Abstract
Staphylococcus aureus is one of the most important human pathogenic bacteria and environmental surfaces play an important role in the spread of the bacterium. Presence of S. aureus on children’s playgrounds and on toys was described in international studies, however, little is known about the prevalence and characteristics of S. aureus at playgrounds in Europe. In this study, 355 samples were collected from playgrounds from 16 cities in Hungary. Antibiotic susceptibility of the isolates was tested for nine antibiotics. Presence of virulence factors was detected by PCR. Clonal diversity of the isolates was tested by PFGE and MLST. The overall prevalence of S. aureus was 2.81% (10/355) and no MRSA isolates were found. Presence of spa (10), fnbA (10), fnbB (5), icaA (8), cna (7), sea (2), hla (10), hlb (2) and hlg (6) virulence genes were detected. The isolates had diverse PFGE pulsotypes. With MLST, we have detected isolates belonging to ST8 (CC8), ST22 (CC22), ST944 and ST182 (CC182), ST398 (CC398), ST6609 (CC45), ST3029 and ST2816. We have identified a new sequence type, ST6609 of CC45. S. aureus isolates are present on Hungarian playgrounds, especially on plastic surfaces. The isolates were clonally diverse and showed resistance to commonly used antibiotics. These data reinforce the importance of the outdoor environment in the spread for S. aureus in the community.
Similar content being viewed by others
Introduction
Staphylococcus aureus is one of the most important human pathogens, causing a wide range of infections from localized suppurative infections as pneumonia, osteomyelitis, endocarditis to bacteremia and sepsis1.
Asymptomatic S. aureus colonization of healthy people is very frequent, especially in the nasopharynx: 15% of the population is continuously colonized with S. aureus, whereas intermittent colonization is described in 70% of individuals. In children, colonization rates are even higher, with peak incidence at 10 years of age2.
Staphylococcus aureus is transmitted from human to human or from animal to human directly with close physical contact and respiratory droplets; or indirectly via contaminated fomites. Inanimate objects serve as potential reservoir and source of infection both for community acquired and hospital acquired S. aureus infections3. S. aureus is remarkably resistant to environmental factors and may persist on surface of fomites for hours, days or even months. Length of the survival is influenced by several factors, including the material of the object, environmental conditions (temperature, humidity), amount of contaminating bacterium introduced to the surface and presence of bodily fluids3.
There are several studies evaluating the prevalence of S. aureus on indoor surfaces. Most of these reports focus on the presence of the pathogen in the hospital environment4,5. Outside of the healthcare system, numerous studies assessed the environmental presence of S. aureus in the households of known carriers of the bacterium and have found that the pathogen can persist on household fomites for up to three months6,7. Public spaces can also be sources of S. aureus transmission. Prevalence of S. aureus on non-hospital public indoor fomites (public transportation, public phones, ATM, door handles, surfaces in schools and universities) varies widely from 6.5% up to 91.4%8.
In spite of the ubiquitous nature and the importance of the bacterium, there is a limited number of studies describing the prevalence of S. aureus on public open-air surfaces, such as playgrounds and to our knowledge there is no data available on the prevalence of S. aureus on playgrounds from Europe.
This study aimed to evaluate the prevalence, antibiotic susceptibility, virulence pattern and clonal composition of S. aureus isolates from public outdoor playgrounds from Hungary.
Material
Environmental sampling
A total of 355 samples were collected from 24 public outdoor playgrounds in 16 cities of Hungary between September 2018 and September 2019 (Fig. 1). All samples have been collected in fair weather conditions (15–25 °C temperature, dry weather). The sampled surfaces were chosen as the most hand touched areas at the sites. Locations were chosen to represent frequently visited playgrounds from both small and large cities from different areas of the country. Surfaces were swabbed with sterile cotton swabs premoistened with sterile 0.9% sodium chloride solution. Samples were transferred to the laboratory in sterile, charcoal containing Amies transport medium (Transwab, Medical Wire & Equipment, Corsham, UK). Specimens were stored and transported at room temperature per the instructions of the manufacturer. Samples were plated within 24 h.
Location of sample collection (modified from9).
