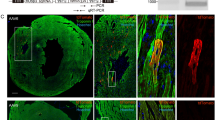
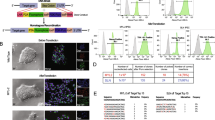
Abstract
Human embryonic stem cells (hESCs) may become important for cardiac repair due to their potentially unlimited ability to generate cardiomyocytes (CMCs). Moreover, genetic manipulation of hESC-derived CMCs would be a very promising technique for curing myocardial disorders. At the present time, however, inducing the differentiation of hESCs into CMCs is extremely difficult and, therefore, an easy and standardizable technique is needed to evaluate differentiation strategies. Vectors driving cardiac-specific expression may represent an important tool not only for monitoring new cardiac-differentiation strategies, but also for the manipulation of cardiac differentiation of ESCs. To this aim, we generated cardiac-specific lentiviral vectors (LVVs) in which expression is driven by a short fragment of the cardiac troponin-I proximal promoter (TNNI3) with a human cardiac α-actin enhancer, and tested its suitability in inducing tissue-specific gene expression and ability to track the CMC lineage during differentiation of ESCs. We determined that (1) TNNI3-LVVs efficiently drive cardiac-specific gene expression and mark the cardiomyogenic lineage in human and mouse ESC differentiation systems (2) the cardiac α-actin enhancer confers a further increase in gene-expression specificity of TNNI3-LVVs in hESCs. Although this technique may not be useful in tracking small numbers of cells, data suggested that TNNI3-based LVVs are a powerful tool for manipulating human ESCs and modifying hESC-derived CMCs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Boheler KR, Czyz J, Tweedie D, Yang HT, Anisimov SV, Wobus AM . Differentiation of pluripotent embryonic stem cells into cardiomyocytes. Circ Res 2002; 91: 189–201.
Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS et al. Embryonic stem cell lines derived from human blastocysts. Science 1998; 282: 1145–1147.
Kehat I, Kenyagin-Karsenti D, Snir M, Segev H, Amit M, Gepstein A et al. Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J Clin Invest 2001; 108: 407–414.
Mummery C, Ward D, van den Brink CE, Bird SD, Doevendans PA, Opthof T et al. Cardiomyocyte differentiation of mouse and human embryonic stem cells. J Anat 2002; 200: 233–242.
Eiges R, Schuldiner M, Drukker M, Yanuka O, Itskovitz-Eldor J, Benvenisty N . Establishment of human embryonic stem cell-transfected clones carrying a marker for undifferentiated cells. Curr Biol 2001; 11: 514–518.
Kay MA, Glorioso JC, Naldini L . Viral vectors for gene therapy: the art of turning infectious agents into vehicles of therapeutics. Nat Med 2001; 7: 33–40.
Bhavsar PK, Brand NJ, Yacoub MH, Barton PJ . Isolation and characterization of the human cardiac troponin I gene (TNNI3). Genomics 1996; 35: 11–23.
Ausoni S, Campione M, Picard A, Moretti P, Vitadello M, De Nardi C et al. Structure and regulation of the mouse cardiac troponin I gene. J Biol Chem 1994; 269: 339–346.
Bonci D, Cittadini A, Latronico MV, Borello U, Aycock JK, Drusco A et al. Advanced' generation lentiviruses as efficient vectors for cardiomyocyte gene transduction in vitro and in vivo. Gene Therapy 2003; 10: 630–636.
Cowan CA, Klimanskaya I, McMahon J, Atienza J, Witmyer J, Zucker JP et al. Derivation of embryonic stem-cell lines from human blastocysts. N Engl J Med 2004; 350: 1353–1356.
Xu C, Police S, Rao N, Carpenter MK . Characterization and enrichment of cardiomyocytes derived from human embryonic stem cells. Circ Res 2002; 91: 501–508.
Mummery C, Ward-van Oostwaard D, Doevendans P, Spijker R, van den Brink S, Hassink R et al. Differentiation of human embryonic stem cells to cardiomyocytes: role of coculture with visceral endoderm-like cells. Circulation 2003; 107: 2733–2740.
Mummery CL, van Achterberg TA, van den Eijnden-van Raaij AJ, van Haaster L, Willemse A, de Laat SW et al. Visceral-endoderm-like cell lines induce differentiation of murine P19 embryonal carcinoma cells. Differentiation 1991; 46: 51–60.
Lemonnier M, Buckingham ME . Characterization of a cardiac-specific enhancer, which directs {alpha}-cardiac actin gene transcription in the mouse adult heart. J Biol Chem 2004; 279: 55651–55658.
Kehat I, Khimovich L, Caspi O, Gepstein A, Shofti R, Arbel G et al. Electromechanical integration of cardiomyocytes derived from human embryonic stem cells. Nat Biotechnol 2004; 22: 1282–1289.
Klug MG, Soonpaa MH, Koh GY, Field LJ . Genetically selected cardiomyocytes from differentiating embryonic stem cells form stable intracardiac grafts. J Clin Invest 1996; 98: 216–224.
Menard C, Hagege AA, Agbulut O, Barro M, Morichetti MC, Brasselet C et al. Transplantation of cardiac-committed mouse embryonic stem cells to infarcted sheep myocardium: a preclinical study. Lancet 2005; 366: 1005–1012.
