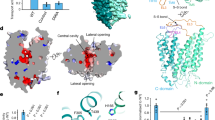
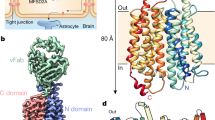
Abstract
Choline is an essential nutrient that the human body needs in vast quantities for cell membrane synthesis, epigenetic modification and neurotransmission. The brain has a particularly high demand for choline, but how it enters the brain remains unknown1,2,3. The major facilitator superfamily transporter FLVCR1 (also known as MFSD7B or SLC49A1) was recently determined to be a choline transporter but is not highly expressed at the blood–brain barrier, whereas the related protein FLVCR2 (also known as MFSD7C or SLC49A2) is expressed in endothelial cells at the blood–brain barrier4,5,6,7. Previous studies have shown that mutations in human Flvcr2 cause cerebral vascular abnormalities, hydrocephalus and embryonic lethality, but the physiological role of FLVCR2 is unknown4,5. Here we demonstrate both in vivo and in vitro that FLVCR2 is a BBB choline transporter and is responsible for the majority of choline uptake into the brain. We also determine the structures of choline-bound FLVCR2 in both inward-facing and outward-facing states using cryo-electron microscopy. These results reveal how the brain obtains choline and provide molecular-level insights into how FLVCR2 binds choline in an aromatic cage and mediates its uptake. Our work could provide a novel framework for the targeted delivery of therapeutic agents into the brain.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The density maps for the OFS and IFS have been deposited into Electron Microscopy Data Bank (EMDB) with accession codes EMD-43684 and EMD-43683, respectively. The structural models for the OFS and IFS have been deposited in the Protein Data Bank (PDB) with accession codes 8VZO and 8VZN, respectively. Raw data from BEND3.1 and iHBECs were deposited in the Gene Expression Omnibus (GEO) under accession GSE244349. Source data are provided with this paper.
References
Zeisel, S. H. & da Costa, K.-A. Choline: an essential nutrient for public health. Nutr. Rev. 67, 615–623 (2009).
Zeisel, S. H. Choline: critical role during fetal development and dietary requirements in adults. Annu. Rev. Nutr. 26, 229–250 (2006).
US Institute of Medicine Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline (National Academies Press, 1998).
Hupe, M. et al. Gene expression profiles of brain endothelial cells during embryonic development at bulk and single-cell levels. Sci. Signal. 10, eaag2476 (2017).
Santander, N. et al. Lack of Flvcr2 impairs brain angiogenesis without affecting the blood-brain barrier. J. Clin. Invest. 130, 4055–4068 (2020).
Kenny, T. C. et al. Integrative genetic analysis identifies FLVCR1 as a plasma-membrane choline transporter in mammals. Cell Metab. 35, 1057–1071.e12 (2023).
Tsuchiya, M., Tachibana, N., Nagao, K., Tamura, T. & Hamachi, I. Organelle-selective click labeling coupled with flow cytometry allows pooled CRISPR screening of genes involved in phosphatidylcholine metabolism. Cell Metab. 35, 1072–1083.e9 (2023).
Gibellini, F. & Smith, T. K. The Kennedy pathway—de novo synthesis of phosphatidylethanolamine and phosphatidylcholine. IUBMB Life 62, 414–428 (2010).
Ueland, P. M. Choline and betaine in health and disease. J. Inherit. Metab. Dis. 34, 3–15 (2011).
Anderson, O. S., Sant, K. E. & Dolinoy, D. C. Nutrition and epigenetics: an interplay of dietary methyl donors, one-carbon metabolism and DNA methylation. J. Nutr. Biochem. 23, 853–859 (2012).
Diamond, I. Choline metabolism in brain: the role of choline transport and the effects of phenobarbital. Arch. Neurol. 24, 333–339 (1971).
Iwao, B. et al. Functional expression of choline transporter like-protein 1 (CTL1) and CTL2 in human brain microvascular endothelial cells. Neurochem. Int. 93, 40–50 (2016).
Kuk, A. C. Y. & Silver, D. L. The cellular supply-side economics for phospholipids. Cell Metab. 35, 909–911 (2023).
Zhang, Y. et al. An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. J. Neurosci. 34, 11929–11947 (2014).
