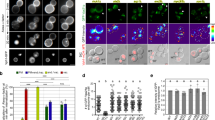
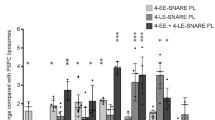
Abstract
Intracellular membrane docking and fusion requires the interplay between soluble factors and SNAREs. The SNARE hypothesis1 postulates that pairing between a vesicular v-SNARE and a target membrane z-SNARE is the primary molecular interaction underlying the specificity of vesicle targeting as well as lipid bilayer fusion. This proposal is supported by recent studies using a minimal artificial system2. However, several observations demonstrate that SNAREs function at multiple transport steps and can pair promiscuously, questioning the role of SNAREs in conveying vesicle targeting3,4,5,6. Moreover, other proteins have been shown to be important in membrane docking or tethering7,8,9. Therefore, if the minimal machinery is defined as the set of proteins sufficient to reproduce in vitro the fidelity of vesicle targeting, docking and fusion as in vivo, then SNAREs are not sufficient to specify vesicle targeting. Endosome fusion also requires cytosolic factors and is regulated by the small GTPase Rab5 (refs 10,11,12,13,14,15,16,17,18,19,20). Here we show that Rab5-interacting soluble proteins can completely substitute for cytosol in an in vivo endosome-fusion assay, and that the Rab5 effector EEA1 is the only factor necessary to confer minimal fusion activity. Rab5 and other associated proteins seem to act upstream of EEA1, implying that Rab5 effectors comprise both regulatory molecules and mechanical components of the membrane transport machinery. We further show that EEA1 mediates endosome docking and, together with SNAREs, leads to membrane fusion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rothman, J. E. Mechanisms of intracellular protein transport. Nature 372, 55–63 (1994).
Weber, T. et al. SNAREpins: Minimal machinery for membrane fusion. Cell 92, 759–772 (1998).
Robinson, L. M. & Martin, T. F. J. Docking and fusion in neurosecretion. Curr. Opin. Cell Biol. 10, 483–492 (1998).
von Mollard, G. F., Nothwehr, S. F. & Stevens T. H. The yeast v-SNARE Vti1p mediates two vesicle transport pathways through interactions with the t-SNAREs Sed5p and Pep12p. J. Cell Biol. 137, 1511–1524 (1997).
Holthuis, J. C., Nichols, B. J., Dhruvakumar, S. & Pelham, H. R. Two syntaxin homologues in the TGN/endosomal system of yeast. EMBO J. 17, 113–126 (1998).
Hay, J. C. et al. Localization, dynamics, and protein interactions reveal distinct roles for ER and Golgi SNAREs. J. Cell Biol. 141, 1489–1502 (1998).
Sapperstein, S. K., Lupashin, V. V., Schmitt, H. D. & Waters, M. G. Assembly of the ER to Golgi SNARE complex requires Uso1p. J. Cell Biol. 132, 755–767 (1996).
Cao, X., Ballew, N. & Barlowe, C. Initial docking of ER-derived vesicles requires Uso1p and Ypt1p but is independent of SNARE proteins. EMBO J. 17, 2156–2165 (1998).
VanRheenen, S. M., Cao, X., Lupashin, V. V., Barlowe, C. & Waters, G. M. Sec35p, a novel peripheral membrane protein, is required for ER to Golgi vesicle docking. J. Cell Biol. 141, 1107–1119 (1998).
Novick, P. & Zerial M. The diversity of Rab proteins in vesicle transport. Curr. Opin. Cell Biol. 9, 496–504 (1997).
Søgaard, M. et al. ARab protein is required for the assembly of SNARE complexes in the docking of transport vesicles. Cell 78, 937–948 (1994).
Lian, J. P., Stone, S., Jiang, Y., Lyons, P. & Ferro-Novick, S. Ypt1p implicated in v-SNARE activation. Nature 372, 698–701 (1994).
Mayer, A. & Wickner, W. Docking of yeast vacuoles is catalyzed by the Ras-like GTPase Ypt7p after symmetric priming by Sec18p (NSF). J. Cell Biol. 136, 307–317 (1997).
Lupashin, V. V. & Waters, M. G. t-SNARE activation through transient interaction with a rab-like guanosine triphosphatase. Science 276, 1255–1258 (1997).
Gorvel, J. -P., Chavrier, P., Zerial, M. & Gruenberg, J. Rab5 controls early endosome fusion in vitro. Cell 64, 915–925 (1991).
Horiuchi, H. et al. Anovel Rab5 GDP/GTP exchange factor complexed to Rabaptin-5 links nucleotide exchange to effector recruitment and function. Cell 90, 1149–1159 (1997).
