Abstract
Study design:
Prospective cohort study.
Objective:
This study was designed to neurophysiologically characterize motor control recovery after spinal cord injury (SCI).
Setting:
University of Louisville, Louisville, Kentucky, USA.
Material:
Eleven acute SCI admissions and five non-injured subjects were recruited for this study.
Methods:
The American Spinal Injury Association Impairment Scale (AIS) was used to categorize injury level and severity at onset. Multimuscle surface electromyography (sEMG) recording protocol of reflex and volitional motor tasks was initially performed between the day of injury and 11 days post onset (6.4±3.6, mean±s.d. days). Follow-up data were recorded for up to 17 months after injury. Initial AIS distribution was as follows: 4 AIS-A; 2 AIS-C; 5 AIS-D. Multimuscle activation patterns were quantified from the sEMG amplitudes of selected muscles using a vector-based calculation that produces separate values for the magnitude and similarity of SCI test-subject patterns to those of non-injured subjects for each task.
Results:
In SCI subjects, overall sEMG amplitudes were lower after SCI. Prime mover muscle voluntary recruitment was slower and multimuscle patterns were disrupted by SCI. Recovery occurred in 9 of the 11 subjects, showing an increase in sEMG amplitudes, more rapid prime mover muscle recruitment rates and the progressive normalization of the multimuscle activation patterns. The rate of increase was highly individualized, differing over time by limb and proximal or distal joint within each subject and across the SCI group.
Conclusions:
Recovery of voluntary motor function can be quantitatively tracked using neurophysiological methods in the domains of time and multimuscle motor unit activation.
Similar content being viewed by others
Introduction
Spinal cord injury (SCI) in humans produces profound changes in the central nervous system sensory and motor function that are known to recover to varying degrees.1 At the time of injury, spinal motor neurons and the interneurons that modulate their excitability suffer significant loss of synaptic connections from interneurons lost within the immediate injury zone and severed long-tract fibers.2 Demyelination of other long-tract fibers disrupts their ability to deliver impulses to the synapses of spinal motor circuitry within and distal to the injury zone.3 Further, neuropathological examination of human SCI reveals that each lesion is highly individualized with regard to severity, and to which and to what extent descending tracts are spared.4 This broad range of injury locations and severity results in considerable diversity in impairment and extent of recovery observed within the population of people with SCI. As a result, detailed characterization of SCI impact on central nervous system motor control processing is a complex and difficult task.
The recovery of voluntary capability after SCI is currently tracked through the use of clinical expert-examiner scales such as the American Spinal Injury Association Impairment Scale (AIS).5 Using the AIS framework, an examiner grades the force produced against resistance by voluntary contraction of selected muscles to derive a categorical classification of severity, A to E. However, the rate of recruitment of the target muscle and the activation of synergistic muscles with concurrent inhibition of antagonistic muscles necessary to efficiently perform functional volitional movement are not captured by such scales. Neurophysiological methods using surface electromyography (sEMG) recorded from multiple muscles have demonstrated an ability to identify and quantify evidence of preserved translesional conduction in chronic, clinically complete , paralyzed SCI subjects.6 Calancie and co-workers7 showed that examiner-judged sEMG amplitudes for selected muscles could be used to describe recovery of voluntary contraction during the first 6 months after SCI. A vector-based method to quantify sEMG to calculate an index relating the multimuscle activation patterns produced to perform volitional movement in a test subject to those of non-injured persons was published,8 and validity was tested in chronic SCI subjects.9 The study reported here was undertaken to neurophysiologically examine and quantify the return of the volitional recruitment of motor units within the domains of time and distribution across multiple muscles to characterize recovery from SCI.
Materials and methods
Assessments were carried out after informed consent was obtained as approved by the Institutional Review Board for human research of the University of Louisville. Persons with concomitant head injury were not enrolled in this study. Medical management strategies were not altered to accommodate this study.
