Abstract
This paper focuses on the maximum speed at which biological evolution can occur. I derive inequalities that limit the rate of evolutionary processes driven by natural selection, mutations, or genetic drift. These rate limits link the variability in a population to evolutionary rates. In particular, high variances in the fitness of a population and of a quantitative trait allow for fast changes in the trait’s average. In contrast, low variability makes a trait less susceptible to random changes due to genetic drift. The results in this article generalize Fisher’s fundamental theorem of natural selection to dynamics that allow for mutations and genetic drift, via trade-off relations that constrain the evolutionary rates of arbitrary traits. The rate limits can be used to probe questions in various evolutionary biology and ecology settings. They apply, for instance, to trait dynamics within or across species or to the evolution of bacteria strains. They apply to any quantitative trait, e.g., from species’ weights to the lengths of DNA strands.
Similar content being viewed by others
Introduction
Fisher’s theorem of natural selection relates the rate of change in the average fitness of a population with the variability in fitness. It holds for evolutionary processes driven by natural selection 1. Fisher’s result suggests that variability serves as a resource by enabling fast evolution. However, the result is of rather limited validity: it does not apply to types that mutate or in the presence of genetic drift 2,3,4. Moreover, Fisher’s focus was on the change in fitness. However, in countless instances biologists are interested in other quantitative attributes—or traits—of individuals in a population. Here, I extend Fisher’s results by studying (a) the rates of arbitrary biological traits and (b) general evolutionary processes that incorporate mutations and genetic drift.
This paper focuses on the rate \(\frac{d\langle A \rangle }{dt}\) at which the average \(\langle A \rangle\) of a quantitative trait A changes. A can represent any measurable trait in a population. For example, A could be breeds’ weights within a species, the maximum CO2 concentration at which different species can survive, or DNA lengths in bacteria strains. The results apply to a range of settings in evolutionary biology and ecology where one is interested in evolution of traits.
Traits’ evolution rates have been studied extensively in quantitative biology. Reference 5 focuses on the biological factors that influence maximum growth rates. References 6,7 and 8,9 study how genetic variance and a population’s structure affects trait evolution, respectively. There’s also extensive data-based work on traits’ evolutionary rates. As one example, Ref. 10 studies the maximum growth rates in mammals.
In the field of applied mathematics, extensions of Fisher’s theorem have also been considered. Reference 11, for example, revises Fisher’s results by studying the effect of mutations on the change in the average fitness of a population (note, though, that it does not focus on other traits). Reference 12 includes a mathematical generalization of Fisher’s theorem to arbitrary traits and dynamics beyond natural selection. References 13,14 rely on uncertainty relations from stochastic thermodynamics to study biological processes.
To derive limits on biological evolutionary rates, I will leverage techniques that have proven useful to study the maximum speed of physical processes. The most related results have appeared in Refs. 15,16. Both articles rely on information theory to bound the evolutionary rates of arbitrary traits. The results in Ref. 15 hold for arbitrary processes. However, they do not discriminate how different evolutionary forces affect rates. In contrast, the results in this article isolate the contributions of natural selection, mutations, and genetic drift to evolutionary rates. The results in Ref. 16 separate contributions from natural selection and mutations, but do not account for genetic drift. Moreover, the results in Refs. 15,16 involve information-theoretic quantities (versions of the Fisher information) that can be hard to evaluate unless one possesses enough knowledge about the dynamics of the system. In contrast, the main results in this article depend on averages and standard deviations that are often more accessible from experimental data.
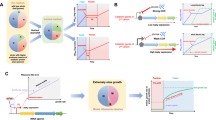
The main outcome of this paper is a set of inequalities that constrain the evolution rate of any quantitative biological trait A (e.g., of a particular phenotype) in terms of simple properties of the system of interest. Specifically, knowledge of expectation values and variances of A and of the fitness f of a population suffices to evaluate the inequalities (see details in Sections “Limits to evolutionary processes with mutations” and “Limits to stochastic evolutionary processes”). In this way, slowly evolving traits can be discriminated from those that can rapidly change without the need to exactly solve the complex dynamics of the system, as pictorially illustrated in Fig. 1.
In Section “Rate limits on biological processes”, I review general limits on traits’ evolutionary rates that hold for any biological system. These results are very general but can sometimes be hard to evaluate. In Sections “Limits to replicator evolutionary processes”, “Limits to evolutionary processes with mutations”, and “Limits to stochastic evolutionary processes”, I derive rate limits that hold for systems driven by (a) natural selection, (b) natural selection and mutations, and (c) natural selection, mutations and stochastic forces (genetic drift), respectively. The results in Sections “Limits to replicator evolutionary processes” and “Limits to stochastic evolutionary processes” are tailored to evolutionary processes and can be evaluated with knowledge of averages and standard deviations. I conclude in Sections “Potential implications to experiments” and “Discussion” with potential connections to experiments and a discussion.
Non-technical summary
How fast can biological evolution occur in nature? What properties of a population enable drastic changes? To what extent are evolutionary rates affected by different driving forces such as natural selection, mutation, or random processes? I address these questions mathematically by deriving inequalities that limit the speed of evolutionary processes. The inequalities take the form of trade-off relations that relate the rate of change of a biological trait with its variance and with the variance in the fitness of a population. In this way, slowly evolving traits can be discriminated from those that can rapidly change. Evaluating the inequalities can be done without exactly solving the complex dynamics of the system. In broad terms, the results in this paper quantify the extent by which variability in a trait can lead to faster evolution.
Evolutionary rate limits. In this work, I derive inequalities that bound the evolutionary rates of quantifiable traits. The rate limits apply to populations that evolve under mutations, natural selection, and random genetic drift. Mutations take a preponderant role in biology, enabling evolutionary changes that can lead to new types. Such new types can have different fitness than their ancestors. Natural selection is the process by which the frequencies of fitter types tend to increase relative to their peers in a given environment (in the illustration, turtles became fitter to survive in a drastically-changed environment.) A population’s evolution can also be affected by chance, known as genetic drift (in the illustration, a random event reduced the population of an ant colony.) The results in this Letter imply that, under natural selection, a trait A with high variability \(\sigma _A\) can evolve faster than a trait B with small variability \(\sigma _B\). At the same time, the trait with higher variability is more susceptible to the effect of random fluctuations in populations due to genetic drift. In this way, the rate limits can be used to discriminate quantifiable traits in terms of their maximum evolutionary rates and their responsiveness to different evolutionary forces.