Zhang, Y. et al. Purification and characterization of progenitor and mature human astrocytes reveals transcriptional and functional differences with mouse. Neuron 89, 37–53 (2016).
Inazu, M. Functional expression of choline transporters in the blood–brain barrier. Nutrients 11, 2265 (2019).
Okuda, T. & Haga, T. High-affinity choline transporter. Neurochem. Res. 28, 483–488 (2003).
Meyer, E. et al. Mutations in FLVCR2 are associated with proliferative vasculopathy and hydranencephaly-hydrocephaly syndrome (Fowler syndrome). Am. J. Hum. Genet. 86, 471–478 (2010).
Kalailingam, P. et al. Deficiency of MFSD7c results in microcephaly-associated vasculopathy in Fowler syndrome. J. Clin. Invest. 130, 4081–4093 (2020).
Quick, M. & Javitch, J. A. Monitoring the function of membrane transport proteins in detergent-solubilized form. Proc. Natl Acad. Sci. USA 104, 3603–3608 (2007).
Yan, N. Structural advances for the major facilitator superfamily (MFS) transporters. Trends Biochem. Sci 38, 151–159 (2013).
Ilcol, Y. O., Uncu, G. & Ulus, I. H. Free and phospholipid-bound choline concentrations in serum during pregnancy, after delivery and in newborns. Arch. Physiol. Biochem. 110, 393–399 (2002).
Muma, N. A. & Rowell, P. P. Brain and serum levels of choline and lecithin resulting from long-term administration. Neurochem. Res. 11, 481–488 (1986).
da Costa, K.-A., Gaffney, C. E., Fischer, L. M. & Zeisel, S. H. Choline deficiency in mice and humans is associated with increased plasma homocysteine concentration after a methionine load. Am. J. Clin. Nutr. 81, 440–444 (2005).
Kasianowicz, J., Benz, R. & McLaughlin, S. The kinetic mechanism by which CCCP (carbonyl cyanidem-Chlorophenylhydrazone) transports protons across membranes. J. Membr. Biol. 82, 179–190 (1984).
Choy, B. C., Cater, R. J., Mancia, F. & Pryor, E. E. Jr A 10-year meta-analysis of membrane protein structural biology: Detergents, membrane mimetics, and structure determination techniques. Biochim. Biophys. Acta 1863, 183533 (2020).
Dominik, P. K. et al. Conformational chaperones for structural studies of membrane proteins using antibody phage display with nanodiscs. Structure 24, 300–309 (2016).
Dominik, P. K. & Kossiakoff, A. A. Phage display selections for affinity reagents to membrane proteins in nanodiscs. Methods Enzymol. 557, 219–245 (2015).
Fellouse, F. A., Wiesmann, C. & Sidhu, S. S. Synthetic antibodies from a four-amino-acid code: a dominant role for tyrosine in antigen recognition. Proc. Natl Acad. Sci. USA 101, 12467–12472 (2004).
Nygaard, R., Kim, J. & Mancia, F. Cryo-electron microscopy analysis of small membrane proteins. Curr. Opin. Struct. Biol. 64, 26–33 (2020).
Wentinck, K., Gogou, C. & Meijer, D. H. Putting on molecular weight: enabling cryo-EM structure determination of sub-100-kDa proteins. Curr. Res. Struct. Biol. 4, 332–337 (2022).
Bärland, N. et al. Mechanistic basis of choline import involved in teichoic acids and lipopolysaccharide modification. Sci. Adv. 8, eabm1122 (2022).
Newstead, S. Recent advances in understanding proton coupled peptide transport via the POT family. Curr. Opin. Struct. Biol. 45, 17–24 (2017).
Parker, J. L., Kato, T., Kuteyi, G., Sitsel, O. & Newstead, S. Molecular basis for selective uptake and elimination of organic anions in the kidney by OAT1. Nat. Struct. Mol. Biol. 30, 1786–1793 (2023).
Mehedint, M. G., Craciunescu, C. N. & Zeisel, S. H. Maternal dietary choline deficiency alters angiogenesis in fetal mouse hippocampus. Proc. Natl Acad. Sci. USA 107, 12834–12839 (2010).
Wang, Y., Surzenko, N., Friday, W. B. & Zeisel, S. H. Maternal dietary intake of choline in mice regulates development of the cerebral cortex in the offspring. FASEB J. 30, 1566–1578 (2016).