Bacterial culture and identification
Samples were inoculated onto Columbia blood agar plates and onto Chromagar Staph aureus (CHROMagar, Paris, France). After an overnight incubation at 37 °C colonies showing typical species-specific morphology were chosen to produce pure cultures and were identified as S. aureus by catalase and latex agglutination test (Pastorex Staph-Plus Kit, Bio-Rad, Marnes-la-Coquette, France). Genotypic identification was based on the PCR detection of the species-specific thermonuclease gene (nucA), using primers described by Laub et al.10. The cycling parameters were as follows: 3 min at 94 °C, then 30 cycles of 94 °C 60 s, 54 °C 60 s, and 72 °C 30 s, and a final extension of 72 °C for 10 min. For all PCR gel electrophoresis, the GeneRuler 100 bp DNA ladder (ThermoFischer Scientific) was used. S. aureus ATCC 29213 was used as positive control. Confirmed S. aureus isolates were stored at − 80 °C on MAST CRYOBANK™ cryobeads (Mast Diagnostica, Bootle, UK).
Antibiotic susceptibility testing
The MIC of the isolates was determined by the agar dilution method using an A400 multipoint inoculator (AQS Manufacturing Ltd., Southwater, UK) on Mueller–Hinton agar plates. Cefoxitin susceptibility was tested by disk diffusion method. The results were interpreted according the EUCAST guidelines11. The following antibiotics were tested: penicillin, cefoxitin, oxacillin, erythromycin, clindamycin, gentamicin, tetracycline, ciprofloxacin, levofloxacin and vancomycin. Inducible resistance to clindamycin was tested by ‘D test’ according to EUCAST guidelines11.
Molecular characterization
PCR was carried out on all S. aureus isolates to detect the presence of the methicillin resistance gene mecA, and the following virulence factor genes: PVL genes (lukS, lukF), haemolysin A, B and G (hla, hlb and hlg) genes, Staphylococcus protein A (spa) gene, exfoliative toxin A and B (eta, etb) genes, toxic shock syndrome toxin (tsst), and Staphylococcus enterotoxin A, B, C (sea, seb, sec) genes as described previously12. For each virulence gene, the appropriate control strain was applied, obtained from the collection of the National Public Health Centre of Hungary. Bacterial DNA was isolated from the bacteria with the Quick-DNA Fungal/Bacterial Miniprep Kit (Zymo Research, California, USA). The DNA samples were stored at – 80 °C until further testing.
All S. aureus isolates were genotyped by pulsed-field gel electrophoresis (PFGE). The preparation of the genomic DNA for PFGE analysis included a 3-h lysis in lysostaphin and lysozyme, followed by an overnight lysis in proteinase K (Sigma Aldrich). The SmaI digestion was performed for 45 min (ThermoScientific FastDigest enzyme). For PFGE gel electrophoresis, we have applied the following running conditions: Pulse times: block 1, 5 s/15 s for 10 h, and block 2, 15 s/60 s for 11 h; at 14 °C. The CHEF DNA Size Standard 48.5–1000 kb Lambda Ladder (Bio-Rad) was used as a molecular size marker.
Multi-locus sequence typing (MLST) was performed on all isolates and Sequence Types (STs) were assigned using organism specific MLST database13. The PCR products for the MLST analysis were purified with the QIAquick PCR Purification Kit (Qiagen, Venlo, Netherlands). The final DNA concentration was measured by a NanoDrop Lite spectrophotometer (Thermo Scientific). PHYLOViZ software v2.0 was used to analyze relatedness of STs and to draw minimum spanning tree.
Ethical approval
The Semmelweis University Regional and Institutional Committee of Science and Research Ethics has confirmed that no ethical approval is required for this study.
Results
Staphylococcus aureus isolates were found in 10 samples (2.81%, 10/355), all positive samples were collected at playgrounds at Budapest (5.49%, 10/182). Seven out of the 10 samples were recovered from plastic surfaces, the remaining three from metal surfaces of playground equipment (swings, slides, monkey bars and seesaws).
Virulence factors
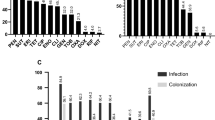
All ten isolates carried the haemolysin A gene (hla), whereas two were positive for haemolysin B (hlb) and six positive for haemolysin G (hlg). Two isolates possessed the staphylococcal enterotoxin A gene (sea). Genes encoding adhesion factors were present in all isolates, ten were positive for spa and fnbA, eight for icaA, seven for cna, and five for fnbB. None of the isolates was positive for pvl (Fig. 2).
