil Biopsies from Patients with a Suspected Diagnosis of vCJD
To further validate the use of 12F10 antibody in an automated procedure for the diagnosis of vCJD, we tested tissue sections from 47 tonsils of patients with suspected vCJD. Among these 47 patients, the final diagnosis was definite vCJD in 7 patients, probable vCJD in 2 and alternative diagnosis in 38. The 12F10 automated fast procedure showed positive results in all patients with definite or probable vCJD (Table 6). No positive staining was observed in the tonsils of patients with an alternative diagnosis. These results matched perfectly those of western blot assay after proteinase K digestion performed on tonsil samples from each patient.
Discussion
Identification of PrPsc in the central nervous tissue by PrP immunohistochemistry is a widely used technique for the diagnosis of human transmissible subacute encephalopathies. In this study, we compared 51 monoclonal antibodies (including the gold standard 3F4 antibody) and we developed a fast, reliable and automated procedure for PrP immunohistochemistry, allowing the diagnosis of the disease in more than 95% of the patients with a 99% specificity. Two brain areas were studied. The cerebellar vermis proved to be more often positive than the frontal isocortex.
It is worth noting that the epitopes of the antibodies that worked with limited pretreatment were located within the 59–161 region of the protein. The epitope 59–89 (4F2, 3B5 and SAF34 antibodies) and epitopes 144-152 (12F10), 157-161 (SAF54) and 141-151 (BAR233) flank the hydrophobic and potentially transmembrane domain of the protein (112–135).
In a previous study, Hainfellner et al24 have described an accumulation of PrP in senile plaques in some patients with AD. We also observed a PrP immunostaining in β-amyloid deposits of AD patients using 4F2, 3B5, SAF34 and SAF54 antibodies (Figure 2). This probably corresponded to nonspecific labelling because (i) all these antibodies produced a strong nonspecific background in the neuropile, and (ii) all the studied AD patients were PrP positive using these antibodies. However, from our experience, 3F4 and 12F10 antibodies can also stain β-amyloid plaques in some AD patients (Figure 4). As previously reported, no PrPsc was detected using western blot in these cases. The mechanics of co-aggregation of PrPc together with β-amyloid plaque compounds remains to be established.
Few monoclonal antibodies were effective when only one epitope-retrieving procedure (hydrated autoclaving) was used. This is less time-consuming and induces less tissue alterations, as illustrated in Figure 5. It also preserves epitopes of other proteins, which is beneficial for double immunostaining. The 12F10 antibody could be used at 37°C in an automated procedure and provided the most sensitive and specific results. By comparison, the 12F10 automated method was, in our hands, more sensitive than the other procedures using the reference 3F4 monoclonal antibody, which is not suitable for this automated system. Our results confirm and extend the results of Kovacs et al,25 who identified 12F10 as a potentially useful antibody for immunodiagnosis, owing to a reduction in the number of pretreatments and a speeding up of the procedure using a fast immunodiagnosis automate. In addition, we demonstrated the usefulness of this procedure in a very large panel of patients affected by a high diversity of human prion disorders.
It is worth noting that PrP immunostaining interpretation is sometimes difficult because PrP deposits may be very focal within the specimen. This is the case for synaptic deposits in the cerebellar or frontal cortex of some sCJD patients with methionine homozygosity and type 1 PrPsc (Figure 6). Of the 169 patients with prion diseases, 12 (7.1%) showed such focal deposits that may be difficult to identify for a pathologist who has not been trained in this technique of PrP immunohistochemistry.
This fast, automated procedure can be used for lymphoid tissue such as tonsil biopsy (nine tonsils from confirmed vCJD patients showed positive results consistently with the presence of type 2B PrPsc on western blot) (Figure 7).
