Abstract
Introduction:
Hirayama disease, a type of cervical flexion myelopathy, is a rare neurological disease characterized by muscular atrophy of the forearms and hands. Generally, the pathology is limited to the gray matter of the anterior horns in the lower cervical spinal cord. However, in rare cases the damage can spread to the white matter and present as long tract signs.
Case Presentation:
We report on a 30-year-old female whose onset presented as unilateral muscle atrophy of the right hand in her teens. Despite conservative treatment using a cervical collar, she developed prolonged bilateral muscle atrophy, sensory disturbance and spastic gait, along with bladder and rectal disturbances. Her hands were frozen into a ‘claw-like’ gesture and her intrinsic muscles were highly atrophic. She was unable to unclench her hands. Although the space available for the spinal cord was large, the spinal cord was highly atrophic. She had local kyphosis with a large (61°) flexion range of motion. During flexion the spinal cord was stretched, resulting in contact with the posterior wall of the vertebrae. Posterior fusion surgery was performed to prevent progression of the myelopathy. After surgery, she gained mild improvement in both muscle strength and her hand’s movement. However, her spastic gait and muscle atrophy remained.
Discussion:
Most cases of cervical flexion myelopathy as represented by Hirayama disease have a self-limiting benign prognosis. However, some cases can develop advanced myelopathy with long tract signs. Long-term follow-up is recommended for these cases because they may require early surgical treatment.
Similar content being viewed by others
Introduction
Hirayama disease, a nonprogressive, juvenile, spinal muscular atrophy of the distal upper limbs is a rare neurological disease characterized by muscular atrophy and weakness of the forearms and hands.1 The disease primarily affects men aged 10–30 years and is regarded as a type of cervical flexion myelopathy.2,3 Its pathology is thought to be due to repeated or sustained flexion causing necrosis of the anterior horns of the lower cervical cord through chronic microcirculatory changes in the area of the anterior spinal artery.3,4
Generally, Hirayama disease is considered to be a benign disorder. While the muscle atrophy initially progresses, spontaneous arrest usually occurs several years after onset. However, there have been a few rare cases of Hirayama disease whose symptoms progressed in adulthood.5 Here, we report on a rare female case whose onset was similar to Hirayama disease and developed advanced myelopathy.
Informed consent
The patient was informed that if she agreed, the authors would like to publish the details of her case in a scientific journal for others to learn from. The types of non-identifying demographic and medical information that would be included was discussed with her, and she gave consent.
Case presentation
The subject was a 30-year-old female with no developmental problems in early childhood. At the age of 12, the patient was aware of muscle weakness of her right hand and experienced difficulty throwing and using a tennis racket. By the age of 20, she frequently incurred burn injuries on her right hand due to impaired temperature sense. At 26, she frequently stumbled and needed to rely on the railing to walk down the stairs and was unable to close a button or use chopsticks. She presented to the neurology department at our hospital at the age of 28. Upon examination, the intrinsic and forearm muscles were observed to be severely atrophic, and the patient demonstrated a spastic gait along with impaired position sense. A neurologist suspected severe Hirayama disease and prescribed a soft collar to prevent flexion. However, as her symptoms gradually deteriorated, she began to present with staggering gait, frequent micturition and constipation. At the age of 30 years, she visited Dr. Keizo Hirayama who proposed a diagnosis of Hirayama disease as a second opinion. According to his diagnosis, her symptoms initially presented as Hirayama disease; however, it was more reasonable to regard her disease as advanced cervical flexion myelopathy. She returned to our hospital and referred to the orthopedics department for surgery.
Examination
Her hands were frozen into a ‘claw-like’ gesture (Figure 1), and a grip and release test6 was very slow (Supplementary Video 1). She also had impaired pain and temperature sense and impaired position sense. She presented with a spastic gait (Supplementary Video 2) and had problems with her micturition and constipation. Her deltoid, biceps and brachioradialis muscles were graded at 4 out of 5 in strength. Wrist extensors and triceps were grade 3/5, whereas wrist flexor, finger extensor and interosseous muscles were grades 1–2/5 in strength. Her right biceps', both brachioradialis and both triceps' tendon reflexes showed hyporeflexia, while left bicep's tendon reflex was normal. An inverted triceps tendon reflex was observed on her left side, which suggested that the left side C5 myotome was preserved. Her patellar and achilles tendon reflexes both showed hyperreflexia. Romberg sign and Babinski reflex were positive. When her neck was placed in a flexion position, she experienced pain that radiated into the upper extremities.