Antibiotic susceptibility
The isolates were susceptible to most of the tested antibiotics. Two isolates were resistant to penicillin, two to erythromycin and clindamycin and four isolates were resistant to ciprofloxacin. All isolates were susceptible to cefoxitin, oxacillin, gentamicin, tetracycline, levofloxacin and vancomycin (Fig. 2).
Clonal diversity
Based on both MLST and PFGE results, our isolates proved to be clonally diverse (Figs. 2 and 3). According to MLST, two isolates belonged to sequence type (ST) 8 of clonal complex (CC) 8. One isolate each belonging to ST22 (CC22), ST398 (CC398), ST182 and ST944 (both in CC182), ST3060 (CC45), ST3029 and ST2816 were found. We have identified a new allelic combination, which was assigned a new sequence type: ST6609 (CC45) by the PubMLST Database, which is a single locus variant of ST3060. One isolate was non-typable by PFGE with SmaI digestion; this isolate belonged to ST398.
Discussion
Staphylococcus aureus is the leading cause of invasive bacterial infections in children. Skin and soft tissue infections (SSTI), including impetigo, folliculitis, cellulitis, and cutaneous abscesses are the most frequent infections caused by S. aureus in the pediatric population, moreover, these localized infections can possibly disseminate to invasive, life-threatening infections14.
Children’s playground are generally high traffic areas where many visitors, adults and children, spend extended time. High-touch zones of playground equipment can easily become contaminated by microbes from humans and from the environment. According to a study evaluating the presence of bacteria and bodily fluids on various public surfaces, the highest contamination rates were found on surfaces from children's playground equipment and daycare centers. Children are especially prone to exposure to environmental contaminations due to frequent hand-to-mouth contact15. Children are exposed to many microbes during their stay at playgrounds and in parks. A study from Mexico found that 2.5% of children had S. aureus on their hand after one hour play at a playground, among other bacteria, viruses and protozoa16.
In a study from Ohio, USA, samples were collected from 10 outdoor playgrounds. S. aureus was found in 31.8% of the samples, 3.9% of the samples were positive for MRSA17. The most prevalent sequence type was ST5/USA100, a common clone in the USA, followed by ST8/USA300 isolates. Five livestock associated ST398 isolates were also found.
A study from Greece demonstrated the importance of the season (temperature, humidity) in the presence of pathogenic bacteria in soil of playgrounds, but not on equipment surface18. Chatziprodromidou et al. detected 5.1 CFU/g S. aureus in soil of playgrounds in cold wet season, however, have found no S. aureus during hot dry season18. Gastroenteritis outbreaks of children were linked to Salmonella enterica and Salmonella typhimurium, associated with playground sand in Austria and in Spain19,20. Another gastroenteritis causing bacterium, Campylobacter jejuni was isolated from 12.5% of the wild bird droppings collected at playgrounds in New Zealand. Authors have linked the frequent environmental presence of the bacterium at playground environment to high rate of campylobacteriosis in children under 5 years of age21. Presence of Escherichia coli is also frequent at playgrounds all over the world. In 2009, an outbreak of diarrhea-associated haemolytic uraemic syndrome, leading to the death of one child, was caused by a shiga toxin-producing E. coli strain in Germany. Likely, the place of the infection was the local playground22.
Whereas several studies have evaluated the presence of other—mostly Gram-negative—bacteria at playgrounds, to our knowledge this is the first study about S. aureus on playground equipment in Europe so far.
In this study, we have found 2.8% prevalence rate of S. aureus on outdoor playground equipment. This rate in much lower compared to a previous study from the USA17. Differences in climate, number of people visiting playgrounds and S. aureus carriage rates in the population may account for this variation.
All S. aureus isolates were recovered from playgrounds of the capital city, Budapest. Higher population density leading to higher numbers of playground visitors is likely to contribute to this result.
Most of our isolates (7/10) derived from plastic surfaces, whereas less S. aureus was found on metal surfaces (3/10) and no isolates were recovered from wooden equipment (Fig. 2). This is in accordance with a previous study testing the survival of MRSA on various surfaces. MRSA survived the longest on plastic and vinyl and for the least amount of time on wood23.
