Abstract
In addition to fatty acids, glucose and lactate are important myocardial substrates under physiologic and stress conditions. They are metabolized to pyruvate, which enters mitochondria via the mitochondrial pyruvate carrier (MPC) for citric acid cycle metabolism. In the present study, we show that MPC-mediated mitochondrial pyruvate utilization is essential for the partitioning of glucose-derived cytosolic metabolic intermediates, which modulate myocardial stress adaptation. Mice with cardiomyocyte-restricted deletion of subunit 1 of MPC (cMPC1−/−) developed age-dependent pathologic cardiac hypertrophy, transitioning to a dilated cardiomyopathy and premature death. Hypertrophied hearts accumulated lactate, pyruvate and glycogen, and displayed increased protein O-linked N-acetylglucosamine, which was prevented by increasing availability of non-glucose substrates in vivo by a ketogenic diet (KD) or a high-fat diet, which reversed the structural, metabolic and functional remodelling of non-stressed cMPC1−/− hearts. Although concurrent short-term KDs did not rescue cMPC1−/− hearts from rapid decompensation and early mortality after pressure overload, 3 weeks of a KD before transverse aortic constriction was sufficient to rescue this phenotype. Together, our results highlight the centrality of pyruvate metabolism to myocardial metabolism and function.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
All data, apart from the western blots, that support the findings of the present study are available from the corresponding author upon reasonable request. Source data for western blots are provided with this paper.
Change history
18 November 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Wende, A. R., Brahma, M. K., McGinnis, G. R. & Young, M. E. Metabolic origins of heart failure. JACC Basic Transl. Sci. 2, 297–310 (2017).
Doenst, T., Nguyen, T. D. & Abel, E. D. Cardiac metabolism in heart failure: implications beyond ATP production. Circ. Res. 113, 709–724 (2013).
Owen, O. E., Kalhan, S. C. & Hanson, R. W. The key role of anaplerosis and cataplerosis for citric acid cycle function. J. Biol. Chem. 277, 30409–30412 (2002).
Des Rosiers, C., Labarthe, F., Lloyd, S. G. & Chatham, J. C. Cardiac anaplerosis in health and disease: food for thought. Cardiovasc. Res. 90, 210–219 (2011).
Pound, K. M. et al. Substrate-enzyme competition attenuates upregulated anaplerotic flux through malic enzyme in hypertrophied rat heart and restores triacylglyceride content: attenuating upregulated anaplerosis in hypertrophy. Circ. Res. 104, 805–812 (2009).
Lommi, J. et al. Blood ketone bodies in congestive heart failure. J. Am. Coll. Cardiol. 28, 665–672 (1996).
Aubert, G. et al. The failing heart relies on ketone bodies as a fuel. Circulation 133, 698–705 (2016).
Ho, K. L. et al. Increased ketone body oxidation provides additional energy for the failing heart without improving cardiac efficiency. Cardiovasc. Res. 115, 1606–1616 (2019).
Huang, Y., Zhou, M., Sun, H. & Wang, Y. Branched-chain amino acid metabolism in heart disease: an epiphenomenon or a real culprit? Cardiovasc. Res. 90, 220–223 (2011).
Marazzi, G., Rosanio, S., Caminiti, G., Dioguardi, F. S. & Mercuro, G. The role of amino acids in the modulation of cardiac metabolism during ischemia and heart failure. Curr. Pharm. Des. 14, 2592–2604 (2008).
Drake, K. J., Sidorov, V. Y., McGuinness, O. P., Wasserman, D. H. & Wikswo, J. P. Amino acids as metabolic substrates during cardiac ischemia. Exp. Biol. Med. 237, 1369–1378 (2012).
Allard, M. F., Schonekess, B. O., Henning, S. L., English, D. R. & Lopaschuk, G. D. Contribution of oxidative metabolism and glycolysis to ATP production in hypertrophied hearts. Am. J. Physiol. 267, H742–H750 (1994).