Rate limits on biological processes
Consider a set of types denoted by indexes \(\{j\}\) with an evolving number of individuals \(n_j = n_j(t)\) and a total (evolving) population \(N = \sum _j n_j\) (I will also analyze results in terms of frequencies of types in the next section). Let r denote the growth rate of the population, with components \(r_j :=\dot{n}_j/n_j\) for type j 17,18. Then, the rate of change in the expectation value \(\langle A \rangle = \sum _j a_j n_j/N\) of a quantitative trait A satisfies
The covariance \(\text {cov}(A,B) :=\left\langle A B \right\rangle - \left\langle A \right\rangle \!\left\langle B \right\rangle\) characterizes the correlations between two quantities A and B 17. See the Supplementary Material for the proof of Eq. (1). Throughout this work, I use da/dt or \(\dot{a}\) interchangeably to denote time derivatives.
In the context of evolutionary biology, Eq. (1) is known as the (time-continuous) Price equation 17,19,20,21. Equation (1) is a mathematical identity that holds under very general assumptions (e.g., differentiability) about the underlying dynamics of the system. Similar equations describe the dynamics of classical systems with evolving probability distributions 22 and of open quantum systems 23. Reference 24 discusses the connections between the Price equation in biology and statistical physics, while Ref. 12 contains a detailed analysis of the way that different biological factors contribute to each term in the Price equation.
The first term in the Price Eq. (1), \(\langle \dot{A} \rangle = \sum _j \dot{a}_j n_j/N\), describes changes in \(\langle A \rangle\) due to explicit time dependence in the values \(a_j\) of the trait. The second term in the Price equation, \(\sum _j a_j \tfrac{d}{dt}(n_j/N) = \text {cov}(A,r)\), corresponds to the change in the average \(\langle A \rangle\) of a trait due to the population changes. Using that the covariance is bounded by the product of standard deviations 17,25, the latter term is constrained by
Here,
are the variances of the trait A and of the growth rate r, respectively. (The standard deviation \(\sigma _A\) is the square root of the variance.)
The inequality in Eq. (2) sets a fundamental trade-off between the rate of change of a biological trait A and (i) the variability \(\sigma _A\) in the trait and (ii) the variability \(\sigma _r\) in the growth rate r with which the populations \(\{n_j\}\) change: fast evolution requires a variable trait and variable population growth rate. I equate the variability of a trait A with its standard deviation \(\sigma _A =\sqrt{\langle A^2 \rangle - \langle A \rangle ^2}\) 25. Note that, from Eq. (1), one concludes that the sign of \(d\langle A \rangle - \langle \dot{A} \rangle\) depends on whether the trait A is positively or negatively correlated with the growth rate r.
Trade-off relations like Eq. (2), typically referred to as speed limits in physics, also constrain the dynamics of quantum 23,26,27 and classical 22 physical systems. In the latter setting, the variance of r is replaced by the Fisher information \(\mathcal {I}_F\). The Fisher information is a measure of the speed with which a probability distribution evolves. For a time-dependent probability distribution \(p_j\), it is given by \(\mathcal {I}_F:=\sum _{j} p_j \! \big ( \frac{\dot{p}_j}{p_j} \big )^2\) 28. Defining \(p_j :=n_j/N\) as the frequency of occurrence of type j, I prove in the Supplementary Material that indeed \(\sigma _r = \sqrt{\mathcal {I}_F}\). Related limits to biological systems in terms of the Fisher information have been considered in Refs. 15,16.
While the constraint in Eq. (2) is extremely general, its practical usefulness may be hindered by the difficulty in relating the variability in the growth rate \(\sigma _r\) (or, equivalently, the Fisher information) to the relevant parameters that govern the dynamics of a concrete system. My goal is to derive bounds (mathematical inequalities) on the rate of change of biological quantities—or rate limits for short—tailored to evolutionary processes.
Limits to replicator evolutionary processes
Under the assumption that mutation rates between types are negligible, the replicator equation,
can be used to model population dynamics 17,29,30 (see Refs. 31,32,33,34 for applications of the replicator equation to various other fields). Here, \(p_j :=n_j/N\) is the frequency of occurrence of type j, and the fitness \(f_j \equiv f_j(\{p_k\},t)\) characterizes whether the frequency \(p_j\) of a type increases or decreases: the populations of types with positive excess fitness, \(f_j \ge \langle f \rangle\), tend to grow relative to their peers 35.
Using Eq. (4), it holds that \(r_j = \dot{p}_j/p_j + \dot{N}/N = f_j - \langle f \rangle + \dot{N}/N\), which in turn leads to \(\sigma _r = \sigma _f\). Thus, Eq. (2) implies that any evolutionary process that can be modeled by the replicator equation is constrained by
where I used that the covariance is invariant under the addition of uniform functions 25. The rate of change in natural selection processes is thus limited by the variability of the fitness of the population and the variability of the quantity of interest.
The rate limit in Eq. (5) for the replicator equation implies constraints on the dynamics of arbitrary quantitative traits A. If A has no explicit time dependence, i.e., if the \(a_j\)’s are constant, then Eq. (5) becomes a bound on the evolutionary rates \(d \langle A \rangle /dt\), discriminating slowly evolving traits from those that can change rapidly. In plain terms, the inequality says that evolution is slow for systems with homogeneous fitness functions, where \(\sigma _f \approx 0\) (neutral selection regime). In contrast, evolution can be faster on systems with a diverse population such that \(\sigma _f\) is large (natural selection regime). This mathematically formalizes and quantifies the common understanding that diversity serves as an evolutionary resource 6,7,36, in this case by enabling fast evolution whenever a trait and fitness have variability across a population. These variabilities can only occur in sufficiently diverse populations.
Equation (5) involves terms that may be reminiscent of the breeder’s equation. In the breeder’s equation, \(\Delta \langle A \rangle = S h^2\), the net change \(\Delta \langle A \rangle\) in a trait is governed by a measure of heritability (h) and the selection coefficient S. The selection coefficient measures covariance between fitness and a trait, so \(S = \text {cov}(A,f)\) 37. Then, Eq. (5) relates S in the breeder’s equation to the trait and fitness variabilities: \(|S| \le \sigma _A \, \sigma _f\). This may be useful in scenarios where the heritability is known but the selection coefficient is not, or, possibly, to study trait changes beyond the regime of applicability of the breeder’s equation 38. (Note, in particular, that the breeder’s equation is less general than Price’s 37.)
It is natural to wonder whether the left-hand and right-hand sides of the inequality (5) are similar (i.e., whether the bound is saturated). When this happens, knowledge of the standard deviations \(\sigma _A\) and \(\sigma _r\) suffices to estimate the evolutionary rate. This happens whenever A has a linear relationship with the growth rate r, i.e., \(a_j \propto r_j + c\) where c is independent of j, in which case \(\text {cov}(A,r)= \sigma _A \, \sigma _r\) 22. This is the case for the fitness function under replicator dynamics, so Eq. (1) yields
This corollary of the general rate limit (5) provides a simple proof of Fisher’s fundamental theorem of natural selection 1,2,3,4. It shows that Fisher’s claim is exact for (i) evolutionary processes modeled by a replicator equation with (ii) fitness functions that are independent of time, in which case \(\frac{d \langle f \rangle }{dt} = \sigma _f^2\). In situations with more general fitness functions \(f_j = f_j(\{p_k\},t)\), Eq. (6) provides a generalized version of Fisher’s theorem whereby the velocity with which fitness changes due to changes in population frequencies equals the fitness variance.