Scidraw. mouse top. Zenodo https://doi.org/10.5281/zenodo.3910057 (2020).
Quick, M. et al. Binding of an octylglucoside detergent molecule in the second substrate (S2) site of LeuT establishes an inhibitor-bound conformation. Proc. Natl Acad. Sci. USA 106, 5563–5568 (2009).
Yang, A. C. et al. Physiological blood–brain transport is impaired with age by a shift in transcytosis. Nature 583, 425–430 (2020).
Li, Y. et al. MFSD7C switches mitochondrial ATP synthesis to thermogenesis in response to heme. Nat. Commun. 11, 4837 (2020).
Sabbagh, M. F. & Nathans, J. A genome-wide view of the de-differentiation of central nervous system endothelial cells in culture. eLife 9, e51276 (2020).
Semesta, K. M., Garces, A. & Tsvetanova, N. G. The psychosis risk factor RBM12 encodes a novel repressor of GPCR/cAMP signal transduction. J. Biol. Chem. 299, 105133 (2023).
Assur, Z., Hendrickson, W. A. & Mancia, F. Tools for coproducing multiple proteins in mammalian cells. Methods Mol. Biol. 801, 173–187 (2012).
Kawate, T. & Gouaux, E. Fluorescence-detection size-exclusion chromatography for precrystallization screening of integral membrane proteins. Structure 14, 673–681 (2006).
Aslanidis, C. & de Jong, P. J. Ligation-independent cloning of PCR products (LIC-PCR). Nucleic Acids Res. 18, 6069–6074 (1990).
Sievers, F. et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 7, 539 (2011).
Rigaud, J.-L., Pitard, B. & Levy, D. Reconstitution of membrane proteins into liposomes: application to energy-transducing membrane proteins. Biochim. Biophys. Acta 1231, 223–246 (1995).
Miller, K. R. et al. T cell receptor-like recognition of tumor in vivo by synthetic antibody fragment. PLoS ONE 7, e43746 (2012).
Cater, R. J. et al. Structural basis of omega-3 fatty acid transport across the blood–brain barrier. Nature 595, 315–319 (2021).
Kim, J. et al. Structure and drug resistance of the Plasmodium falciparum transporter PfCRT. Nature 576, 315–320 (2019).
Mukherjee, S. et al. Engineered synthetic antibodies as probes to quantify the energetic contributions of ligand binding to conformational changes in proteins. J. Biol. Chem. 293, 2815–2828 (2018).
Suloway, C. et al. Automated molecular microscopy: the new Leginon system. J. Struct. Biol. 151, 41–60 (2005).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Bepler, T. et al. Positive-unlabeled convolutional neural networks for particle picking in cryo-electron micrographs. Nat. Methods 16, 1153–1160 (2019).
Kim, K. kookjookeem/kmeans-beamtilt: v1.0.0. Zenodo https://doi.org/10.5281/zenodo.8373629 (2023).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010).
Acknowledgements
The authors thank members of the Mancia and Arnold laboratories and the Columbia Cryo-EM facility for assistance; and F. F. Gonzalez and R. Pennington for help with in vivo radioactive uptake assays. This work was supported by National Institutes of Health (NIH) grants (R21 NS129105-01 to T.A., F.M. and R.J.C.). R.J.C. has received support through an NIH K99 Fellowship (HL166866-01) and an Australian Research Council Discovery Early Career Researcher Award (DE230101536). The contribution of F.M. is supported by NIGMS grant R35GM132120. Some of the work was performed at the Center for Membrane Protein Production and Analysis (COMPPÅ; NIH P41 GM116799 to W. A. Hendrickson), previously located at the New York Structural Biology Center.