Kagaya, Y. et al. Effects of long-term pressure overload on regional myocardial glucose and free fatty acid uptake in rats. A quantitative autoradiographic study. Circulation 81, 1353–1361 (1990).
Karwi, Q. G., Uddin, G. M., Ho, K. L. & Lopaschuk, G. D. Loss of metabolic flexibility in the failing heart. Front. Cardiovasc. Med. 5, 68 (2018).
Chandramouli, C. et al. Myocardial glycogen dynamics: new perspectives on disease mechanisms. Clin. Exp. Pharm. Physiol. 42, 415–425 (2015).
Leong, H. S., Brownsey, R. W., Kulpa, J. E. & Allard, M. F. Glycolysis and pyruvate oxidation in cardiac hypertrophy—why so unbalanced? Comp. Biochem Physiol. A Mol. Integr. Physiol. 135, 499–513 (2003).
Lopaschuk, G. D., Wambolt, R. B. & Barr, R. L. An imbalance between glycolysis and glucose oxidation is a possible explanation for the detrimental effects of high levels of fatty acids during aerobic reperfusion of ischemic hearts. J. Pharm. Exp. Ther. 264, 135–144 (1993).
Nascimben, L. et al. Mechanisms for increased glycolysis in the hypertrophied rat heart. Hypertension 44, 662–667 (2004).
Comte, B. et al. A 13C mass isotopomer study of anaplerotic pyruvate carboxylation in perfused rat hearts. J. Biol. Chem. 272, 26125–26131 (1997).
Peuhkurinen, K. J., Nuutinen, E. M., Pietilainen, E. P., Hiltunen, J. K. & Hassinen, I. E. Role of pyruvate carboxylation in the energy-linked regulation of pool sizes of tricarboxylic acid-cycle intermediates in the myocardium. Biochem. J. 208, 577–581 (1982).
Pisarenko, O. I., Solomatina, E. S. & Studneva, I. M. The role of amino acid catabolism in the formation of the tricarboxylic acid cycle intermediates and ammonia in anoxic rat heart. Biochim. Biophys. Acta 885, 154–161 (1986).
Gibala, M. J., Young, M. E. & Taegtmeyer, H. Anaplerosis of the citric acid cycle: role in energy metabolism of heart and skeletal muscle. Acta Physiol. Scand. 168, 657–665 (2000).
Opie, L. H. & Mansford, K. R. L. The value of lactate and pyruvate measurements in the assessment of the redox state of free nicotinamide-adenine dinucleotide in the cytoplasm of perfused rat heart. Eur. J. Clin. Invest. 1, 295–306 (1971).
Lazo, P. A. & Sols, A. Pyruvate dehydrogenase complex of ascites tumour. Activation by AMP and other properties of potential significance in metabolic regulation. Biochem. J. 190, 705–710 (1980).
Comte, B., Vincent, G., Bouchard, B. & Des Rosiers, C. Probing the origin of acetyl-CoA and oxaloacetate entering the citric acid cycle from the 13C labeling of citrate released by perfused rat hearts. J. Biol. Chem. 272, 26117–26124 (1997).
Bricker, D. K. et al. A mitochondrial pyruvate carrier required for pyruvate uptake in yeast, Drosophila, and humans. Science 337, 96–100 (2012).
Halestrap, A. P. The mitochondrial pyruvate carrier: has it been unearthed at last? Cell Metab. 16, 141–143 (2012).
Herzig, S. et al. Identification and functional expression of the mitochondrial pyruvate carrier. Science 337, 93–96 (2012).
Vanderperre, B. et al. Embryonic lethality of mitochondrial pyruvate carrier 1 deficient mouse can be rescued by a ketogenic diet. PLoS Genet. 12, e1006056 (2016).
Gray, L. R. et al. Hepatic mitochondrial pyruvate carrier 1 is required for efficient regulation of gluconeogenesis and whole-body glucose homeostasis. Cell Metab. 22, 669–681 (2015).