Limits to evolutionary processes with mutations
Mutations are a crucial driving force in realistic evolutionary processes 39,40,41. Mutations between types can be described by the replicator-mutator, or quasispecies model:
\(Q_{kj} \ge 0\) is a dimensionless transition matrix that models mutations between types, which satisfies \(\sum _{j} Q_{kj} = 1\) 42,43,44,45. The replicator Eq. (4) is recovered when the mutation matrix is the identity, \(Q_{kj} = \delta _{kj}\).
The general inequality (2) holds in this case, too. Note, though, that while for the replicator dynamics the variance in the growth rate equals the variance in fitness, \(\sigma _r^2 = \sigma _f^2\), this is no longer the case for dynamics with mutations. Relating the variance in the growth rate (or equivalently, the Fisher information) to biologically relevant quantities in concrete settings remains an interesting problem to be explored.
Alternatively, I define the mutation-driven distribution
I interpret \(\Pi\) as the frequency with which a given type would hypothetically occur in the future if evolution were only driven by mutations, or, possibly more biologically relevant, in regimes where strong mutation dominate over natural selection processes 39. Note that \(\Pi = p\) in the mutation-less regime.
Then, I prove in the Supplementary Material one of the main results of this work:
\(\langle A \rangle _\Pi :=\sum _j \Pi _j a_j\) and \(\sigma _A^\Pi :=\sqrt{\langle A^2 \rangle _\Pi - \langle A \rangle _\Pi ^2}\) are the average and the standard deviation evaluated in the mutation-driven distribution \(\Pi\). I emphasize that the general result in Eq. (2) does not imply the rate limit (9). The replicator-mutator evolutionary model, given by Eq. (7), was crucial to derive the latter bound.
The rate of change of any trait A is thus constrained by the quantity’s standard deviation evaluated in the mutation-driven distribution \(\Pi\), and the standard deviation of the fitness of the system. As in the mutation-less setting, diversity in the population is seen to give rise to less constrained evolution rates.
One could be puzzled by the appearance of a term in Eq. (9) that directly depends on the average fitness \(\langle f \rangle\) of the population and not just on relative fitness values. After all, shifting the fitness by an additive constant \(f_j \rightarrow f_j + c\) in the replicator Eq. (4) does not affect population dynamics. However, this is not the case in the mutator-replicator Eq. (7), where an additive constant c on the fitness function leads to a change in \(\dot{p}_j\) of \(c(\Pi _j - p_j)\), which depends on how the distribution p and mutation-driven distribution \(\Pi\) differ. The absolute values of the fitness function play a dynamical role in a system with mutations, and this is manifested in the rate limit (9).
To simplify interpretation, let us momentarily consider the case when A does not explicitly depend on time, i.e., the \(a_j\)’s are constant. Then, Eq. (9) and the triangle and reverse triangle inequalities imply upper and lower rate limits,
Here, one can identify two distinct sources that contribute to the evolutionary rates of the system. One of the sources, \(\sigma ^\Pi _A \, \sigma _f\), involves the standard deviation of the fitness function, and the standard deviation of the trait of interest evaluated in the mutation-driven distribution. The remaining term depends on the averages of fitness and the quantity of interest. This is somewhat reminiscent of the speed limits for open quantum systems, where two distinct sources to the dynamics of a system lead to additive contributions to the ultimate speed with which a quantity can evolve, which in turn allows to derive lower bounds on speed 23. A different bound on evolutionary rates under natural selection and mutations was derived in Ref. 16 in terms of contributions to the Fisher information (i.e., more in the spirit of Section “Rate limits on biological processes”). In contrast, evaluating Eqs. (9) and (10) only requires knowledge of averages and standard deviations.
The results in this article can be used to discriminate traits with fast or slow evolutionary rates and how different evolutionary forces affect them. Given knowledge of expectation values \(\{ \langle f \rangle , \langle A \rangle , \langle A \rangle _\Pi \}\) and standard deviations \(\{ \sigma _f, \sigma _A, \sigma ^\Pi _A \}\), the right-hand side of Eqs. (10a) and (10b) constrain the evolutionary rates of a trait A. The rate limits allow mathematically identifying two extremes of evolutionary regimes: (i) The maximum evolution rate of a trait that is robust to mutations, with \(\langle A \rangle _\Pi \approx \langle A \rangle\), is bounded by the variabilities in the trait and fitness; (ii) Meanwhile, a population with uniform fitness \(f_j \approx f_k\), for which \(\sigma _f \approx 0\), leads to constrained dynamics where \(\left| d \langle A \rangle /dt \right| \approx \langle f \rangle \big | \langle A \rangle _\Pi - \langle A \rangle \big |\). Natural selection dominates in case (i), often identified as the weak or neutral selection regime 46, while mutations dominate in case (ii). I illustrate this in Fig. 2. (Also see the Supplementary Material for a model illustrating these claims and the inequalities derived in this article.)
We can use Eqs. (9) and (10) to study the evolution rates of quantities often considered to characterize populations. The entropy \(S :=-\sum _j p_j \ln p_j\), also known as the Shannon-Wiener index in ecology, has been used to measure the diversity in a population 47. While \(S \approx 0\) if only one type k occurs with \(p_k \approx 1\), one has \(S = \ln N\) if N types are equally likely to occur. Taking \(\{ a_j \equiv I_j :=-\ln p_j\}\) and using that \(\dot{S} = -\sum _j \dot{p}_j \ln p_j\) from conservation of probability, Eq. (9) implies that
Here, the relative entropy (sometimes called the Kullback-Leibler divergence) \(S\big ( p \Vert \Pi \big ) :=-\sum _j p_j \ln \left( \frac{\Pi _j}{p_j} \right)\) serves as a proxy for the distance between the distribution p and the mutation-driven distribution \(\Pi\) 48,49,50,51. When \(S\big ( p \Vert \Pi \big )\) is small it is hard to distinguish p from \(\Pi\). Note that \(\sigma ^\Pi _I \ge 0\) but it is unbounded from above. In cases with an homogeneous population (\(\sigma _f \approx 0\)) and finite \(\sigma ^\Pi _I\), mutations drives variability since the entropy only evolves due to the mismatch between the two distributions, with a rate \(\frac{dS}{dt} = \langle f \rangle \, S\big (p \Vert \Pi \big )\). In the mutation-less case in which \(p = \Pi\), Theorem 8 of Ref. 52 implies that \(\sigma ^\Pi _I = \sigma _I \le \sqrt{\tfrac{1}{4} \ln ^2(N-1) + 1}\). Then, the maximum entropy rate scales as \(\max \big | \dot{S} \big | \approx \ln N \, \sigma _f/2\) for large N.