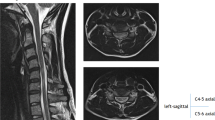
A plain radiograph showed 31° of local kyphosis at C4/C7 (Figure 2a). Range of motion was very large (Figure 2b and c). Especially, the extent of flexional range was 61°. A neutral MRI revealed a highly atrophic spinal cord between C4 and C7 (Figure 3a) with the presence of a ‘snake-eye’ appearance (Figure 3b). In flexion, the MRI showed that the spinal cord was stretched, resulting in contact with the posterior wall of the vertebrae (Figure 3c). There was no tethered cord, and a CT myelogram in flexion showed that there was a space between the spinal cord and the dorsal part of dura membrane (Figure 4).
A magnetic resonance image (MRI) revealing atrophy of the spinal cord resulting in a ‘snake-eye’ appearance. (a) Sagittal MRI showing the area of lesion. (b) Axial MRI showing ‘snake-eye’ appearance (open arrow). (c) MRI taken during flexion, showing a stretched spinal cord, resulting in contact with the posterior wall of the vertebrae (open arrow).
Both motor and sensory nerve conduction tests showed that peripheral nerve function was within the normal range. However, there was a significant decrease in the frequency of F waves of the median nerves, and the amplitude of the compound muscle action potential of the ulnar nerve was significantly decreased. An electromyogram revealed severe neurogenic changes to muscles innervated by the C5 to T1 myotomes.
Her history, examination and electromyogram suggested that the spinal cord damage was progressive with signs of ascending bilateral anterior horn cell involvement, as well as pyramidal, spinothalamic and posterior column long tracts. At the beginning, she was reluctant to undergo surgery because we informed her that the recovery was limited and that she had become highly adapted to her capability of the hands. However, after discussion, she consented for surgery to prevent progression of myelopathy. We chose a posterior fixation from C4 to C7 (Figure 5). We performed neither laminectomy nor duraplasty because MRI in the neutral position showed that the space for the cord was large, and flexion CT myelogram showed the dural membrane was not tight. Bony union was achieved 1 year after surgery. She gained mild improvement in biceps, wrist extensor and triceps and was able to unclench her left hand (Supplementary Video 3). The radiating pain of upper extremities during flexion decreased. However, her spastic gait and muscle atrophy remained.
Discussion
To the best of our knowledge, there have not been any reported cases of flexion myelopathy in females that developed both gait and urinary disturbances. Generally most cases of cervical flexion myelopathy as represented by Hirayama disease have a self-limiting benign prognosis. While the muscle atrophy initially progresses, spontaneous arrest usually occurs several years after onset. However, this patient developed advanced myelopathy with long tract signs. We speculated that the reason why the patient had such a severe myelopathy was because she had a very large flexional ROM of 61°, with normal female volunteers in their 30s generally showing a ROM in flexion of 27°.7 The local kyphosis with this large flexion ROM provided a mechanism by which the severe spinal cord damage was induced.
Flexion myelopathy as a type of cervical myelopathy
Cervical spondylotic myelopathy, which is normally seen in middle-aged and older patients is the most common type of cervical myelopathy. In cervical spondylotic myelopathy, the spinal cord is compressed during extension by a pincer mechanism, and long tract signs are more common than segmental-sign-like muscle atrophy. On the other hand, cervical spondylotic amytrophy usually presents with muscle atrophy in the upper extremities with little sensory disturbance or long tract signs.8,9 This pathology is thought to be due to selective damage of the anterior root and/or anterior horn. Because the peak age of onset of cervical spondylotic amytrophy is different from that of flexion myelopathy (Hirayama disease), these three diseases, cervical spondylotic myelopathy, cervical spondylotic amytrophy and Hirayama disease, are clinically differentiated. However, there have been reports of a few cases of Hirayama disease that developed symptoms of gait disturbance and/or proximal upper-extremity muscle atrophy in middle age after temporary arrest of symptoms in their teens.10,11 These cases, in addition to ours, suggest that cervical spondylotic myelopathy, cervical spondylotic amytrophy and flexion myelopathy can overlap and should be treated as a kind of myelopathy.12
Pathomechanism of flexion myelopathy
There are several theories regarding the pathology of flexion myelopathy. The most common one is the ‘over-stretch’ theory.13,14 In this theory, there is a disproportion of growth between the spine and the spinal cord, making the spinal cord relatively short compared to the spine.15,16 During flexion, this results in the cord and dura being stretched by 17.6% between C2 and T1 at maximum.17–19 As the spinal cord takes the shortest pathway through the spinal canal,20 this results in the spinal cord not only being stretched but also pressed into the posterior wall of the vertebrae during flexion.4 In addition, some researchers have proposed the tight dural canal theory.2,21 In this theory, a nonelastic dural membrane compresses the spinal cord into the vertebral body. In support of this, some studies have reported that microscopic examination revealed that the dural membrane harvested from flexion myelopathy shows a loss of both elastic fiber and normal wavy structure.22, 23
Some reports have shown that the spinal cord was damaged only by maintaining the neck in a flexed position.24–26 This mechanism is supported by a three-dimensional finite element model.27 During mild flexion, the stresses are concentrated in the gray matter, especially the anterior and posterior horns. This model may correspond to typical Hirayama disease. On the other hand, during severe flexion, in addition to the stresses placed on gray matter, there are also high levels of stress on the white matter.