Nineteen subjects were recruited from acute traumatic SCI admissions to the University of Louisville hospital. Eleven who completed follow-up studies at least 1 month after injury were analyzed for this report (Table 1). Initial neurophysiological information was recorded as early as was judged medically appropriate by attending physicians, between 1 and 11 days post onset (6.4±3.6 days, mean±s.d.). Seven underwent surgical stabilization before initial testing and two were stabilized between initial and follow-up recordings. Two saw no surgical intervention. All subjects received rehabilitation treatment after discharge from the acute care facility. Follow-up data were recorded between 1 and 17 months (206±137 days) post injury. Two subjects were female and ages ranged from 24 to 63 (45±12) years at the time of injury. Neurological injury levels ranged from C1 to T6. Initial AIS distribution was 4 AIS-A, 2 AIS-C and 5 AIS-D. Only one subject in this study, number 5, was taking antispasticity medication during follow-up recording. One female and four male non-injured subjects, age 20–59 (39±18) years, underwent the neurophysiological recording to provide normative data.
Clinical assessment was carried out on admission to the hospital using the AIS. The AIS provides a subjective estimation of voluntary contraction strength for five upper limb and five lower limb muscles, representing C5 to T1 and L2 to S1 levels.5 Initial AIS examinations were performed between the same day (n=4) and 5 (1.7±1.8) days before neurophysiological recording in 10 subjects. In one subject who was intubated, a complete initial AIS examination was not performed until 33 days following the neurophysiological recording. Final follow-up AIS exams were performed on the same day in four subjects, and between 4 and 360 (87.0±116.3) days before the final neurophysiological recording in the others.
Neurophysiological assessment was carried out using an expanded brain motor control assessment protocol10 with upper-limb tasks added to the published lower-limb tasks. The brain motor control assessment includes volitional and reflex motor tasks carried out with the subject in the supine position using published standards for administration and analysis. Following skin preparation, pairs of sEMG electrodes, spaced 2 cm apart, were placed, oriented parallel to the long axis of the muscle, over the right and left upper trapezius (UT), biceps brachi (BB), triceps brachi (TB), wrist extensors (WE), wrist flexors (WF), rectus abdominus (para-umbilical) (RA), quadriceps (Qd), adductor femoris (Add), hamstrings (H), tibialis anterior (TA) and soleus (Sol). Five minutes of relaxation was followed by three repetitions each of reinforcement maneuvers; unilateral voluntary movement of elbow, wrist, hip and knee and ankle joints; passive stretch, ankle clonus and plantar stimulation. For voluntary tasks, subjects were cued by an audible 5-s tone for each phase and instructed to ‘move and hold for the duration of the tone.’ Signals were recorded on a 32-channel AXON Eclipse Neuromonitoring System (AXON Systems Inc., Hauppauge, NY, USA) with a sampling rate of 2 kHz per channel and a band pass of 30–1 kHz.
Data reduction
sEMG was quantified using a root-mean-square algorithm that produced values in μV sec−1. Values from 5-s windows, the published standard for this analysis method, were averaged for each muscle from three trials each of elbow flexion and extension, wrist extension, hip and knee flexion and ankle dorsiflexion and plantar flexion. In some recordings, analysis windows for elbow extension and plantar flexion tasks were <5 s based on event marks. Hip and knee extension was not presented because it is not a true extension task, but rather an eccentric flexion when performed in the supine position. Background activity and noise were measured from a 1-s window before the motor task and subtracted from the average value for each channel. These values were used to calculate overall magnitude and the similarity index (SI), which compares the relative distribution across an appropriate set of muscles chosen for the task with that of a non-injured subject group.8 Response vectors (RVs) with one element per muscle were calculated for each task. RVs for elbow flexion and extension were calculated from right and left upper trapezius, biceps brachi and triceps brachi muscles. The wrist extension RV was calculated from right and left upper trapezius, biceps brachi, triceps brachi, wrist extensors and wrist flexors muscles. The elements for the hip and knee flexion RV were from the right and left quadriceps, adductor femoris and hamstring muscles. Right and left quadriceps, adductor femoris, hamstrings, tibialis anterior and soleus muscle values made up the RVs for ankle dorsiflexion and plantar flexion. The distal muscles were not included in elbow or hip and knee task prototypes because instructions to the subject did not specify how they were to be moved during those tasks. To generate the prototype response vectors (PRVs), the healthy subject RVs were first normalized and then averaged. The PRV is presented in dimensionless units for each task. The Euclidian length of the RV or PRV provided the magnitude value. The SI, a numerical expression of the relationship of the RV to the PRV, is computed as the cosine of the solid angle between the two vectors. A value of 1.0 for the SI means that the angle was zero and that the test subject's RV had an identical distribution of sEMG activity across muscles as did the non-injured subject group PRV for that task.