As a second example, consider the rate of change of the average fitness function for dynamics that incorporate mutations. Equation (9) becomes
which coincides with Eq. (6) in the mutation-less regime or for neutral mutations that do not influence the fitness landscape, since then \(\langle f \rangle _\Pi = \langle f \rangle\) and \(\sigma ^\Pi _f = \sigma _f\).
Equation (12) imposes the most stringent constraints on evolution when the fitness is completely certain when evaluated in the distribution p or in the mutation-driven distribution \(\Pi\), i.e., when \(\sigma ^\Pi _f = 0\) or \(\sigma _f = 0\). This is the neutral selection strong mutation regime (center column of Fig 2), when types have comparable fitness and natural selection is not a strong driver of evolution. In these cases, dynamics are due to the difference in fitness between the two distributions, and \(d\langle f \rangle / dt - \big \langle \dot{f} \big \rangle = \langle f \rangle \big ( \langle f \rangle _\Pi - \langle f \rangle \big )\), that is, mutations dominate. This also illustrates that beneficial and deleterious mutations can be naturally characterized by \(\langle f \rangle _\Pi - \langle f \rangle \ge 0\) and \(\langle f \rangle _\Pi - \langle f \rangle \le 0\), respectively, depending on the change in the average fitness of the population. Note that the contribution to the fitness rate depends on the population’s average fitness but not on its standard deviation—Fisher’s theorem of natural selection does not hold in this regime.
In contrast to the neutral selection regime, faster evolutionary rates are possible for systems with variable fitness landscapes. In this way, rate limits can be used to mathematically formalize evolutionary regimes according to the dynamical forces in action. I will explore this further at the end of the next section.
Discriminating evolutionary regimes. The rate limits derived in this work constrain the evolution of quantifiable traits of populations driven by natural selection, mutations, or genetic drift. The inequalities depend on (i) the expectation values \(\{ \langle f \rangle , \langle A \rangle , \langle A \rangle _\Pi \}\) and (ii) standard deviations \(\{ \sigma _f, \sigma _A, \sigma ^\Pi _A \}\) of the trait of interest and of the fitness profile of the population, and (iii) on the maximum strength \(\Vert \gamma \Vert _\infty\) of genetic drift stochastic forces. Here, \(\langle A \rangle _\Pi\) and \(\sigma ^\Pi _A\) are evaluated in the distribution \(\Pi _j\) defined in Eq. (8), which characterizes type’s frequencies if the system only evolved due to mutations. Equation (15) can be used to mathematically delineate three distinct evolutionary regimes: [(a)—Strong selection regime] When \(\sigma ^\Pi _A \sigma _f \, \gg \, \big \{ \langle f \rangle | \langle A \rangle _\Pi \!-\! \langle A \rangle |, \Vert \gamma \Vert _\infty \sigma _A \big \}\), the contributions of mutations or genetic drift to the changes in a trait are negligible. This can be identified as the regime where natural selection is the dominant evolutionary force. In it, the maximum rate of a trait is constrained by its standard deviation \(\sigma _A^\Pi\) evaluated in the mutation-driven distribution \(\Pi\): smaller trait variability implies smaller maximum rates. [(b)—Strong mutation regime] Natural selection and genetic drift contributions are negligible when \(\langle f \rangle | \langle A \rangle _\Pi - \langle A \rangle | \, \gg \, \big \{ \sigma ^\Pi _A \sigma _f, \Vert \gamma \Vert _\infty \sigma _A \big \}\). Then, the rate of change of a trait is determined by the change induced by the mutation-driven distribution, and Eq. (9) implies that \(d\langle A \rangle /dt - \langle \dot{A} \rangle \approx \langle f \rangle (\langle A \rangle _\Pi \!-\! \langle A \rangle )\). [(c) – Genetic drift regime] The variability of a trait determines its susceptibility to changes due to genetic drift. In the regime \(\Vert \gamma \Vert _\infty \sigma _A \, \gg \, \big \{ \sigma ^\Pi _A \sigma _f, \langle f \rangle | \langle A \rangle _\Pi \!-\! \langle A \rangle | \big \}\) where the stochastic force dominates, the total change in a trait over a time \(\tau\) is bounded by the integrated variance of the trait and the maximum drift strength.
Limits to stochastic evolutionary processes
Equations (9) and (12) provide generalizations of Fisher’s fundamental theorem of natural selection to replicator-mutator dynamics in terms of universal constraints on evolutionary rates. However, the replicator and replicator-mutator equations are only simplified deterministic models for the dynamics of infinite populations. In realistic scenarios, stochastic forces typically known as genetic drift play a preponderant role in evolutionary processes 53,54.
To account for genetic drift, I consider a stochastic replicator-mutator equation,
as a model for stochastic evolutionary processes. The first two terms coincide with the replicator-mutator Eq. (7) and describe natural selection and mutation dynamics. The last term models genetic drift driven by noise terms \(dW_j\).
In the stochastic difference Eq. (13), the population changes by \(dp_j\) during a time-step dt, over which a stochastic Wiener noise \(dW_j\) randomly affects the system. The strength of the genetic drift of population j is characterized by the ‘volatility parameter’ \(\gamma _j\) 55, while the term \(\sum _l\! \gamma _l p_l dW_l\) ensures that the frequencies \(p_j\) remain normalized. Following the rules of Itô calculus, the zero-mean Wiener noises \(dW_j\) satisfy \(dW_j^2 = dt\) and \(\overline{dW_j dW_k} = \delta _{jk} dt\), where \(\overline{F}\) represents the average of a function F(p) over realizations of the stochastic noise 56.
Dynamics are thus driven by natural selection, mutations, and random genetic drift. Note that the latter dominate dynamics for infinitesimally short times 56. Therefore, in this case, it is more meaningful to focus on integrated changes \(\tfrac{1}{\tau }\int _0^\tau \big ( d \langle A \rangle - \langle dA \rangle \big )\) during a time interval \(\tau\) rather than on rates of change. The prefactor \(\tfrac{1}{\tau }\), which makes the quantity a time-average, ensures the same units as in Eqs. (5) and (9).
Then, I prove in the Supplementary Material that,
where \(\Vert \gamma \Vert _\infty :=\max _j {\{\gamma _j\}}\). Equation (14) shows that the noise-averaged change in \(\langle A \rangle\) with respect to the change \(\big [ \int _0^\tau \sum _{jk} p_k Q_{kj}\big (f_k\!-\!\langle f \rangle \big ) a_j dt \big ]\) due to natural selection and mutations, is bounded by the variability \(\sigma _A\) and by the maximum strength \(\Vert \gamma \Vert _\infty\) of the stochastic forces. (One can apply the same techniques I used to derive Eq. (14) to other Wiener-noise dynamical models 53,57,58, see the Supplementary Material for one such example.)
