Abstract
Study design:
Cross-sectional and comparative investigation using quadriplegics (QP) and nondisabled subjects (ND).
Objective:
To evaluate cardiorespiratory responses during passive walking-like exercise (PWE) in QP.
Setting:
National Rehabilitation Center for Persons with Disabilities in Japan.
Method:
The subjects were seven male QP with complete lesion (age: 27.0±5.4, injured level: C6–C7) and six male ND (age: 26.3±4.5). Cardiorespiratory responses were measured until voluntary fatigue during PWE, the rhythmical activity of paralyzed lower limbs synchronized with arm movements.
Results:
There were no significant differences in oxygen consumption (V̇O2), pulmonary ventilation (V̇E), heart rate (HR) and oxygen pulse (O2 pulse) between QP and ND during PWE. ND showed increased ventilatory equivalent for oxygen (V̇E/V̇O2 ratio) during exercise, while QP showed a significantly greater respiratory rate (RR) during exercise than ND (P<0.05).
Conclusion:
PWE elicited an increase in V̇O2 with workload increment in QP similar to ND. However, higher RR suggested the intrinsic dysfunction of RR control during submaximal exercise in QP. From these results, it was thought that respiratory response would be the restriction factor of efficient oxygen transportation during PWE in QP.
Similar content being viewed by others
Introduction
For individuals with spinal cord injury (ISCI), it is difficult to improve their cardiorespiratory function and to activate the oxygen supply function by exercise training such as wheelchair or arm swinging exercise because of the characteristics of the obstacles.1, 2 In general, the peak oxygen uptake (peak V̇O2) is dependent on the level of spinal cord injury (SCI), and quadriplegics (QP) and high lesion paraplegics (PP) show a lower peak V̇O2 than nondisabled (ND) or low lesion PP.3, 4, 5 In addition, during submaximal exercise, QP and high lesion PP showed the reduced ventilation efficiency, stroke volume, venous return and sympathetic activity in comparison to ND or low lesion PP.5, 6
For such ISCI with low physiological responses to exercise, the exercise posture is an important factor enhancing the effect of aerobic training. McLean et al7 investigated the influence of body posture in training on aerobic capacity in ISCI and indicated that although improvements in aerobic capacity could be achieved by training in either a supine or a sitting posture, the supine posture had more effect on aerobic training than the sitting posture. One reason is that the supine posture is advantageous to ISCI circulation, because there is small amount of blood in the paralyzed leg compared to the sitting posture due to the lower effect of gravity.
In contrast, in a sitting posture during exercise, venous blood pools in the legs and abdomen, causing reduced filling pressure and diminished ventricular volume. Therefore, when QP and PP perform exercise in a sitting posture, blood is not efficiently redistributed to the working muscles.8, 9, 10, 11 To improve venous blood pooling during exercise in a sitting posture, some researchers have investigated the effect of passive leg exercise on circulation.9, 12 This exercise passively moves the paralyzed lower limbs, the blood stored in lower limbs returns to the heart, and stroke volume increases conjointly with the law of Frank-starling.9, 12
It is reported that functional electrical stimulation (FES) is also a useful method to decrease venous blood pooling in the legs, because FES increases the activity of muscle pumping and vasoconstriction in the legs.13, 14, 15 Bhambhani et al16 measured the deoxygenation of the quadriceps muscle during FES using QP and PP. They indicated that the muscle deoxygenation of paralyzed muscles occurred more quickly with metabolic responses in the paralyzed muscles. Mutton et al17 reported a significant increase in peak V̇O2 during hybrid exercise18, 19, 20 that combined upper arm exercise with FES in QP and PP when compared with only FES. Moreover, Raymond et al18 showed a higher oxygen intake and lower heart rate (HR) during hybrid exercise including arm-swinging exercise at a workload of 65% maximal oxygen uptake in PP. In addition, Hooker et al21 compared the respiratory and circulatory responses during hybrid exercise with those during submaximal arm-swinging exercise and FES leg-cycle exercise in QP. They showed higher pulmonary ventilation (V̇E), oxygen uptake (V̇O2) and carbon dioxide elimination during hybrid exercise than the other two exercises, but a higher stroke volume than only arm-swinging exercise. They concluded that hybrid exercise using whole-body exercise, including the paralyzed muscles, is effective in improving the cardiorespiratory function of ISCI.