Among 161 patients with transmissible subacute encephalopathies, only 8 showed negative immunohistochemical results (one patient with FFI, two fCJD patients associated with the D178N-129M or the V180I mutation and five patients with sCJD). The low level of PrPsc accumulation in FFI patients is a well-documented phenomenon.26 In all the eight transmissible subacute encephalopathy patients with negative immunohistochemical results, the western blot study yielded a positive result. Different methods have been developed, such as conformation-dependent immunoassay (CDI)27 or protein misfolding cyclic amplification (PMCA),28, 29 to increase the sensitivity of PrPsc detection. How these methods could improve routine diagnosis procedure in human pathology has to be confirmed using large case–control series. In terms of diagnosis of human transmissible subacute encephalopathies, developing highly sensitive methods such as CDI and PMCA may be more adapted to prion detection in peripheral tissue where very low PrPsc levels do occur (human blood). Considering our results using a fast (2 h), automated and highly sensitive procedure, more complex protocols would only slightly increase the sensitivity of detection. In addition, this procedure can be routinely applied in a general pathology laboratory.
In conclusion, after screening a large panel of anti-PrP monoclonal antibodies, we developed a fast, reliable and automated procedure for PrPsc immunohistochemistry in human tissue sections. It allows a high sensitivity and specificity for the diagnosis of human prion diseases. The morphological information is preserved thanks to a simplified antigen-retrieving procedure. This procedure was effective to detect PrPsc in the brain of almost all forms of human transmissible subacute encephalopathies. It can be successfully applied in lymphoid tissue.
References
Ironside JW . Prion diseases in man. J Pathol 1998;186:227–234.
Kretzschmar HA, Ironside JW, Dearmond SJ, et al. Diagnostic criteria for sporadic Creutzfeldt–Jakob disease. Arch Neurol 1996;53:913–920.
Kitamoto T, Ogomori K, Tateishi J, et al. Formic acid pre-treatment enhances immunostaining of cerebral and systemic amyloids. Lab Invest 1987;57:230–236.
DeArmond SJ, Mobley WC, Demott DL, et al. Changes in the localization of brain prion proteins during scrapie infection. Neurology 1987;37:1271–1280.
Guiroy D, Yanagihara R, Gajdusek D . Localization of amyloidogenic proteins and sulfated glycosaminoglycans in nontransmissible and transmissible cerebral amyloidoses. Acta Neuropathol 1991;82:87–92.
Doi-Yi R, Kitamoto T, Tateishi J . Immunoreactivity of cerebral amyloidosis is enhanced by protein denaturation treatments. Acta Neuropathol 1991;82:260–265.
Kitamoto T, Shin RW, Dohura K, et al. Abnormal isoform of prion proteins accumulates in the synaptic structures of the central nervous system in patients with Creutzfeldt–Jakob disease. Am J Pathol 1992;140:1285–1294.
Hashimoto K, Mannen T, Nukina N . Immunohistochemical study of kuru plaques using antibodies against synthetic prion protein peptides. Acta Neuropathol 1992;83:613–617.
Haritani M, Spencer YI, Wells GA . Hydrated autoclave pre-treatment enhancement of prion protein immunoreactivity in formalin-fixed bovine spongiform encephalopathy-affected brain. Acta Neuropathol (Berl) 1994;87:86–90.
Hayward P, Bell J, Ironside J . Prion protein immunocytochemistry: reliable protocols for the investigation of Creutzfeldt–Jakob disease. Neuropathol Appl Neurobiol 1994;20:375–383.
Macdonald ST, Sutherland K, Ironside JW . A quantitative and qualitative analysis of prion protein immunohistochemical staining in Creutzfeldt–Jakob disease using four anti prion protein antibodies. Neurodegeneration 1996;5:87–94.
Kascsak RJ, Rubenstein R, Merz PA, et al. Mouse polyclonal and monoclonal antibody to SAF (PrP) protein. J Virol 1987;61:3688–3693.
Bell JE, Gentleman SM, Ironside JW, et al. Prion protein immunocytochemistry—UK five centre consensus report. Neuropathol Appl Neurobiol 1997;23:26–35.
Privat N, Sazdovitch V, Seilhean D, et al. PrP immunohistochemistry: different protocols, including a procedure for long formalin fixation, and a proposed schematic classification for deposits in sporadic Creutzfeldt–Jakob disease. Microsc Res Tech 2000;50:26–31.