Staphylococcus aureus isolates in this study were generally susceptible to the tested antibiotics, however, some isolates had resistance to commonly used antibiotics, such as penicillin, erythromycin, clindamycin and ciprofloxacin (Fig. 2). Antibiotic resistance rates of isolates in this study were lower compared to clinical isolates, and similar to that found in a study describing S. aureus isolates asymptomatically colonizing Hungarian children24. The majority of our isolates (8/10) were susceptible to penicillin. Clinical isolates of S. aureus are generally resistant to penicillin; however, the proportion of penicillin susceptible S. aureus (PSSA) isolates has increased globally in recent years25. Macrolides are one of the most frequently used antibiotics in community-acquired infections of children; hence erythromycin and clindamycin resistance of environmental S. aureus isolates may lead to failure of empirical antibiotic therapy26.
All of our isolates carried the haemolysin A gene (hla), staphylococcal protein A gene (spa) and fibronectin binding protein A gene (fnbA) (Fig. 2). Several isolates have carried additional virulence factor genes, encoding for cytotoxins (hlb, hlg) and adhesion factors (icaA, cna, fnbA). Prevalence of virulence factor genes was lower compared to results of a study describing clinical isolates from S. aureus bloodstream infection in Hungary12. It is of particular interest that two isolates carried the sea gene encoding for staphylococcal enterotoxin A, potentially causing staphylococcal food poisoning.
It is well established that the population structure of S. aureus is clonal, the most dominant clones globally are clonal complexes (CC) CC5, CC8, CC22, CC30, and CC4527,28. Isolates of this study were diverse both by PFGE and MLST, they belonged to nine sequence types (ST) (Figs. 2 and 3). One isolate belonging to ST398 was non-typable with PFGE. This is a well-described characteristic of ST398 due to the methylation of the genome of this particular sequence type at the restriction site of SmaI enzyme, making ST398 resistant to standard PFGE typing29.
We have detected isolates of the most frequent S. aureus clones, including ST22, which is widespread all over Europe and has been increasingly reported from Asia and the Middle East in the recent years. ST22 is more prevalent in invasive infections compared to asymptomatic colonization30. Globally, isolates of CC45 are also very common, especially among nasal commensal isolates31. ST45 was described as the most frequent S. aureus strain from asymptomatic nasal colonization of Hungarian children24. In our study, we have found two CC45 isolates. One isolate belonged to ST3060, a single-locus variant of ST45. In the scientific literature, ST3060 S. aureus has been described on two occasions so far, both times from wild animals. A ST3060-mecA-MRSA isolate was recently identified from a hedgehog from Hungary. Hedgehogs are frequent carriers of S. aureus and they are prevalent in both rural and populated areas, have access to human food waste and playgrounds as well, and they may serve an important role in transmission of bacteria between wildlife, environment and humans. ST3060 S. aureus was also described from white storks exposed to human waste in Spain32. Migratory birds, such as storks play an important role in international and intercontinental dissemination of human pathogenic bacteria33 as their feces contaminates environment of humans, including playgrounds21. Various Staphylococci were described in feces of wild birds, suggesting their role in the carriage and dissemination of resistant bacteria into the environment34. Furthermore, in this study, we have identified a new sequence type, ST6609, a single locus variant of ST3060, belonging to CC45.
Two isolates of this study belonged to ST8 that is among the most prevalent clones in North America and from there has spread to all over the world35.
Two of our isolates belonged to CC182 (ST182 and ST944). ST182 has worldwide distribution and has been identified from asymptomatic colonization of adults and children, as well as invasive infections and animals36. ST944 is a single-locus variant of ST182 that has been described from asymptomatic carrier adults and schoolchildren and clinical isolates in China and in Portugal37,38.
Moreover, we have identified one isolate belonging to ST398 of CC398. Methicillin resistant strains of ST398 are important livestock associated pathogens of worldwide distribution, whereas methicillin susceptible ST398 isolates are adapted to humans, are primarily transmitted from human to human, with the majority of the infections reported from Europe and China39. However, wild animals, including the migratory white storks have also been described to carry MSSA-CC39840.
We have identified two singleton S. aureus isolates (ST2816 and ST3029), that differ from all other existing STs by at least two alleles, and do not belong to any existing CCs. ST2816 has been described from clinical isolates and asymptomatic colonization in Kuwait and in Saudi Arabia41,42. ST3029 has only been isolated in Malaysia, from skin infection so far43.