More recently, it has been reported that standing gait exercise with orthoses has a good influence on cardiorespiratory function in ISCI.22 Faghri et al13, 23 investigated the physiological reaction of a standing posture on ISCI with and without FES. They showed stable cardiac output, stroke volume and total peripheral resistance (TPR) during 30 min standing with FES in both QP and PP. In contrast, during passive standing without FES, QP demonstrated significantly higher TPR and significantly lower systolic blood pressure and mean arterial pressure than PP. Faghri et al13, 23 also indicated that standing without FES was disadvantageous to the regulation of hemodynamics during posture change in QP.
When ISCI passively walked on a treadmill using body weight support equipment, they showed a similar electromyographic pattern in the paralyzed muscles to ND.24 Furthermore, Colombo et al25 obtained the same result during passive stepping using driven gait orthosis for C3 (incomplete) and C5 (complete). It is naturally expected that passive walking with arm exercise increases energy expenditure and oxygen supply to the arm is elevated. However, as far as we know, there are no studies investigating the cardiorespiratory responses of QP during passive walking-like exercise (PWE) when standing.
The purpose of this study, therefore, was to clarify respiratory and circuratory responses during PWE by a stepwise incremental method and to compare the results of QP with those of ND.
Methods
Subjects
Seven male patients with complete chronic QP and six ND male subjects volunteered to participate in this study. Table 1 lists their physical characteristics. The lesion in SCI was located between C6 and C7. All subjects regularly performed wheelchair sports, such as twin basketball, quad rugby and distance running, for more than 60 min a day and more than twice a week. No subject had a history of cardiovascular, metabolic, or pulmonary disease. Informed consent was obtained from all subjects before their participation in this study. The subject refrained from food, caffeine and nicotine for at least 3 h before testing. The study was approved by the Ethical Research Committee in the National Rehabilitation Center for Persons with Disabilities.
Testing protocols
All subjects performed an incremental exercise test on an Easy Stand Glider 6000 (Altimate Medical Int., Morton, MN, USA). The Easy Stand Glider 6000 is designed to strengthen both the upper and lower extremities while standing. It has a safety belt around the waist, a chest pad, hip guide and knee support. When the subject swings his arms back and forth, his legs simultaneously move passively just like walking. Tests were performed with the push and pull handle horizontal to the level of the shoulder joint and elbows slightly flexed at the point of maximal arm extension (Figure 1).
The subjects remained seated in a wheelchair or chair for at least 30 min. Baseline physiological measurements were recorded for the last 5 min during seating. They were subsequently guided with a metronome for reciprocal movement of the arms and legs while standing. Exercise commenced by swinging the arms back and forth at 20 times/min for 2 min. The swings were then increased to 10 times/min every 2 min until 50 times/min, and then 5 times/min every 2 min until exhaustion. The incremental exercise test was terminated when voluntary fatigue was attained. Voluntary fatigue was defined as the point at which the subject could no longer keep pace and his RPE was over 15. The experiment was carried out in a room with ambient temperature and relative humidity maintained at 22–25°C and 30–50%, respectively.
Cardiorespiratory measurements
Cardiorespiratory measurements were continuously monitored during the test using the gas analyzer of the metabolic system (Model AT-3000, Anima, Tokyo, Japan). The gas analyzer was calibrated using standard gas concentrations (16.1% oxygen, 5.01% carbon dioxide). The volume transducer was calibrated using a syringe calibrated to 2 l. The gas analyzer was programmed to present the following results: absolute V̇O2 (l/min), relative V̇O2 (ml/kg/min), respiratory rate (RR, times/min) and V̇E (l/min). The following variables were calculated from the oxygen pulse (O2 pulse, ml/beat) as the ratio between absolute V̇O2 and HR (beats/min), and the ventilatory equivalent for oxygen (V̇E/V̇O2 ratio, l/ml) as the ratio between V̇E and absolute V̇O2.