In the present case, there was ‘snake-eye’ appearance between the C4 and C7 segments, suggesting cystic necrosis in the gray matter. However, the spread of the cystic necrosis to the white matter was unclear because of the severe atrophy of the spinal cord.
The growth spurt associated with puberty is thought to be associated with the onset of flexion myelopathy.12 This may be the reason why men are predominantly affected. Normally, the motoneuron pools in the anterior horn are distributed through a few upper and lower segments. As a result, transverse damage limited to a single segmental level does not induce muscle atrophy because compensatory reinnervation prevents damage. This explains how the onset of flexion myelopathy usually lags a few years behind puberty-associated growth spurts, as muscle atrophy only becomes obvious after the damage spreads to a few upper and lower segments.12
Treatment options
In a small number of cases, the slow process of the disease and limited response to therapy make it difficult to achieve a consensus for treatment. Normally, a cervical collar is the first choice for flexion myelopathy.28 However, while a cervical collar is thought to be effective during the early phase of the disease when muscle atrophy is mild, surgery is usually the preferred method for treating advanced cases. Surgery can be considered as fusion surgery (anterior or posterior) and duraplasty. Although there were some differences in severity of symptoms, a review of the literature revealed 19 authors reporting 62 surgically treated cases5,14,29–44 (Table 1). In most of these cases, the primary symptom was unilateral muscle atrophy. However, there were several reported cases showing mild, long tract signs such as hyperreflexia of the lower extremities, while only four cases showed gait disturbance.5,14,29,34
In a comparison of operative methods, 7 surgeons performed posterior fusion29,31,34,36,3940,43 (13 cases), 5 surgeons performed anterior fusion32,35,4142,44 (16 cases) and 5 surgeons performed duraplasty5,23,30,33,38 (16 cases). Additionally, two surgeons performed either posterior (5 cases) or anterior fusion (12 cases). The choice of surgery was mainly based on the surgeons’ preference and/or concept of the pathogenesis rather than clinical characteristics like age, severity of myelopathy and radiographic parameters. Most of the reports found that surgery improved grip power, although muscle atrophy remained.
Conclusion
Most cases of cervical flexion myelopathy as represented by Hirayama disease have a self-limiting benign prognosis. However, some cases can develop advanced myelopathy with long tract signs. Long-term follow-up is recommended for these cases because they may require early surgical treatment.
References
Hirayama K, Tsubaki T, Toyokura Y, Okinaka S . Juvenile muscular atrophy of unilateral upper extremity. Neurology 1963; 13: 373–380.
Iwasaki Y, Tashiro K, Kikuchi S, Kitagawa M, Isu T, Abe H . Cervical flexion myelopathy: a ‘tight dural canal mechanism’. Case report. J Neurosurg 1987; 66: 935–937.
Hirayama K, Tomonaga M, Kitano K, Yamada T, Kojima S, Arai K . Focal cervical poliopathy causing juvenile muscular atrophy of distal upper extremity: a pathological study. J Neurol Neurosurg Psychiatry 1987; 50: 285–290.
Henderson FC, Geddes JF, Vaccaro AR, Woodard E, Berry KJ, Benzel EC . Stretch-associated injury in cervical spondylotic myelopathy: new concept and review. Neurosurgery 2005; 56: 1101–1113.
Fujimoto Y, Oka S, Tanaka N, Nishikawa K, Kawagoe H, Baba I . Pathophysiology and treatment for cervical flexion myelopathy. Eur Spine J 2002; 11: 276–285.
Hosono N, Sakaura H, Mukai Y, Kaito T, Makino T, Yoshikawa H . A simple performance test for quantifying the severity of cervical myelopathy. J Bone Joint Surg Br 2008; 90: 1210–1213.
Yukawa Y, Kato F, Suda K, Yamagata M, Ueta T . Age-related changes in osseous anatomy, alignment, and range of motion of the cervical spine. Part I: radiographic data from over 1,200 asymptomatic subjects. Eur Spine J 2012; 21: 1492–1498.