In plain terms, Eq. (14) provides a criteria to discriminate situations in which biological quantities are resilient against genetic drift. A trait’s high variability can make its dynamics more susceptible to changes due to stochastic forces. In contrast, traits with small variability \(\sigma _A \approx 0\) over a period \(\tau\) evolve as if natural selection and mutations were the only driving evolutionary forces.
The final result of this paper singles out the contributions of natural selection, mutations, and genetic drift to an inequality on the noise-averaged change of a trait over a time interval \(\tau\),
There are many ways in which evolutionary regimes can be (often phenomenologically) classified depending on their predominant driving forces on a given model 46,59,60,61,62. Equation (15) yields a formal way to mathematically demarcate such regimes by comparing the relative strengths between the sources that dominate the maximum evolutionary rate:
-
(a)
Strong selection regime
$$\begin{aligned}\sigma ^\Pi _A \sigma _f \, \gg \, \Big \{ \langle f \rangle \big | \langle A \rangle _\Pi - \langle A \rangle \big | \,, \, \Vert \gamma \Vert _\infty \sigma _A \Big \}, \end{aligned}$$ -
(b)
Strong mutation regime
$$\begin{aligned}\langle f \rangle \big | \langle A \rangle _\Pi - \langle A \rangle \big | \, \gg \, \Big \{ \sigma ^\Pi _A \sigma _f \,, \, \Vert \gamma \Vert _\infty \sigma _A \Big \},\end{aligned}$$ -
(c)
Genetic drift regime
$$\begin{aligned} \Vert \gamma \Vert _\infty \sigma _A \, \gg \, \Big \{ \sigma ^\Pi _A \sigma _f \,, \, \langle f \rangle \big | \langle A \rangle _\Pi - \langle A \rangle \big | \Big \}. \end{aligned}$$
In words, the characteristic regimes depend on the values of three quantities: the product of the fitness and trait standard deviations (\(\sigma ^\Pi _A \sigma _f\)), the mutation-driven change in a trait (\(\langle f \rangle \big | \langle A \rangle _\Pi - \langle A \rangle \big |\)), and the genetic drift’s intensity times the trait standard deviation (\(\Vert \gamma \Vert _\infty \sigma _A\)). (a) The strong selection regime occurs when the product of the fitness and trait standard deviations are significantly larger than the other two factors. Under this regime, mutation and genetic drift can be ignored. (b) The strong mutation regime occurs when the mutation-driven change in a trait (weighed by the average fitness) is the larger term. In it, natural selection and genetic drift are negligible. (c) Finally, the genetic drift regime is identified by the intensity of the genetic drift times the trait’s standard deviation being the significantly larger term. In it, the trait’s evolution is solely due to random events.
In each of the three regimes detailed above, dynamics of a trait is provably due to the corresponding biological drive. Note that these are trait-dependent criteria — as conveyed by Fig. 2, certain traits are more responsive than others to a given evolutionary force.
Potential implications to experiments
In this section, I discuss how the theoretical inequalities derived in Sections “Rate limits on biological processes” and “Limits to stochastic evolutionary processes” could be contrasted with the evolution of concrete biological systems. I hope to bridge the results in this article, which borrow ideas from physics, to questions of interest in biology.
In any experiment where one has access to the frequencies \(p_j = n_j/N\) with which types occur and to the variability of a trait, evaluating the results in Section “Rate limits on biological processes” is straightforward. In this case, evaluating the left- and right-hand sides of Eq. (2) from experimental data would inform whether a trait evolves as fast as possible. A trait that saturates the inequality is exploiting all biological resources to evolve as fast as nature allows.
Evaluating the rate limits on systems driven predominantly by natural selection and mutations (in Section “Limits to evolutionary processes with mutations”) will involve purposely designed experiments. Some of the quantities in Eq. (9) are likely to be directly available from experimental data (e.g., \(\langle A \rangle\) and \(\langle f \rangle\)). However, \(\langle A \rangle _\Pi\) and \(\sigma _a^\Pi\) are the trait average and standard deviation evaluated in the mutation-driven frequency distribution \(\Pi\) [Eq. (8)]. Evaluating them would require an experiment where the intensity of mutations can be “cranked up” relative to that of natural selection. Experiments with microbial evolution and growth arena (MEGA) plates, introduced in Ref. 63, could be used for this purpose. Reference 64 discusses another method to influence the fitness landscape and favour certain mutations.
Finally, Section “Limits to stochastic evolutionary processes” includes the effect of random stochastic influences (genetic drift). Climate change is believed to affect the occurrence and impact of extreme weather events 65,66. Let us consider modeling the effects of extreme events on a population by stochastic forces, as in “Limits to stochastic evolutionary processes”. Then, we can ask: What properties of a biological system make it resilient against the effects of climate change? Relying on the conclusions drawn after Eq. (14), one can argue that traits with less variability will remain unaffected by extreme events for longer. This is because the right-hand side of Eq. (14) is small for traits with low variability. Traits with high variability can be affected more. Note that this doesn’t necessarily mean that the former case yields more resilience against climate change than the latter: it is possible that populations will need to evolve fast to survive and, if so, Eq. (14) suggests that variability may help 67,68.
Discussion
Recent works, mostly within the fields of quantum physics 23,27,69,70 and classical statistical mechanics 71,72,73,74,75,76, but also biology 14,15,16, derived trade-off relations that constrain the dynamics of observables. I find it remarkable that constraints that bound speed, while being extremely general, are saturated in certain paradigmatic cases. In stochastic thermodynamics, for example, the rate at which heat is exchanged with a system and the rate at which a system’s entropy changes saturates their speed limits for Gibbs states with (arbitrarily) time-dependent temperature 22. In an unrelated setting, certain quantum algorithms have been shown to compute as fast as allowed by speed limits on quantum annealing 77.
Here, replicator dynamics—a model often used to describe mutation-less population dynamics in evolutionary biology—have been found to saturate the rate limit for the fitness of a population (the left and right-hand sides of Eq. (5) coincide for \(A=f\)). Higher variability in fitness leads to higher evolutionary rates. This last fact was known by Fisher, who connected the rate of change of the average fitness to the variability in the fitness of a population.
More generally, I have shown that connections between the variability in fitness and evolutionary rates hold for dynamics that incorporate mutations and genetic drift. Moreover, these connections can be made for the evolution of arbitrary traits.