HR was recorded during the last 10 s at each work stage using a wireless monitor (Life Scope 8/Two, Nihon Koden, Tokyo, Japan). Blood was sampled from the earlobe during rest and exercise and blood lactate accumulation (LA, mmol/ml) was measured using a simplified blood lactate test meter (Lactate Pro™ LT-1710, Arckly, Inc., Kyoto, Japan). Blood sampling was conducted immediately after rest and within 30 s after each workload. The sampling time was within 20 s.
Statistical analysis
All variables were expressed as the mean±SD. Two-way repeated analysis of variance was used to compare the difference between groups. P-values <0.05 were considered significant.
Results
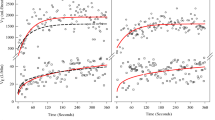
Figure 2a shows the relationship between V̇O2 and workload in QP and ND. V̇O2 within the workload of 60 times/min varied little and almost no rise was observed. At a workload of 65 times/min, V̇O2 started to increase rapidly. At any workload, there was no significant difference between the groups. Changes in V̇E and LA over time during exercise were similar to those in V̇O2. No significant differences were found in V̇E and LA between QP and ND.
Change in oxygen consumption (a), heart rate (b), and oxygen pulse (c) during passive walking with arm swinging exercise in persons with Quadriplegics (•) and nondisabled (▵). Although there was no difference of oxygen consumption between QP and ND, oxygen transportation of QP was inferior to ND during the exercise. *P<0.05: compared with nondisabled subjects
HR increased with the workload increment in both groups (Figure 2b). When the workloads were between 40 and 70 times/min, there was little increase in HR for QP and ND. Although QP showed a higher HR than ND during rest and exercise, significant difference was only found at a workload of 30 times/min. Figure 2c indicates the O2 pulse over the time course of exercise. In ND, the O2 pulse remained unchanged from the beginning of exercise to a workload of 60 times/min. Subsequently, the O2 pulse of QP increased rapidly. In contrast, QP showed a gradual increase in the O2 pulse linearly with the workload. The O2 pulse of QP was higher than that of ND at any workload and significant difference was only found at the beginning of exercise (20 times/min).
Figure 4a illustrates the relationship of HR to V̇O2 in QP and ND. With increasing V̇O2, HR linearly and significantly increased in both groups. When V̇O2 was around 500 ml, HR of QP was apparently greater than ND.
There existed a great difference in RR between QP and ND during exercise (Figure 3a). ND had almost unchanged RR during exercise. In contrast to ND, QP showed increased RR over the time course of exercise. There were significant differences in RR between the groups at any workload except the lowest (20 times/min) and the highest (95 times/min). Figure 3b shows the V̇E/V̇O2 ratio during the incremental exercise test in QP and ND. The V̇E/V̇O2 ratio of QP was higher than that of ND at any workload and significant differences were found at higher workloads of 75 and 85 times/min.
Change in respiratory rate (a) and ventilation for oxygen consumption (b) during passive walking with arm swinging exercise in persons with Quadriplegics (•) and nondisabled (▵). Ventilation efficiency showed significant decrease in QP as compared with ND. *P<0.05, #P<0.01: compared with nondisabled subjects
The relationship of RR to V̇E is illustrated in Figure 4b with regression lines. The regression line of QP shifted to the upper side of ND, indicating that QP required more RR to achieve the same V̇E as ND.