McCommis, K. S. et al. Loss of mitochondrial pyruvate carrier 2 in the liver leads to defects in gluconeogenesis and compensation via pyruvate–alanine cycling. Cell Metab. 22, 682–694 (2015).
Rauckhorst, A. J. et al. The mitochondrial pyruvate carrier mediates high fat diet-induced increases in hepatic TCA cycle capacity. Mol. Metab. 6, 1468–1479 (2017).
Sharma, A. et al. Impaired skeletal muscle mitochondrial pyruvate uptake rewires glucose metabolism to drive whole-body leanness. eLife 8, e45873 (2019).
Fernandez-Caggiano, M. et al. Analysis of mitochondrial proteins in the surviving myocardium after ischemia identifies mitochondrial pyruvate carrier expression as possible mediator of tissue viability. Mol. Cell Proteom. 15, 246–255 (2016).
Abel, E. D. et al. Cardiac hypertrophy with preserved contractile function after selective deletion of GLUT4 from the heart. J. Clin. Invest. 104, 1703–1714 (1999).
Krebs, H. A. & Gascoyne, T. The redox state of the nicotinamide-adenine dinucleotides in rat liver homogenates. Biochem. J. 108, 513–520 (1968).
Ruiz, M., Gelinas, R., Vaillant, F., Lauzier, B. & Des Rosiers, C. Metabolic tracing using stable isotope-labeled substrates and mass spectrometry in the perfused mouse heart. Methods Enzymol. 561, 107–147 (2015).
Halestrap, A. P. & Denton, R. M. The specificity and metabolic implications of the inhibition of pyruvate transport in isolated mitochondria and intact tissue preparations by alpha-cyano-4-hydroxycinnamate and related compounds. Biochem. J. 148, 97–106 (1975).
Contreras-Baeza, Y. et al. Monocarboxylate transporter 4 (MCT4) is a high affinity transporter capable of exporting lactate in high-lactate microenvironments. J. Biol. Chem. 294, 20135–20147 (2019).
Lindblom, P. et al. Isoforms of alanine aminotransferases in human tissues and serum: differential tissue expression using novel antibodies. Arch. Biochem. Biophys. 466, 66–77 (2007).
O’Donnell, J. M., Kalichira, A., Bi, J. & Lewandowski, E. D. In vivo, cardiac-specific knockdown of a target protein, malic enzyme-1, in rat via adenoviral delivery of DNA for non-native miRNA. Curr. Gene Ther. 12, 454–462 (2012).
Lahey, R. et al. Enhanced redox state and efficiency of glucose oxidation with miR based suppression of maladaptive NADPH-dependent malic enzyme 1 expression in hypertrophied hearts. Circ. Res. 122, 836–845 (2018).
Funk, A. M. et al. Effects of deuteration on transamination and oxidation of hyperpolarized 13C-pyruvate in the isolated heart. J. Magn. Reson. 301, 102–108 (2019).
Brooks, G. A. The science and translation of lactate shuttle theory. Cell Metab. 27, 757–785 (2018).
Chen, Y. J. et al. Lactate metabolism is associated with mammalian mitochondria. Nat. Chem. Biol. 12, 937–943 (2016).
Taylor, E. B. Functional properties of the mitochondrial carrier system. Trends Cell Biol. 27, 633–644 (2017).
Ferron, M., Denis, M., Persello, A., Rathagirishnan, R. & Lauzier, B. Protein O-GlcNacylation in cardiac pathologies: past, present, puture. Front. Endocrinol. 9, 819 (2018).
Arad, M. et al. Glycogen storage diseases presenting as hypertrophic cardiomyopathy. N. Engl. J. Med. 352, 362–372 (2005).
Puchalska, P. & Crawford, P. A. Multi-dimensional roles of ketone bodies in fuel metabolism, signaling, and therapeutics. Cell Metab. 25, 262–284 (2017).