Author information
Authors and Affiliations
Contributions
The project was conceived by R.J.C., T.A. and F.M. Mouse studies and cell-based assays were designed and performed by D.M., N.S., K.K., K.M.G., S.W.Y. and T.A. N.S. and T.A. analysed expression data. A.L. performed the retro-orbital injection of radioactive choline into mice. A.K. and K.K. collected mouse brains and stained them. E.J.H. prepared human brain specimens. D.M. and T.A. imaged and analysed the data. SPA and liposomal uptake assays were designed and performed by E.G.-I., R.J.C., T.C. and M.Q. Cloning was performed by T.C. and B.K. R.J.C. designed and performed expression screening experiments, produced baculovirus, and optimized protein expression and purification with assistance from T.C., A.R. and B.C.C. S.K.E., T.G. and A.A.K. identified and purified the Fabs, and performed epitope binning assays. R.J.C. prepared the sample for structure analysis, screened and optimized sample vitrification, and generated cryo-EM data with assistance from Z.Z. and A.R. Initial cryo-EM datasets were analysed by R.J.C. and Z.Z. High-resolution cryo-EM data analysis and model building was performed by R.J.C. with guidance from O.B.C. S.W.J.M. assisted with model building and performed structure validation. R.J.C., D.M., S.W.J.M., T.A. and F.M. wrote the manuscript with input from B.K. and A.A.R. R.J.C., D.M., S.W.J.M., T.A., B.K. and N.S. prepared the figures.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Camilo Perez, Alexandre Prat and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer review reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Expression profiles of bona fide and putative choline transporters.
a, Expression profiles14,15 showing that Flvcr2 is enriched in brain endothelial cells in both human and mouse. Other putative and validated choline transporters are not specifically enriched at the BBB. Abbreviations on the horizontal axes indicate: astrocytes (astro), neuron, oligodendrocyte progenitor cells (OPC), immature oligodendrocytes (Olig1), mature oligodendrocytes (Olig2), microglia (MG), endothelial cells (EC), foetal astrocytes (FetalAC), maternal astrocytes (MatAC), and oligodendrocytes (Olig). Expression in endothelial cells is shown in orange; all other cell types are shown in green. Note the differences in vertical axis scale between transporters. b, Affinities of putative and validated choline transporters16. c, RNA sequencing expression profiles from multiple in vitro BBB models compared to acutely isolated brain endothelial cells (Brain EC): primary culture of brain EC, iHBEC, BEND3, HEK293, human iPSC-derived neurons, and HeLa cells (with and without Flvcr1 knockout), showing expression of putative and validated choline transporters in these cells. Flvcr1 expression level in Flvcr1 knockout HeLa cells is coloured grey to denote gene knockout by Crispr/Cas9. d, RT-qPCR performed with RNA extracted from iHBEC cells transfected with either scr siRNA (control) or Flvcr2 siRNA. Human Gapdh expression was used as the internal loading control. e, Fixed sections from Flvcr2HA/HA mice immuno-stained for HA (cyan) and vascular markers ICAM2 (luminal; pink) and COL IV (abluminal; dark blue). Projected Z-stack images (15 μm thick) and scale bars of 10 µm are shown. Each representative image was taken from three different biological samples and data analysed from four to five images from each brain. The right panel shows percentage co-localisation of FLVCR2-HA with ICAM2 and COL IV. f, Fixed human brain tissues were immuno-stained for FLVCR2 (cyan) using an FLVCR2 specific antibody and pan-endothelial markers CD34 (luminal; pink) and COL IV (abluminal; dark blue). Projected Z-stack images (15 μm thick) and scale bars of 10 µm are shown. Each representative image was taken from five different biological samples and data analysed from four to five images from each brain. The right panel shows percentage co-localisation of FLVCR2 with CD34 and COL IV. Graphical data represent mean ± s.e.m. ****P < 0.0001, ***P < 0.001, **P < 0.01, and *P < 0.05. Statistical tests used and exact P and n values are provided in Supplementary Tables 1 and 2.
Extended Data Fig. 2 Generation and validation of Flvcr2 HA-tagging, knock-out, and knock-down.