Discussion
In this study, there were no significant differences in V̇O2 and V̇E during standing exercise between QP and ND (Figure 2a). Some investigators showed that the cardiorespiratory responses of QP during arm exercise were relatively lower than ND and PP,4, 5, 26 suggesting that the cardiorespiratory responses of ISCI are largely dependent on the level of SCI.3, 6, 27 These results were obtained from arm-swinging exercise or wheelchair ergometer exercise requiring mainly upper limb activity in a sitting posture. It may be considered that the exercise while sitting, using only the upper limbs, influences the V̇O2 of ISCI. Hopman et al28 demonstrated that peak V̇O2 significantly increased in a supine posture during maximal arm-swinging exercise in comparison with a sitting posture. In addition, McLean et al7 compared the power output (PO) of QP during an intermittent progressive peak exercise test in a sitting posture with that in a supine posture. As a result, they reported higher PO in a sitting than supine posture. These investigations suggest that cardiorespiratory responses during exercise are affected by exercise postures in QP.
Cardiorespiratory responses during exercise in QP change with passive exercise by the paralyzed limbs in addition to the exercise posture. Pitetti et al29 carried out arm-swinging exercise in ISCI and ND with lower body positive pressure (LBPP). They found a significant increase in V̇O2, V̇E and work rate during arm-swinging exercise with LBPP compared to without LBPP. Furthermore, there were no differences in V̇O2, V̇E and work rate between ISCI and ND during exercise with LBPP. From these results, Pitetti et al29 suggested that for ISCI, LBPP augmented the exercise capacity by preventing the redistribution of blood to the lower extremities. Hopman et al27, 28 investigated the effects of exercise posture, wearing an antigravity suit (anti-G suit), elastic stocking and abdominal binder, and FES on blood redistribution and circulatory responses in QP and PP. They demonstrated that V̇O2 and HR decreased by wearing an anti-G suit and increased by FES, and increased by wearing elastic stockings and FES during submaximal exercise. In contrast, during maximal exercise, only FES increased V̇O2 and HR. From these results, Hopman et al27, 28 suggested that these methods of circulatory redistribution have different working mechanisms and the effects are dependent on the SCI level probably because of differences in active muscle mass, sympathetic impairment and blood pressure values. Furthermore, V̇O2 increased significantly during FES exercise of lower limbs in comparison with at rest30 and V̇O2 during hybrid exercise was higher than that during arm-swinging exercise or leg cycle exercise by FES.17, 21, 31
The findings using the passive activity of paralyzed lower limbs and FES in addition to arm exercise clearly demonstrated good effects on cardiorespiratory responses and improving the efficiency of their oxygen utilization in ISCI. However, these studies were mostly performed in a sitting posture. If ISCI perform exercise in a standing posture, the cardiorespiratory responses may be different from those in a sitting posture. Nash et al32 showed significant increases in V̇O2, V̇E and HR during PWE when standing by using lobotic-assisted locomotion in QP with a lesion level of C3–C4. In addition, Dietz et al24 identified electromyographic activities of the musculus tibialis anterior and musculus soleus during passive walking on a treadmill in QP and PP. These investigations suggested that PWE in a standing posture with arm exercise in QP facilitated cardiorespiratory responses. Our study showed the same V̇O2 between QP and ND, indicating that the rhythmical activity of paralyzed limb increased V̇O2.
In the present study, QP showed a significantly higher RR from 30 to 90 times/min (Figure 3b), and QP increased RR to the equivalent V̇E of the ND (Figure 4b). Coutts et al6 found lower respiratory parameters such as V̇E and ventilation equivalent in QP than in PP during submaximal arm-swinging exercise. The ventilation equivalent is generally considered to be a measure of breathing efficiency, and decreases during submaximal exercise are associated with increased tidal volume and relative decreases in dead space ventilation.6 Bhambhani et al16 demonstrated that the V̇E/V̇O2 ratio in ISCI, including QP, is lower during FES cycle exercise than ND, indicating that the ventilatory efficiency of ISCI is inferior to that of ND. In good agreement with the data of Bhambhani et al,16 we found a lower V̇E/V̇O2 ratio of QP in comparison with ND. There was no difference in LA between QP (2.9±1.1 mmol/l) and ND (3.0±1.6 mmol/l) during peak exercise, indicating that the increase in RR was not related to metabolic factors, because LA stimulates the respiratory center and consequently increases the elimination of carbon dioxide. Furthermore, it has been reported that expiratory muscle contraction is influenced by sympathetic nerve activity more than muscle metaboreflex.33
In AB, the impulse from the motor area of the cerebral cortex via the center of breathing adjusts the ventilation equivalent to the exercise intensity during exercise.34 In addition, it has been demonstrated that respiration is regulated by transmitting the afferent information from activity muscles to the center.35 In QP, the afferent information from the agonist of the upper limbs was transmitted to the center during exercise; however, the nerve impulse of the ventilatory regulation corresponding to the workload is not sent to the respiratory muscles. That is, it is believed that the afferent information to the center increased excessively in our tests. Restrictive ventilatory impairment may disturb respiratory regulation during exercise in QP.