Heng BC, Cao T, Tong GQ, Ng SC . Potential utility of cell-permeable transcription factors to direct stem cell differentiation. Stem Cells Dev 2004; 13: 460–462.
Keller G . Embryonic stem cell differentiation: emergence of a new era in biology and medicine. Genes Dev 2005; 19: 1129–1155.
Xiong C, Tang DQ, Xie CQ, Zhang L, Xu KF, Thompson WE et al. Genetic engineering of human embryonic stem cells with lentiviral vectors. Stem Cells Dev 2005; 14: 367–377.
Beltrami AP, Barlucchi L, Torella D, Baker M, Limana F, Chimenti S et al. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 2003; 114: 763–776.
Oh H, Bradfute SB, Gallardo TD, Nakamura T, Gaussin V, Mishina Y et al. Cardiac progenitor cells from adult myocardium: homing, differentiation, and fusion after infarction. Proc Natl Acad Sci USA 2003; 100: 12313–12318.
Matsuura K, Nagai T, Nishigaki N, Oyama T, Nishi J, Wada H et al. Adult cardiac Sca-1-positive cells differentiate into beating cardiomyocytes. J Biol Chem 2004; 279: 11384–11391.
Urbanek K, Torella D, Sheikh F, De Angelis A, Nurzynska D, Silvestri F et al. Myocardial regeneration by activation of multipotent cardiac stem cells in ischemic heart failure. Proc Natl Acad Sci USA 2005; 102: 8692–8697.
Messina E, De Angelis L, Frati G, Morrone S, Chimenti S, Fiordaliso F et al. Isolation and expansion of adult cardiac stem cells from human and murine heart. Circ Res 2004; 95: 911–921.
Armstrong MT, Lee DY, Armstrong PB . Regulation of proliferation of the fetal myocardium. Dev Dyn 2000; 219: 226–236.
Barile L, Chimenti I, Gaetani R, Forte E, Miraldi F, Frati G et al. Cardiac stem cells: isolation, expansion and experimental use for myocardial regeneration. Nat Clin Pract Cardiovasc Med 2007; 4 (Suppl 1): S9–S14.
Sen A, Dunnmon P, Henderson SA, Gerard RD, Chien KR . Terminally differentiated neonatal rat myocardial cells proliferate and maintain specific differentiated functions following expression of SV40 large T antigen. J Biol Chem 1988; 263: 19132–19136.
Dani C, Smith AG, Dessolin S, Leroy P, Staccini L, Villageois P et al. Differentiation of embryonic stem cells into adipocytes in vitro. J Cell Sci 1997; 110 (Part 11): 1279–1285.
Savatier P, Lapillonne H, Jirmanova L, Vitelli L, Samarut J . Analysis of the cell cycle in mouse embryonic stem cells. Methods Mol Biol 2002; 185: 27–33.
Fraichard A, Chassande O, Bilbaut G, Dehay C, Savatier P, Samarut J . In vitro differentiation of embryonic stem cells into glial cells and functional neurons. J Cell Sci 1995; 108 (Part 10): 3181–3188.
Dull T, Zufferey R, Kelly M, Mandel RJ, Nguyen M, Trono D et al. A third-generation lentivirus vector with a conditional packaging system. J Virol 1998; 72: 8463–8471.
Ma Y, Ramezani A, Lewis R, Hawley RG, Thomson JA . High-level sustained transgene expression in human embryonic stem cells using lentiviral vectors. Stem Cells 2003; 21: 111–117.
Acknowledgements
This work was supported by grants from the Italian Ministry of University and Research, the European Community (EU FP6 Grant LSHM-CT-2005-018833, EUGeneHeart) for the part related to lentiviral vector generation, and private funds to GC. We thank the technical help from Antonio Addario, Alessandra Benetello, Marco Santonastasi and Domenico Vignone and Angela Pitisci. We are grateful to Lucio Barile, Elisa Messina and Alessandro Giacomello for the use of cardiac progenitor cells.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Gene Therapy website (http://www.nature.com/gt)
Rights and permissions
About this article
Cite this article
Gallo, P., Grimaldi, S., Latronico, M. et al. A lentiviral vector with a short troponin-I promoter for tracking cardiomyocyte differentiation of human embryonic stem cells. Gene Ther 15, 161–170 (2008). https://doi.org/10.1038/sj.gt.3303017
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3303017
Keywords
This article is cited by
-
CaMKII inhibition rectifies arrhythmic phenotype in a patient-specific model of catecholaminergic polymorphic ventricular tachycardia
Cell Death & Disease (2013)
-
Post-natal cardiomyocytes can generate iPS cells with an enhanced capacity toward cardiomyogenic re-differentation
Cell Death & Differentiation (2012)
-
Lentiviral vectors and cardiovascular diseases: a genetic tool for manipulating cardiomyocyte differentiation and function
Gene Therapy (2012)
-
Sigma-2 Receptor as Potential Indicator of Stem Cell Differentiation
Molecular Imaging and Biology (2012)
-
Physiological and tissue-specific vectors for treatment of inherited diseases
Gene Therapy (2011)