Quantification of the voluntary recruitment rate focused on four muscles considered to be the prime movers for their motor task: biceps brachi for elbow flexion, wrist extensors for wrist extension, quadriceps for hip flexion and tibialis anterior for ankle dorsiflexion. Elbow extension and plantar flexion were not evaluated for this measure because they were performed from an active state as the second phase of a two-phase movement. The sEMG from these muscles was processed into a root-mean-square envelope and filtered at 20 Hz to enhance envelope peak recognition. Prime mover activation times were measured as the time between the appearance of the first motor unit firing and the peak of the root-mean-square envelope. Again, three trials were averaged for each task.
Statistical analysis
Data sets were tested for normal distribution using skewness, kurtosis and omnibus normality tests. For normally distributed sets, two-tailed unpaired t-test calculations were carried out to compare non-injured with SCI group onset-to-peak, SI and magnitude values. Paired t-test analysis was used to compare initial with final session results for each variable. For sets that were not normally distributed, the Wilcoxon and Mann–Whitney tests were used to compare paired and non-paired data sets, respectively. Spearman's rank correlation coefficient (ρ) calculations were used to determine the strength of the relationships between non-parametric data sets, specifically between AIS motor scores and brain motor control assessment parameters. Significance was reached at the P<0.05 level. This analysis was conducted using NCSS/Pass software (v.2002, NCSS, Kaysville, UT, USA).
Results
Recovery of voluntary activation
The sEMG patterns were highly individualized as was their rate of change over time. Two subjects, numbers 10 and 11, were unable to activate motor units in muscles below their respective T1 and T6 lesions for the period covered by serial recordings at 4 and 17 months, respectively. They were unchanged throughout and therefore not included in the calculated values presented below. For the remaining nine subjects, unilateral voluntary motor tasks were analyzed for a maximum of 18 responding limbs for each task. Two subjects progressed from complete paralysis of both arms and legs to having differing degrees of ability in follow-up recordings. One subject, #7, developed the ability to perform only elbow flexion on one side. The other, #8, recovered for all tested tasks. Although the occurrence and degree of recovery varied across and within subjects and follow-up recording periods differed, the prevalence of volitional activation in this group of nine increased from 67 to 100% for elbow flexion, 44–94% for elbow extension, 56–89% for wrist extension, 69–87% for hip and knee flexion and 75–87% for ankle dorsiflexion, but plantar flexion was 75% throughout.
Prime mover onset-to-peak recruitment time
Figure 1 illustrates progressive increase in the volitional activation of the biceps brachi muscle in one representative subject. Initial prime mover onset-to-peak times from the SCI group were highly variable but significantly delayed in comparison with those of the non-injured group (P<0.01) (Table 2). Envelope peak amplitudes showed no significant difference between the two groups. One subject, #2, developed continuous background motor unit activity, which interfered with the determination of sEMG onset. Onset-to-peak times decreased significantly with recovery in the remaining eight subjects (P<0.05).