Then, the main strengths of our results are that (i) Eq. (9) generalizes the constraints on evolutionary rates to arbitrary quantifiable traits and not just the fitness function, (ii) the inequality in Eq. (12) concisely shows how fitness variability influences dynamics, in the spirit of Fisher’s theorem, and that (iii) Eq. (14) accounts for stochastic evolutionary forces. See Refs. 11,15,16 for related work generalizing Fisher’s theorem of natural selection, and deriving distinct speed limits on evolutionary processes driven by natural selection and mutations. Speed limits that incorporate stochastic dynamics, however, are mostly unexplored.
There is an extensive literature devoted to understanding the factors that limit biological evolutionary rates 5,6,7,8,9,10. However, the complex nature of this question implies that most of such work is devoted to observations on particular traits within particular types or species, based on experimental observations, and sometimes phenomenological. In contrast, the results in this paper take the role of general mathematical theorems that hold for any biological system whose dynamics can be described by the models considered [Eqs. (4), (7), or (13)]. The techniques introduced here open a path to derive rate limits for modifications of these stochastic dynamical models, too.
It is worth emphasizing that the main results in this work, which constrain rates for dynamics with mutations [Eqs. (9–12)] and which discriminate quantities that are resilient against stochastic driving forces [Eqs. (14, 15)], are not implied by the general speed limits derived in Ref. 22 nor by recent works generalizing the fundamental theorem of natural selection 15,16. Making use of the structure of the differential equations that model biological evolutionary processes was crucial in deriving the new rate limits. In particular, speed limits on stochastic noisy dynamics such as Eq. (13) are mostly unexplored. It would be interesting to study applications of the methods used here to other models of evolutionary processes that account for genetic drift and finite size effects 54,78,79,80, other stochastic drives 81,82, or to quantify the environment’s effect on trait plasticity 83. At the same time, while I have argued and illustrated in Fig. 2 how the rate limits are useful in discriminating observable traits in terms of their evolutionary rates, it would be insightful to test these ideas in biological systems from experimental data.
The overarching aim of this work was to mathematically formalize the common knowledge that biological diversity influences evolutionary processes. The results shown here suggest and quantify ways in which variability, as measured by the standard deviations of fitness and of the biological traits of interest, can serve as a resource by allowing for faster evolution. It is tempting to ponder about the implications to related fields 84,85,86.
Data availability
The simulations used in this work are available upon request to the corresponding author.
References
Fisher, R. A. The genetical theory of natural selection (The Clarendon Press, 1930). https://doi.org/10.5962/bhl.title.27468.
Price, G. R. Fisher’s ‘fundamental theorem’ made clear. Ann. Hum. Genet. 36, 129. https://doi.org/10.1111/j.1469-1809.1972.tb00764.x (1972).
Edwards, A. The fundamental theorem of natural selection. Theor. Popul. Biol. 61, 335. https://doi.org/10.1006/tpbi.2002.1570 (2002).
Baez, J. C. The fundamental theorem of natural selection. Entropy 23, 1436. https://doi.org/10.3390/e23111436 (2021).
Dmitriew, C. M. The evolution of growth trajectories: What limits growth rate?. Biol. Rev. 86, 97. https://doi.org/10.1111/j.1469-185X.2010.00136.x (2011).
Blowsand, M. W. & Hoffmann, A. A. A reassessment of genetic limits to evolutionary change. Ecology 86, 1371. https://doi.org/10.1890/04-1209 (2005).
Hoffmann, A. et al. Evolutionary limits and constraints. Princet. Guide Evol.https://doi.org/10.1515/9781400848065-034 (2014).
Frean, M., Rainey, P. B. & Traulsen, A. The effect of population structure on the rate of evolution. Proc. R. Soc. B Biol. Sci. 280, 20130211. https://doi.org/10.1098/rspb.2013.0211 (2013).
Tkadlec, J., Pavlogiannis, A., Chatterjee, K. & Nowak, M. A. Population structure determines the tradeoff between fixation probability and fixation time. Commun. Biol. 2, 138. https://doi.org/10.1038/s42003-019-0373-y (2019).
Evans, A. R. et al. The maximum rate of mammal evolution. Proc. Natl. Acad. Sci. 109, 4187. https://doi.org/10.1073/pnas.1120774109 (2012).
Basener, W. F. & Sanford, J. C. The fundamental theorem of natural selection with mutations. J. Math. Biol. 76, 1589. https://doi.org/10.1007/s00285-017-1190-x (2018).
Lion, S. Theoretical approaches in evolutionary ecology: Environmental feedback as a unifying perspective. Am. Nat. 191, 21. https://doi.org/10.1086/694865 (2018).
Marsland, R. III., Cui, W. & J. M. Horowitz,. The thermodynamic uncertainty relation in biochemical oscillations. Journal of the Royal Society Interface16, 20190098. https://doi.org/10.1098/rsif.2019.0098 (2019).
Genthonand, A. & Lacoste, D. Universal constraints on selection strength in lineage trees. Phys. Rev. Res. 3, 023187. https://doi.org/10.1103/PhysRevResearch.3.023187 (2021).
Adachi, K., Iritani, R. & Hamazaki, R. Universal constraint on nonlinear population dynamics. Commun. Phys. 5, 1. https://doi.org/10.1038/s42005-022-00912-4 (2022).
Hoshino, M., Nagayama, R., Yoshimura, K., Yamagishi, J. F. & Ito, S. Geometric speed limit for acceleration by natural selection in evolutionary processes. Phys. Rev. Res. 5, 023127. https://doi.org/10.1103/PhysRevResearch.5.023127 (2023).
Rice, S. H. Evolutionary Theory: Mathematical and Conceptual Foundations 1st edn. (Sinauer Associates, Inc., 2004).
Vandermeer, J. How populations grow: The exponential and logistic equations. Nat. Educ. Knowl. 3, 15 (2010).
Price, G. R. Selection and covariance. Nature 227, 520. https://doi.org/10.1038/227520a0 (1970).
Frank, S. A. Natural selection. IV. The price equation. J. Evol. Biol. 25, 1002. https://doi.org/10.1111/j.1420-9101.2012.02498.x (2012).
Queller, D. C. Fundamental theorems of evolution. Am. Nat. 189, 345. https://doi.org/10.1086/690937 (2017).
Nicholson, S. B., García-Pintos, L. P., del Campo, A. & Green, J. R. Time-information uncertainty relations in thermodynamics. Nat. Phys. 16, 1211 (2020).
García-Pintos, L. P., Nicholson, S. B., Green, J. R., del Campo, A. & Gorshkov, A. V. Unifying quantum and classical speed limits on observables. Phys. Rev. X 12, 011038. https://doi.org/10.1103/PhysRevX.12.011038 (2022).
Frank, S. A. & Bruggeman, F. J. The fundamental equations of change in statistical ensembles and biological populations. Entropy 22, 1395. https://doi.org/10.3390/e22121395 (2020).
Colquhoun, D. Lectures on Biostatistics: An Introduction to Statistics with Applications in Biology and Medicine (David Colquhoun, 1971).