Brain WR, Northfield D, Wilkinson M . The neurological manifestations of cervical spondylosis. Brain 1952; 75: 187–225.
Keegan JJ . The cause of dissociated motor loss in the upper extremity with cervical spondylosis. J Neurosurg 1965; 23: 528–536.
Hashiguchi S, Ogasawara N, Watanabe A, Kawachi Y, Miki N . Cervical spondylotic amyotrophy associated with Hirayama's disease. Intern Med 1997; 36: 647–650.
Kameyama T, Ando T, Mimatsu K, Sofue G . [Delayed exacerbation of cervical myelopathy in a case of juvenile muscular atrophy of unilateral distal upper extremity]. Rinsho Shinkeigaku 1997; 37: 60–63.
Kameyama T . Relationship between cervical flexion myelopathy and cervical spondylotic amyotrophy. Sekitsui sekizui J 2009; 22: 1147–1155.
Yada K, Tachibana S, Okada K .Spinal cord lesion due to relative imbalance of cervical spine and cervical cord. 1981 Annual Report of prevention and Treatment For The Congenital Anomalies of the Spine and Spinal Cord, The Ministry of Health Welfare of Japan. Vol. S56, 1982, pp 48–55 .
Mitsui K, Iida H, Tachibana S, Yada K . Surgical treatment for flexion myelopathy with overstretch syndrome. Nihon Seikeigeka Gakkai Zasshi 1990; 64: 325.
Kohno M, Takahashi H, Yagishita A, Tanabe H . ‘Disproportion theory’ of the cervical spine and spinal cord in patients with juvenile cervical flexion myelopathy. A study comparing cervical magnetic resonance images with those of normal controls. Surg Neurol 1998; 50: 421–430.
Pradhan S, Gupta RK . Magnetic resonance imaging in juvenile asymmetric segmental spinal muscular atrophy. J Neurol Sci 1997; 146: 133–138.
Breig A, el-Nadi AF . Biomechanics of the cervical spinal cord. Relief of contact pressure on and overstretching of the spinal cord. Acta Radiol Diagn (Stockh) 1966; 4: 602–624.
Breig A, Turnbull I, Hassler O . Effects of mechanical stresses on the spinal cord in cervical spondylosis. A study on fresh cadaver material. J Neurosurg 1966; 25: 45–56.
Reid JD . Effects of flexion-extension movements of the head and spine upon the spinal cord and nerve roots. J Neurol Neurosurg Psychiatry 1960; 23: 214–221.
Penning L, van der Zwaag P . Biomechanical aspects of spondylotic myelopathy. Acta Radiol Diagn 1966; 5: 1090–1103.
Kikuchi S, Shinpo K, Niino M, Higashi T, Tashiro K . Cervical myelopathy due to a ‘tight dural canal in flexion’ with a posterior epidural cavity. Intern Med 2002; 41: 746–748.
Yoshiyama Y, Tokumaru Y, Arai K . Flexion-induced cervical myelopathy associated with fewer elastic fibers and thickening in the posterior dura mater. J Neurol 2010; 257: 149–151.
Mochizuki M, Goto S, Kita T, Kobayashi K, Sodeyama T, Murata J et al. [Evaluation of the dynamicity of spinal cord and dura mater until neck full flexion in flexion myelopathy and the histopathologhy of its dura mater]. Seikeigeka 1996; 47: 162–166.
Kaye KL, Ramsay D, Young GB . Cervical flexion myelopathy after valproic acid overdose. Spine (Phila Pa 1976) 2001; 26: E459–E461; discussion E462.
Morandi X, Riffaud L, Amlashi SF, Brassier G . Extensive spinal cord infarction after posterior fossa surgery in the sitting position: case report. Neurosurgery 2004; 54: 1512–1515;discussion 1515-1516.
Rau CS, Liang CL, Lui CC, Lee TC, Lu K . Quadriplegia in a patient who underwent posterior fossa surgery in the prone position. Case report. J Neurosurg 2002; 96 (1 Suppl): 101–103.
Kato Y, Kataoka H, Ichihara K, Imajo Y, Kojima T, Kawano S et al. Biomechanical study of cervical flexion myelopathy using a three-dimensional finite element method. J Neurosurg Spine 2008; 8: 436–441.
Tokumaru Y, Hirayama K . [Cervical collar therapy for juvenile muscular atrophy of distal upper extremity (Hirayama disease): results from 38 cases]. Rinsho Shinkeigaku 2001; 41: 173–178.