Green36 found that excitations of the stretch receptors stimulated by the stretch reflex (the Hering–Breuer reflex) in the lung were relayed via the vagus nerve to the medullary respiratory center, leading to a reflex decrease in tidal volume. This reflex was not found in normal adults and in babies with undeveloped respiratory-related muscles and in some animals.36
We hypothesized that QP with restrictive ventilatory impairment might show a condition similar to that of the babies in Green's study. To compensate for the decrease in tidal volume by the Hering–Breuer reflex and to achieve the same ventilation as ND, QP increased RR from the commencement of exercise. Specifically, the increase in RR in QP could be a result of the increased afferent information from the agonist to the respiratory center and the increased reflex induced by restrictive ventilatory impairment.
Some investigators have shown a lower maximal HR below 110 beats/min in QP than that of PP and ND.4, 37 Bhambhani et al16 found no significant difference of HR in ISCI including QP during FES exercise from that at rest. A lower HR during exercise is naturally expected in QP because of sympathetic activity dysfunction controlling the heart. In this study, however, during peak exercise, the HR of QP was higher than the data of Bhambhani et al,16 and there were no significant differences of HR between QP and ND similar to V̇O2 (Figure 2b). Muraki et al12 reported a significant increase in stroke volume and cardiac output without a rise of HR during passive leg cycle exercise in PP. They suggested the promotion of venous return related to the lengthening and shortening of the paralyzed muscle without tension in the lower limbs.
In this study, HR in QP increased during PWE. This is not consistent with the findings of Faghri et al,23 who found no increase of HR in QP during FES when standing. On the other hand, they reported increased TPR, which could be a compensatory mechanism to control the significant drop in blood pressure occurring during standing in QP. Hooker et al21 investigated cardiorespiratory responses during arm-swinging exercise, FES leg-cycle exercise and hybrid exercise in QP, and revealed that V̇O2, V̇E and HR were higher during hybrid exercise than the other two exercises and there was no significant difference in stroke volume between the hybrid exercise and FES leg-cycle exercise. Dela et al14 reported that although HR increased immediately after the commencement of FES and attained a steady state in ND, QP showed a delay in the HR increment. HR responses in QP may be attributable to arterial baroreceptors that elevate HR in QP with the lower blood pressure developed during exercise.14
In this study, there was no significant difference in HR between QP and ND during peak passive walking. On the other hand, during submaximal exercise, a clear difference in HR was found between QP and ND. In ND, HR increased linearly with workload increment, while it increased in QP from the commencement of exercise to 40 times/min and HR increased gradually, showed a steady state between 50 and 75 times/min, increasing remarkably after 80 times/min (Figure 2b). It could be expected that HR in QP increased by the activation of arterial baroreceptors in compensation for deficient blood distribution from the beginning of exercise to 40 times/min, while between 50 and 75 times/min, HR showed a steady state because blood was distributed sufficiently to agonists (Figure 2c).
In conclusion, PWE, the rhythmical activity of paralyzed lower limbs synchronized with arm movements, elicited an increase in V̇O2 in QP similar to ND. However, higher RR suggested the intrinsic dysfunction of RR control during submaximal exercise in QP. From these results, it was thought that respiratory responses would restrict the efficiency of oxygen transportation during PWE in QP.