Mandelstamand, L. & Tamm, I. The uncertainty relation between energy and time in non-relativistic quantum mechanics. In Selected Papers 115–123 (Springer, 1991). https://doi.org/10.1007/978-3-642-74626-0_8
Deffnerand, S. & Campbell, S. Quantum speed limits: From Heisenberg’s uncertainty principle to optimal quantum control. J. Phys. A Math. Theor. 50, 453001. https://doi.org/10.1088/1751-8121/aa86c6 (2017).
Cramér, H. Mathematical Methods of Statistics Vol. 43 (Princeton University Press, 1999).
Maynard Smith, J. The theory of games and the evolution of animal conflicts. J. Theor. Biol. 47, 209. https://doi.org/10.1016/0022-5193(74)90110-6 (1974).
Schusterand, P. & Sigmund, K. Replicator dynamics. J. Theor. Biol. 100, 533. https://doi.org/10.1016/0022-5193(83)90445-9 (1983).
Sato, Y. & Crutchfield, J. P. Coupled replicator equations for the dynamics of learning in multiagent systems. Phys. Rev. E 67, 015206. https://doi.org/10.1103/PhysRevE.67.015206 (2003).
Safarzyńska, K. & van den Bergh, J. C. Evolutionary models in economics: A survey of methods and building blocks. J. Evolut. Econ. 20, 329. https://doi.org/10.1007/s00191-009-0153-9 (2010).
Cressmanand, R. & Tao, Y. The replicator equation and other game dynamics. Proc. Natl. Acad. Sci. 111, 10810. https://doi.org/10.1073/pnas.1400823111 (2014).
Bloembergen, D., Tuyls, K., Hennes, D. & Kaisers, M. Evolutionary dynamics of multi-agent learning: A survey. J. Artif. Intell. Res. 53, 659. https://doi.org/10.1613/jair.4818 (2015).
Smith, J. M. Evolutionary Genetics (Oxford University Press, 1998).
Naeem, S., Duffy, J. E. & Zavaleta, E. The functions of biological diversity in an age of extinction. Science 336, 1401. https://doi.org/10.1126/science.1215855 (2012).
Heywood, J. S. An exact form of the breeder’s equation for the evolution of a quantitative trait under natural selection. Evolution 59, 2287. https://doi.org/10.1111/j.0014-3820.2005.tb00939.x (2005).
Morrissey, M. B., Kruuk, L. E. & Wilson, A. J. The danger of applying the breeder’s equation in observational studies of natural populations. J. Evol. Biol. 23, 2277. https://doi.org/10.1111/j.1420-9101.2010.02084.x (2010).
Gregory, T. R. Understanding natural selection: Essential concepts and common misconceptions. Evol. Educ. Outreach 2, 156. https://doi.org/10.1007/s12052-009-0128-1 (2009).
Carlin, J. Mutations are the raw materials of evolution. Nat. Educ. Knowl. 3, 10 (2011).
Hershberg, R. Mutation-the engine of evolution: studying mutation and its role in the evolution of bacteria. Cold Spring Harb. Perspect. Biol. 7, a018077. https://doi.org/10.1101/cshperspect.a018077 (2015).
Eigenand, M. & Schuster, P. Stages of emerging life-five principles of early organization. J. Mol. Evol. 19, 47. https://doi.org/10.1007/BF02100223 (1982).
Hofbauer, J. The selection mutation equation. J. Math. Biol. 23, 41. https://doi.org/10.1007/BF00276557 (1985).
Wilke, C. O. Quasispecies theory in the context of population genetics. BMC Evol. Biol. 5, 1. https://doi.org/10.1186/1471-2148-5-44 (2005).
Nowak, M. A. Evolutionary Dynamics: Exploring the Equations of Life (Harvard University Press, 2006). https://doi.org/10.2307/j.ctvjghw98.
Wildand, G. & Traulsen, A. The different limits of weak selection and the evolutionary dynamics of finite populations. J. Theor. Biol. 247, 382. https://doi.org/10.1016/j.jtbi.2007.03.015 (2007).
Spellerberg, I. F. & Fedor, P. J. A tribute to Claude Shannon (1916–2001) and a plea for more rigorous use of species richness, species diversity and the ‘Shannon–Wiener’ index. Glob. Ecol. Biogeogr. 12, 177 (2003).
Weibull, J. W. Evolutionary Game Theory (MIT Press, 1997).
Karev, G. P. Replicator equations and the principle of minimal production of information. Bull. Math. Biol. 72, 1124. https://doi.org/10.1007/s11538-009-9484-9 (2010).
Frank, S. A. Natural selection. V. How to read the fundamental equations of evolutionary change in terms of information theory. J. Evol. Biol. 25, 2377. https://doi.org/10.1111/jeb.12010 (2012).
Baez, J. C. & Pollard, B. S. Relative entropy in biological systems. Entropy 18, 46. https://doi.org/10.3390/e18020046 (2016).
Reeb, D. & Wolf, M. M. Tight bound on relative entropy by entropy difference. IEEE Trans. Inf. Theory 61, 1458. https://doi.org/10.1109/TIT.2014.2387822 (2015).
Fosterand, D. & Young, P. Stochastic evolutionary game dynamics. Theor. Popul. Biol. 38, 219. https://doi.org/10.1016/0040-5809(90)90011-J (1990).
Czupponand, P. & Traulsen, A. Understanding evolutionary and ecological dynamics using a continuum limit. Ecol. Evol. 11, 5857. https://doi.org/10.1002/ece3.7205 (2021).
Bedford, T. & Hartl, D. L. Optimization of gene expression by natural selection. Proc. Natl. Acad. Sci. 106, 1133. https://doi.org/10.1073/pnas.0812009106 (2009).
Jacobs, K. Stochastic Processes for Physicists: Understanding Noisy Systems (Cambridge University Press, 2010). https://doi.org/10.1017/CBO9780511815980.
Cabrales, A. Stochastic replicator dynamics. Int. Econ. Rev. 41, 451. https://doi.org/10.1111/1468-2354.00071 (2000).
Feng, T.-J., Li, C., Zheng, X.-D., Lessard, S. & Tao, Y. Stochastic replicator dynamics and evolutionary stability. Phys. Rev. E 105, 044403. https://doi.org/10.1103/PhysRevE.105.044403 (2022).
Rouzine, I. M., Wakeley, J. & Coffin, J. M. The solitary wave of asexual evolution. Proc. Natl. Acad. Sci. 100, 587. https://doi.org/10.1073/pnas.242719299 (2003).
Desaiand, M. M. & Fisher, D. S. Beneficial mutation-selection balance and the effect of linkage on positive selection. Genetics 176, 1759. https://doi.org/10.1534/genetics.106.067678 (2007).