Ruan, H. B. & Crawford, P. A. Ketone bodies as epigenetic modifiers. Curr. Opin. Clin. Nutr. Metab. Care 21, 260–266 (2018).
Horton, J. L. et al. The failing heart utilizes 3-hydroxybutyrate as a metabolic stress defense. JCI Insight 4, e124079 (2019).
Fernandez-Caggiano, M. et al. Mitochondrial pyruvate carrier abundance mediates pathological cardiac hypertrophy. Nat. Metabol. https://doi.org/10.1038/s42255-020-00276-5 (2020).
McCommis, K. S. et al. Nutritional modulation of heart failure in mitochondrial pyruvate carrier–deficient mice. Nat. Metabol. https://doi.org/10.1038/s42255-020-00296-1 (2020).
Ye, J. et al. Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinform. 13, 134 (2012).
Chong, J., Wishart, D. S. & Xia, J. Using MetaboAnalyst 4.0 for comprehensive and integrative metabolomics data analysis. Curr. Protoc. Bioinformatics 68, e86 (2019).
Riehle, C. et al. PGC-1β deficiency accelerates the transition to heart failure in pressure overload hypertrophy. Circ. Res. 109, 783–793 (2011).
Vaillant, F. et al. Ivabradine and metoprolol differentially affect cardiac glucose metabolism despite similar heart rate reduction in a mouse model of dyslipidemia. Am. J. Physiol. Heart Circ. Physiol. 311, H991–H1003 (2016).
Huang, X. et al. X13CMS: global tracking of isotopic labels in untargeted metabolomics. Anal. Chem. 86, 1632–1639 (2014).
Acknowledgements
This work was supported by: American Heart Association (AHA; grant nos. 16SFRN31810000 to E.D.A. and 15POST22940024 to Y.Z.); Montreal Heart Institute Foundation (CDR); National Institutes of Health (NIH; grant nos. OD019941 to R.M.W., and R01 DK104998 and R00 AR059190 to E.B.T.); T32 (grant no. HL007638 to A.J.R.); American Diabetes Association (grant no. 1-18-PDF-060 (to A.J.R.); and NIH (grant nos. F32 DK101183 to L.R.G., U54DK110858, 1S10OD021505 and 1S10OD018210 to J.E.C., R01HL113057, R01HL132525 and R01HL049244 to E.D.L. and DK091538 to P.A.C.).
Author information
Authors and Affiliations
Contributions
Y.Z. and E.D.A. designed the research. Y.Z., P.V.T., J.D.C., I.R.-F., J.M.M., J.S., A.D.P., F.T., L.M.T., A.J.R., L.R.G., P.P., T.R.F., R.M., K.Z., W.J.K., T.C., S.H., K.L., K.M.K., J.L.S., L.H., R.M.W. and J.E.C. performed the research. E.B.T. and J.R. provided materials and methodology support. Y.Z., E.D.L., P.A.C., C.D.R. and E.D.A. analysed the data. P.A.C., E.D.L. and C.D.R. contributed to the writing. Y.Z. and E.D.A. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Primary Handling Editor: George Caputa.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
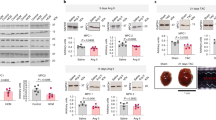
Extended Data Fig. 1 Mitochondrial Characterization of cMPC1-/- hearts.
a, b, MPC protein levels were determined by Western blots in whole heart lysates from 4-week-old cMPC1-/- mice. Images are representative of n = 8 per group. c, Palmitoyl-carnitine driven oxygen consumption by seahorse respirometry in isolated mitochondria (n = 6 both groups). d, Expression of selected electron transport chain (ETC) subunits (from Complex I-V) and VDAC by Western blot in heart lysates from control and cMPC1-/- mice. Images are representative of n = 3 per group. e, mtDNA copy number determined by qPCR analysis and normalized to the nuclear gene RPL13A in 8-week-old cMPC1-/- hearts (Control, 12; cMPC1-/-, 10). f, Representative TEM images of cMPC1-/- hearts from 4 and 8-week-old mice. Images are representative of n = 9 (4-week-old control and cMPC1-/-)/6(8-week-old control)/12(8-week-old cMPC1-/-). g, Quantification of mitochondrial number, volume density and size (4 weeks: Control, 9; cMPC1-/-, 9; 8 weeks: Control, 6; cMPC1-/-, 12). Data are presented as mean ± SEM and analyzed by two-tailed unpaired Student’s t-test.