References
Hjeltnes N, Wallberg-Henriksson H . Improved work capacity but unchanged peak oxygen uptake during primary rehabilitation in tetraplegic patients. Spinal Cord 1998; 36: 691–698.
Birk TJ, Nieshoff E, Gray G, Steeby J, Jablonski K . Metabolic and cardiopulmonary responses to acute progressive resistive exercise in a person with C4 spinal cord injury. Spinal Cord 2001; 39: 336–339.
Bernard PL, Mercier J, Varry A, Prefaut C . Influence of lesion level on the cardioventilatory adaptations in paraplegic wheelchair athletes during muscular exercise. Spinal Cord 2000; 38: 16–25.
Coutts KD, Rhodes EC, McKenzie DC . Maximal exercise responses of tetraplegics and paraplegics. J Appl Physiol 1983; 55: 479–482.
Schimid A et al. Catecholamines, heart rate, and oxygen uptake during exercise in persons with spinal cord injury. J Appl Physiol 1998; 85: 635–641.
Coutts KD, Rhodes EC, McKenzie DC . Submaximal exercise responses of tetraplegics and pareplegics. J Appl Physiol 1985; 59: 237–241.
McLean KP, Skinner JS . Effect of body training position on outcomes of an aerobic training study on individuals with quadriplegia. Arch Phys Med Rehabil 1995; 76: 139–150.
Davis GM . Exercise capacity of individuals with paraplegia. Med Sci Sports Exerc 1993; 25: 423–432.
Hopman MT . Circulatory responses during arm exercise in individuals with paraplegia. Int J Sports Med 1994; 15: 126–131.
Mathias CJ, Frankel HL . Cardiovascular control in spinal man. Ann Rev Physiol 1988; 50: 577–592.
Olive JL, Mccully KK, Dudley GA . Blood flow response in individuals with incomplete spinal cord injuries. Spinal Cord 2002; 40: 639–645.
Muraki S, Yamasaki M, Ehara Y, Kikuchi K, Seki K . Cardiovascular and respiratory responses to passive leg cycle exercise in people with spinal cord injuries. Eur J Appl Physiol 1996; 74: 23–28.
Faghri PD, Yount J . Electrically induced and voluntary activation of physiologic muscle pump: a comparison between spinal cord-injured and able-bodied individuals. Clin Rehabil 2002; 16: 878–885.
Dela F et al. Cardiovascular control during exercise insights from spinal cord-injury humans. Circulation 2003; 107: 2127–2133.
Kjaer M, Pott F, Mohr T, Linkis P, Tornoe P, Secher NH . Heart rate during exercise with leg vascular occlusion in spinal cord-injured humans. J Appl Physiol 1999; 86: 806–811.
Bhambhani Y, Tuchak C, Burnham R, Jeon J, Maikala R . Quadriceps muscle deoxygenation during functional electrical stimulation in adults with spinal cord injury. Spinal Cord 2000; 38: 630–638.
Mutton DL, Scremin AME, Barstow TJ, Scott MD, Kunkel CF, Cable TG . Physiologic responses during functional electrical stimulation leg cycle and hybrid exercise in spinal cord injured subjects. Arch Phys Med Rehabil 1997; 78: 712–718.
Raymond J, Davis GM, Fahey A, Climstein M, Sutton JR . Oxygen uptake and heart rate responses during arm versus combined arm+ES-induced leg exercise in people with paraplegia. Spinal Cord 1997; 35: 680–685.
Laskin JJ et al. Electrical stimulation-assisted rowing exercise in spinal cord injured people. A pilot study. Paraplegia 1993; 31: 534–541.
Wheeler GD et al. Functional electric stimulation-assisted rowing: increasing cardiovascular fitness through functional electric stimulation rowing training in persons with spinal cord injury. Arch Phys Med Rehabil 2002; 83: 1093–1099.