Sniegowski, P. D. & Gerrish, P. J. Beneficial mutations and the dynamics of adaptation in asexual populations. Philos. Trans. R. Soc. B Biol. Sci. 365, 1255. https://doi.org/10.1098/rstb.2009.0290 (2010).
Levy, S. F. et al. Quantitative evolutionary dynamics using high-resolution lineage tracking. Nature 519, 181. https://doi.org/10.1038/nature14279 (2015).
Baym, M. et al. Spatiotemporal microbial evolution on antibiotic landscapes. Science 353, 1147. https://doi.org/10.1126/science.aag0822 (2016).
Carpenter, A. C., Feist, A. M., Harrison, F. S., Paulsen, I. T. & Williams, T. C. Have you tried turning it off and on again? Oscillating selection to enhance fitness-landscape traversal in adaptive laboratory evolution experiments. Metab. Eng. Commun. 17, e00227. https://doi.org/10.1016/j.mec.2023.e00227 (2023).
Franco, B. C. et al. Climate change impacts on the atmospheric circulation, ocean, and fisheries in the southwest South Atlantic Ocean: A review. Clim. Change 162, 2359. https://doi.org/10.1007/s10584-020-02783-6 (2020).
Trenberth, K. E., Fasullo, J. T. & Shepherd, T. G. Attribution of climate extreme events. Nat. Clim. Chang. 5, 725. https://doi.org/10.1038/nclimate2657 (2015).
Poloczanska, E. S. et al. Responses of marine organisms to climate change across oceans. Front. Mar. Sci. 3, 180581. https://doi.org/10.3389/fmars.2016.00062 (2016).
Pecl, G. T. et al. Biodiversity redistribution under climate change: Impacts on ecosystems and human well-being. Science 355, e9214. https://doi.org/10.1126/science.aai9214 (2017).
Gong, Z. & Hamazaki, R. Bounds in nonequilibrium quantum dynamics. Int. J. Mod. Phys. Bhttps://doi.org/10.1142/S0217979222300079 (2022).
Carabba, N., Hörnedal, N. & del Campo, A. Quantum speed limits on operator flows and correlation functions. Quantum 6, 884. https://doi.org/10.22331/q-2022-12-22-884 (2022).
Uffink, J. & Van Lith, J. Thermodynamic uncertainty relations. Found. Phys. 29, 655. https://doi.org/10.1023/A:1018811305766 (1999).
Barato, A. C. & Seifert, U. Thermodynamic uncertainty relation for biomolecular processes. Phys. Rev. Lett. 114, 158101. https://doi.org/10.1103/PhysRevLett.114.158101 (2015).
Vo, V. T., Van Vu, T. & Hasegawa, Y. Unified approach to classical speed limit and thermodynamic uncertainty relation. Phys. Rev. E 102, 062132. https://doi.org/10.1103/PhysRevE.102.062132 (2020).
Horowitz, J. M. & Gingrich, T. R. Thermodynamic uncertainty relations constrain non-equilibrium fluctuations. Nat. Phys. 16, 15 (2020).
Dechant, A. & Sasa, S.-I. Improving thermodynamic bounds using correlations. Phys. Rev. X 11, 041061. https://doi.org/10.1103/PhysRevX.11.041061 (2021).
Das, S. & Green, J. R. Speed limits on deterministic chaos and dissipation. Phys. Rev. Res. 5, L012016. https://doi.org/10.1103/PhysRevResearch.5.L012016 (2023).
García-Pintos, L. P., Brady, L. T., Bringewatt, J. & Liu, Y.-K. Lower bounds on quantum annealing times. Phys. Rev. Lett. 130, 140601. https://doi.org/10.1103/PhysRevLett.130.140601 (2023).
Crow, J. F. & Morton, N. E. Measurement of gene frequency drift in small populations. Evolution 9, 202. https://doi.org/10.2307/2405589 (1955).
Traulsenand, A. & Hauert, C. Stochastic evolutionary game dynamics. In Reviews of Nonlinear Dynamics and Complexity, Chap. 2 (ed. Schuster, H. G.) 25–61 (John Wiley & Sons, Ltd, Hoboken, 2009). https://doi.org/10.1002/9783527628001.ch2.
Tataru, P., Simonsen, M., Bataillon, T. & Hobolth, A. Statistical inference in the Wright–Fisher model using allele frequency data. Syst. Biol. 66, e30. https://doi.org/10.1093/sysbio/syw056 (2016).
Huang, W., Hauert, C. & Traulsen, A. Stochastic game dynamics under demographic fluctuations. Proc. Natl. Acad. Sci. 112, 9064. https://doi.org/10.1073/pnas.1418745112 (2015).
Vasconcelos, V. V., Santos, F. P., Santos, F. C. & Pacheco, J. M. Stochastic dynamics through hierarchically embedded Markov chains. Phys. Rev. Lett. 118, 058301. https://doi.org/10.1103/PhysRevLett.118.058301 (2017).
Berg, M. P. & Ellers, J. Trait plasticity in species interactions: A driving force of community dynamics. Evol. Ecol. 24, 617. https://doi.org/10.1007/s10682-009-9347-8 (2010).
Whitley, D. A genetic algorithm tutorial. Stat. Comput. 4, 65. https://doi.org/10.1007/BF00175354 (1994).
Yu, X. & Gen, M. Introduction to Evolutionary Algorithms (Springer Science & Business Media, 2010). https://doi.org/10.1007/978-1-84996-129-5.
Hodgsonand, G. M. & Knudsen, T. Generative replication and the evolution of complexity. J. Econ. Behav. Org. 75, 12. https://doi.org/10.1016/j.jebo.2010.03.008 (2010).
Acknowledgements
I thank José Ignacio Arroyo, Jake Bringewatt, Adolfo del Campo, Lucas Fernandes, Daniel Freire, Jason Green, Schuyler Nicholson, Lodovico Scarpa, and Nicolás Rubido for discussions related to this work. This work was supported by the Beyond Moore’s Law project of the Advanced Simulation and Computing Program at Los Alamos National Laboratory (LANL) managed by Triad National Security, LLC, for the National Nuclear Security Administration of the U.S. Department of Energy under contract 89233218CNA000001. I also acknowledge support by the DOE Office of Science, Office of Advanced Scientific Computing Research, Accelerated Research for Quantum Computing program, Fundamental Algorithmic Research for Quantum Computing (FAR-QC) project. Work at LANL was carried out under the auspices of the US DOE and NNSA under contract No. DEAC52-06NA25396.
Author information
Authors and Affiliations
Contributions
L.P.G.P. was the sole contributor to all aspects of this work.
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
García-Pintos, L.P. Limits on the evolutionary rates of biological traits. Sci Rep 14, 11314 (2024). https://doi.org/10.1038/s41598-024-61872-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-61872-z
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.