Biceps brachi sEMG during three repeated trials of voluntary elbow flexion (auditory cue at up arrow) and extension (down arrow) performed in the supine position by subject #5 (C4, AIS-D). Serial recordings made 9, 22 and 36 days after injury show typical characteristics of changing motor control in the prime mover muscle for the task. Note that, at first, only a few motor units can be fired; however, with recovery, an increase in motor unit firing occurs with a progressive decrease in the time from the onset of activity to the peak of activation. Also note the antagonistic co-activation of this muscle during the elbow extension phase of the motor task that developed over time.
sEMG amplitudes
Mean sEMG envelope amplitudes were significantly lower for SCI group elbow flexion and extension, wrist extension and ankle dorsiflexion (Table 3). Amplitudes increased between initial and final recordings but reached significance only for upper-limb tasks. During hip and knee flexion, contralateral hamstrings activity, an important component of the non-injured pattern, also increased significantly between initial and final recordings.
Multimuscle activation patterns
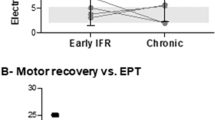
Variance within the non-injured group prototypes for the tested tasks was similar to those previously reported.8, 9 In initial recordings, SCI group mean SI and magnitude values were lower than those of the non-injured group (Table 3). The degree of recovery was indicated by increased SI and magnitude values at the end of the follow-up period. Figure 2 illustrates progressive change in the multimuscle activation patterns from no volitional activation through a pattern that included co-activation of antagonistic and distant muscles to the appropriate pattern for ankle dorsiflexion within an individual subject. Increases in SI and magnitude values occurred at different rates in different limb segments, yielding a different index profile for each of the motor tasks within each individual (Figure 3) and across the SCI group (Figure 4).
sEMG from the right (R) and left (L) quadriceps (Qd), adductor (Add), hamstrings (H), tibialis anterior (TA) and soleus (Sol) muscles for voluntary right ankle dorsiflexion performed by a subject (#9) with an initial injury level at C5, who improved to the level of L2. Serial recordings made 11, 27, 45 and 135 days post onset show the evolution of control in which the prime mover (RTA) amplitude increases, at first along with co-activation of other muscles, followed by the reduction of activation in all but the prime mover. The cue mark is 5 s.
Involuntary muscle activation
Within the group of nine people with SCI, five were able to relax completely during their initial examination, whereas four produced low-amplitude continuous activation of one or more muscles. This form of activity appeared on follow-up in three others and disappeared in two by the final recording. No ‘spontaneous’ episodic spasms were observed. Responses were recorded from the muscles stretched in three of the eight subjects in whom passive movements were performed during the initial examination. However, of the 10 muscles stretched in each study, only 1–3 responded in these three subjects. Responsiveness to stretch was found in follow-up recordings in six subjects, again not appearing in all 10 muscles tested. This responsiveness disappeared by the final recording in all but three of the nine subjects. Further, volitional activation of spasms distal to the injury was not seen in any initial recordings and developed on follow-up in only four subjects. Long-duration ankle clonus was present in only one subject on initial recording and disappeared on follow-up. Finally, on initial examination, eight of the nine responded to plantar stimulation and four of those were able to volitionally reduce the response. Final recordings showed asymmetrical results with 5 of 18 limbs not responding, 10 responding but which could be volitionally suppressed and 3 that could not be suppressed.
American Spinal Injury Association impairment scale
AIS motor score data were available for eight of these nine subjects covering a follow-up period of 25–420 (184±120) days post injury. Group mean motor scores increased significantly for elbow flexor and extensor, wrist extensor and hip flexor muscles (P<0.01) (Table 3). Ankle plantar flexor motor scores also increased significantly (P<0.05) but ankle dorsiflexor motor score increase was not significant. Only five recordings in four subjects were performed within a day of the AIS assessment. Within these recordings, AIS motor scores correlated significantly (P<0.01) with sEMG amplitude recorded from the prime mover muscle (ρ=0.82). Further, SI and magnitude values correlated with AIS motor scores (ρ=0.84 both; P<0.01). No subjects showed a decrease in AIS letter grade. For the nine in whom both upper and lower limbs were studied, AIS grades increased by one letter in three subjects: #2, 60 days before recording; #3, 90 days before recording; #6, day of recording. In these three subjects, magnitude and SI values for both upper- and lower-limb tasks increased in follow-up recordings (Table 4). Neurological injury level moved caudally in six subjects, rostrally in one, did not change in one and disappeared in one. Both magnitude and SI values increased for all (Table 4).