Hooker SP et al. Metabolic and hemodynamic responses to concurrent voluntary arm crank and electrical stimulation leg cycle exercise in quadriplegics. J Rehabil Res Dev 1992; 29: 1–11.
Kawashima N, Sone Y, Nakazawa K, Akai M, Yano H . Energy expenditure during walking with weight-bearing control (WBC) orthosis in thoracic level of paraplegic patients. Spinal Cord 2003; 41: 506–510.
Faghri PD, Yount JP, Pesce WJ, Seetharama S, Votto JJ . Circulatory hypokinesis and functional electric stimulation during standing in persons with spinal cord injury. Arch Phys Med Rehabil. 2001; 82: 1587–1595.
Dietz V, Nakazawa K, Wirz M, Erni T . Level of spinal cord lesion determines locomotor activity in spinal man. Exp Brain Res 1999; 128: 405–409.
Colombo G, Wirz M, Dietz V . Driven gait orthosis for improvement of locomotor training in paraplegic patients. Spinal Cord 2001; 39: 252–255.
Irizawa M, Yamasaki M, Muraki S, Komura T, Seki K, Kikuchi K . Relationship between heart rate and oxygen uptake during submaximal arm cranking in paraplegics and quadriplegics. Ann Physiol Anthropol 1994; 13: 275–280.
Hopman MT, Monroe M, Dueck C, Phillips WT, Skinner JS . Limits to maximal performance in individuals with spinal cord injury. Int J Sports Med 1998; 19: 98–103.
Hopman MT, Monroe M, Dueck C, Phillips WT, Skinner JS . Blood redistribution and circulatory responses to submaximal arm exercise in persons with spinal cord injury. Scand J Rehabil Med 1998; 30: 167–174.
Pitetti KH, Barrett PJ, Campbell KD, Malzahn DE . The effect of lower body positive pressure on the exercise capacity of individuals with spinal cord injury. Med Sci Sports Exerc 1994; 24: 463–468.
Figoni SF et al. Physiologic responses of paraplegics and quadriplegics to passive and active leg cycle ergometry. J Am Paraplegia Soc 1990; 13: 33–39.
Raymond J, Davis GM, Climstein M, Sutton JR . Cardiorespiratory responses to arm cranking and electrical stimulation leg cycling in people with paraplegia. Med Sci Sports Exerc 1999; 31: 822–828.
Nash MS, Jacobs PL, Johnson BM, Field-Fote’ E . Metabolic and cardiac responses to robotic-assisted locomotion in motor-complete tetraplegia: a case report. J Spinal Med 2004; 27: 78–82.
Derchak PA, Shell AW, Morgan BJ, Dempsey JA . Effects of expiratory muscle work on muscle sympathetic nerve activity. J Appl Physiol 2002; 92: 1539–1552.
Krough A, Lindhard J . The reguration of respiration and circulation during the initial stages of muscular work. J Physiol 1913; 47: 112–136.
Morikawa T et al. Afferent and cardiodynamic drives in the early phase of exercise hyperpnea in humans. J Appl Physiol 1989; 67: 2006–2013.
Green JH . The Autonomic Nervous System and Exercise. Chapman & Hall: London 1990, pp 126–128.
Van Loan MD, McCluer S, Loftin JM, Boileau RA . Comparison of physiological responses to maximal arm exercise among able-bodied, paraplegics and quadriplegics. Paraplegia 1987; 25: 397–405.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Higuchi, Y., Kitamura, S., Kawashima, N. et al. Cardiorespiratory responses during passive walking-like exercise in quadriplegics. Spinal Cord 44, 480–486 (2006). https://doi.org/10.1038/sj.sc.3101875
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101875
Keywords
This article is cited by
-
Results of the first interim analysis of the RAPPER II trial in patients with spinal cord injury: ambulation and functional exercise programs in the REX powered walking aid
Journal of NeuroEngineering and Rehabilitation (2017)
-
Functional Electrical Stimulation: Cardiorespiratory Adaptations and Applications for Training in Paraplegia
Sports Medicine (2015)