A limitation of our study is that all samples were collected within Hungary, therefore no global European picture can be extracted. Further studies are needed to get a better view on the prevalence and characteristics of S. aureus isolates on European playground and their potential role in colonization and infections of children.
Conclusions
The presence of S. aureus isolates on playground equipment highlights the role of public outdoor playgrounds in the transmission of S. aureus in the community. Antibiotic resistance and the many virulence factors of the identified S. aureus isolates underline their potential as pathogens of pediatric infections. In line with the One Health concept, not only humans, but also wild animals, such as migratory birds may introduce human pathogenic bacteria onto playground surfaces, and these environmental reservoirs can serve as contact points for exchange of pathogenic bacteria between humans, animals and the ecosystem. The S. aureus isolates in this study showed high clonal diversity: both representatives of important international S. aureus clones and minor, rarely identified STs were present simultaneously with no dominant clone identified, emphasizing the importance of public open-air playgrounds introduction of different bacterial clones into the community.
Data availability
All data generated or analysed during this study are included in this article.
References
Lowy, F. D. Staphylococcus aureus infections. N. Engl. J. Med. 339(8), 520–532. https://doi.org/10.1056/nejm199808203390806 (1998).
Lee, A. S. et al. Methicillin-resistant Staphylococcus aureus. Nat. Rev. Dis. Prim. 4, 18033. https://doi.org/10.1038/nrdp.2018.33 (2018).
Jaradat, Z. W. et al. Methicillin resistant Staphylococcus aureus and public fomites: A review. Pathog. Glob. Health 114(8), 426–450. https://doi.org/10.1080/20477724.2020.1824112 (2020).
Mody, L. et al. Multidrug-resistant organisms in hospitals: What is on patient hands and in their rooms?. Clin. Infect. Dis. 69(11), 1837–1844. https://doi.org/10.1093/cid/ciz092 (2019).
Veloso, J. O. et al. Methicillin-resistant and vancomycin-intermediate Staphylococcus aureus colonizing patients and intensive care unit environment: Virulence profile and genetic variability. APMIS 127(11), 717–726. https://doi.org/10.1111/apm.12989 (2019).
Shankar, N., Soe, P. M. & Tam, C. C. Prevalence and risk of acquisition of methicillin-resistant Staphylococcus aureus among households: A systematic review. Int. J. Infect. Dis. 92, 105–113. https://doi.org/10.1016/j.ijid.2020.01.008 (2020).
Eells, S. J. et al. Persistent environmental contamination with USA300 methicillin-resistant Staphylococcus aureus and other pathogenic strain types in households with S. aureus skin infections. Infect. Control Hosp. Epidemiol. 35(11), 1373–1382. https://doi.org/10.1086/678414 (2014).
Lin, J. et al. Non-hospital environment contamination with Staphylococcus aureus and methicillin-resistant Staphylococcus aureus: Proportion meta-analysis and features of antibiotic resistance and molecular genetics. Environ. Res. 150, 528–540. https://doi.org/10.1016/j.envres.2016.06.040 (2016).
Map of Hungary. https://en.wikipedia.org/wiki/File:Hungary_location_map.svg (2023).
Laub, K., Tothpal, A., Kardos, S. & Dobay, O. Epidemiology and antibiotic sensitivity of Staphylococcus aureus nasal carriage in children in Hungary. Acta Microbiol. Immunol. Hung. 64(1), 51–62. https://doi.org/10.1556/030.64.2017.001 (2017).
The European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters, version [cited 2023 Jan 13]. Avalaible from: https://www.eucast.org/clinical_breakpoints/ (2023).
Horváth, A. et al. Characterisation of antibiotic resistance, virulence, clonality and mortality in MRSA and MSSA bloodstream infections at a tertiary-level hospital in Hungary: A 6-year retrospective study. Ann. Clin. Microbiol. Antimicrob. 19(1), 17. https://doi.org/10.1186/s12941-020-00357-z (2020).
Jolley, K. A., Bray, J. E. & Maiden, M. C. J. Open-access bacterial population genomics: BIGSdb software, the PubMLST.org website and their applications. Wellcome Open Res. 3, 124. https://doi.org/10.12688/wellcomeopenres.14826.1 (2018).
Kalu, I. C., Kao, C. M. & Fritz, S. A. Management and prevention of Staphylococcus aureus infections in children. Infect. Dis. Clin. North Am. 36(1), 73–100. https://doi.org/10.1016/j.idc.2021.11.006 (2022).