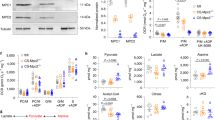
Extended Data Fig. 2 Glycolysis enzymes and intermediates in cMPC1-/- hearts.
a, Glycolysis-derived metabolic intermediates in 8-week-old control and cMPC1-/- hearts were determined by GC-MS. Mice were random fed before sacrifice. (Control, 15; cMPC1-/-, 11). b, hexokinase I (HK I), GAPDH, O-GlcNAc transferase (OGT) and glycogen synthase (GS) blots were performed in lysates of cardiomyocytes isolated from 8-week-old control and cMPC1-/- mice. Protein quantification was normalized to total Coomassie blue staining. (n = 5 both groups). Data are presented as mean ± SEM and analyzed by two-tailed unpaired Student’s t-test.
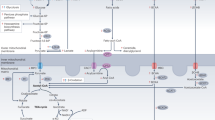
Extended Data Fig. 3 Flux scheme of 13C-labeled substrate utilization in cMPC1-/- hearts.
Schematic depicting metabolic fate of uniformly labeled glucose ([U-13C6]-glucose) into glycolysis, the pentose phosphate pathway (PPP), serine biosynthesis pathway (SBP) and the Citric Acid Cycle (CAC). Closed and open circles represent 13C-labeled and 12C-labeled carbons respectively.
Extended Data Fig. 4 13C-MPE for pyruvate, lactate, alanine and serine from [U-13C6]-glucose perfusion.
13C-isotopomer labeling pattern of pyruvate (a), lactate (b), alanine (c) and serine (d) following [U-13C6]-glucose perfusion (Control, 7; cMPC1-/-, 9). Data are presented as mean ± SEM and P values were determined by two-tailed unpaired Student’s t-test.
Extended Data Fig. 5 13C-isotopomer labeling pattern of CAC intermediates from 13C-β-HB perfusion.
13C-labeled CAC intermediates analysis of Langendorff-perfused hearts perfused with [2,4-13C2]-β-HB and unlabeled glucose, palmitate and lactate. Fractional enrichment 13C-labeled isotopomers of citrate (a), glutamate (b), succinate (c), α-ketoglutarate (KG) (d), malate (e), aspartate (f) and β-hydroxybutyrate (HB) (g) were determined by LC-MS (Control, 3; cMPC1-/-, 5). Data are presented as mean ± SEM and P values were determined by two-tailed unpaired Student’s t-test.
Extended Data Fig. 6 Expression levels of hypertrophic markers and selected transcripts encoding metabolic genes in the cMPC1-/- hearts on 2920X and ketogenic diet.
The cMPC1-/- mice under protocol 2 were analyzed for gene expression after 8-week-feeding on Ketogenic (Keto) Diet or control diet (2920X). Sample sizes: n = 4 (Control-2920X), n = 4 (cMPC1-/–2920X), n = 5 (Control-Keto), n = 6 (cMPC1-/–Keto). Data are presented as mean ± SEM and P values were determined by two-way ANOVA followed by Tukey multiple comparison test.
Extended Data Fig. 7 Cardiac function of cMPC1-/- after switching ketogenic diet to regular chow.