Discussion
The neurophysiological data presented here described recovery in terms of significant increases in (1) the ability to activate, on command, motor units within the prime mover muscle for each specific task; (2) the rate at which those motor units were recruited; (3) the ability to appropriately organize the relative distribution of motor-unit activation across prime mover, antagonist and distant muscles. The first of these three aspects of motor control is characterized by the AIS examination, which produces a motor score from which a severity grade (A to E) and neurological injury level are determined. All of these increased for upper-limb muscles during the follow-up term as did SI and magnitude. However, it should be pointed out that the pattern of motor unit firings in multiple muscles during unloaded movements as in the neurophysiological examination and the forces perceived by an examiner during resisted contractions are related but not equivalent. The second and third parameters quantify a previously unmeasured aspect of motor control and the analytic approaches that generate them avoid the statistical variance induced by subject-to-subject differences known to exist in sEMG raw amplitudes and the inherent inter and intrarater inconsistencies of clinical scales.
Potential mechanisms of recovery from SCI elucidated in animal models include the following: remyelination of long-tract fibers the axons and synaptic connections to interneurons and motor neurons of which remained intact but were deprived of their myelin cover within the injury zone;3 new synaptic connections formed to reoccupy space vacated at the time of injury from surviving long-tract axons;11, 12 new connections produced by sprouting from peripheral nerve fibers.13 The latter mechanism would likely be counterproductive for the recovery of voluntary motor control as it may increase responsiveness to muscle and cutaneous afferents and increase spasticity.13 In the current study, increased responsiveness to muscle stretch was transient, of low amplitude, in only a few muscles tested, and disappeared in all but three subjects. Although some developed increased responsiveness to cutaneous input, most also developed the ability to volitionally inhibit the response. Therefore, increasing voluntary control found in the data reported here was more likely a result of improvements within the long-tract systems of the spinal cord. Although any neurons with axons ending near spinal motor neurons or interneurons with vacated synaptic space would be candidates for postinjury synaptogenesis, the functional EMG method used here minimizes the participation of vestibulospinal, reticulospinal and propriospinal systems by testing in the fully supported supine position. Thus, the method as applied is likely to be most sensitive to corticospinal system function. In fact, within chronic SCI subjects, the responsiveness of the corticospinal system to transcranial magnetic stimulation was related to higher SI values for dorsiflexion14 and improvement of gait after training.15 In both studies, thresholds for the elicitation of motor-evoked potentials from muscles caudal to the lesion were the lowest when motor control was the best.
A final component of the recovery process must certainly be the functional reorganization that cortical motor circuitry undergoes16 as it ‘learns’ to make the ‘best use’ of surviving and new corticospinal and corticobulbospinal architecture. Synergistic, co-activated, multimuscle patterns reflecting the characteristics of the musculoskeletal system and the constraints to movement due to the physical environment are useful in the control of often-repeated movements.17 However, in disordered control as is characteristic of incomplete SCI, inappropriate co-activation decreases the forces developed during voluntary movement attempts.18 Although not specifically described in the literature, such co-activation would likely reduce endurance and contribute significantly to a person's SCI-induced disability. Mechanisms of inhibitory control that are affected by SCI include presynaptic inhibition of Ia inputs from muscles being stretched;19 enhanced fusimotor activity of the static γ-motor neurons; non-reciprocal Ib inhibition;20 reciprocal or ‘disynaptic’ inhibition;21 and recurrent or Renshaw inhibition.21 All but the Renshaw inhibitory process would be involved in the refinement of volitional control after SCI. Further, the reduction in responsiveness to passive stretch and cutaneous input suggests an increase in the efficacy of inhibitory control.