Reynolds, K. A., Watt, P. M., Boone, S. A. & Gerba, C. P. Occurrence of bacteria and biochemical markers on public surfaces. Int. J. Environ. Health Res. 15(3), 225–234. https://doi.org/10.1080/09603120500115298 (2005).
Martínez-Bastidas, T. et al. Detection of pathogenic micro-organisms on children’s hands and toys during play. J. Appl. Microbiol. 116(6), 1668–1675. https://doi.org/10.1111/jam.12473 (2014).
Thapaliya, D. et al. Prevalence and molecular characterization of Staphylococcus aureus and methicillin-resistant S aureus on children’s playgrounds. Pediatr. Infect. Dis. J. 38(3), e43–e47. https://doi.org/10.1097/inf.0000000000002095 (2019).
Chatziprodromidou, I. P., Chatziantoniou, S., Vantarakis, G. & Vantarakis, A. Risk factor analysis of children’s exposure to microbial pathogens in playgrounds. Risk Anal. https://doi.org/10.1111/risa.13752 (2021).
Lucerón, C. O. et al. Notes from the Field: An outbreak of Salmonella typhimurium associated with playground sand in a Preschool Setting—Madrid, Spain, September-October 2016. MMWR Morb. Mortal Wkly. Rep. 66(9), 256–257. https://doi.org/10.15585/mmwr.mm6609a3 (2017).
Staff, M., Musto, J., Hogg, G., Janssen, M. & Rose, K. Salmonellosis outbreak traced to playground sand, Australia, 2007–2009. Emerg. Infect. Dis. 18(7), 1159–1162. https://doi.org/10.3201/eid1807.111443 (2012).
French, N. P. et al. Molecular epidemiology of Campylobacter jejuni isolates from wild-bird fecal material in children’s playgrounds. Appl. Environ. Microbiol. 75(3), 779–783. https://doi.org/10.1128/aem.01979-08 (2009).
Nielsen, S. et al. Desperately seeking diarrhoea: Outbreak of haemolytic uraemic syndrome caused by emerging sorbitol-fermenting shiga toxin-producing Escherichia coli O157:H-, Germany, 2009. Zoonoses Public Health 58(8), 567–572. https://doi.org/10.1111/j.1863-2378.2011.01405.x (2011).
Coughenour, C., Stevens, V. & Stetzenbach, L. D. An evaluation of methicillin-resistant Staphylococcus aureus survival on five environmental surfaces. Microb. Drug Resist. 17(3), 457–461. https://doi.org/10.1089/mdr.2011.0007 (2011).
Laub, K. et al. High prevalence of Staphylococcus aureus nasal carriage among children in Szolnok, Hungary. Acta Microbiol. Immunol. Hung. 65(1), 59–72. https://doi.org/10.1556/030.65.2018.001 (2018).
Reynolds, G., Crawford, S., Cuenca, J., Ghosh, N. & Newton, P. Penicillin versus anti-staphylococcal beta-lactams for penicillin-susceptible Staphylococcus aureus blood stream infections: A retrospective cohort study. Eur. J. Clin. Microbiol. Infect. Dis. 41(1), 147–151. https://doi.org/10.1007/s10096-021-04330-2 (2022).
D’Amore, C. et al. Use of multiple metrics to assess antibiotic use in Italian children’s hospitals. Sci. Rep. 11(1), 3543. https://doi.org/10.1038/s41598-021-83026-1 (2021).
Turner, N. A. et al. Methicillin-resistant Staphylococcus aureus: An overview of basic and clinical research. Nat. Rev. Microbiol. 17(4), 203–218. https://doi.org/10.1038/s41579-018-0147-4 (2019).
Monecke, S. et al. A field guide to pandemic, epidemic and sporadic clones of methicillin-resistant Staphylococcus aureus. PLoS One. 6(4), e17936. https://doi.org/10.1371/journal.pone.0017936 (2011).
Bens, C. C., Voss, A. & Klaassen, C. H. Presence of a novel DNA methylation enzyme in methicillin-resistant Staphylococcus aureus isolates associated with pig farming leads to uninterpretable results in standard pulsed-field gel electrophoresis analysis. J. Clin. Microbiol. 44(5), 1875–1876. https://doi.org/10.1128/jcm.44.5.1875-1876.2006 (2006).