a, Targeting strategy to generate Flvcr2HA mice. Pink flag denotes HA tag placed downstream of G4S linker (orange) and immediately upstream from 3’ untranslated region (UTR). Mice carrying the HA insertion were selected based on screening PCR (not shown) and confirmed using Southern blot (right panel; twelve mice diagnosed with correct Flvcr2-HA insertion, and two WT controls). b, Strategy adopted to generate endothelial cell-specific Flvcr2 conditional knockouts (Flvcr2 cKO). c, RT-qPCR performed with RNA extracted from adult mouse brain showing control (Flvcr2Fl/+:Cdh5CreER) vs. Flvcr2 cKO (Flvcr2Fl/Fl:Cdh5CreER). Mouse β-actin expression was used as the internal loading control. d, Brains from control (Flvcr2Fl/+:Cdh5CreER) and Flvcr2 cKO (Flvcr2Fl/Fl:Cdh5CreER) mice stained for HA (Rpl22HA recombination reporter; pink, upper panel) and ERG (red, lower panel) to stain endothelial cell nuclei and CD31 (blue) to delineate blood vessels. Scale bar is 100 µm. Each representative image was taken from three biologically different samples and data analysed from four to five images from each brain. e, Measurement of vascular density vessel length density and total number of endothelial nuclei per unit length of vessel in control vs Flvcr2 cKO. Graphical data represent mean ± s.e.m. ***P < 0.001, **P < 0.01, *P < 0.05, and ns = not significant. Statistical tests used and exact P and n values are provided in Supplementary Tables 1 and 2.
Extended Data Fig. 3 Expression, purification, and Fab complex formation of MmFLVCR2.
a, Fluorescent size exclusion chromatography elution profiles of six FLVCR2 orthologs fused to green fluorescent protein (GFP). Orthologs screened are from Homo sapiens (HsFLVCR2; NCBI – NP_060261.2), Mus musculus (MmFLVCR2; NCBI – NP_663422.1), Eptesicus fuscus (EfFLVCR2; NCBI – XP_027997147.2), Canis lupus dingo (CldFLVCR2; NCBI – XP_025299773.1), Bos taurus (BtFLVCR2; NCBI – NP_001179072.1), and Danio rerio (DrFLVCR2; NCBI – XP_693589.3). b, Representative SDS-PAGE gel of MmFLVCR2 purified in DDM/CHS. c, EC50 evaluation of select purified Fabs binding to MmFLVCR2 incorporated into MSP1D1 nanodisc. Data points represent the mean of two data points; approximate EC50 values are provided. d, Normalised high-performance liquid chromatography elution profiles of MmFLVCR2 reconstituted into MSP1D1 nanodisc alone (black) and in complex with Fab FLV23 (blue) and Fab FLV9 (orange). Numbers indicate the retention time (mins) of each peak. e, Mass photometry analysis of nanodisc-reconstituted MmFLVCR2 purified via size exclusion chromatography in the presence (orange) and absence (purple) of Fab FLV23. The peaks differ by 51 kDa which corresponds to the molecular weight of Fab FLV23. Note that negative masses are artefacts caused by non-protein unbinding events. f, Single-point phage ELISA for binding of MmFLVCR2-specific Fab-phage in the presence (orange) and absence (blue) of 1 μM purified Fab FLV32, which was used as an epitope masking reagent. Note that FLV32 was confirmed to have the same binding epitope as FLV23 via preliminary cryo-EM experiments (data not shown). Only Fab FLV9 produced the same level of ELISA signal in the presence and absence of the Fab FLV32.
Extended Data Fig. 4 FLVCR2 and FLVCR1 multiple sequence alignment.
Four FLVCR2 and FLVCR1 variants were aligned using Clustal Omega46 and visualised and coloured using MView, colouring by property colour scheme. The sequences aligned are: Homo sapiens (HsFLVCR2; NCBI – NP_060261.2), Mus musculus (MmFLVCR2; NCBI – NP_663422.1), Canis lupus dingo (CldFLVCR2; NCBI – XP_025299773.1), Bos taurus (BtFLVCR2; NCBI – NP_001179072.1), Homo sapiens (HsFLVCR1; NCBI – NP_054772.1), Mus musculus (MmFLVCR1; NCBI – NP_001074728.1), Canis lupus familiaris (ClfFLVCR1; NCBI – XP_003639227.1), and Bos taurus (BtFLVCR1; NCBI – NP_001192948.1). Secondary structural elements are shown as cylinders, labelled, and coloured as in Fig. 2. Red circles and orange triangles denote choline-coordinating residues and gating residues respectively. Purple diamonds denote residues at which mutations are associated with PVHH. Blue squares represent residues surrounding the substrate-adjacent ion-like cryo-EM densities.