In humans, no methods have been developed to independently monitor changes specific to each of the excitatory and inhibitory processes listed above. However, it is possible to monitor net effects as they work in parallel to increase long-tract synaptic connections to spinal motor circuits. For example, in a coarse manner, the AIS can be used to recognize large changes as Marino and co-workers did in 1999 when they reported that, of 775 initially motor-complete SCI subjects, 16% became incomplete within the first year after injury.22 Neurophysiological measures offer greater sensitivity to this net change. For example, >60% of persons clinically categorized as motor-complete lesions, AIS-A or B, have been shown to have residual translesional ability to modulate spinal motor excitability.6 In the current report, the rate of recruitment of prime mover muscle motor units would be difficult to measure without sEMG. Further, the co-activation of prime mover and distant muscles during voluntary movement attempts, a common feature within the AIS-C and D categories,18 can only be captured through neurophysiological examination.
The neurophysiological quantification of recovery or intervention effect offers the potential for greater sensitivity and reliability, but additional study is needed. First, the functional EMG protocol used here should be reduced to focus on specific body regions. Each phase of the voluntary movement tasks should be performed independently from as nearly complete relaxation as is possible. In addition, they should be performed self-paced, as in this study, and repeated under instruction to do so as fast as possible to provide true reaction-time information and test the maximum rate or prime mover recruitment. Further, better temporal synchronization of functional EMG and clinical assessment scale sessions is needed to better relate findings to currently recognized measures. Finally, functional EMG parameters introduced here need to be compared with clinically relevant measures of function to examine clinical and predictive capacity. Regardless, the findings presented here introduce new parameters that are objectively derived from non-invasively acquired spinal motor output with which clinicians and clinical researchers can measure and track changes in spatiotemporal aspects of motor control recovery that may result from spontaneous recovery as in the current study, or that may result from intervention or additional disease processes.
References
Yilmaz F, Sahin F, Aktug S, Kuran B, Yilmaz A . Long-term follow-up of patients with spinal cord injury. Neurorehabil Neural Repair 2005; 19: 332–337.
Illis LS . The motor neuron surface and spinal shock. Mod Trends Neurol 1967; 4: 53–68.
Sasaki M, Li B, Lankford KL, Radtke C, Kocsis JD . Remyelination of the injured spinal cord. Prog Brain Res 2007; 161: 419–433.
Kakulas BA . Neuropathology: the foundation for new treatments in spinal cord injuryLudwig Guttman Lecture). Spinal Cord 2004; 42: 549–563.
Marino RJ, Barros T, Biering-Sorensen F, Burns SP, Donovan WH, Graves DE et al. International standards for neurological classification of spinal cord injury. J Spinal Cord Med 2003; 26 (Suppl 1): S50–S56.
McKay WB, Lim HK, Priebe MM, Stokic DS, Sherwood AM . Clinical neurophysiological assessment of residual motor control in post-spinal cord injury paralysis. Neurorehabil Neural Repair 2004; 18: 144–153.
Calancie B, Molano MR, Broton JG . EMG for assessing the recovery of voluntary movement after acute spinal cord injury in man. Clin Neurophysiol 2004; 115: 1748–1759.
Lee DC, Lim HK, McKay WB, Priebe MM, Holmes SA, Sherwood AM . Toward an objective interpretation of surface EMG patterns: a voluntary response index (VRI). J Electromyogr Kinesiol 2004; 14: 379–388.
Lim HK, Sherwood AM . Reliability of surface electromyographic measurements from subjects with spinal cord injury during voluntary motor tasks. J Rehab Res Dev 2005; 42: 413–422.