Sunagar, R. et al. Prevalence and genotype distribution of methicillin-resistant Staphylococcus aureus (MRSA) in India. J. Glob. Antimicrob. Resist. 7, 46–52. https://doi.org/10.1016/j.jgar.2016.07.008 (2016).
Wertheim, H. F. et al. Associations between Staphylococcus aureus genotype, infection, and in-hospital mortality: A nested case-control study. J. Infect. Dis. 192(7), 1196–1200. https://doi.org/10.1086/444427 (2005).
Gómez, P. et al. Detection of MRSA ST3061-t843-mecC and ST398-t011-mecA in white stork nestlings exposed to human residues. J. Antimicrob. Chemother. 71(1), 53–57. https://doi.org/10.1093/jac/dkv314 (2016).
Elsohaby, I. et al. Migratory wild birds as a potential disseminator of antimicrobial-resistant bacteria around Al-Asfar Lake, Eastern Saudi Arabia. Antibiot. (Basel). 10, 3. https://doi.org/10.3390/antibiotics10030260 (2021).
Tareen, A. R. & Zahra, R. Community acquired methicillin resistant Staphylococci (CA-MRS) in fecal matter of wild birds—a “one health” point of concern. J. Infect. Public Health 16(6), 877–883. https://doi.org/10.1016/j.jiph.2023.04.002 (2023).
Strauß, L. et al. Origin, evolution, and global transmission of community-acquired Staphylococcus aureus ST8. Proc. Natl. Acad. Sci. U. S. A. 114(49), E10596–E10604. https://doi.org/10.1073/pnas.1702472114 (2017).
Campbell, S. J. et al. Genotypic characteristics of Staphylococcus aureus isolates from a multinational trial of complicated skin and skin structure infections. J. Clin Microbiol. 46(2), 678–684. https://doi.org/10.1128/jcm.01822-07 (2008).
Blumental, S. et al. Dynamic pattern and genotypic diversity of Staphylococcus aureus nasopharyngeal carriage in healthy pre-school children. J. Antimicrob. Chemother. 68(7), 1517–1523. https://doi.org/10.1093/jac/dkt080 (2013).
Chen, X. et al. Antimicrobial resistance and molecular characteristics of nasal Staphylococcus aureus isolates from newly admitted inpatients. Ann. Lab. Med. 36(3), 250–254. https://doi.org/10.3343/alm.2016.36.3.250 (2016).
Bouiller, K., Bertrand, X., Hocquet, D. & Chirouze, C. Human infection of methicillin-susceptible Staphylococcus aureus CC398: A review. Microorganisms. 8, 11. https://doi.org/10.3390/microorganisms8111737 (2020).
Abdullahi, I. N. et al. Staphylococcus aureus carriage in the nasotracheal cavities of white stork nestlings (Ciconia ciconia) in Spain: Genetic diversity, resistomes and virulence factors. Microb. Ecol. 86(3), 1993–2002. https://doi.org/10.1007/s00248-023-02208-8 (2023).
Boswihi, S. S., Udo, E. E. & Al-Sweih, N. Shifts in the clonal distribution of methicillin-resistant Staphylococcus aureus in Kuwait Hospitals: 1992–2010. PLoS One. 11(9), e0162744. https://doi.org/10.1371/journal.pone.0162744 (2016).
El-Mahdy, T. S., Al-Agamy, M. H., Emara, M., Barakat, A. & Goering, R. V. Complex clonal diversity of Staphylococcus aureus nasal colonization among community personnel, healthcare workers, and clinical students in the Eastern Province, Saudi Arabia. Biomed. Res. Int. 2018, 4208762. https://doi.org/10.1155/2018/4208762 (2018).
Staphylococcus aureus MLST site https://pubmlst.org/saureus/ (2023).
Funding
Open access funding provided by Semmelweis University. The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by A.H., Á.T., S.H-K., A.H., J. S-T. and O.D. The first draft of the manuscript was written by A.H. and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Horváth, A., Tormássi, Á., Hajósi-Kalcakosz, S. et al. High clonal diversity of Staphylococcus aureus isolates from children’s playgrounds in Hungary. Sci Rep 14, 10021 (2024). https://doi.org/10.1038/s41598-024-60481-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-60481-0
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.