Feraudet C, Morel N, Simon S, et al. Screening of 145 anti-PrP monoclonal antibodies for their capacity to inhibit PrPSc replication in infected cells. J Biol Chem 2005;280:11247–11258.
Morel N, Simon S, Frobert Y, et al. Selective and efficient immunoprecipitation of the disease-associated form of the prion protein can be mediated by nonspecific interactions between monoclonal antibodies and scrapie-associated fibrils. J Biol Chem 2004;279:30143–30149.
Reik LM, Maines SL, Ryan DE, et al. A simple, non-chromatographic purification procedure for monoclonal antibodies. Isolation of monoclonal antibodies against cytochrome P450 isozymes. J Immunol Methods 1987;100:123–130.
Budka H, Aguzzi A, Brown P, et al. Neuropathological diagnostic criteria for Creutzfeldt–Jakob disease (CJD) and other human spongiform encephalopathies (prion diseases). Brain Pathol 1995;5:459–466.
Parchi P, Castellani R, Capellari S, et al. Molecular basis of phenotypic variability in sporadic Creutzfeldt–Jakob disease. Ann Neurol 1996;39:767–778.
Schulz-Schaeffer WJ, Giese A, Windl O, et al. Polymorphism at codon 129 of the prion protein gene determines cerebellar pathology in Creutzfeldt–Jakob disease. Clin Neuropathol 1996;15:353–357.
Hauw JJ, Sazdovitch V, Laplanche JL, et al. Neuropathologic variants of sporadic Creutzfeldt–Jakob disease and codon 129 of PrP gene. Neurology 2000;54:1641–1646.
Hilton DA, Fathers E, Edwards P, et al. Prion immunoreactivity in appendix before clinical onset of variant Creutzfeldt–Jakob disease. Lancet 1998;352:703–704.
Hilton DA, Ghani AC, Conyers L, et al. Prevalence of lymphoreticular prion protein accumulation in UK tissue samples. J Pathol 2004;203:733–739.
Hainfellner J, Wanschitz J, Jellinger K, et al. Coexistence of Alzheimer-type neuropathology in Creutzfeldt–Jakob disease. Acta Neuropathol 1998;96:116–122.
Kovacs GG, Head MW, Hegyi I, et al. Immunohistochemistry for the prion protein: comparison of different monoclonal antibodies in human prion disease subtypes. Brain Pathol 2002;12:1–11.
Montagna P, Gambetti P, Cortelli P, et al. Familial and sporadic fatal insomnia. Lancet Neurol 2003;2:167–176.
Safar JG, Geschwind MD, Deering C, et al. Diagnosis of human prion disease. Proc Natl Acad Sci USA 2005;102:3501–3506.
Castilla J, Saa P, Soto C . Detection of prions in blood. Nat Med 2005;11:982–985.
Saborio GP, Permanne B, Soto C . Sensitive detection of pathological prion protein by cyclic amplification of protein misfolding. Nature 2001;411:810–813.
Acknowledgements
This work was supported in part by the GIS Prions and the EU Network of Excellence Neuroprion. We thank Agueda Matos and Emilie Morain for helpful technical assistance and Professor Gerhard Hunsmann (German Primate Centre, Goettingen, Germany) for hybridoma cells producing monoclonal antibodies directed against human recombinant PrP (4F2, 3B5, 12F10, 14D3, 8G8 and 11C6).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Privat, N., Laffont-Proust, I., Faucheux, B. et al. Human prion diseases: from antibody screening to a standardized fast immunodiagnosis using automation. Mod Pathol 21, 140–149 (2008). https://doi.org/10.1038/modpathol.3800994
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800994
Keywords
This article is cited by
-
Neuropathology of iatrogenic Creutzfeldt–Jakob disease and immunoassay of French cadaver-sourced growth hormone batches suggest possible transmission of tauopathy and long incubation periods for the transmission of Abeta pathology
Acta Neuropathologica (2018)
-
Prion protein
AfCS-Nature Molecule Pages (2009)
-
Cerebral amyloid angiopathy with co-localization of prion protein and beta-amyloid in an 85-year-old patient with sporadic Creutzfeldt–Jakob disease
Acta Neuropathologica (2008)