Sherwood AM, McKay WB, Dimitrijevic MR . Motor control after spinal cord injury: assessment using surface EMG. Muscle Nerve 1996; 19: 966–979.
Hill CE, Beattie MS, Bresnahan JC . Degeneration and sprouting of identified descending supraspinal axons after contusive spinal cord injury in the rat. Exp Neurol 2001; 171: 153–169.
Fouad K, Tse A . Adaptive changes in the injured spinal cord and their role in promoting functional recovery. Neurol Res 2008; 30: 17–27.
McCough GP, Austin GM, Liu CN, Liu CY . Sprouting as a cause of spasticity. J Neurophysiol 1958; 21: 205–216.
McKay WB, Lim HK, Priebe MM, Stokic DS, Sherwood AM . Neurophysiological examination of the corticospinal system and voluntary motor control in motor-incomplete human spinal cord injury. Exp Brain Res 2005; 163: 379–387.
Thomas SL, Gorassini MA . Increases in corticospinal tract function by treadmill training after incomplete spinal cord injury. J Neurophysiol 2005; 94: 2844–2855.
Crawley AP, Jurkiewicz MT, Yim A, Heyn S, Verrier MC, Fehlings MG et al. Absence of localized grey matter volume changes in the motor cortex following spinal cord injury. Br Res 2004; 1028: 19–25.
Tresch MC, Jarc A . The case for and against muscle synergies. Curr opin neurobiol 2009; 19: 601–607.
Lim HK, Lee D, McKay WB, Priebe MM, Holmes SA, Sherwood AM . Neurophysiological assessment of lower-limb voluntary control in incomplete spinal cord injury. Spinal Cord 2005; 43: 283–290.
Knikou M . Effects of hip joint angle changes on intersegmental spinal coupling in human spinal cord injury. Exp Brain Res 2005; 167: 381–393.
McCrea DA . Neuronal basis of afferent-evoked enhancement of locomotor activity. Ann N Y Acad Sci 1998; 16: 216–225.
Nielsen JB, Crone C, Hultborn H . The spinal pathophysiology of spasticity—from a basic science point of view. Acta Physiol (Oxf) 2007; 189: 171–180.
Marino RJ, Ditunno JF, Donovan WH, Maynard F . Neurologic recovery after traumatic spinal cord injury: data from the model spinal cord injury systems. Arch Phys Med Rehabil 1999; 80: 1291–1396.
Acknowledgements
The authors express their appreciation to the subjects who participated in this study, to Anne Watson, RN, for project management support and Renee Ford for obtaining informed consent and coordination of study scheduling. We are also grateful to Yangsheng Chen for data conversion and Dongchul Lee, and Matthew Nitzken for their preparation of tools used in the calculation of the voluntary response index. In addition, we would like to acknowledge the neurosurgery residents at the University of Louisville Hospital for providing the clinical assessments reported in this study. This study was supported by NIH NINDS funded project #NS049954-01.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
McKay, W., Ovechkin, A., Vitaz, T. et al. Neurophysiological characterization of motor recovery in acute spinal cord injury. Spinal Cord 49, 421–429 (2011). https://doi.org/10.1038/sc.2010.145
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2010.145
Keywords
This article is cited by
-
Properties of the surface electromyogram following traumatic spinal cord injury: a scoping review
Journal of NeuroEngineering and Rehabilitation (2021)
-
Motor recovery after activity-based training with spinal cord epidural stimulation in a chronic motor complete paraplegic
Scientific Reports (2017)
-
Reliability of surface EMG as an assessment tool for trunk activity and potential to determine neurorecovery in SCI
Spinal Cord (2015)
-
Spinal cord injury and physical activity: preservation of the body
Spinal Cord (2012)
-
Respiratory Motor Control Disrupted by Spinal Cord Injury: Mechanisms, Evaluation, and Restoration
Translational Stroke Research (2011)