Gournier, H., Stenmark, H., Rybin, V., Lippe, R. & Zerial, M. Two distinct effectors of the small GTPase Rab5 cooperate in endocytic membrane fusion. EMBO J. 17, 1930–1940 (1998).
Simonsen, A. et al. EEA1 links phosphatidylinositol 3-kinase function to Rab5 regulation of endosome fusion. Nature 394, 494–498 (1998).
Emans, N. et al. Annexin II is a major component of fusogenic endosomal vesicles. J. Cell Biol. 120, 1357–1369 (1993).
Colombo, M. I., Beron, W. & Stahl, P. D. Calmodulin regulates endosome fusion. J. Biol. Chem. 272, 7707–7712 (1997).
Rodriguez, L., Stirling, C. J. & Woodman, P. G. Multiple N-ethylmaleimide-sensitive components are required for endosomal vesicle fusion. Mol. Biol. Cell 5, 773–783 (1994).
Colombo, M. I., Taddese, M., Whiteheart, S. W. & Stahl, P. D. Apossible predocking attachment site for N-ethylmaleimide-sensitive fusion protein. Insights from in vitro endosome fusion. J. Biol. Chem. 271, 18810–18816 (1996).
Wiedemann, C. & Cockcroft, S. Vesicular transport: sticky fingers grab a lipid. Nature 394, 426–427 (1998).
Patki, V. et al. Identification of an early endosomal protein regulated by phosphatidylinositol 3-kinase. Proc. Natl Acad. Sci. USA 94, 7326–7330 (1997).
Barnard, R. J. O., Morgan, A. & Burgoyne, R. D. Stimulation of NSF ATPase activity by alpha-SNAP is required for SNARE complex disassembly and exocytosis. J. Cell Biol. 139, 875–883 (1997).
Mayer, A., Wickner, W. & Haas, A. Sec18p (NSF)-driven release of Sec17p (α-SNAP) can precede docking and fusion of yeast vacuoles. Cell 85, 83–94 (1996).
Sonnichsen, B. et al. Arole for giantin in docking COPI vesicles to Golgi membranes. J. Cell Biol. 140, 1013–1021 (1998).
Barlowe, C. Coupled ER to golgi transport reconstituted with purified cytosolic proteins. J. Cell Biol. 139, 1097–1108 (1997).
Mu, F. et al. EEA1, an early endosome-associated protein. J. Biol. Chem. 270, 13503–13511 (1995).
Stenmark, H., Aasland, R., Toh, B. H. & D'Arringo, A. Endosomal localization of the autoantigen EEA1 is mediated by zinc-binding FYVE finger. J. Biol. Chem. 271, 24048–24054 (1996).
Acknowledgements
We thank R. Lippe for the supply of recombinant Rabaptin-5/Rabex-5 complex; H.Stenmark for the GST–Rab5 cDNA construct; J. Rothman for pQE9 α-SNAP; A. Giner for technical assistance; V. Rybin for helpful discussions on technical points; E. Nielsen and B.Sonnichsen for the supply of rhodamine-labelled early endosomes; P. Scheiffele, B. Sonnichsen and members of the laboratory for discussions and critical reading of the manuscript; and K. Ashman and M. Wilm form providing mass spectroscopy data. S.C. is supported by an EU TMR fellowship. H.M.M. is recipient of an Alexander vonHumboldt Stiftung. This work was supported by the Max Planck Gesellschaft, and by grants from the Human Frontier Science Program, EU TMR and Biomed (to M.Z.). This work is dedicated to the memory of Thomas Kreis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Christoforidis, S., McBride, H., Burgoyne, R. et al. The Rab5 effector EEA1 is a core component of endosome docking. Nature 397, 621–625 (1999). https://doi.org/10.1038/17618
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/17618
This article is cited by
-
Rab GTPases and phosphoinositides fine-tune SNAREs dependent targeting specificity of intracellular vesicle traffic
Nature Communications (2024)
-
M6PR interacts with the HA2 subunit of influenza A virus to facilitate the fusion of viral and endosomal membranes
Science China Life Sciences (2024)
-
The Role of Rab GTPases in the development of genetic and malignant diseases
Molecular and Cellular Biochemistry (2024)
-
Lysosomal cathepsins act in concert with Gasdermin-D during NAIP/NLRC4-dependent IL-1β secretion
Cell Death & Disease (2022)
-
Spatial snapshots of amyloid precursor protein intramembrane processing via early endosome proteomics
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.