Extended Data Fig. 5 Cryo-EM workflow and analysis of the outward-facing state of MmFLVCR2.
a, Flow chart outlining cryo-EM image acquisition and processing performed to obtain a structure of detergent-purified MmFLVCR2 in complex with the Fab FLV23. A representative micrograph and 2D class averages are shown. All processing was performed using CryoSPARC v.4.1.253 (see Methods for details). b, Euler angle distribution plot of the final three-dimensional reconstruction of the MmFLVCR2-Fab FLV23 complex. c, Fourier shell correlation (FSC) curves for the MmFLVCR2-Fab FLV23 complex. d, Local resolution map of the MmFLVCR2-Fab FLV23 complex, with an orthogonal view indicating the location of the clipping plane. Density is coloured by resolution from 2.0 (blue) to 3.0 Å (red).
Extended Data Fig. 6 Cryo-EM density fit of the outward-facing FLVCR2 model.
Cryo-EM densities (semi-transparent surface) are superimposed on the structural elements of MmFLVCR2 including TM helices 1–12 and bound choline. All elements are shown in stick representation and coloured as in Fig. 2.
Extended Data Fig. 7 Structural features of FLVCR2 and human mutations in FLVCR2 associated with PVHH.
a, The N- and C-domains of FLVCR2 as viewed from within the extracellular cavity of the OFS (top panel) and the intracellular cavity of the IFS (bottom panel). For both domains, the electrostatic potential is shown as a surface coloured from red (negative) to blue (positive) with the structure overlayed in ribbon representation and coloured as in Fig. 2. b, Interaction site of Fab FLV23 with FLVCR2 in the OFS. TM2, TM11, and Fab FLV23 are coloured red, blue, and grey respectively, with interacting residues highlighted in stick representation. c, The N- and C-domains of FLVCR2 viewed in the same manner as in panel a, with the protein shown in ribbon representation and coloured by sequence conservation across mammalian FLVCR2 species using a gradient from most (maroon) to least (cyan) conserved. d, Ion-like cryo-EM densities observed close to the choline binding site in the OFS (left) and IFS (right). The protein is coloured as in Fig. 2 with choline shown in grey stick representation and the ion-like cryo-EM densities and cryo-EM density of surrounding residues and choline shown as a grey semi-transparent surface. Positions of the ion-like densities are indicated by the arrows. Note that the ion-like density in the OFS has a higher occupancy than choline. e, Known human mutations in FLVCR2 that are associated with PVHH, their equivalent sites in MmFLVCR2, and observed phenotypes associated with these mutations. Postnatal phenotype refers to a mild phenotype, whereas foetal phenotype refers to subjects who died during birth or by compassionate abortion. f,g, Position of human mutations in FLVCR2 mapped onto the structure of MmFLVCR2 in the f, OFS and g, IFS. Left panel shows a front-on view of the protein and the right panel shows the N- and C-domains as viewed from within the central cavity. All views are in the plane of the membrane. Equivalent sites of human mutations are coloured in purple and shown in stick representation.
Extended Data Fig. 8 Cryo-EM workflow and analysis of the inward-facing state of MmFLVCR2.
a, Flow chart outlining cryo-EM image acquisition and processing performed to obtain a structure of detergent-purified MmFLVCR2 in complex with the Fab FLV9. A representative micrograph and 2D class averages are shown. All processing was performed using CryoSPARC v.4.1.253 (see Methods for details). b, Euler angle distribution plot of the final three-dimensional reconstruction of the MmFLVCR2-Fab FLV9 complex. c, Fourier shell correlation (FSC) curves for the MmFLVCR2-Fab FLV9 complex. d, Local resolution map of the MmFLVCR2-Fab FLV9 complex, with an orthogonal view indicating the location of the clipping plane. Density is coloured by resolution from 2.25 (blue) to 3.25 Å (red).
Extended Data Fig. 9 Cryo-EM density fit of the inward-facing MmFLVCR2 model.
Cryo-EM densities (semi-transparent surface) are superimposed on structural elements of MmFLVCR2 including TM helices 1–12 and bound choline. All elements are shown in stick representation and coloured as in Fig. 2.
Supplementary information
Supplementary Information
Supplementary Figs. 1–2 and Supplementary Tables 1–2.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cater, R.J., Mukherjee, D., Gil-Iturbe, E. et al. Structural and molecular basis of choline uptake into the brain by FLVCR2. Nature 629, 704–709 (2024). https://doi.org/10.1038/s41586-024-07326-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-024-07326-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.