Okamoto Y, Kawahara N, Murakami H, Demura S, Yoshioka K, Tomita K . Cervical flexion myelopathy: two cases report. Tyubu seisaishi 2010; 53: 205–206.
Arrese I, Rivas JJ, Esteban J, Ramos A, Lobato RD . A case of Hirayama disease treated with laminectomy and duraplasty without spinal fusion. Neurocirugia (Astur) 2009; 20: 555–558; discussion 558.
Patel TR, Chiocca EA, Freimer ML, Christoforidis GA . Lack of epidural pressure change with neck flexion in a patient with Hirayama disease: case report. Neurosurgery 2009; 64: E1196–E1197; discussion E1197.
Watanabe K, Hasegawa K, Hirano T, Endo N, Yamazaki A, Homma T . Anterior spinal decompression and fusion for cervical flexion myelopathy in young patients. J Neurosurg Spine 2005; 3: 86–91.
Ito H, Takai K, Taniguchi M . Cervical duraplasty with tenting sutures via laminoplasty for cervical flexion myelopathy in patients with Hirayama disease: successful decompression of a ‘tight dural canal in flexion’ without spinal fusion. J Neurosurg Spine 2014; 21: 743–752.
Sakai K, Ono K, Okamoto Y, Murakami H, Yamada M . Cervical flexion myelopathy in a patient showing apparent long tract signs: a severe form of Hirayama disease. Joint Bone Spine 2011; 78: 316–318.
Shiohara K, Morishita M, Kanzaki K, Saito H, Ochiai J . An experience of antrior decompression fixation for cervical flexion myelopathy. Higashinihon Seisai 2003; 15: 604–607.
Inasaka R, Horii H, Iwamura Y, Ishii M . Posterior cervical fusion for flexion myelopathy. Kanagawa Igakukaizasshi 1999; 26: 343.
Kohno M, Takahashi H, Ide K, Yamakawa K, Saitoh T, Inoue K . Surgical treatment for patients with cervical flexion myelopathy. J Neurosurg 1999; 91 (1 Suppl): 33–42.
Konno S, Goto S, Murakami M, Mochizuki M, Motegi H, Moriya H . Juvenile amyotrophy of the distal upper extremity: pathologic findings of the dura mater and surgical management. Spine (Phila Pa 1976) 1997; 22: 486–492.
Ohnari K, Yamada K, Hachiya M, Fujii H, Fujisita A, Okubo T et al. Pathophysiology of cervical flexion myelopathy in young patients. Rinsho Seikeigeka 1993; 28: 1119–1127.
Kondo S, Ohnari K, Saito Y, Mitsuhashi T, Iwamura Y, Toguchi A et al. Posterior fusion in patients with cervical flexion myelopathy: a report of two cases. Kanto J Orthop Traumatol 1991; 22: 32–39.
Ogasa T, Taniguchi M, Maeda A, Yamamoto K, Samejima S, Aoki Y et al. A case report of anterior spinal fusion for Hirayama's disease. Seikeigeka 1992; 43: 83–87.
Katayama K, Umefuji T, Satou Y, Onodera N, Yasuda M, Harada Y . A case of similar Hirayama's disease with long tract sign. Rinsho Seikeigeka 1990; 25: 672–677.
Masaki T, Hashida H, Sakuta M, Kunogi J . [A case of flexion myelopathy presenting juvenile segmental muscular atrophy of upper extremities--a successful treatment by cervical spine immobilization]. Rinsho Shinkeigaku 1990; 30: 625–629.
Okamoto A, Okawa A, Nakai O, Yamaura I . A case of cervical flexion myelopathy. East Jpn J Clin Orthop 1989; 1: 13–17.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Spinal Cord Series and Cases website
Supplementary information
Rights and permissions
About this article
Cite this article
Fujimori, T., Tamura, A., Miwa, T. et al. Severe cervical flexion myelopathy with long tract signs: a case report and a review of literature. Spinal Cord Ser Cases 3, 17016 (2017). https://doi.org/10.1038/scsandc.2017.16
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/scsandc.2017.16
This article is cited by
-
Hirayama disease associated with neurogenic bladder dysfunction
Acta Neurologica Belgica (2023)
-
Finger trembling improvement after surgery in Hirayama disease: a case report
Spinal Cord Series and Cases (2022)
-
Impact of various cervical surgical interventions in patients with Hirayama’s disease—a narrative review and meta-analysis
Neurosurgical Review (2021)
-
Expert’s Comment concerning Grand Rounds case entitled “Hirayama disease” by D. C. Kieser, P. J. Cox, S. C. J. Kieser (Eur Spine J; 2018: doi:10.1007/s00586-018-5545-9)
European Spine Journal (2018)