10-week-old control and cMPC1-/- mice were fed a Keto Diet for 8 weeks and then 50% of mice of each genotype were switched to regular chow for 6 weeks. The feeding scheme is shown in panel (a). LV mass (b) and ejection fraction (c) were determined via echocardiography at the age of 22 weeks (4 weeks after chow switch). Heart weight and tibia length (d) was determined at the age of 24 weeks. Sample sizes: n = 10 (Control-2920X), n = 8 (cMPC1-/–2920X), n = 9 (Control-Keto), n = 8 (cMPC1-/–Keto). Data are presented as mean ± SEM and P values were determined by two-way ANOVA followed by Tukey multiple comparison test.
Extended Data Fig. 8 Effects of ketogenic diet feeding on pressure overload-induced cardiac remodeling in WT mice.
a–c, 8-week-old WT C57Bl6/J mice were fed with chow and Keto Diet 1 day before TAC surgery. LV mass (a) and ejection fraction (b) were measured by echocardiography prior to surgery and 3 weeks post TAC. Heart weight normalized to tibia length (c) was determined at the time of sacrifice. (n = 5 for both groups). d–f, 12-week-old WT C57Bl6/J mice were fed with chow and Keto Diet 1 day before sham/TAC surgery. LV mass (d) and ejection fraction (e) were measured by echocardiography prior to surgery and 3 weeks post TAC. Heart weight normalized to tibia length (f) was determined at the time of sacrifice. (n = 10 for Keto Diet-TAC group and n = 5 for other groups). Data are presented as mean ± SEM and P value was determined by two-way ANOVA followed by Tukey multiple comparison test.
Extended Data Fig. 9 mRNA level of ME isoforms and ALT activity in cMPC1-/- hearts.
a, mRNA level of three malic enzyme isoforms were determined by qPCR in the hearts from control and cMPC1-/- mice (Control, 6; cMPC1-/-, 5). b, c, ALT activity (Cayman 700260) were determined in the hearts from 8-week-old and 18-week-old control and cMPC1-/- mice (8-week-old group: Control, 6; cMPC1-/-, 4. 18-week-old group: Control, 7; cMPC1-/-,7). Data are presented as mean ± SEM and P values were determined by two-tailed unpaired Student’s t-test.
Extended Data Fig. 10 13C-labeled CAC intermediates analysis from [U-13C5]-glutamine perfusion.
13C-labeled CAC intermediates analysis of Langendorff-perfused hearts perfused with 0.5 mM [U-13C5]-glutamine and unlabeled substrates (10 mM glucose, 0.4 mM palmitate, 0.5 mM lactate, and 0.1 mM β-HB). 13C-MPE of glutamine (a), glutamate (b), alpha ketoglutarate (a-KG) (c), citrate (d), malate (e), succinate (f) and pyruvate (g) were determined by GC-MS (Control, 6; cMPC1-/-, 6). Data are presented as mean ± SEM and P values were determined by two-tailed unpaired Student’s t-test.
Supplementary information
Supplementary Tables
Supplementary Tables 1 and 2
Source data
Source Data Fig. 1
Unprocessed western blots.
Source Data Extended Data Fig. 1
Unprocessed western blots.
Source Data Extended Data Fig. 2
Unprocessed western blots.
Rights and permissions
About this article
Cite this article
Zhang, Y., Taufalele, P.V., Cochran, J.D. et al. Mitochondrial pyruvate carriers are required for myocardial stress adaptation. Nat Metab 2, 1248–1264 (2020). https://doi.org/10.1038/s42255-020-00288-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-020-00288-1
This article is cited by
-
Control of NAD+ homeostasis by autophagic flux modulates mitochondrial and cardiac function
The EMBO Journal (2024)
-
From Energy Metabolic Change to Precision Therapy: a Holistic View of Energy Metabolism in Heart Failure
Journal of Cardiovascular Translational Research (2024)
-
Untargeted mass spectrometry-based metabolomics approach unveils biochemical changes in compound probiotic fermented milk during fermentation
npj Science of Food (2023)
-
Metabolic Messengers: ketone bodies
Nature Metabolism (2023)
-
Hallmarks of cardiovascular ageing
Nature Reviews Cardiology (2023)